Abstract
Appropriate end-points are integral to the refinement of laboratory animal experiments. Our recent experience has highlighted that ambiguity around end-points is hampering their adoption in experiments that cause severe suffering to fish. In toxicology, the term endpoint (single word) refers to the response variable to the treatment that is measured and analysed. This differs to usage within laboratory animal experimentation, where end-point (hyphenated) refers to the time-point when exposure of the animal(s) to the treatment (and suffering) ends. Within laboratory animal experimentation, standardised terminology is needed for different types of early end-point which are based on the condition of the animal(s) or progress of the experiment. We propose that those involved in regulating and conducting animal experiments consider seven distinct types of early end-point (aim, technical error, biological error, mortality, moribundity, prognostic humane, non-prognostic humane) in addition to the planned experimental end-point (i.e. maximum duration). Moribundity (not morbidity) refers to an animal in a severely debilitated state close to death. Moribundity in fish is not yet defined, so we propose identification via a lack of response to external stimuli, loss of equilibrium (i.e. loss of righting reflex), and a slow opercular ventilation rate. As these clinical signs equate to those of deep/surgical anaesthesia, this moribundity end-point cannot be considered a humane end-point as the fish is likely to be unconscious and have passed the point of maximum suffering. We believe that identification of earlier humane end-points based on clinical signs and wider recognition of other types of early end-point can reduce suffering in experiments.
Introduction
Users of animals in laboratory experiments are legally and/or ethically obliged to minimise suffering in experimental animals via consideration and application of the 3Rs: replacement, reduction, refinement.1–3 Prescribed regulatory tests for fish, such as those assessing the acute toxicity of chemicals 4 and the efficacy of vaccines, 5 are still based around a mortality end-point and are amongst the most severe experiments that use fish. 6 As opportunities for replacement and reduction in such prescribed tests are limited or have already been addressed,7–9 the current focus is on refinement.
A key refinement required by European animal experimentation legislation is that ‘Death as the end-point of a procedure shall be avoided as far as possible and replaced by early and humane end-points’. 2 The need to identify and apply such end-points to reduce suffering has been highlighted. 10 However, terminology for different types of early end-point is variable11,12 and potentially confusing. Although the recently published PREPARE guidelines 13 highlight the need for consideration of humane end-points during study planning, potential other early end-points are overlooked.
Here we aim to clarify different types of early end-point for animal experiments. We believe that such elucidation is needed, based upon our recent experience: involvement in fish disease studies at the Cefas Weymouth Laboratory; 14 participation in the latest review of the Fish, Acute Toxicity Test (OECD Test Guideline 203, TG203);4,8 and attendance at three recent meetings (Severity and Humane Endpoints in Fish Research, Bergen, 4 October 2019; Third RSPCA international meeting, Focus on severe suffering: Avoiding mortality, Athens, 10–11 October 2019; Establishing score sheets and defining endpoints in fish experiments, Bergen, 28 January 2020). The benefits of clarifying end-points are likely to extend beyond fish to other experimental animals.
What is an end-point?
The hyphenated term end-point is used synonymously with the single word (endpoint) and un-hyphenated term (end point).2,3,10,13,15–19 The term is being used with two distinct meanings:
Within laboratory animal experimentation, end-point refers to the point at which exposure of the animal(s) to the treatment (and associated suffering) ends (sensu Wolfensohn and Lloyd).16 In toxicology, endpoint is a long-standing term referring to the response variable measured to assess treatment effects.19,20 Acute toxicology studies (< 7 d) have traditionally assessed mortality as the endpoint to determine LC50 or LD50 (i.e. the lethal dose/concentration of a compound required to kill half the organisms in the study) but may also assess lesions/injuries to the skin or eye as endpoints.19,20 Chronic (longer) toxicology studies typically assess multiple endpoints, which can be either dichotomous or continuous variables, and various descriptive prefixes have been used, for example mechanistic, sensitive, non-lethal, sublethal, apical, endocrine-specific endpoints.21 Response variables in health studies may similarly be termed endpoints, surrogate endpoints or clinical biomarkers.22,23
Parallel usage of the two distinct meanings has developed because: mortality and moribundity end-points refer to both the response variable and the point at which exposure to the treatment ends;
24
and the responses to a treatment (adverse effects) being used to determine the point at which exposure of the animal ends.
25
For future clarity, we recommend that:
the hyphenated term ‘end-point’ is used to refer to points at which exposure of the animal(s) to the treatment ends (consistent with the EU),2 and differentiated from the single word ‘endpoint’ which is restricted to response variables, as originally and still commonly used.
What categories of laboratory animal experimentation end-point are there?
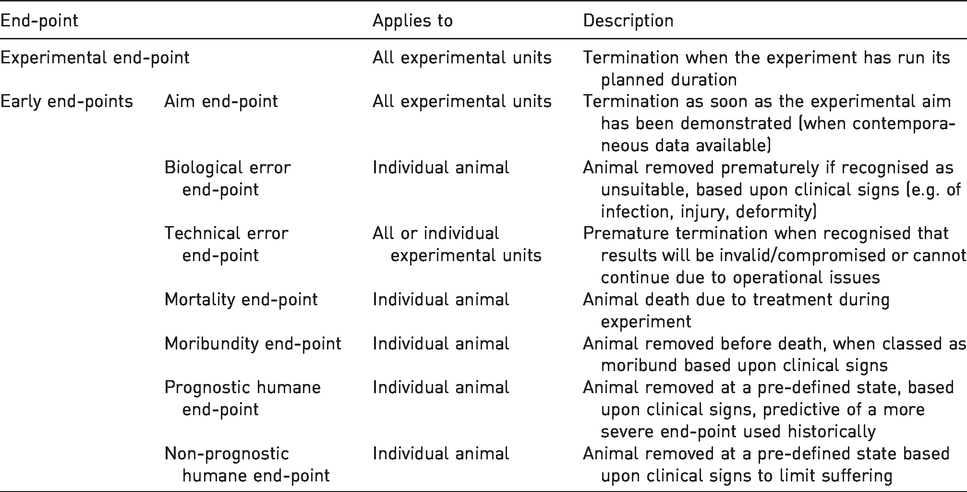
In their handbook of laboratory animal welfare, Wolfensohn and Lloyd 16 refer to three different types of end-point: experimental, error and humane. Morton 12 suggested five different categories of (humane) end-point. End-points can therefore be based on time, the progress of the experiment, or the condition of the animals. However, there does not appear to be standardised terminology and a wide range of other idioms is being used to refer to different types of end-point, for example objective-achieved, surrogate (pre-lethal), absolute, predictive, clinical, predetermined operational, welfare and scientific.11,26,27 We suggest differentiation of eight distinct categories of end-point (Table 1).
What is the experimental end-point?
Following Wolfensohn and Lloyd, 16 the experimental end-point is the pre-defined duration of the experiment, based on time and/or the progress of responses to the treatment. An example of an experimental end-point is the 96 h planned duration of TG203. 4 All other types of end-point occur before the experimental end-point and can therefore be considered as categories of early end-point.
Please note that the National Academy of Sciences 28 terms the experimental end-point when ‘the scientific aims and objectives have been reached’. This matches what we propose calling the aim end-point (see below) and suggest that use of experimental end-point is restricted to define the maximum duration.
What is the aim end-point?
Experimental study plans should have a clear aim, as well as a maximum duration. Once the aim has been achieved (e.g. demonstration of protection from disease), researchers are obliged to intervene 11 to end the experiment rather than continuing to the experimental end-point. This will only be possible when response data is available contemporaneously.
TG203 aims to assess the toxicity of a chemical over an experimental end-point (duration) of 96 h, but requires calculation of LC50 values for 24, 48, 72 and 96 h. 4 If treatment-related mortality occurs before 96 h and an earlier LC50 value indicative of toxicity can be calculated (e.g. LC50 at 24 h), then has the aim been achieved obliging early termination of the remaining fish?
What are error end-points?
Wolfensohn and Lloyd 16 described the error end-point as early termination when it is recognised that the experimental result will be invalid. We suggest dividing into two types: biological and technical. Biological error end-points are when individual animals develop a physiological (or psychological state) that may affect the response to the treatment, 12 such as infection with a non-target pathogen, injury or distressed behaviour that then may affect physiology or dosing. Examples of technical error end-point are: human error resulting in incorrect treatment doses being administered; system failures due to loss of power, food or water supply; insufficient animals remaining to conduct a statistically valid test. Prior consideration of action to take in the event of such occurrences will reduce any delay in interventions.
What is the mortality end-point?
If an animal dies due to the treatment before the experimental end-point, it has reached the mortality end-point. The mortality end-point was developed nearly 100 years ago, associated with the LD50 method of standardising drug toxicity assessment. 20 The mortality end-point has proved popular because ‘it is objective, is not time-consuming, and can be monitored by nonskilled staff’. 29 Regulatory toxicity tests involving mammals no longer require the mortality end-point which has been refined by use of humane end-points on ethical grounds.3,20 However, TG203 still requires the mortality end-point for fish, to enable calculation of LC50 values. 4
Death in fish can be difficult to confirm, so it has been defined as ‘no visible movement (e.g. gill movements) and if touching the caudal peduncle produces no reaction’. 4 TG203 requires that ‘mortalities are recorded, and dead fish are removed as soon as they are observed’, but does not state that death should be confirmed prior to disposal. To conform to the principles of humane experimentation, it is recommended that when such fish are removed, death is confirmed, for example, by destruction of the brain or presence of rigor mortis, as required for completing euthanasia. 2
What is the moribundity end-point?
Moribundity (i.e. the state of being moribund) generally refers to an animal ‘close to death’ 18 or in a ‘severely debilitated state that precedes imminent death’.7,30 (Moribundity has also been defined as ‘being in state of dying or inability to survive, even if treated’ 3 which is a broader state and reflects progression beyond the point of no return (see below).)
The term moribundity must not be confused with morbidity, which is also mentioned in relation to end-points, for example ‘use morbidity rather than mortality as the endpoint’; 18 ‘Death or severe morbidity as an endpoint should always be challenged in any field of research’. 31 Morbidity has a very different meaning, referring to ‘a departure… from a state of physical or mental well-being’. 32
There is a lack of clarity around moribundity in fish (as with higher vertebrates) and a definition is needed.4,24,30 In a discussion of regulatory testing of fish vaccines, Midtlyng et al.
7
suggest that the ‘moribund state is relatively easy to identify’ and that clinical signs in infected fish progress with: very sick fish becoming unresponsiveness to external stimuli, and usually float at the surface or sink to the bottom of the tank. This can progress to a loss of equilibrium, with fish floating ‘belly up’ as respiration rate slows. At this stage mortality usually occurs.
When considering moribundity in fish, it is pertinent to consider the progressive clinical signs of anaesthesia in fish (Table 2). The behavioural clinical signs we propose to identify the moribundity end-point match those of the deep/surgical anaesthesia stage in fish, which (imminently) precede the medullary collapse and death stages during overdose (Table 2). Fish in deep/surgical anaesthesia are considered unconscious, due to the loss of reaction to stimuli. Moribund fish are therefore similarly likely to be unconscious and incapable of suffering,18,30 and the moribundity end-point cannot be considered as a refinement of the mortality end-point. Although it is recommended that moribund fish are removed for humane killing, they can be assumed to have already experienced maximal suffering.7,30
Progression of clinical signs through anaesthesia stages in fish, collated from Brown, 33 Martins et al., 34 Ross et al., 35 and Tytler and Hawkins. 36 Please note that classification of stages and associated signs differ slightly between sources. Indication of assumed level of consciousness added, based on response to stimuli.
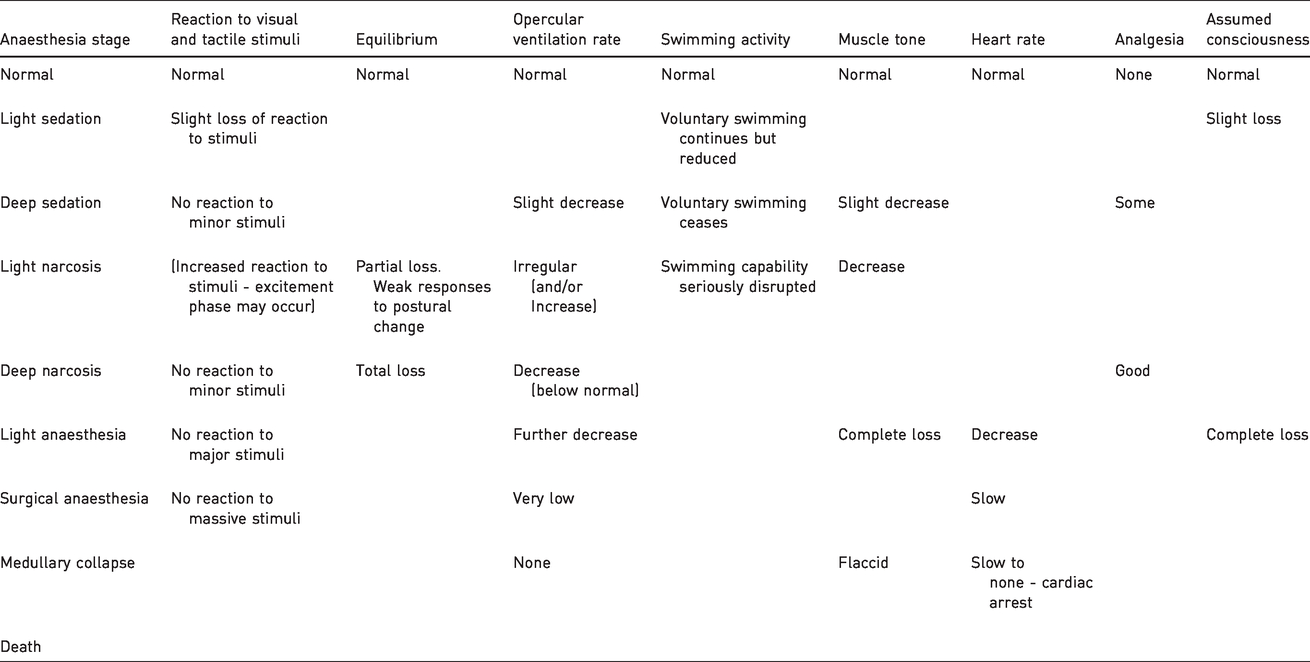
Early end-points based upon clinical signs, have been applied within TG203 in those countries where animal experimentation legislation requires that the mortality end-point is avoided. 4 Rufli 24 assessed the available data on clinical signs and five alternative definitions for the moribundity end-point (Table 3). However, the use of the term moribundity to classify these states appears to be a misnomer because: a clinical sign that is present for 2 d is not indicative of imminent death within a 4 d experiment; strong ventilation, and severe scores for swimming behaviour which include hyper-activity and abnormal swimming patterns, do not indicate a severely debilitated state. We therefore consider that the use and analysis of such early end-points by Rufli 24 represents progression towards introducing humane (rather than moribundity) end-points.
The clinical signs proposed previously by Rufli 24 for five different definitions of moribundity end-point assessed for the fish acute toxicity test TG203.

What is a humane end-point?
A humane end-point is the point when suffering of an individual animal due to a treatment is ended (typically by humane killing), based upon its clinical signs reaching pre-defined criteria that precede those of moribundity and mortality (sensu Olfert and Godson).
37
Given their importance to refinement, it is surprising that there is not already a consistent definition of humane end-point: • the point at which an experimental animal’s pain and/or distress can be terminated, minimised, or reduced by actions such as killing the animal humanely, terminating a painful procedure, or providing treatment to relieve pain and/or distress;11,18 • the earliest indicator in an animal experiment of severe pain, severe distress, suffering, or impending death;3 • the animal is killed at a defined point to limit its suffering;16 • clear, predictable and irreversible criteria which substitute for more severe experimental outcomes such as advanced pathology or death;38 • criteria that can serve as the basis for ending a test procedure earlier in order to terminate or avoid pain and distress associated with the traditional study endpoint, while still allowing attainment of study objectives;39 • the earliest indicator in an animal experiment of (potential) pain and/or distress that, within the context of moral justification and scientific endpoints to be met, can be used to avoid or limit pain and/or distress by taking actions such as humane killing or terminating or alleviating the pain and distress.40
Based on these variable definitions and our understanding, we consider that humane end-points can be divided into two distinct categories: non-prognostic and prognostic (Table 1). Non-prognostic humane end-points reflect cases where the researcher accepts that the experimental aim can be achieved without animals progressing to a more severe state associated with greater suffering. Prognostic humane end-points reflect cases where clinical sign data are analysed to identify those that are predictive of a historically used, more severe end-point; 39 Morton 12 termed such cases surrogate humane end-points. EU researchers are required to employ an experimental design and observations to minimise the animal suffering whilst achieving the experimental aim. 2 Humane end-points therefore need to be considered for all experimental procedures, regardless of severity.
A conceptual model is presented to illustrate the theoretical application of a humane end-point in a severe acute toxicity test where the conventional end-point was mortality (Figure 1); analogous stages can be considered for a severe disease challenge. There are various points and stages in the time-line leading to death of a fish (Figure 1(a)):
start of exposure to a toxic dose of a chemical; an initial period where there are likely to be physiological and molecular responses to the chemical, but no changes in appearance or behaviour due to toxicity are visible. Fish may however show transient aversive behaviour or hyperactivity if the chemical is a chemosensory irritant.41 A period when clinical signs (visible abnormalities in fish behaviour or appearance) occur due to toxicity which are assumed to intensify with duration of exposure due to increased toxic damage; the point of no return (PNR),16 that is, a theoretical point when damage to the fish exceeds the capacity to recover, and death is therefore inevitable at a future point; moribundity; death.
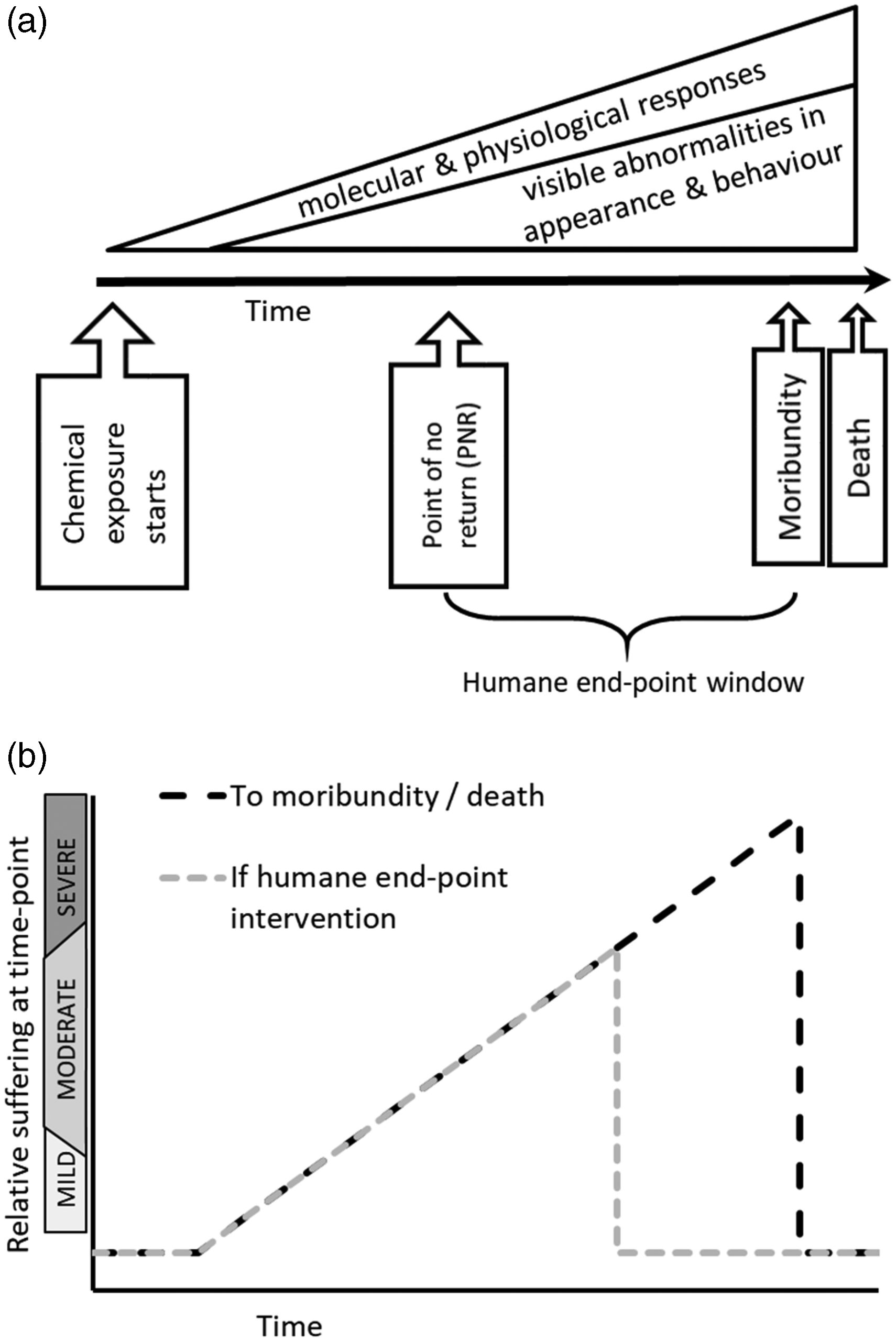
Conceptual model to illustrate the application of a humane end-point during the time course of a severe acute toxicity test, adapted from Wolfensohn and Lloyd, 16 and, Brønstad. 42 (a) Time-line of stages between initiation of exposure and death. (b) Suffering of individual fish at any time-point. Please note that severity bands have been included for illustrative purposes only. See text for further explanation.
This conceptual model can be expanded to visualise suffering (Figure 1(b)) based upon the assumptions that the level of suffering at any time-point increases as the test progresses, correlating with the degree of abnormality of the clinical signs, and moribund fish are past the point of suffering as discussed above.
A humane end-point, based upon visible abnormalities in behaviour and appearance (clinical signs), can therefore be applied between the PNR and moribundity, which reduces the suffering experienced. The theoretical model also illustrates that consideration should be given to continual refinement, that is, to reduce suffering by moving towards earlier, more humane end-points within the intervention window where possible.
Please note that the theoretical model (Figure 1(b)) illustrates the level of suffering at any time-point; the area under the line indicates the integrated suffering experienced to that point. 16 There is some ambiguity on severity assessment within the EU Directive 2 due to the requirement to consider ‘cumulative suffering within a procedure’. Our understanding is that: it should be based upon ‘the most severe effects likely to be experienced’, that is the maximum level of suffering at any time-point, and not the total suffering integrated over time; the requirement to consider ‘cumulative’ suffering is meant to reflect (usually) additive causes of suffering at any time-point, for example: handling to select a defined size range for the experiment + food deprivation + transfer to a barren test tank + abnormal social grouping + chemical exposure.
Discussion
End-points are integral to ethical animal experimentation, and there is often a legal duty to apply appropriate end-points when animals are used for scientific purposes. 2 However, terminology around end-points is confusing as highlighted in the Introduction. Our aim in writing this paper is to clarify the variable terminology to avoid further misunderstanding and encourage application. We have suggested division of early end-points into seven different types (Table 1). We hope that this framework and associated text does clarify, rather than further confuse, and its utility will be determined by uptake. We also expect our proposed framework to evolve further. For example, when clinical signs reach a specific stage in some animal models, it may be possible to make therapeutic interventions (e.g. fluid replacement, pain relief) to reduce suffering without ending the experiment. 29 Another example is the appearance of an unexpected phenotype in genetically modified animals; 28 rather than being identified as a biological error end-point and euthanised, these may be held under modified conditions due to intrinsic scientific value. As use of the animal continues in such cases, separate terminology may need to be developed for intervention points. 43
Prior consideration of the various early end-points has been introduced as part of study planning and ethical review process in our establishment; this reinforces the need to consider refinement and contributes to our culture of care. Application of the aim end-point has reduced suffering in our research studies of disease transmission and treatment efficacy. Moribundity, humane and biological error end-points are routinely applied by study staff via checks for general and treatment-specific clinical signs. 14 Wider appreciation of different early end-points by regulators and experimenters is likely to have broader refinement benefits for fish and other species.
There is growing acceptance that regulatory tests requiring mortality and severe suffering are ethically unacceptable. 22 With specific reference to TG203, discussions have been ongoing for a number of years to introduce an early end-point to refine the mortality end-point.4,24 The focus of debate was to use clinical signs to identify a prognostic humane end-point. Due to perceived reluctance to move substantially away from the mortality end-point (which reflects the environmental protection goal of acute toxicity testing) the term moribundity end-point was introduced. However, this term generated confusion and debate, which demonstrates that clarity in terminology is needed for progress at international level. Our aim in clarifying the moribundity end-point is to enable progression towards genuine humane end-points in regulatory tests using fish.
Momentum is building to implement humane end-points in severe eco-toxicity and fish disease experiments.7,8 To identify humane end-points, score sheets are needed for recording clinical signs, 44 and have been proposed for fish (Annex 4 in TG203;4 Appendix B in Midtlyng et al. 7 ). Laboratory fish researchers and technicians should already be trained and competent to recognise clinical signs 18 due to familiarity with the progressive stages of anaesthesia in fish (Table 2). This combination provides an important foundation from which to identify and implement humane end-points in fish. Katsiadaki et al. 8 discuss practical and regulatory issues associated with implementation of humane end-points in fish toxicity testing. The refinement discussions for TG203 have also expanded to include addressing a separate early end-point, the aim end-point, that is early termination where a toxicity result is derived before the 96 h experimental end-point. 8
Footnotes
Acknowledgements
The manuscript benefited from comments from Hans Rufli, Chris Green, three anonymous referees and the journal editors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Defra, International Chemicals (How can we produce internationally standardised guidance for assessing sublethal clinical signs of toxicity in fish to improve animal welfare in chemical safety tests?); the Veterinary Medicines Directorate (VM0519: Can humane end-points be introduced into fish disease challenges for fish medicine development?).