Abstract
An otherwise healthy two-month-old female C57BL/6J mouse presented with a left-sided head tilt. Differential diagnoses included idiopathic necrotizing arteritis, bacterial otitis media/interna (Pasteurella pneumotropica, Pseudomonas aeruginosa, Streptococcus sp., Mycoplasma pulmonis and Burkholderia gladioli), encephalitis, an abscess, neoplasia, a congenital malformation and an accidental or iatrogenic head trauma. Magnetic resonance imaging (MRI) revealed a large space-occupying right olfactory lobe intra-axial lesion with severe secondary left-sided subfalcine herniation. Following imaging, the animal was euthanized due to poor prognosis. Histopathologic examination revealed a unilateral, full-thickness bone defect at the base of the cribriform plate and nasal conchae dysplasia, resulting in the herniation of the olfactory bulb into the nasal cavity. There was also a left midline-shift of the frontal cortex and moderate catarrhal sinusitis in the left mandibular sinus. The MRI and histopathologic changes are consistent with a congenital malformation of the nasal cavity and frontal aspect of the skull known as an ethmoidal meningoencephalocele. Encephaloceles are rare abnormalities caused by herniation of contents of the brain through a defect in the skull which occur due to disruption of the neural tube closure at the level anterior neuropore or secondary to trauma, surgical complications, cleft palate or increased intracranial pressure. The etiology is incompletely understood but hypotheses include genetics, vitamin deficiency, teratogens, infectious agents and environmental factors. Ethmoidal encephaloceles have been reported in multiple species including humans but have not been reported previously in mice. There are multiple models for spontaneous and induced craniofacial malformation in mice, but none described for ethmoidal encephaloceles.
Case report
A two-month-old female C57BL/6J (The Jackson Laboratory, Bar Harbor, ME, USA) mouse presented with a moderate left-sided head tilt. The mouse was obese but otherwise unremarkable and was housed with four healthy cagemates. The mouse had been provided a purified high-fat diet (60%) diet (AIN-93) ad libitum as part of an IACUC approved study since its arrival from the vendor two weeks prior to presentation. All mice in the cohort were housed at Weill Cornell Medicine/Memorial Sloan Kettering Cancer Center in an AAALAC-accredited facility in accordance with the Guide for the Care and Use of Laboratory Animals: Eighth Edition 1 and maintained in individually ventilated cages (no. 9, Thoren Caging Systems, Hazelton, PA, USA), and provided autoclaved aspen chip bedding (PWI Industries, Quebec, Canada) and acidified reverse osmosis purified water (pH 2.5–2.8, with hydrochloric acid). The room was ventilated with 100% filtered air at 10 to 15 air changes hourly, maintained on a 12:12-h light:dark cycle, with a relative humidity of 30–70%, and a room temperature of 22.2 ± 1.1°C. The vendor’s and the recipient colony are free of mouse hepatitis virus, Sendai virus, mouse parvovirus, minute virus of mice, pneumonia virus of mice, Theiler meningoencephalitis virus, mouse rotavirus (epizootic diarrhea of infant mice virus), ectromelia virus, reovirus 3, lymphocytic choriomeningitis virus, K virus, mouse adenovirus types 1 and 2, polyoma virus, mouse cytomegalovirus, mouse thymic virus and Hanta virus; Mycoplasma pulmonis, Citrobacter rodentium, Salmonella spp., Filobacterium rodentium and Clostridium piliforme; and fur mites (Myobia musculi, Myocoptes musculinis and Radfordia affinis), pinworms (Syphacia spp. and Aspiculuris tetraptera) and Encephalitozoon cuniculi.
Possible differential diagnoses for the head tilt included idiopathic necrotizing arteritis, bacterial otitis media/interna (e.g. Pasteurella pneumotropica, M. pulmonis and Burkholderia gladioli), meningoencephalitis, an abscess, neoplasia, a congenital malformation and accidental or iatrogenic head trauma. Magnetic resonance imaging (MRI; 7.0 Tesla Avance III BioSpec 70/30 USR, Bruker Biospin MRI, Inc., Billerica, MA, USA) was performed the day after clinical presentation. Only T2-weighted (T2W) transverse images were acquired and revealed a space-occupying intra-axial lesion in the right olfactory lobe, T2W-isointense compared to the grey matter, with severe left sided-displacement of the longitudinal cerebral fissure and secondary compression of the left olfactory lobe (Figure 1). The caudal extent of this intra-axial right forebrain lesion was poorly defined, with a mass effect in the right aspect of the thalamus/ventral aspect of the right lateral ventricle, faintly T2W iso-to-hyperintense, with secondary compression and effacement of the right lateral ventricle. Left subfalcine herniation of the falx cerebri could also be noted secondary to this right-sided intra-axial lesion (Figure 1(a)–(f)).
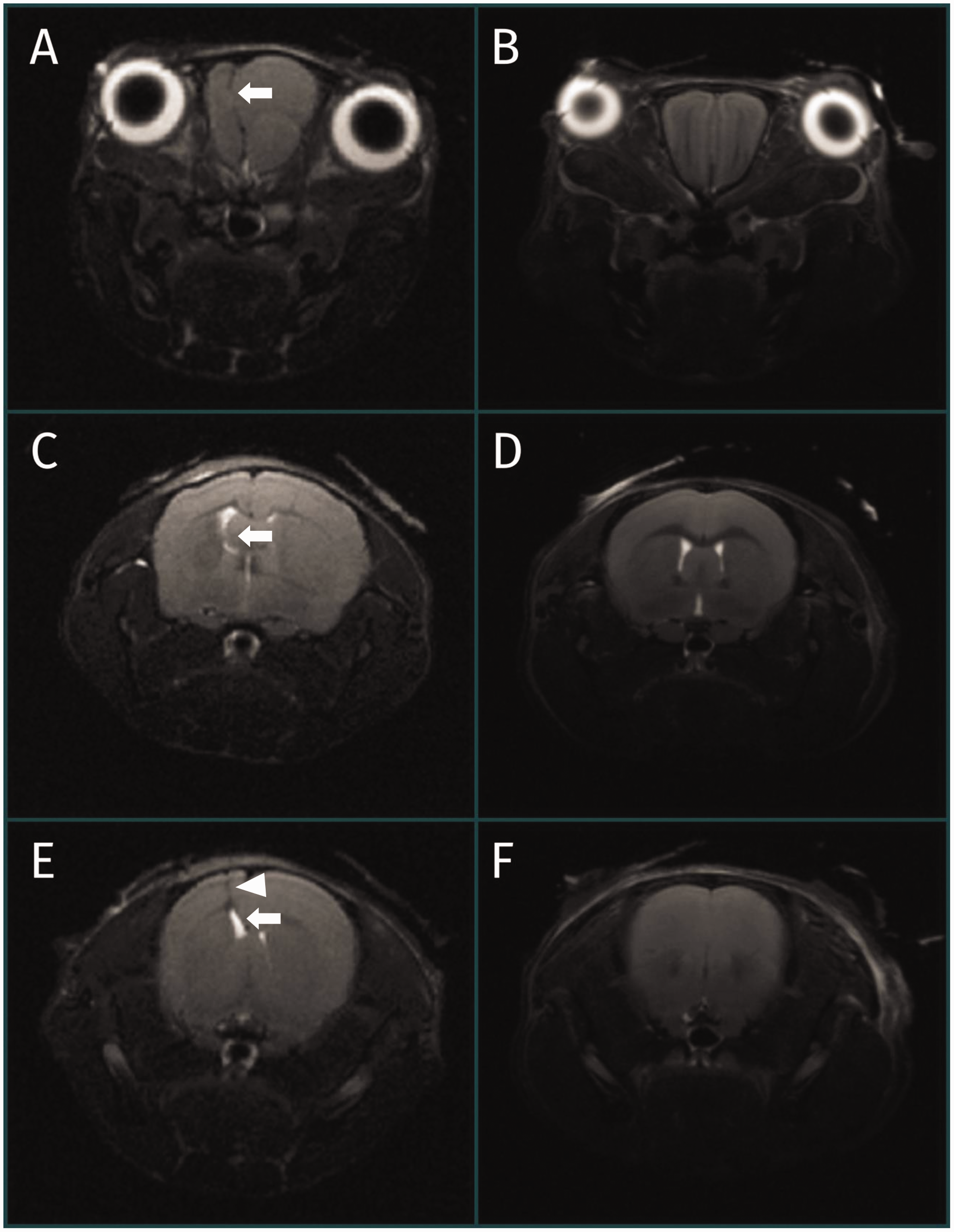
Sequential, T2-weighted (T2W), transverse magnetic resonance images through the brain of a C57BL/6J mouse diagnosed with a fronto-ethmoidal meningoencephalocele ((a), (c) and (e)) and a normal mouse ((b) (d) and (f)). (a) and (b) are at the level of the olfactory bulbs, with (c)–(f) progressing caudally to the level of the lateral ventricles and parietal lobes. A space-occupying intra-axial lesion is noted in the right olfactory lobe on (a), isointense compared to the grey matter, with severe left-sided displacement of the longitudinal cerebral fissure and secondary compression of the left olfactory lobe ((a), arrow). The caudal extent of this intra-axial right forebrain lesion is poorly defined, with a mass effect in the right aspect of the thalamus/ventral aspect of the right lateral ventricle, faintly T2W iso-tohyperintense, with secondary compression and effacement of the right lateral ventricle ((c) and (e), arrows). Left subfalcine herniation of the falx cerebri can also be noted on (e) (arrowhead).
Following MRI, the mouse was euthanized via carbon dioxide asphyxiation in accordance with AVMA Guidelines for euthanasia and the head was submitted for complete necropsy. 2 No abnormalities were noted on gross necropsy and externally, the calvarium was within normal limits. The entire head was left intact, fixed in 10% buffered formalin, decalcified, trimmed into coronal sections at four levels (olfactory bulbs, thalamus and hippocampus, mid-cerebellum) and paraffin embedded. Five-micron thick sections at multiple levels were cut and slides stained with hematoxylin and eosin. Histopathological examination revealed asymmetry of the olfactory bulb with a left midline shift. The right olfactory bulb appeared more rounded and larger than normal, and impinged into the left side occupying approximately two-thirds of the cranial vault. The contralateral olfactory bulb and meninges were herniated through a focal defect (aplasia) of the cribriform lamina, resulting in encephalomalacia (Figure 2(a) and (c)). There was loss of symmetry and bilaterally, the nasal conchae were abnormally shaped (dysplasia) or absent, the left side being more severe, with asymmetric thickness of the diploe and cortices of the dorsal portion of the nasal bone as well as osteoclastic resorption. There was partial loss of olfactory epithelium and nerves lining the left conchae and mild submucosal edema. The nasal septum was shifted to the right of midline. The caudal half of the left ethmoid and maxillary sinuses were partially filled with nuclear and cellular debris, admixed with proteinaceous material and small numbers of neutrophils, the left nasolacrimal duct was obstructed with proteinaceous fluid and rare macrophages (Figure 2(a), (c), (e) and (f)). The MRI and histopathologic findings were consistent with a fronto-ethmoidal meningoencephalocele (naso-ethmoidal subtype).
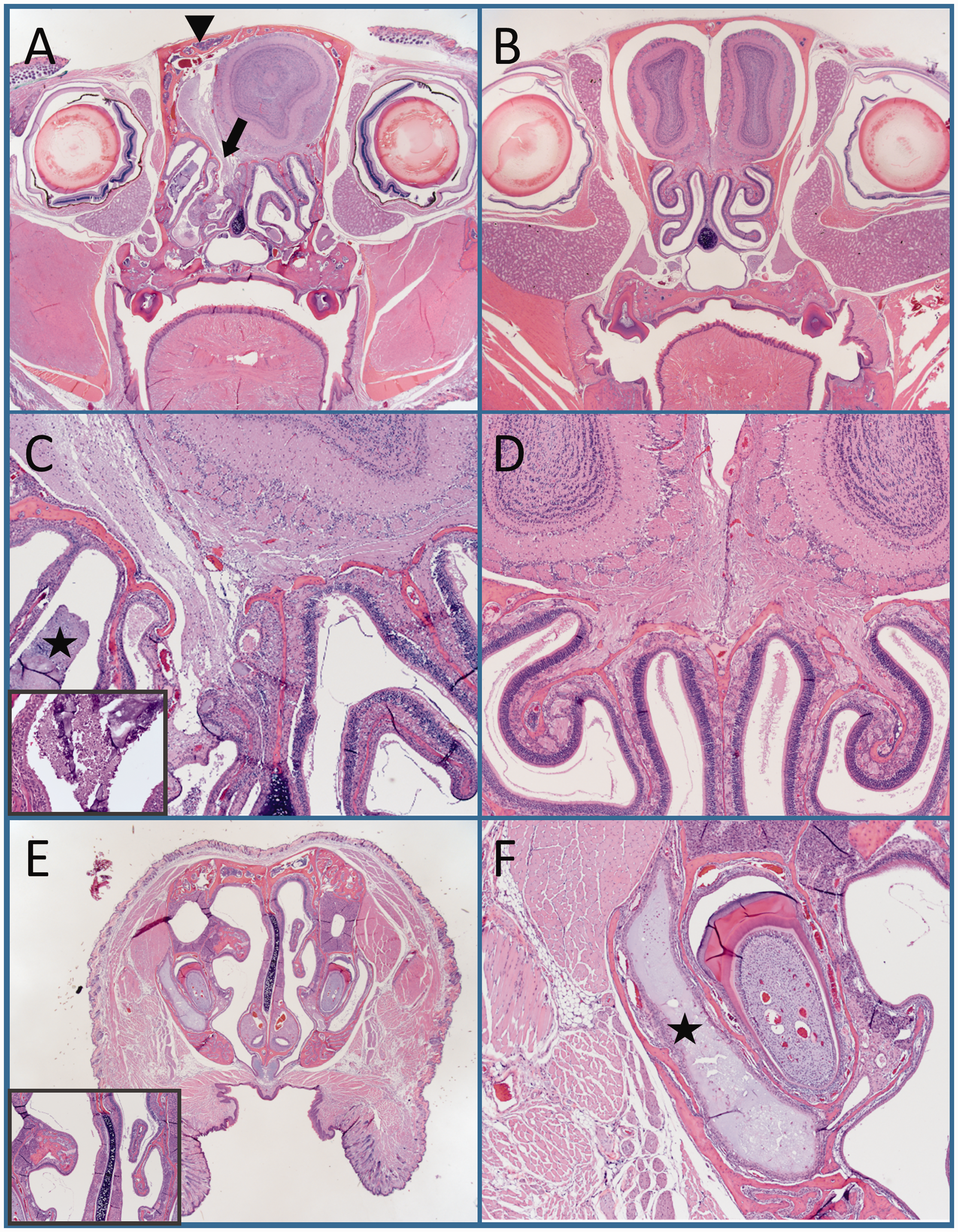
Fronto-ethmoidal meningoencephalocele, C57BL/6J mouse, histopathological photomicrographs. (a) Asymmetry of the olfactory bulb with left midline shift. The right olfactory bulb appears larger and the contralateral olfactory bulb is smaller and has evidence of rare faction. The cribriform plate has a full-thickness bone defect (aplasia) and the ipsilateral bulb is pictured herniating through it (black arrow). The nasal conchae are asymmetric, and the left nasal conchae are abnormally shaped (dysplasia). The nasal bone has asymmetric thickness of the diploe and cortices of the dorsal portion of the nasal bone (black arrowhead). Hematoxylin and Eosin stain (H&E); magnification ×12.5. (b) Coronal section of a mouse with normal symmetric olfactory bulbs and nasal conchae formation. H&E; ×12.5. (c) Herniation of the left olfactory bulb and detachment of ethmoidal lamina from the nasal septum with nasal conchae dysplasia and maxillary sinusitis (black star) with inset of higher magnification showing cellular debris and proteinaceous material with small numbers of neutrophils. H&E; ×200 and ×400 (inset). (d) Coronal sections of a mouse with an intact cribriform plate. H&E; ×200. (E) Right midline shift of the nasal septum and inset with higher magnification of the deviation. H&E; ×12.5 and ×200 (inset). (f) Obstruction of the nasolacrimal duct with fluid build-up and mild dacryocystitis (black star). H&E; ×200.
Discussion
This report describes the clinical presentation as well as MRI and histopathologic findings of a mouse with a fronto-ethmoidal meningoencephalocele. Given the young age of this mouse, the histopathologic findings, and absence of other lesions that may explain herniation (iatrogenic causes, trauma, tumor, etc.) a congenital defect is the most probable cause.
The cribriform plate or lamina cribrosa, a small perforated cup of ethmoid bone that separates the nasal cavity from the olfactory bulb, is unique to mammals. 3 Cribriform plate foramina serve as passageways for olfactory sensory neuron axons traveling from the nasal mucosa to the brain. 3 During embryologic development, neural crest cells migrate to the forebrain following closure of the neural tube forming the bone and connective tissue associated with the ethmoid, cribriform, vomeronasal and maxillary regions.4–6 This event is followed by the invagination of the paired nasal placodes, which are surrounded by neural crest cells, forming the nasal cavities, conchae, and olfactory mucosa of the ethmoidal labyrinth.4,5
Cephaloceles are rare congenital abnormalities characterized by herniation of cranial contents through a defect in the skull. 7 Classification is made based on their contents with meningoencephaloceles consisting of brain parenchyma and meninges and meningoceles containing only meninges. 7 They can also be classified as primary or secondary. 7 Primary cephaloceles occur congenitally, as a result of disruption of the neural tube closure at the level of the anterior neuropore, while secondary causes happen as a sequela to trauma, surgical complications, cleft palate, or increased intracranial pressure.7,8 In humans, meningoencephaloceles are divided into groups based on their anatomic location as occipital, parietal, basal and sincipital.7,9–11 Swanwela and Swanwela further classified sincipital meningoencephaloceles into fronto-ethmoidal (subdivided into naso-frontal, naso-ethmoidal and naso-orbital), inter-frontal and cranio-facial clefts.7,9,11 The location and the contents of the encephalocele predicts prognosis with occipital defects and those containing large amounts of cerebral cortex being associated with worse neurologic signs and outcomes compared to more rostrally located herniation containing small amounts of tissue.10,12 Herniation typically occurs cranially through a defect in the skull at the foramen cecum where the frontal and ethmoid bones meet and posteriorly by the crista galli. 7 The naso-ethmoidal defect occurs between the nasal bone and nasal cartilages, as was the case in this mouse. 7
In Western countries, occipital herniation is the most common site whereas fronto-ethmoidal sites are more common in Southeast Asia and some parts of India. 13 The reported incidence of ethmoidal meningoencephaloceles in Western countries is between 1:35,000 to 1:40,000 live births, whereas in Thailand the incidence is as high as 1:5000 live births. 13 Clinical features of fronto-ethmoidal meningoencephaloceles reported in humans depend on the amount of herniated tissue and the age of the person. 14 Clinical manifestations vary from none to epiphora, dacrocystitis, cerebrospinal fluid (CSF) rhinoliquorrhoea, facial and ocular deformities, meningitis, neurofibromatosis, seizures and mental changes.7,10,13–15 The exact etiology is not completely understood. Genetics, vitamin deficiency, teratogens, infectious agents, and environmental factors have been suggested as potential causes.7,9,10,13,16 Due to the lack of familial cases, genetics is less likely to play a primary role, although inheritance of a dominant gene with low penetrance has been suggested. 16 David et al. studied 25 cases of fronto-ethmoidal meningoencephaloceles and found an increase in parental age suggesting the role of an autosomal dominant gene mutation. 9 Thu and Kyu observed an increased interval between the birth of the affected child and the previous birth which they speculated may be due to an unfavorable uterine environment and found an increased prevalence in rural areas of Southeast Asia where rice is grown, as well a high prevalence in children conceived in this region during the warm rainy season.7,9,16 Their results suggested a fungal agent with teratogenic affects present in contaminated rice or cereal may be responsible.7,16,17 This theory is supported by researchers demonstrating specific mutagenic agents can incite developmental defects at the anterior neural tube in laboratory animals. 16 One particular study demonstrated that intraperitoneal injection of the aflatoxin, ochratoxin A, into pregnant mice on day 7–12 of gestation resulted in multiple congenital malformations including exencephaly, cleft palates and anophthalmia.17,18
Encephaloceles have also been reported in various other species including pigs, dogs, horses, cats, calves, rats, rabbits and goats.4,12,19–24 Heredity was attributed in a litter of Burmese cats. 22 In rats, encephaloceles as well as microphthalmia and maxillary hypoplasia were induced by hyperthermia during gastrulation. 23 Griseofulvin administration has been shown to cause multiple congenital malformations including encephaloceles in cats. 21 Clinical signs in domestic species include generalized or focal seizures, abnormal behavior, mentation changes, circling, vestibular ataxia, cerebellar ataxia, proprioceptive deficits, absent to decreased menace response, unilateral decreased nasal sensation, strabismus, intermittent positional nystagmus and hyperesthesia of the head and cervical spine.4,12,20,24 The only presenting clinical sign in this mouse was a head tilt, but the animal was otherwise unremarkable. The sinusitis and nasolacrimal duct obstruction was likely secondary to outflow obstruction of both structures resulting from either herniation of the olfactory bulb into the nasal cavity, nasal conchae dysplasia, or both. Histologic and MRI findings show a larger and rounder right olfactory bulb occupying most of the rostral cranial vault in addition to compression of the left hemisphere, with subfalcine herniation. The underlying cause of these morphological changes are not determined, but they suggest either a congenital or acquired anomaly intricately linked to the ethmoidal meningoencephalocele. Secondary infections have been reported as a complication in many different species with encephaloceles, including rhinitis and meningoencephalitis as a sequela due to direct communication of the nasal cavity with the central nervous system 4 ,13,15,25 No secondary meningitis or encephalitis was appreciated in this case. The cause of the mouse’s head tilt is not readily apparent. In a dog, vestibular ataxia and intermittent nystagmus was associated with a parietal meningoencephalocele which localized to the cerebellum. 12 Compulsive circling to the right following a series of seizures has also been reported in a puppy with an ethmoidal meningoencephalocele involving the olfactory bulb and prefrontal cortex. On gross and histopathologic examination absence of a cribriform plate and protrusion of the olfactory bulbs into the nasal cavity were the most significant findings. 20 In this case, there was no histologic evidence of morphologic changes to the peripheral vestibulocochlear nerve or the cerebellum. Based on evidence of compression seen on MRI and histopathology, traction or damage to the vestibulocochlear nerve or vestibular nuclei (not evident on histopathology) is the most likely explanation.
Many spontaneous craniofacial malformations have been described in mice. Indeed, Palmer et al. identified and characterized 43 mouse models of spontaneous craniofacial deformities and discovered a causative gene in 33 of these. They proposed using spontaneous mouse models instead of genetically manipulated mice because craniofacial disorders typically occur as part of a syndrome involving multiple genes and not from the disruption of a specific gene or pathway. In addition, facial bones in mice are comparable to those in humans. 26 Thus, mice play an important role in understanding the mechanisms behind development of the cribriform plate and associated structures, as well as serve as animal models for exploring the pathophysiology behind congenital craniofacial defects. To the authors’ knowledge, this is the first report of a spontaneous and congenital fronto-ethmoidal meningoencephalocele in a mouse. Spontaneous defects, such as this, could be a valuable tool for studying the pathophysiology behind congenital craniofacial malformations.
Footnotes
Acknowledgements
The authors would like to thank Dr Neil S. Lipman and Dr Alexandre Le Roux for their assistance with manuscript preparation and the staff of the Laboratory of Comparative Pathology for their technical assistance with tissue harvest and preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was partially funded by MSKCC NCI Cancer Center Support Grant P30 CA008748.
