Abstract
In recent decades, imaging devices have become indispensable tools in the basic sciences, in preclinical research and in modern drug development. The rapidly evolving high-resolution in vivo imaging technologies provide a unique opportunity for studying biological processes of living organisms in real time on a molecular level. State of the art small-animal imaging modalities provide non-invasive images rich in quantitative anatomical and functional information, which renders longitudinal studies possible allowing precise monitoring of disease progression and response to therapy in models of different diseases. The number of animals in a scientific investigation can be substantially reduced using imaging techniques, which is in full compliance with the ethical endeavours for the 3R (reduction, refinement, replacement) policies formulated by Russell and Burch; furthermore, biological variability can be alleviated, as each animal serves as its own control. The most suitable and commonly used imaging modalities for in vivo small-animal imaging are optical imaging (OI), ultrasonography (US), computed tomography (CT), magnetic resonance imaging (MRI), and finally the methods of nuclear medicine: positron emission tomography (PET) and single photon emission computed tomography (SPECT).
Preclinical imaging
Prior to the concept of ‘small-animal imaging’, experimental animals could only be imaged using clinical instrumentation. 1 However, preclinical research mostly utilizes rodent models (mice or rats), which in many instances are too small to be imaged accurately by clinical imaging devices. Low spatial resolution, low sensitivity and poor tissue contrast proved to be most limiting. Overcoming these limitations and improving overall imaging performance was achieved by introducing specific refinements and alterations to instrumentation. 1 Despite these adjustments, imaging small laboratory animals remains a major challenge. The size of the imaged subjects, the total volume to be evaluated, the spatial resolution necessary to detect anatomical or functional changes and the total time that may be dedicated to acquiring an image are fundamentally different for a 20–30 g mouse than for a 70 kg human. 2 The primary challenge is to obtain the highest possible signal-to-noise ratio (SNR), and to localize the signal as accurately as possible with good temporal resolution with a minimal amount of molecular probe. 3
Main characteristics of in vivo imaging techniques for small animals.
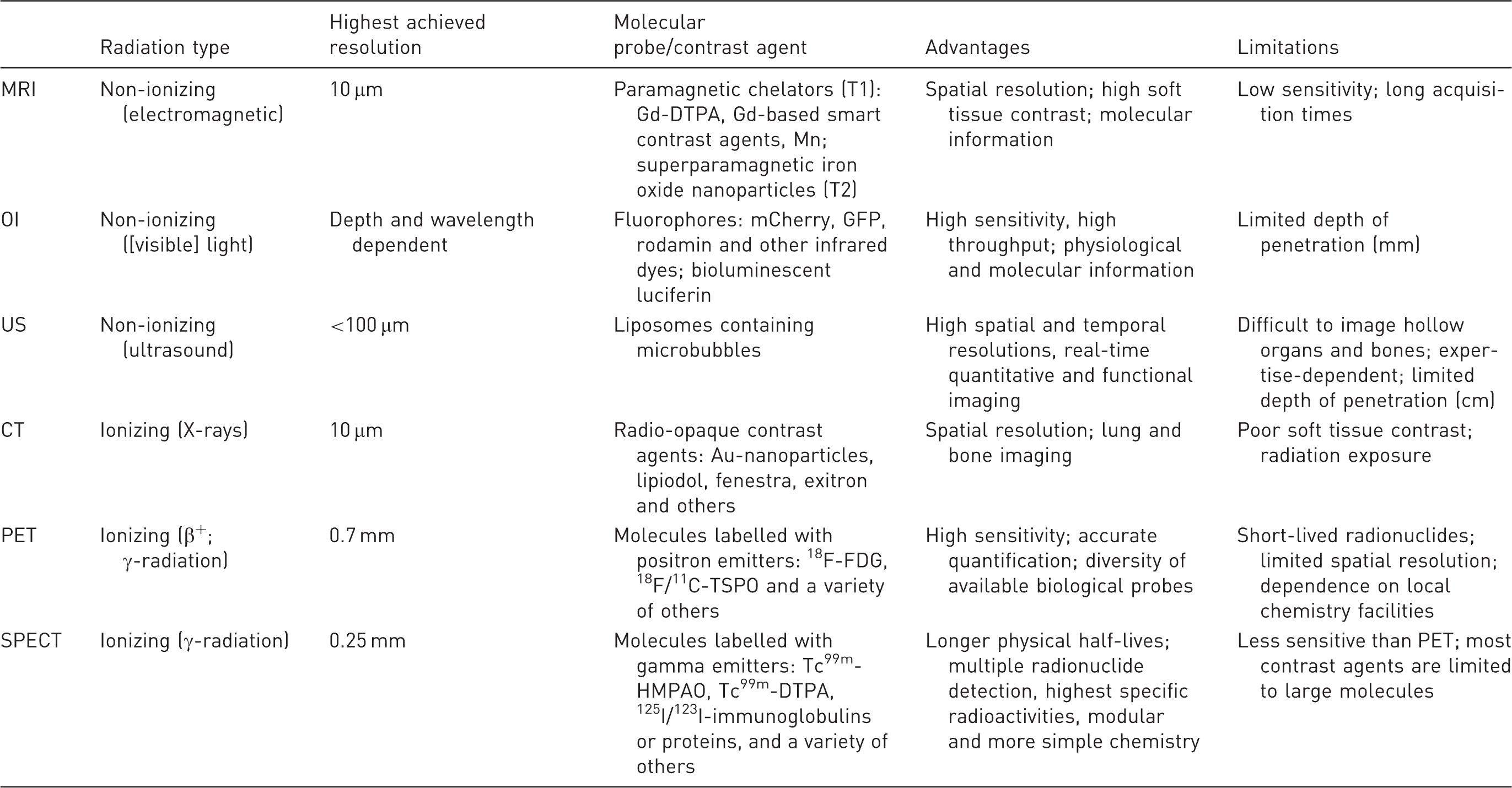
MRI: magnetic resonance imaging, OI: optical imaging, US: ultrasonography, CT: computed tomography, PET: positron emission tomography, SPECT: single photon emission computed tomography, Gd-DTPA: gadolinium-diethylenetriamine pentaacetic acid, Mn: manganese, GFP: green fluorescent protein, Au: gold, 18F-FDG: fluorodeoxyglucose, TSPO: translocator protein, HMPAO: hexamethylpropyleneamine oxime.
Micro-CT
Computed tomography and MRI provide detailed anatomical cross-sectional images. These two groundbreaking technologies have virtually transformed modern healthcare services. In recognition of their importance the Nobel Prize was jointly awarded to Allan M Cormack and Godfrey N Hounsfield for the invention of CT in 1979 and to Paul C Lauterbur and Sir Peter Mansfield for the invention of MRI in 2003. The evolution of electronics and engineering led to the development of miniaturized versions of CT (micro-CT) and MRI (micro-MRI), which are now commercially available and are optimized for small-animal imaging in preclinical research. 6
Computed tomography calculates the attenuation of X-ray for single tissue volume elements (voxels) and creates three dimensional (3D) matrix images, from which sectional or spatial images can be reconstructed. A typical CT device consists of an X-ray tube and a 2D X-ray detector assembled in opposing positions in a gantry, which rotates around the examined object. Some preclinical systems, mainly used for imaging ex vivo specimens, have a static radiation source and an X-ray detector, while the examined object rotates within the circular apparatus.7,8 X-ray attenuation mainly depends on tissue density and the atomic number of the elements contained in the voxels. Therefore, tissues with higher atomic numbers and higher densities, such as bone (Z(Ca) = 20), yield excellent images with sharp contrast. On the other hand, soft tissues associated with lower atomic numbers and lower densities have very poor contrast, and are therefore often required to be accentuated by using certain contrast agents (Z(I) = 53, Z(Br) = 35).
The components of micro-CT devices have been fine-tuned to achieve maximal spatial resolution and to optimize SNR: the properties of the X-ray tubes were modified, as were pixel pitch and matrix size of the detectors, the source detector distance and size of the focal spot. 9 In contrast to a minimum focal spot size of 300 µm in conventional clinical CT systems, focal spot size could be downscaled to 1–10 µm in micro-focus CT scanners which significantly diminished penumbra blurring and led to images with much sharper contours.9,10 Due to these technical innovations micro-CT provides tomographic data at microscopic spatial resolution (10–200 µm) and a temporal resolution in a minute timescale. 11
Micro-CT is ideal for imaging osseous structures or the lung, due to distinct differences in density, and it is also well suited for imaging cardiac function and tumour growth. 12 Despite the sharp air-to-tissue contrast, preclinical imaging of the lung is cumbersome because of the minuscule size and rapid physiological movement of the structures in question (the mean respiratory rate of a laboratory rat is 85/min). 11 The introduction of cardiorespiratory gating systems in recent years has led to substantial improvements in image quality and contrast.13,14
However, similar densities make soft tissue differentiation with CT difficult. The use of contrast agents increases sensitivity by highlighting the vascular system, specific organs or tissues. For a long time, conventional water soluble intravenous iodide-based contrast agents were not applicable in rodents, due to the much smaller voxel size and much faster circulation, 15 which diminish the contrast medium in seconds. 16 Since then, faster X-ray detectors have been developed with readouts of between 30 and 60 frames per second (fps), which allows for intravenous bolus imaging. 17 Blood-pool contrast agents have a longer half-life, either because they are macromolecules with lower elimination rate or they are bound to plasma proteins. These contrast agents allow experimental setups with longer scan times; however, the level of contrast is generally lower than with conventional contrast agents.17,18 Newer liposome-based blood-pool contrast agents containing high concentrations of iodine or metal nanoparticles (e.g. gold [Au] or barium [Ba]) have a longer half-life, while offering improved detail perceptibility particularly in small anatomical structures.19–22
The main advantages of micro-CT are high spatial resolution, detailed morphological information and relatively short scanning times (10–15 min). Nevertheless, the volume of contrast agents used to enhance the contrast resolution of soft tissue becomes a potential limitation, particularly when imaging a series. Furthermore, a longitudinal experimental setup requires balancing image contrast and resolution with the radiation burden associated with CT imaging. 23 Although radiation levels used in micro-CT are not lethal to animals, the levels may still surpass the threshold associated with changes in immune response and other biological pathways that may alter experimental outcomes. 24
Micro-MRI
Three-dimensional imaging based on MR might be recognized as the most innovative, versatile and useful application of quantum physics in medicine. MRI is capable of whole-body in vivo imaging, producing high-contrast images of soft tissue without the need for ionizing radiation. Clinical 1.5–3.0 T MRI scanners could also be used for targeted imaging in medium-sized animals such as rabbits, albeit with limited spatial resolution. 25 However, modern micro-MRI devices are suited for high-resolution (below 100 μm) imaging of small-bodied rodents with excellent contrast for soft tissue differentiation. 26
Magnetic resonance imaging is based on the interaction of nuclear spin (a quantum-physical property) and an external magnetic field. The dominant nuclear particle in MRI is the proton in hydrogen (1H), the spin of which is in precession about the magnetic field direction. Atomic spin can be manipulated with a combination of magnetic fields resulting from radiofrequency (RF) impulses. Imaging rests on the ability to detect the bulk precession of the hydrogen spins in water, fat and other organic molecules, and to determine the time for spin magnetization to rebuild along the direction of the static magnetic field (T1 relaxation) or the time until dephasing of clusters of spins (T2 relaxation). 27 Signal strength correlates with proton density, and signal pattern is influenced by the microenvironment of the protons in individual tissues.28,29 To produce images with much finer resolution, micro-MRI uses stronger magnets, specific receiver coils and RF receiver chains as well as stronger gradient sets compared with clinical instrumentation. Visualizing fine tissue anatomy details or blood flow changes with good MR resolution requires high SNR. For these applications MR systems operating at or over 7 T field strengths are optimal. For many molecular imaging applications such as chemical exchange saturation transfer (CEST), even with 1 T or 3 T translational field strength, small-animal systems offer the appropriate performance. 23 Typical MRI contrast agents are paramagnetic chelators (i.e. gadolinium – T1, manganese – T2 and superparamagnetic iron oxide [SPIO] – T2), which lead to higher target tissue contrast by altering the magnetic micro-environment. 26
Furthermore, minor modification of the measuring technique and analysis of data by the introduction of special algorithms enables MRI to provide valuable and unique information on biological processes via, for example:
The differentiation between oxyhaemoglobin and deoxyhaemoglobin based on their different magnetic properties (the former being diamagnetic, the latter paramagnetic). Functional MRI (fMRI) assesses blood oxygenation by determining the haemoglobin concentration of blood, fMRI is thus capable of indirectly revealing and highlighting activated brain structures via their higher oxygen transport by blood.
30
Imaging based on intravenous bolus injection of exogenous contrast agents is called dynamic contrast-enhanced imaging (DCE–MRI), which can also be used to determine vascular permeability with contrast molecules of varying sizes. Recently, non-invasive perfusion measurements have also become entirely possible: arterial spin labelling (ASL) for instance utilizes arterial blood water magnetization itself as an endogenous contrast agent.
31
Diffusion weighted imaging (DWI) shows the different properties of diffusion in tissues by detecting the motion of protons (mainly water), which is restricted by various intercellular structures resulting in certain diffusion patterns which reflect the conditions of tissue structure or cell density. This method is applied to detect neuronal pathways (tractography), for tumour differentiation or early recognition of cerebral ischaemia.23,32 Magnetic resonance spectroscopy (MRS) provides information about tissue biochemistry by determining the concentration of different chemicals or metabolites. This technique mostly assesses molecules rich in hydrogen (choline, creatinine, lipids and amino acids
33
); nevertheless, chemicals composed of atoms (such as phosphorus [31P], carbon [13C], lithium [7Li], fluorine [19F] or sodium [23Na]34) can also be detected by modifying the coils, generators and assessment sequences. MRS is mainly applied to examine brain metabolism, to differentiate tumorous tissues or to assess drug pharmacodynamics.33,35,36 Hyperpolarized nuclear MRI relies on MR spectroscopic maps obtained from acquisitions yielding time-domain information. In particular, it is possible to monitor the biochemical transformations of specific substrates that are delivered to subjects. As this method provides data on the kinetics of the conversion of substrates into metabolites, MRS of the carbon nuclei (13C) is one of the most powerful techniques of investigating intermediary metabolism. Specially prepared solutions of hyperpolarized 13C-containing substrates such as hyperpolarized 13C-pyruvate are usually applied both in animal models of cancer and clinical cancer patients for the in situ, in vivo detection of the Warburg effect by imaging the increase in endogenous lactate levels in tissues. Other hyperpolarized nuclear MRS methods using 129Xe or 3He noble gases have also found translational applications recently.37,38
Micro-MRI produces morphological images with high resolution and excellent tissue contrast. Furthermore, it provides information regarding tissue composition, perfusion, oxygenation, tissue elasticity, metabolism, and also enables detection of molecular probes; all within a single acquisition session without any radiation exposure. Thus, MRI is arguably the most versatile modality. 39 Micro-MRI is an essential part of preclinical cardiovascular research, providing valuable information regarding tissue structure, function and perfusion, both on cellular and at molecular levels.13,40 In oncological research, micro-MRI is an excellent modality for monitoring tumour growth and development, tumour differentiation, perfusion imaging, oxygenation and angiogenesis, and particularly for monitoring tumour growth and degradation in response to therapy.25,35,41 Assessment of osseous structures remains a challenge because of the low water content (15%) of bone volume, nevertheless, micro-MRI has also been successfully applied in the assessment of musculoskeletal tissue structures such as tendons, cartilage, menisci, and ligaments. 28 The lower sensitivity of MRI molecular methods can be compensated for by the application of complementary or hybrid methods (e.g. SPECT or PET, see below).39,42
Nuclear medicine or molecular imaging
Imaging in nuclear medicine rests on two core principles: (1) A variety of biomolecules can be labelled with radioactive isotopes which are referred to as radiopharmaceuticals or radioactive tracer molecules. (2) Radiotracers tend to accumulate in certain localizations due to physiological processes (such as active or passive transport mechanisms, metabolism, phagocytosis and microembolization) according to the principle of the dynamic state of body constituents. Therefore, metabolic processes can be quantified topographically measuring tracer concentrations within anatomical structures (standardized uptake value [SUV]) as well as temporally assessing the time course of tracer concentrations (Logan et al. 43 or Gjedde 44 and Patlak 45 plots). Thus, biochemical and physiological processes become detectable and traceable in real time from cellular and molecular levels up to organ or organism levels without interfering with normal physiology. 46 The modalities of nuclear imaging are ‘planar scintigraphy’ yielding a 2D image, SPECT, which is the 3D version of the former, and PET. The downscaled models of each (micro-SPECT, micro-PET) are well established modalities of preclinical research.
Micro-SPECT and planar scintigraphy
Micro-SPECT and planar scintigraphy are based on the detection of γ-photons originating from the decay of atomic nuclei (radionuclides, mainly: 99mTc, 67Ga, 201Tl, 111In or 123,125,131I).47,48 The gamma-camera consists of a detector crystal and a collimator, which is made of lead or tungsten and manufactured with multiple elongated holes, which are positioned in such a way that only photons which are travelling in a parallel orientation to the camera face can interact with the crystal. Conventional planar scintigraphy allows for the acquisition of the 2D projection of tracer distribution. 49 In SPECT imaging, the gamma-camera rotates around (or completely surrounds) the object and a number of projections at different angles are merged to produce 3D tomographic images.50,51
Preclinical SPECT systems apply pinhole collimation to obtain better spatial resolution by pinhole magnification. The use of multiple pinholes or other multiple collimation aperture formats also enhances imaging sensitivity.
52
To further increase resolution and sensitivity, different crystal materials to the most widespread scintillation detector crystal NaI[Tl] (e.g. [CsI(Tl)], [CsI(Na)] or [LaBr3(Ce)]) have been introduced.
53
Modern systems are able to detect minute (<10−10 molar) tracer concentrations (0.1 nanomol) with submillimetre resolution (0.5–0.7 mm) in vivo.54,55 Some preclinical SPECT systems incorporate semiconductor detector materials (CdTe or CdZnTe [CZT]) offering the possibility of higher energy discrimination efficiency, which are important for low-energy radionuclides (125I) and dual isotope applications.53,56 Gamma-photon-emitting isotopes eligible for SPECT imaging are usually elements with higher atomic numbers, which are suitable for labelling several molecules: from simple organic chelates to proteins, antibodies, hormones or selectins. However, diffusion and clearance of the resulting larger-molecule radiopharmaceuticals require a relatively long time due to the larger size of these molecules; thus radionuclides with a relatively longer half-life (hours) are to be used.
54
Micro-SPECT is widely used in key areas of preclinical research: it is a well-established modality for cardiovascular research (e.g. to monitor myocardial function, perfusion, necrosis or angiogenesis40,57,58), stem-cell research,59,60 oncology (e.g. detection of in vivo gene expression and metastases, or molecular pathways of tumour formation61,62), neurological research (e.g. research in Parkinson's and Alzheimer's diseases63,64) as well as for the development of new drugs26,65 (Figure 1).
In vivo single photon emission computed tomography/computed tomography (SPECT/CT) imaging with radiolabelled human serum albumin. Radiolabelled human serum albumin (125I-HSA) was administered directly before SPECT/CT imaging. Osseous structures are clearly visible in the single modality CT images (a). SPECT images show distribution of 125I-HSA (b). The SPECT/CT fusion image allows exact anatomical localization of higher HSA concentration and identification of structures, e.g. carotid arteries, heart, aorta, liver, kidneys and urinary bladder (c). NanoSPECT/CT PLUS (Mediso, Budapest, Hungary). See Szigeti K, Horváth I, Veres DS, et al. A novel SPECT-based approach reveals early mechanisms of central and peripheral inflammation after cerebral ischemia. J Cereb Blood Flow Metab 2015; 35: 1921–1929.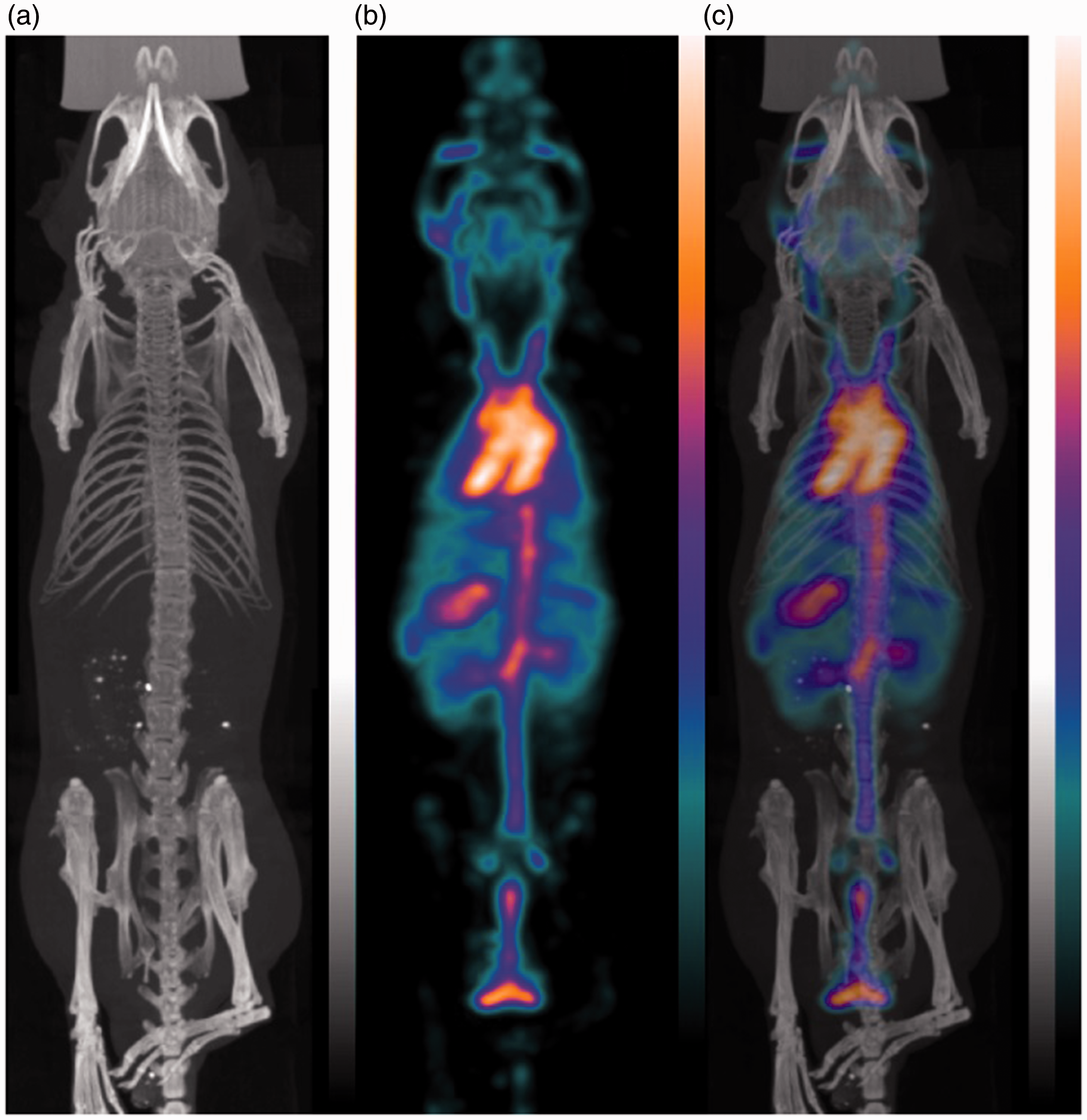
Micro-PET
Positron emission tomography is also based on the detection of γ-photons. However, the tracers emit positrons (unstable subatomic e+-particles), which travel until they collide with an electron, their respective antiparticle. The distance travelled (‘positron range’) depends on the kinetic energy of the emitted positron (usually a few millimetres). The collision of a positron and an electron results in a process called annihilation: the annihilated particles are replaced by two γ-photons of fixed energy ( = 511 keV), which travel along the same line but in opposite directions (at a 180 ° angle). 66 Therefore, a PET system must contain a pair of opposing detectors at a minimum, but the best overall sensitivity can be achieved by a full ring of detectors. Since photons travel at the speed of light, the opposing detectors will receive a signal almost simultaneously. 67 Thus, the line determined by two photons which are detected within a short time window (usually 4–20 ns) determines the trajectory of the annihilation point, which is referred to as an electronic collimation. The absence of a physical collimator significantly improves the sensitivity of PET, which can be up to 100 times greater than SPECT, especially when 3D data are obtained. 66 However, improving spatial resolution above a certain threshold is physically impossible (in contrast to SPECT) due to the methodical background of a positron range. 68 Modern preclinical micro-PET systems achieve spatial resolution values of around 1.0 mm under optimal conditions (resolution of clinical systems is 6–10 mm) 68 partially owing to multiple advancements such replacing old bismuth germanate (BGO) scintillators by improved detector blocks made of inorganic scintillators, for example lutetium oxyorthosilicate (LSO), lutetium–yttrium oxyorthosilicate (LYSO) or gadolinium oxyorthosilicate (GSO).69,70 New compact silicate detectors, based on semiconductor technology, facilitate synchronous PET and MRI signal acquisitions, thus significantly reducing SNR and examination time.71,72
The most commonly used positron emitters are 11C (carbon), 13N (nitrogen), 15O (oxygen) and 18F (fluorine). The biggest advantage of carbon, nitrogen or oxygen isotopes is the possibility of labelling nearly any organic molecule without changing its chemical properties. However, the very short half-life (minutes) of these radiopharmaceuticals necessitates in situ radioisotope production and compound labelling, which limits their availability to dedicated facilities equipped with cyclotrons. The somewhat longer half-life (110 minutes) of 18F already allows external production and transportation.
39
Recently, other isotopes with longer half-life (13h to a few days), such as 60,61,64Cu, 76Br, 94Tc, 89Zr, or 124I, are gaining increasing attention and are also applied to clinical diagnosis or therapy follow-up studies.
23
These radionuclides offer the advantages of relatively easy production and transportation, as well as the potential to transfer SPECT radiochemistry to PET, thus taking advantage of a number of already evaluated SPECT tracers.
66
Micro-PET is one of the most frequently used preclinical imaging modalities, and plays an important role in oncological research (by tracking metabolism, gene expression, cell proliferation, angiogenesis, hypoxia or apoptosis73–76), cardiology (heart function and localizing cardiac infarction
77
), neurology (measuring cerebral metabolism and perfusion78,79), as well as drug development29,65,80 (Figure 2).
In vivo investigation of metabolic activity by fusion positron emission tomography/magnetic resonance imaging (PET/MRI). The first two images show single modality (a) morphological MR and (b) highly sensitive PET images of the lower limb of a mouse in which arthritis was induced by administration of the K/BxN mouse serum.The glucose analogue, fluorodeoxyglucose (18F-FDG), used for PET imaging exhibits increased uptake kinetics in high-glucose-using cells of inflamed tissue. The PET/MR fusion image (c) shows the anatomic regions of higher glucose uptake, i.e. the joints due to inflammation and the liver due to physiologically high metabolism. NanoScan PET/MRI (Mediso, Budapest, Hungary). See Botz B, Bölcskei K, Kereskai L, et al. Differential regulatory role of pituitary adenylate cyclase-activating polypeptide in the serum-transfer arthritis model. Arthritis Rheumatol 2014; 66: 2739–2750.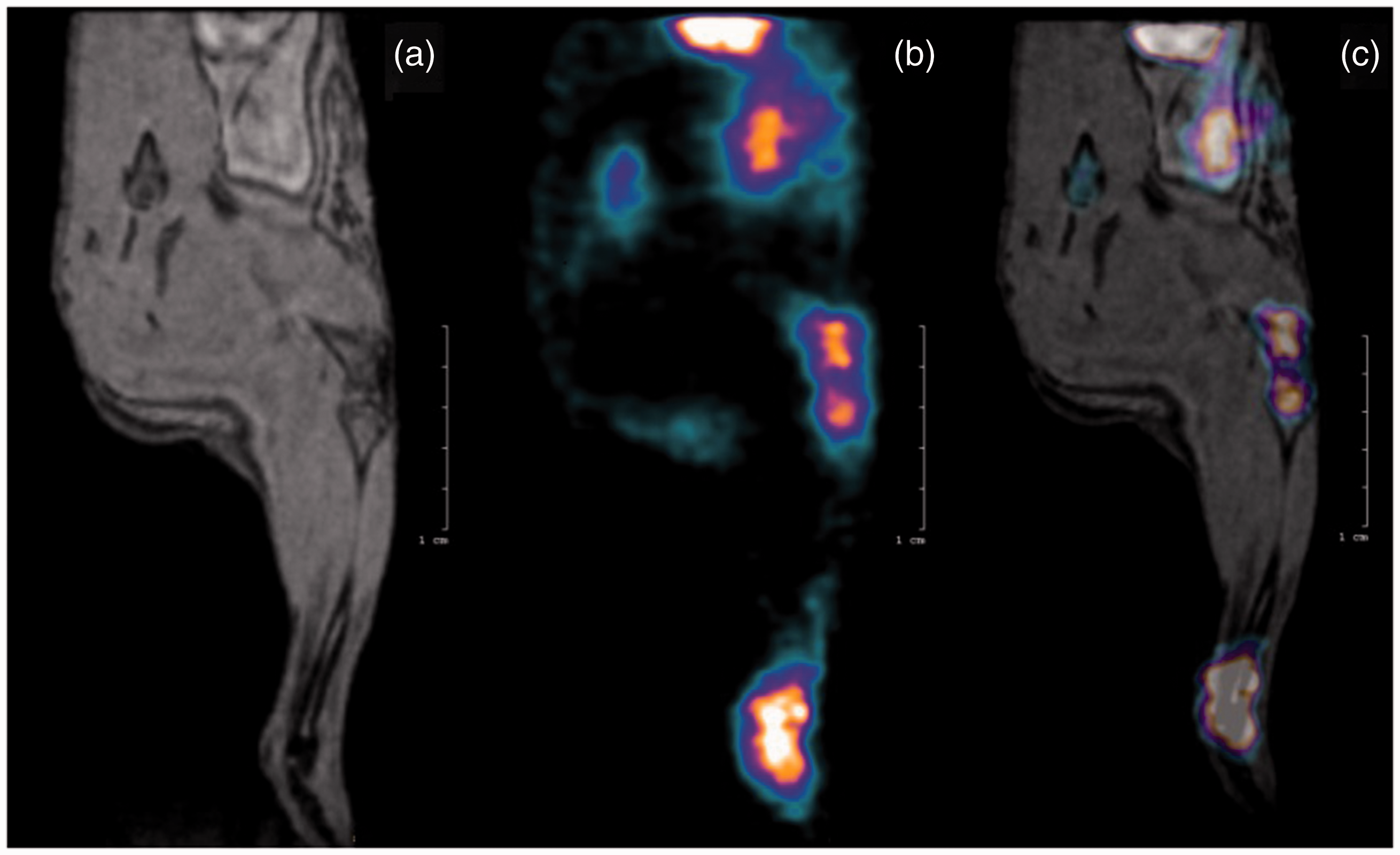
Micro-SPECT and micro-PET
The greatest advantage of PET, besides the somewhat higher sensitivity, is provided by the possibility of constructing physiological biomarkers labelled with positron emitting isotopes 11C, 13N, 15O, or 18F, with which physiological and pathological processes can be monitored virtually without interference.13,40 However, the physical properties of beta radiation (positron range, photon non-collinearity and random events) limit the achievable spatial resolution (1–2.2 mm).68,81 The spatial resolution of modern micro-SPECT devices, by contrast, ranges between 0.35 mm and 0.7 mm.54,55 The isotopes used for SPECT imaging have a longer half-life, are chemically stable and emit photons with specific energies determined by the isotope in use. Therefore, radiopharmaceuticals are easily transportable if they are not synthesized on site; also longer periods of data acquisition and the use of multiple tracers are possible. Meanwhile in the case of PET, tracer differentiation is not plausible due to the fixed photon energy of annihilation. 23
Due to the effects of partial volume and spill-over of signals, achieving the desired quantifiable image contrast with an appropriate resolution for small-animal imaging usually requires a 20-fold higher tracer concentration per gram of tissue than with human applications. 82 However, the amount of chemical probe compound applicable per body mass is limited; excessive amounts of probe molecules might interfere with the physiological dynamic state of body constituents (e.g. by occupying more than 5% of the total receptor sites) and threatening the validity of the tracer principle. 83 Furthermore, the maximal injected volume should not exceed 10% of the total blood volume in order to obviate adverse effects on circulation (in the case of laboratory mice, intravenous injections should not exceed 200 µL). 84 Both micro-PET and micro-SPECT provide unique functional information on a molecular level. However, this information is sometimes difficult to interpret because the images lack correlation with anatomical structures. The absence of detailed anatomical information can be readily eliminated by combining imaging modalities via a hybrid or fusion imaging (such as PET/MR, PET/CT, SPECT/CT and SPECT/MR). 66 An especially interesting hybrid recently being introduced into the market is the micro-SPECT/MRI system. With this combination, the versatility and high chemical sensitivity of SPECT meets the superb soft tissue contrast and high resolution of MRI. This system brings novel possibilities of therapeutic research in nephrology, immunology, transplantation and oncology where studies with longer monitoring intervals in high resolution have long been sought.85,86
Optical imaging
Optical imaging in medicine utilizes special molecules which either act as a light source per se (luminescence) or can be excited externally to emit photons (fluorescence) and so enhance image contrast and monitor molecular processes. 87 Bioluminescence occurs as luciferases (special enzymes synthesized by protists, fungi, insects or bacteria 88 ) which catalyze the oxidation of luciferins (substrate), creating non-reactive oxyluciferins while releasing photons of light. 89 In recent years, advances in genomics and genetic engineering have led to the isolation of different luciferase genes that can be transferred into single cells, cell cultures or entire organisms by way of DNA vectors. 88 Following intraperitoneal or intravenous injection, luciferin is oxidized and the affected cells are lit up for 10–12 min, which can then be detected by highly sensitive detectors. 90 This method is very sensitive, even single cells can be detected, and is mainly used in oncological research.
The phenomenon of fluorescence differs from luminescence. Certain substances or molecules are capable of absorbing energy from an external light at a certain wavelength, while emitting photons with a typical (increased) shifted wavelength, which may be captured by charged-coupled detector (CCD) cameras. 91 OI either utilizes fluorescent properties of endogenous substances (collagen or haemoglobin) or external fluorescent agents administered to the organism. These agents may be intravenous dyes (indocyanine green [ICG]), fluorescent-labelled antibodies, or artificially expressed molecules by genetically-modified cells (green fluorescent protein [GFP]).87,92
The main limitation of OI is the intraparenchymatous absorption and scattering of photons within a few millimetres, thereby strongly reducing penetration depth, and producing images which are not quantitative and have a suboptimal effective resolution. 93 Light in the near infrared range penetrates tissue slightly better; nevertheless, this method is used predominantly in preclinical research (with a few exceptions, such as breast cancer imaging). 94 By using multiple source-detector pairs reconstructed 3D images can be obtained (diffuse optical tomography [DOT]) with excellent sensitivity and good spatial resolution, which, in contrast to planar images, allows for accurate quantification and volumetric localization.95,96 The outstanding sensitivity renders OI a modality quite frequently utilized in small-animal studies, especially in oncological research, in vivo stem-cell studies, or more recently, even to detect vascular endothelial growth factor (VEGF), integrin or matrix metalloprotease activity.97–99
The term OI also includes other techniques which utilize optical signals (besides luminescence or fluorescence). Laser speckle imaging, for example, is based on the acquisition (by CCD cameras) and analysis of interference patterns (‘speckles’) which appear when coherent light is scattered from a random medium. The movement of scattering particles such as erythrocytes causes phase shifts in the scattered light, changing the random interference pattern, which makes this method a powerful tool for perfusion imaging applications in tissues, such as the retina, skin and brain. Laser speckle imaging has become a standard tool in a variety of vascular diseases including dermatological complications, neurotrauma clinical therapy and preclinical research. 100
A more recent technology combines the advantages of OI and US. Photoacoustic tomography (PAT) obtains images with high spatial resolution and improved contrast by detecting acoustic signals generated by pulsed laser light.
101
A fraction of incident light pulse energy is absorbed by the target tissue causing thermal expansion, which propagates as a sound wave and can thus be detected by an acoustic detector, providing both structural and compositional information with depth penetration from over several millimetres to centimetres.102,103 PAT can be used for imaging oxygenation and perfusion at the microscopic level,104–106 molecular tissue composition,107,108 and is capable of highly-specific molecular and functional imaging with extrinsically administered optical or optoacoustic agents.109,110 As emerging concepts are translated into robust tools, this modality will fill important gaps in basic and translational research111,112 (Figure 3).
Fluorescent imaging of mCherry protein expression in U251 human glioma cells orthotopically injected to a mouse brain (strain: Crl:NU(NCr)-Foxn1nu/nu). The newly formed tumour is well visualized as the image of fluorescence is superimposed on a grey-scale image of the mouse. FOBI fluorescent optical imaging system (NeoScience Co Ltd, Suwon, Korea). See Dico AL, Costa V, Martelli C, et al. MiR675-5p acts on HIF-1α to sustain hypoxic responses: a new therapeutic strategy for glioma. Theranostics 2016; 6: 1105–1118.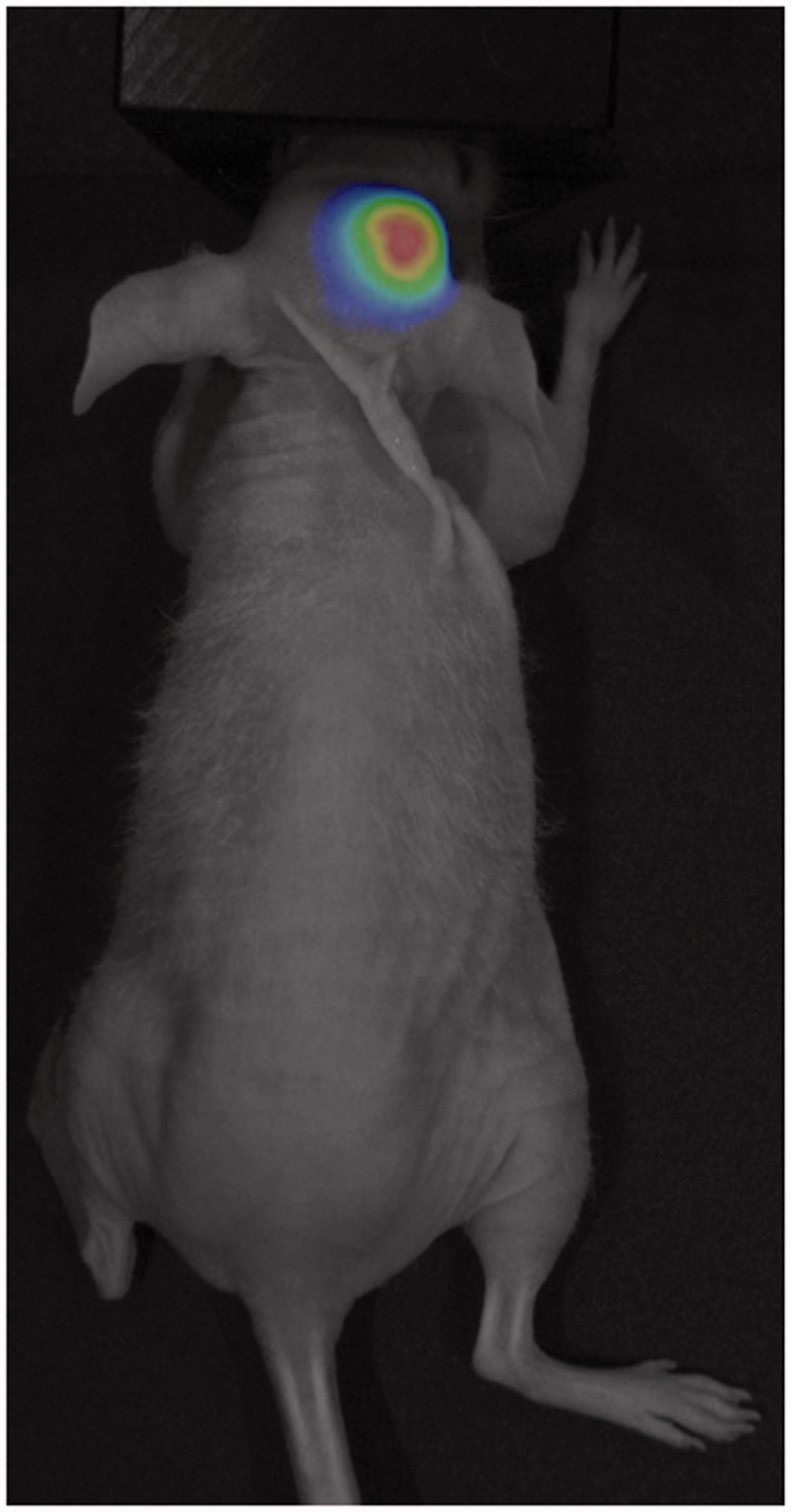
Ultrasonography
Ultrasonic imaging (echography, sonography, US) in small-animal studies is a fast and cost-efficient method, with high spatial and temporal resolutions, providing images with high sensitivity and contrast. 113 US is based on the differing acoustic properties of tissue. Ultrasound waves, usually generated by piezoelectric crystals, are reflected on the border of adjacent tissue layers (with different acoustic impedance) creating an echo of sound wave, which can be detected and converted into electric signals by piezoelectric crystals.114,115 Preclinical ultrasound devices use much higher frequencies than commonly used ultrasound diagnostic systems (20–50 MHz versus 2–15 MHz). The higher the frequency, the higher the spatial resolution achieved, but at the expense of higher tissue attenuation, which results in a more superficial penetration.116,117 The introduction of dedicated US contrast agents further enhances image contrast and enables the analysis of biological and functional processes on a molecular level. 118 Ultrasound biomicroscopy is able to produce real-time images with microscopic spatial resolutions (30–60 μm) at ultrahigh frequencies (60–100 MHz). High-resolution ultrasound biomicroscopy is able to portray the beating heart of in utero embryonic mice, and is frequently used in cancer research or during the detection of human conjunctival melanomas.119,120 Ultrasound imaging has several extremely advantageous features: high spatial and temporal resolutions, real-time and functional imaging without ionizing radiation, broad availability and cost efficiency. However, to achieve images of high quality and reproducibility requires the expertise of a well-trained sonographer. 115 However, US is incapable of imaging bone and gas-filled structures; and therefore it cannot be used routinely in brain, spinal cord or lung imaging. 121
Conclusions
Preclinical imaging offers prodigious advantages over conventional ex vivo techniques, such as real-time monitoring, due to its non-destructive and non-invasive nature, and the possibility of simultaneously acquiring molecular, functional and anatomical information. Each individual imaging modality has its intrinsic advantages and disadvantages, yielding different kinds of information, which are most often complementary. The combination of high-resolution modalities (micro-MRI, micro- or nano-CT) and techniques bearing highly sensitive functional information (micro-PET or micro-SPECT) has become a major cornerstone in preclinical research and modern drug development.
Footnotes
Acknowledgements
Luisa Ottobrini (University of Milan, Italy) and Anna Maria Rapisarda (National Institutes of Health, USA) are gratefully acknowledged for the U251-mCherry cell line.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: A Szijártó and K Szigeti were supported by the ‘Janos Bolyai Research Fellowship Program’ of the Hungarian Academy of Sciences. This study was funded by the Hungarian Research Grants OTKA-115607 and by the National Research, Development and Innovation Office – NKFIH, grant No. K115607. The research leading to these results has received funding from the European Union Seventh Framework Programme FP7/2007-2013 under grant agreement No. 305311 (INSERT) and HEALTH-F2-2011-278850 (INMiND).
