Abstract
The increasing potential for radiation exposure from nuclear accidents or terrorist activities has intensified the need to develop pharmacologic countermeasures against injury from total body irradiation (TBI). Many initial experiments to develop and test these countermeasures utilize murine irradiation models. Yet, the route of drug administration can alter the response to irradiation injury. Studies have demonstrated that cutaneous injuries can exacerbate damage from radiation, and thus surgical implantation of osmotic pumps for drug delivery could adversely affect the survival of mice following TBI. However, daily handling and injections to administer drugs could also have negative consequences. This study compared the effects of subcutaneous needlesticks with surgical implantation of osmotic pumps on morbidity and mortality in a murine model of hematopoietic acute radiation syndrome (H-ARS). C57BL/6 mice were sham irradiated or exposed to a single dose of 7.7 Gy 60Co TBI. Mice were implanted with osmotic pumps containing sterile saline seven days prior to irradiation or received needlesticks for 14 days following irradiation or received no treatment. All irradiated groups exhibited weight loss. Fewer mice with osmotic pumps survived to 30 days post irradiation (37.5%) than mice receiving needlesticks or no treatment (70% and 80%, respectively), although this difference was not statistically significant. However, mice implanted with the pump lost significantly more weight than mice that received needlesticks or no treatment. These data suggest that surgical implantation of a drug-delivery device can adversely affect the outcome in a murine model of H-ARS.
Introduction
The hematopoietic system is sensitive to damage by ionizing radiation, and high-dose total body irradiation (TBI), as a result of accidental exposure or planned clinical exposure, can result in a potentially fatal hematopoietic acute radiation syndrome (H-ARS).1–3 In response to ionizing radiation, white blood cells and many hematopoietic progenitors primarily undergo apoptosis in response to DNA damage, 4 while erythrocytes undergo radiation-induced hemolysis,5–7 leading to leukopenia and anemia. The reduction in the white blood cell population can encourage opportunistic infections as well as coagulation dysfunction.8,9 Due to radiation damage to the bone marrow, there is an insufficient response by hematopoietic progenitors, leading to a failure to replace the mature blood cell population.7,8,10,11 Thus, mortality from H-ARS is associated with hematopoietic insufficiency.
The development of countermeasures to prevent radiation toxicity, especially H-ARS, has been the subject of research for more than 50 years. There is an acute need to be able to provide therapeutics to people exposed to radiation after nuclear accidents, such as occurred at the Fukushima Nuclear Power Plant, or terrorist attacks. However, there are currently only three radiation countermeasures approved by the Food and Drug Administration (FDA) for H-ARS. All three of these are drugs that must be administered after radiation exposure but prior to the manifestation of clinical symptoms (termed radiation mitigators). FDA-approved radiation mitigators filgrastim and pegfilgrastim are two preparations of granulocyte colony-stimulating factor, and sargramostim is a preparation of granulocyte/macrophage colony-stimulating factor.11 These mitigators must be administered via several subcutaneous or intramuscular injections, usually over a period of several days.12,13 While these drugs have been demonstrated to be effective for H-ARS in mice and nonhuman primates, the administration of multiple injections over several days is likely impractical for a mass-casualty event.
The search for novel radiation mitigators and the necessity to understand the mechanisms of these potential mitigators require significant animal experimentation. Although oral administration of drugs is the preferred clinical route, particularly for the large number of people who could be affected in a mass-casualty setting, it is often easier for preclinical studies to administer drugs subcutaneously. When drug efficacy requires daily administration over a prolonged period, many investigators choose to implant osmotic minipumps for drug delivery. This provides for consistent drug delivery without the need for daily injections and the necessary stress that this daily procedure might cause the rodents. Indeed, it has been shown that increased post-irradiation manipulations resulted in decreased survival of mice. 14 However, it is not known whether implantation of an osmotic minipump would adversely affect survival after irradiation, as studies have demonstrated that morbidity and mortality from radiation is increased in the presence of cutaneous injuries.15,16 The exposure to radiation together with a wound is termed “combined injury.” Murine studies have demonstrated that the presence of a wound can increase mortality from a 35% lethal dose of radiation at 30 days (LD35/30) to 85% lethality (LD85/30). 17 Additionally, radiation increases the closure time for a skin wound. 18 The increased mortality in combined injury is associated with increased opportunistic bacterial infection (from multiple Gram-positive and Gram-negative species) together with some indications of sepsis and decreased recovery of mature blood cells.17,19
In our search for novel radiation mitigators, we needed to determine whether it would be better to deliver the drugs through daily subcutaneous injection or through implantation of an osmotic minipump. We were unable to find any prior literature on this topic. We therefore decided to compare the effects of minipump implantation directly with 14 daily needlesticks or no manipulations to determine whether either of these manipulations altered the morbidity associated with irradiation in mice. We chose these parameters because they were similar to the parameters for an experimental drug test we were preparing. As post-irradiation wounds were previously demonstrated to result in increased morbidity and mortality, we investigated the effects of implantation of an osmotic minipump prior to irradiation, allowing sufficient time for recovery of the skin incision. In order to dissociate any potential minipump effects from any drug effects, we filled the minipump with physiologic sterile saline solution. The needlesticks did not inject any substance into the skin. Our data indicate that multiple needlesticks had a minimal effect on weight loss and mortality from TBI, but that implantation of an osmotic pump before TBI led to significantly increased weight loss and nonsignificant decreased survival.
Methods
Mice
This study was performed in compliance with the recommendations in the Guide for the Care and Use of Laboratory Animals of the National Academies of Sciences. 20 The protocol (PHA-18-064) was approved by the Institutional Animal Care and Use Committee of the Department of Laboratory Animal Resources (USUHS, Bethesda, MD). Female C57BL/6J mice were from a high SPF barrier (Barrier AX4; The Jackson Laboratory, Bar Harbor, ME). Barrier health reports were consulted to show that the purchased mice were free from common viruses, bacteria, and mycoplasma (Supplemental Data S1), and the mice were housed in individually ventilated cages. Mice were 12–14 weeks old (weight 18–23 g) at the time of irradiation. Female mice have traditionally been used for irradiation studies, as they are less aggressive and therefore do not usually have wounds from fighting that could form a combined injury after irradiation and alter the study significantly. Mice were housed four to five per cage in a facility accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. Animal rooms were maintained at 21 ± 2°C, 50 ± 10% humidity, and 12-hour light/dark cycle. Commercial rodent ration (Harlan Teklad Rodent Diet 8604) and water were freely available. Mice were assigned to a specific group (pump implantation, daily needlestick, or no intervention; n = 9–10/irradiated group, n = 4/sham group) by randomly assigning two cages to each treatment group. No cage-specific effects were noted during the progression of the experiment. A total of 41 mice were used in this study. From our previous experience with survival studies after irradiation with this strain of mice, 10 mice/group is sufficient for LD50 analysis (where lethality is determined by time of euthanasia indicated by morbidity scoring). All mice were individually identified by tail marking. For all procedures except injection, mice were picked up by the tail. Outcome measurements were weight and survival. Weight was taken as indicated (every four days before the critical period and every two days from day 14 to day 30). A morbidity score sheet was used daily during the critical period (days 14–28 post irradiation) to determine whether the mice could be considered moribund and euthanized. This score sheet has been published previously. 21 Mice were scored twice per day according to their appearance, respiratory rate, general behavior and movement, provoked behavior, and weight loss. A score of 12 (maximum score) in any one category or a combined total score of 12 in all categories was used as the indication for euthanasia. In general, animals were euthanized in response to the morbidity score, and only in rare instances were animals found dead in the cage. Two animals out of 41 in these experiments were found dead, while 19 were euthanized at time points determined by scoring.
Irradiation
Each irradiated mouse received a single-dose TBI of 7.7 Gy 60Co at a 0.615 Gy/min dose rate while inside “jigs” (Plexiglas holders) in a bilateral gamma radiation field at the Armed Forces Radiobiology Research Institute (AFRRI) 60Co facility, as previously described. 22 Irradiation was performed at the same time each day (morning) to provide consistent outcomes between experiments and to accommodate the circadian rhythm of bone-marrow cell cycles. 23 Sham-irradiated mice were placed in “jigs” for the same time periods as mice that were irradiated, but they did not receive radiation.
Osmotic minipump implantation
Alzet Osmotic minipumps (1.5 cm long×0.6 cm diameter, 0.4 g weight; #1004; Durect, Cupertino CA) were filled and equilibrated with sterile saline 24 hours before implantation. Seven days prior to irradiation, in the morning, mice were anesthetized with isoflurane (3.5% initial and 2% maintenance in oxygen; Baxter Healthcare Corp., Deerfield, IL), and the dorsal area of the neck was shaved and cleaned with betadine scrub followed by 70% ethanol. Minipumps were implanted subcutaneously under sterile conditions. Buprenorphine for analgesia (0.01 mg/kg, injected subcutaneously in the caudal shank) was administered immediately before pump implantation. A 1 cm incision was made through the skin on the neck, and space was made for the pumps by blunt dissection with sterile forceps from the incision site until under the skin of the left flank. Skin was sutured with sterile nonabsorbable ETHILON 6.0 sutures (ETHICON, San Lorenzo, PR), and triple antibiotic ointment (polymixin B sulfate, bacitracin zinc and neomycin sulfate; G&W Laboratories, South Plainfield, NJ) was applied b.i.d. for seven days. This preparation contains tocopheryl acetate, which can act as a mild analgesic. No additional anti-inflammatory agents were used, as these could prevent systemic hematopoietic recovery following TBI. Mice were kept on electric heating pads throughout the procedure and until complete recovery from anesthesia when they were returned to their home cage.
Needlesticks
Mice received daily subcutaneous needlesticks each morning with 25 G sterile needles to simulate injection injury. Mice were scruffed at the neck before the hind-limb area was cleaned with 70% isopropanol and needlesticks given. Needlesticks were administered from days 2 through 16 post irradiation or sham irradiation, and were alternated from the left to right side from day to day. Empty needlesticks were used, as it was previously demonstrated that the injection of saline alone could influence survival advantage. 14 Mice that did not receive needlesticks were not handled daily, but only when weighed.
Euthanasia
Mice that were scored to be moribund were immediately euthanized using carbon dioxide or by intraperitoneal injection of ketamine/xylazine according to current AVMA guidelines. Mice that survived until 30 days after irradiation were euthanized by ketamine/xylazine administration followed by exsanguination. After euthanasia, the animals were skinned, and the skins were reversed and examined for petechiation, hemorrhage, or other skin lesions that could be concealed by fur and skin pigmentation.
Statistical analysis
Kaplan–Meier plots were analyzed using Fisher’s exact tests to assess the differences in survival between the groups after irradiation (GraphPad Prism v7.1; GraphPad Software, La Jolla, CA). A p-value of ≤0.05 (two-tailed) was considered statistically significant. Survival among the three groups was analyzed using a log-rank test. Weights from day 3 through day 30 (post irradiation) were compared to baseline weights (day –7) after pump implantation (where relevant), and weight changes were calculated. A mixed model for repeated measures was used in the analysis to account for some missing data points, and the model allowed for correlated observations over time within each animal using a first-order autoregressive structure. Groups were compared overall and at each time point using post-hoc tests with Sidak’s correction for multiple comparisons.
Results
Effect of needlesticks and surgical implantation of osmotic minipumps on weight loss following TBI
We implanted the osmotic minipumps prior to irradiation, allowing a week for recovery of the site of incision prior to radiation exposure. Our laboratory and others have demonstrated that cachexia has a significant effect of TBI, and can be used as an indication of radiation-induced morbidity.24,25 We therefore compared the effects of pump implantation with needlesticks or no treatment on weight loss after irradiation. Body weights were obtained for all animals, starting seven days prior to TBI (day –7) to provide individual baselines. Subsequent measurements were obtained on days –2, 0, 3, 7, 11, 14, 16, 18, 20, 22, 24, 26 and 28 post irradiation. Sham-irradiated mice with osmotic minipumps, needlesticks or no treatment gained an average of 1 g over the 37-day experiment, with no significant differences between groups (Figure 1). In contrast, mice exposed to 7.7 Gy (0.6 Gy/min) TBI exhibited weight loss, with nadirs of weights between 18 and 22 days post irradiation (Figure 1). There were significant differences among the three different groups (F(2, 31) = 6.60, p = 0.004), significant changes over time (F(10, 162) = 19.68, p < 0.001) and a significant group–time interaction (F(20, 162) = 1.94, p = 0.013). Post hoc tests of the main effect of group showed that with averaging over time, there was significantly greater weight loss in the pump group than in the needlestick group (p = 0.003) but no significant overall differences in the group with no manipulation after irradiation. Separate comparisons between groups at each time point showed significant differences between the needlestick and pump groups at 14, 16, 18, 20, 22, 24 and 26 days. Comparisons also showed a significant difference between the needlestick and no manipulation groups on day 22 and between the pump and no manipulation groups on days 18 and 20. Thus, mice with the implanted minipump lost significantly more weight than either the mice receiving daily needlestick or no manipulation.
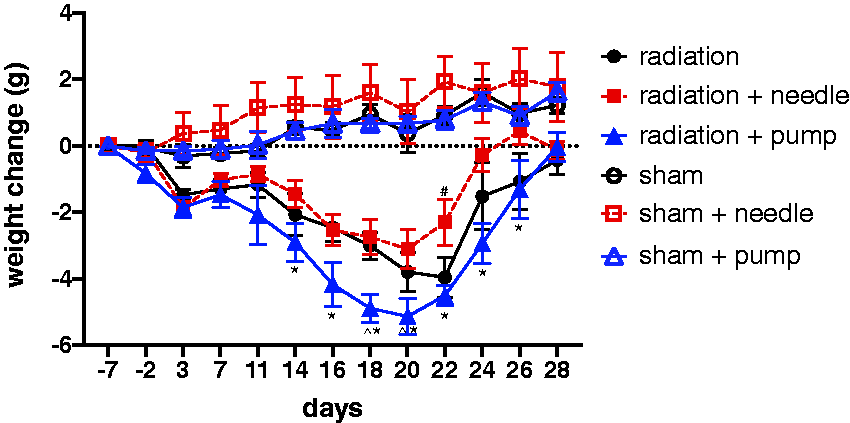
Time-dependent change in body weight in nonirradiated control sham mice or mice before and after exposure to 7.7 Gy 60Co total body irradiation (TBI). Measurements were initiated seven days before irradiation just before minipump implantation. Data represent the change in body weight relative to the initial weight seven days before irradiation ± standard error of the mean (n = 9–10 mice/group irradiated, n = 4/group for sham). *p < 0.05 compared to radiation+needle mice. ^p < 0.05 compared to radiation alone mice.
Effect of needlesticks and surgical implantation of osmotic minipumps on survival following TBI
Previous studies have demonstrated that wounds increase the lethality of radiation. Death can also occur earlier in combined-injury animals compared to animals exposed to radiation alone. 18 We investigated the rates of mortality in animals receiving subcutaneous osmotic pumps compared to animals receiving needlesticks or no treatment (Figure 2). Our findings indicate that implantation of osmotic pumps resulted in only 37.5% survival in animals exposed to 7.7 Gy TBI. In contrast, animals that received needlesticks displayed 70% survival, and untreated animals had 80% survival. Thus, the implantation of osmotic pumps resulted in ∼40% greater mortality. However, survival among the three groups was not significantly different based on a log-rank test (χ2 (2) = 3.077, p = 0.535). Implantation of osmotic minipumps did not result in earlier initial times of death (initial mortality at 10–11 days post irradiation in all groups), but did show a trend of greater numbers of animals dying at later time points (days 18–22 post irradiation). No skin lesions, hemorrhage or petechiation was noted in any groups postmortem.
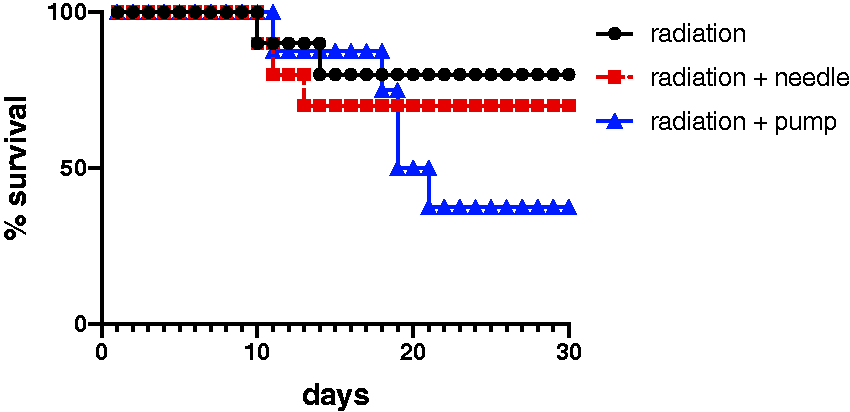
Kaplan–Meier curves of the effects of osmotic minipump implantation or daily needlesticks on survival from TBI. Female C57BL/6 mice 12–14 weeks old were exposed to total body 60Co irradiation (7.7 Gy at 0.6 Gy/min). Minipumps were implanted seven days before irradiation. Needlesticks were administered from days +2 through +16 post irradiation (n = 9–10/group).
Discussion
Understanding the effect that the route of administration of medical countermeasures has on the response to irradiation is critically important. We initiated these studies to compare two routes of administration, both of which we hypothesized could have a negative impact on the response to irradiation: daily injections or implantation of an osmotic minipump. Studies in murine models of H-ARS have demonstrated that the presence of wounds increase morbidity and mortality induced by radiation.17,18 We found that implantation of an osmotic minipump seven days before irradiation can lead to significantly reduced body weight of mice and a nonsignificant decreased survival. However, the stress of daily handling and receiving needlesticks alone did not lead to any differences from mice receiving no manipulations after irradiation.
We originally hypothesized that implanting the osmotic minipump a week before irradiation would allow time for the surgical wound to heal, and therefore would not necessarily cause the “combined injury” problem of wounds plus irradiation that leads to worse survival. However, although the skin wound did appear to have healed, the poorer outcomes in the pump group suggest that internal inflammation may still have been occurring and contributing to the greater weight loss and reduced survival. Additionally, because of the greater weight loss, the pumps were a greater percentage of body area, and their shapes were more clearly visible through the skin of mice that lost the most weight (data not shown). This also may have contributed to the general discomfort of irradiated mice. In this case, the osmotic minipumps may be useful in a larger species where the pump is less of the percentage of the animals’ total subcutaneous space and weight.
We sought to use the osmotic minipumps to spare the mice the stress of daily handling and injections. Surprisingly, daily needlesticks in the hind limbs for 14 days did not result in any increased weight loss or reduced survival. These data suggest that there was no significant detriment in body weight or survival from the stress associated with this daily procedure. We alternated the needlestick between limbs, and we did not see any bleeding around the insertion area, suggesting that this was not producing a combined injury. We also did not see any petechiation on the pelts of the mice. However, in a subsequent study from our group, we have found that daily injections (even in the control saline group) do decrease survival in comparison to uninjected mice (data not shown). Mice in this later experiment were irradiated at a slightly higher dose (7.85 Gy). So, it is possible that daily injections are detrimental with a stronger irradiation injury.
There are two caveats to this pilot study. First, our study did not include a group of mice with anesthesia alone to control for the effects of anesthesia on survival following radiation. Because anesthesia was performed one week prior to radiation exposure, we expect that the anesthesia would have minimal effects, as the inhaled anesthesia would no longer be present in the tissues by the time of irradiation. Although anesthesia has been demonstrated to increase hypoxia during radiation, which can increase tumor resistance to radiation damage, 26 no study has demonstrated an effect of anesthesia when applied seven days prior to radiation exposure. Second, this study utilized only female mice. In general, male mice have a stronger acute inflammatory response than female mice. 27 So, we assumed that irradiated male mice with an implanted pump would have a worse survival rate than the females in this experiment. For this reason, we did not feel justified in repeating this experiment in male mice.
Although we observed a decreased survival with the female mice implanted with the minipumps, these data were not significant. This experiment was a pilot study, designed to inform us as to the best way to administer a compound to mice that required daily administration after irradiation. Given the effect sizes we saw, our study was underpowered to achieve significance. Yet, as we did observe reduced survival, we did not feel that repeating the study with additional animals to gain significance warranted the use of additional mice in a survival study. These data provided us with sufficient data to avoid implanting osmotic minipumps in mice in irradiation experiments. These observations may also be relevant to other rodent irradiation studies. We therefore elected to administer the necessary drugs in subsequent experiments by subcutaneous injection after irradiation.
Supplemental Material
sj-pdf-1-lan-10.1177_0023677220939991 - Supplemental material for Comparison of the effects of osmotic pump implantation with subcutaneous injection for administration of drugs after total body irradiation in mice
Supplemental material, sj-pdf-1-lan-10.1177_0023677220939991 for Comparison of the effects of osmotic pump implantation with subcutaneous injection for administration of drugs after total body irradiation in mice by Amory L Koch, Milan Rusnak, Katherine Peachee, Akira Isaac, Elizabeth A McCart, W Bradley Rittase, Cara H Olsen, Regina M Day and Aviva J Symes in Laboratory Animals
Footnotes
Acknowledgements
Some of the authors are employees of the U.S. government. This work was prepared as part of their official duties. Title 17 U.S.C. §105 provides that ‘Copyright protection under this title is not available for any work of the United States Government.’ Title 17 U.S.C. §101 defines a U.S. government work as a work prepared by a military service member or employees of the U.S. government as part of that person’s official duties. The views in this article are those of the authors and do not necessarily reflect the views, official policy, or position of the Uniformed Services University of the Health Sciences, the Armed Forces Radiobiology Research Institute, Department of the Navy, Department of Defense or the U.S. Federal Government.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported a Department of Defense JPC-7 grant (0130‐18‐0003‐00022) to A.J.S. and R.M.D. There are no financial or nonfinancial conflicts of interest for any of the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
