Abstract
Urethane is a widely used anesthetic in animal lower urinary tract research. Our objective was to investigate the quality of anesthesia and the correlation between bladder (voiding) contractions, micturition pressure, bladder capacity and urethane dosage and body weight. Urethane was given subcutaneously and/or intraperitoneally (1.0–1.2 g/kg). The bladder was filled with saline and the bladder pressure was recorded continuously. Animals in which the subcutaneous/intraperitoneal ratio was higher needed less urethane. Heavier animals needed less extra urethane. In animals, in which no bladder contractions could be evoked, the total amount of urethane given was similar to that in those that did show contractions. In the animals that did void, the bladder never emptied completely and residual volumes remained. There was no relationship between animal weight or total amount of urethane and mean capacity. Anesthesia lasted up till 14 h, during which bladder (voiding) contractions could be recorded. Considering all results, we conclude that urethane is a well suited anesthetic for acute lower urinary tract physiological research in the intact rat.
Voiding in rats is a common model for studying the physiology of the lower urinary tract. For this purpose animals are usually anesthetized. Over the years, many anesthetics have been applied, among which urethane is one of the most commonly used. It has been shown to have limited influence on the autonomic nervous system, and both excitatory and inhibitory reflex responses in a variety of organs and systems are well preserved. 1 The literature shows that urethane is administered in doses varying from 0.56 to 1.5 g/kg via different routes of administration: subcutaneous, intraperitoneal or intravenous. It induces a prolonged and relatively stable anesthesia during which reflex voiding is maintained for up to 24 h.2–5
Our ongoing research on the lower urinary tract in rats involves the study of urodynamic parameters such as bladder pressure and bladder capacity, and also the activity of the bladder innervating nerves.6,7 These experiments require a long-lasting anesthesia that affects neither urinary bladder contraction nor the activity of the nerves. In 121 animals, all anesthetized with urethane, we studied the relationship between administered urethane (dose and route of administration), animal weight, and cystometric parameters such as bladder (voiding) contractions and bladder capacity. The aim was to define a uniform, optimum anesthesia protocol for lower urinary tract research in rats.
Animals
All animal procedures were reviewed and approved by the Erasmus Medical Center Animal Care and Use Committee. The animals were housed in a temperature-controlled (25℃) facility with a 12 h light/dark cycle and given free access to food and water. A total of 121 male Wistar rats weighing 412 ± 46 g (range: 279–510 g) were included in this study. At the end of the experiments, animals were euthanized by injecting potassium chloride (KCl, 0.2 M) into the heart.
Materials and methods
Animals used from 2011–2015 were pooled in this analysis. The results on nerve activity are or will be published elsewhere.8–10 The animals were all 8–9 weeks old, and handled in the same way by the same investigator.
The animals were anesthetized with 1.0 or 1.2 g/kg urethane (ethyl carbamate, 50% w/v solution in water; Sigma–Aldrich, St Louis, MO, USA). The first part of the dose (57 ± 10% of the calculated amount) was given intraperitoneally (i.p.) after briefly (2 to 3 min) anesthetizing the animal with isoflurane inhalation (induction box, 5%, 0.5 L/min O2). After about 2 h, the remaining part of the dose (36 ± 8%) was injected subcutaneously (s.c.) in the nape of the neck. If necessary, extra urethane was given i.p. during the experiment (7 ± 9% of the total amount). In eight of the 121 animals, all the urethane was given i.p. About 3 h after the first injection of urethane, the animal was put in a supine position on a heated underground. The depth of anesthesia was checked by the plantar reflex. The abdominal skin was shaved and locally anesthetized with xylocaine spray (10%). If the animal still reacted to pinching of the foot or cutting of the abdominal skin, extra urethane was given i.p. (0.13 ± 0.09 mL, range 0.05–0.55 mL [n = 62]). In the first 22 animals, a tracheotomy was performed. In all the animals the abdominal wall was opened and the bladder was dissected from the underlying tissue. A 23 G catheter was inserted into the dome of the bladder both for saline filling and for bladder pressure recording. The bladder was filled with 0.9% sodium chloride (NaCl) at 0.11 mL/min in the first 67 and at 0.06 mL/min in the remaining 46 animals. The catheter was attached to a disposable pressure transducer connected to a Statham SP1400 blood pressure monitor and to a pump (Harvard Apparatus Model 2202; Harvard Apparatus, Holliston, MA, USA or Hospal K10 infusion pump; Hospal Dasco, Medolla, Italy). Measurements were started ∼5 h after the first injection of urethane. The pump was stopped when a voiding contraction occurred or when a volume of 1.2 mL was reached without voiding. Bladder pressure was sampled at 25 Hz and recorded using custom written software in LabVIEW® (National Instruments, Austin, TX, USA). The data were processed and analyzed using MATLAB® (MathWorks, Natick, MA, USA) and Excel® (Microsoft Excel 2010). Both the basal pressure pbasal at the start of filling and the maximum pressure pmax just before voiding were determined in 50 animals. The micturition pressure pmict was calculated by subtracting pbasal from pmax. Functional bladder capacity was calculated as filling time × filling rate in 32 animals. On average, in each animal, five micturition cycles were evoked and the mean capacity of all consecutive fillings (1–5) was calculated. Measurements lasted from 4 up to 14 h. In 10 animals extra urethane was given i.p. during the measurements. Urethane administration was quantified as: (a) the calculated amount given at the start, based on the animal’s weight (2.4 × (animal weight)/1000), partly given i.p and s.c.; and (b) the total amount = calculated amount + extra urethane (given i.p. during the measurements when necessary).
Animals were killed at the end of the measurements by injection of 0.2 M KCl into the heart, postmortem examinations were not performed. Not all 121 animals could be used for all analyses because of failures in equipment set-up or recording such as catheter displacement, the presence of air bubbles or movement of the rat (see Figure 1).
Flow diagram of the animals used for different procedures. Of the 121 animals, eight received urethane intraperitoneally only, which are not shown in the diagram. Animals are divided into groups based on whether the trachea is opened or closed, the flow rate, the amount of extra urethane and the occurrence of voiding contractions.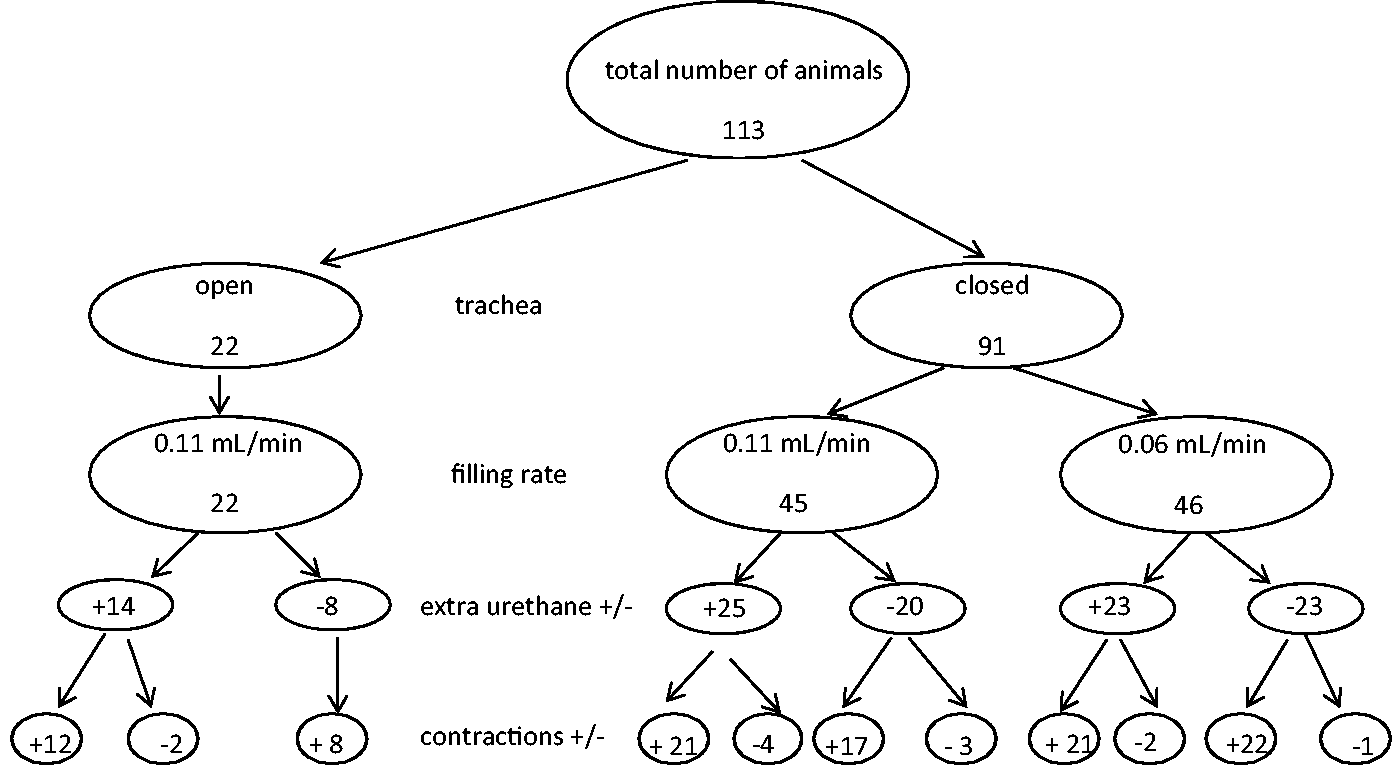
Statistics
Data are reported as mean ± standard deviation. Statistical analysis was performed using SPSS statistical package (version 21.0; SPSS Inc, Chicago, IL, USA). To compare data, analysis of variance (ANOVA) was used for normally distributed data, the Mann–Whitney U-test (MWU) for non-normally distributed data and Spearman’s rho test for correlations. The Shapiro–Wilk test was used to check for normality. P < 0.05 was considered to be statistically significant.
Results
One animal died immediately after the first injection of urethane. After dissection we found that the urethane was accidentally injected into the upper part of the intestine, and not i.p. as intended.
Tracheotomy
Because urethane has been reported to enhance the pulmonary airway resistance, the trachea was opened to facilitate breathing. 11 In four animals, small blood vessels running over the trachea were damaged during the dissection, and small amounts of blood leaked into the trachea causing breathing problems. Mucus had to be removed from the trachea in two (other) animals only. After undertaking this in 22 animals, the opening of the trachea was omitted from the procedure.
Intraperitoneal and subcutaneous
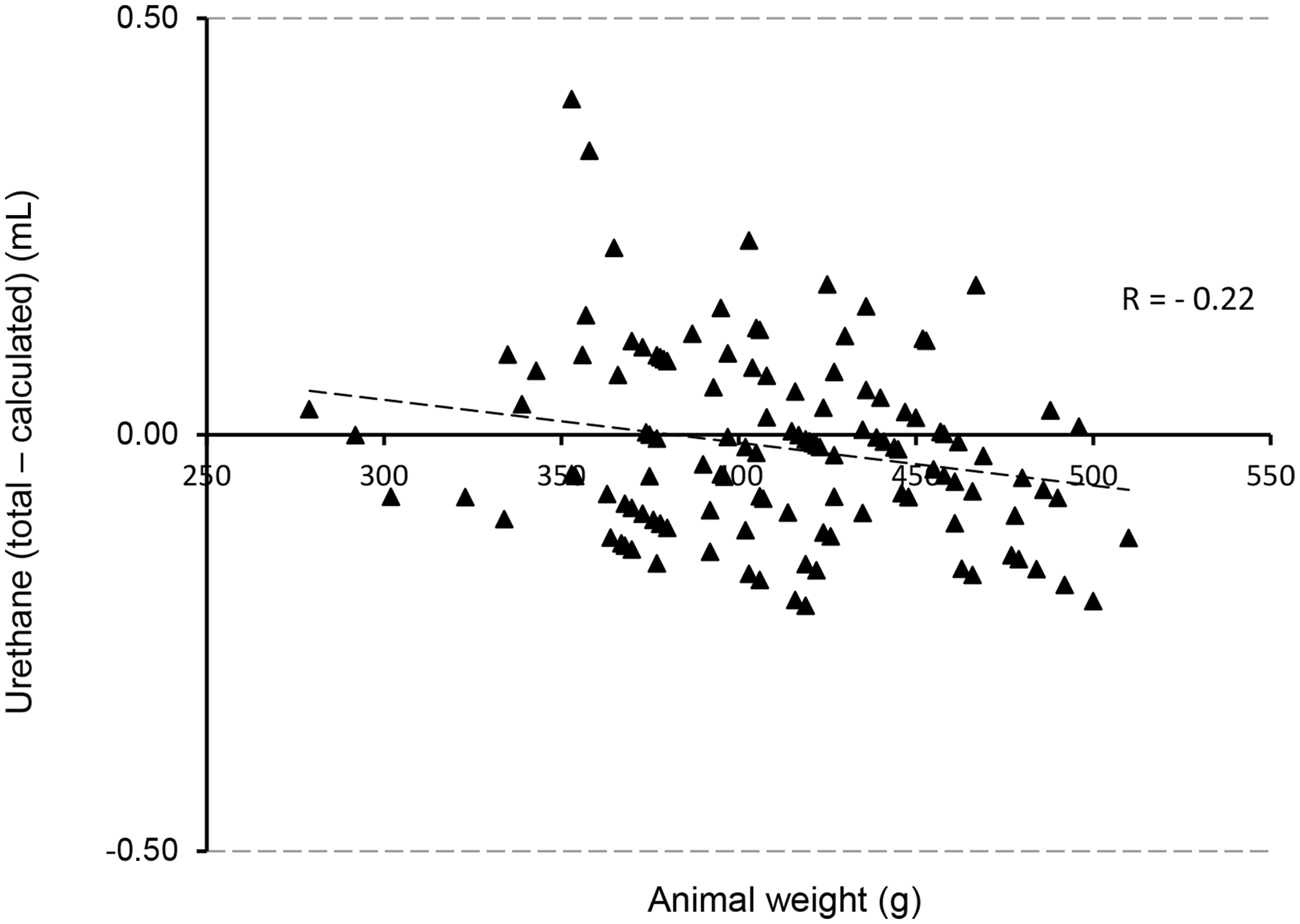
In Figure 2, the calculated and the total amount of urethane are plotted as a function of the animal’s weight. The correlation coefficients are respectively 0.99 and 0.63. When plotting the total minus the calculated amount against weight, a negative relationship occurred, which means that heavier animals needed less extra urethane (R = – 0.22, P < 0.05, Figure 3). The eight animals to which urethane was given i.p. only, needed more urethane than the ones which received urethane i.p. and s.c: 1.10 ± 0.08 mL vs 0.97 ± 0.13 (MWU, P < 0.05), seven out of the eight were given extra urethane. pmict did not differ between the two groups.
The relationship between animal weight and (a) the calculated amount of urethane (R = 0.99) and (b) the total amount of urethane administered during the experiment (R = 0.63). n = 121. The relationship between animal weight and the amount of extra urethane given (total – calculated amount) (R = – 0.20). Heavier animals need less extra urethane. n = 121.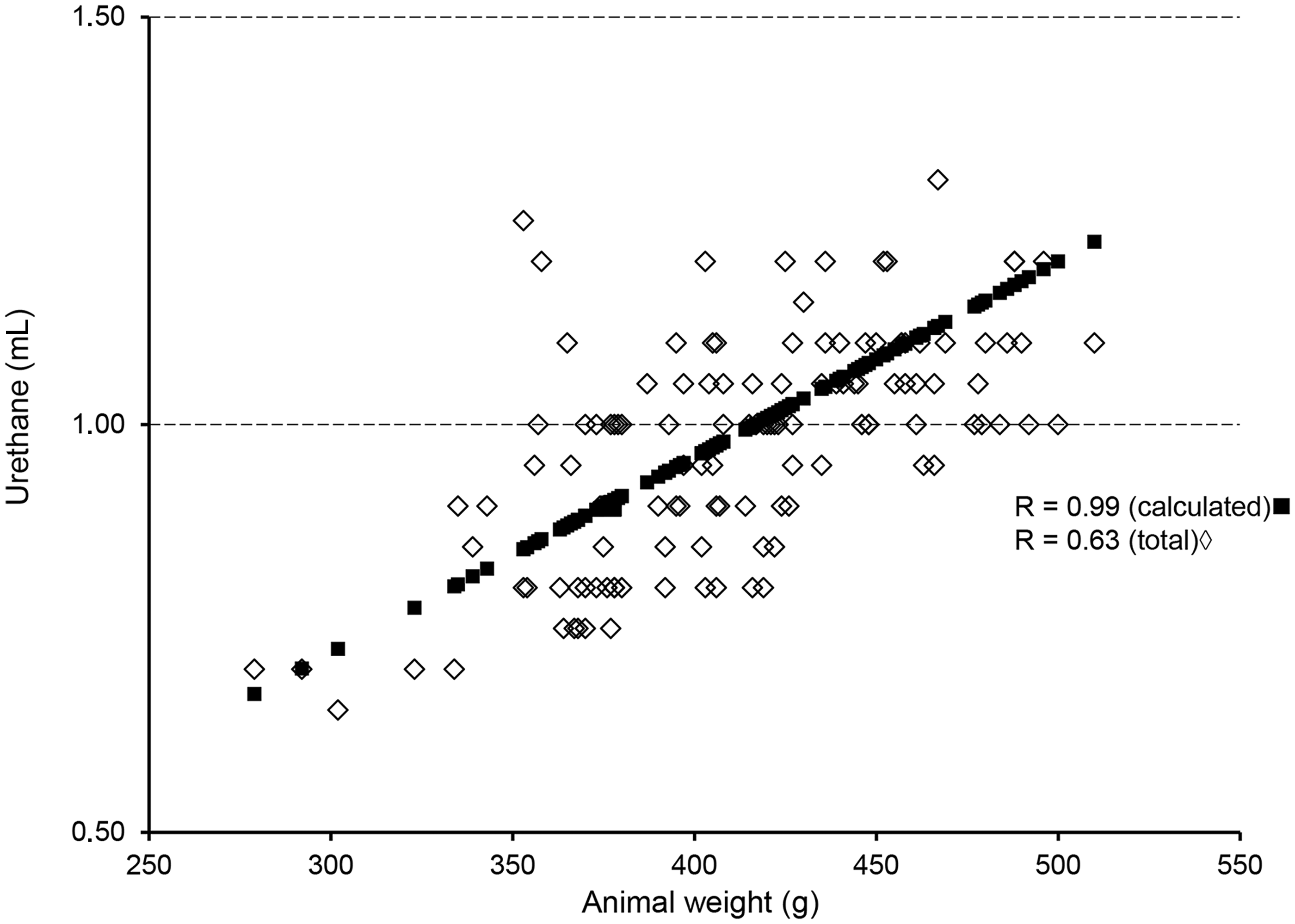
Animals in which the ratio s.c./i.p. was higher needed less urethane (Spearman’s correlation coefficient −0.19, P < 0.05). There was no significant relationship between animal weight and this ratio (Spearman’s correlation coefficient 0.010, P = 0.915).
Emptied and continuous filling
Comparison of micturition pressure (pmict) and basal pressure (pbasal) and capacity between different groups of animals.
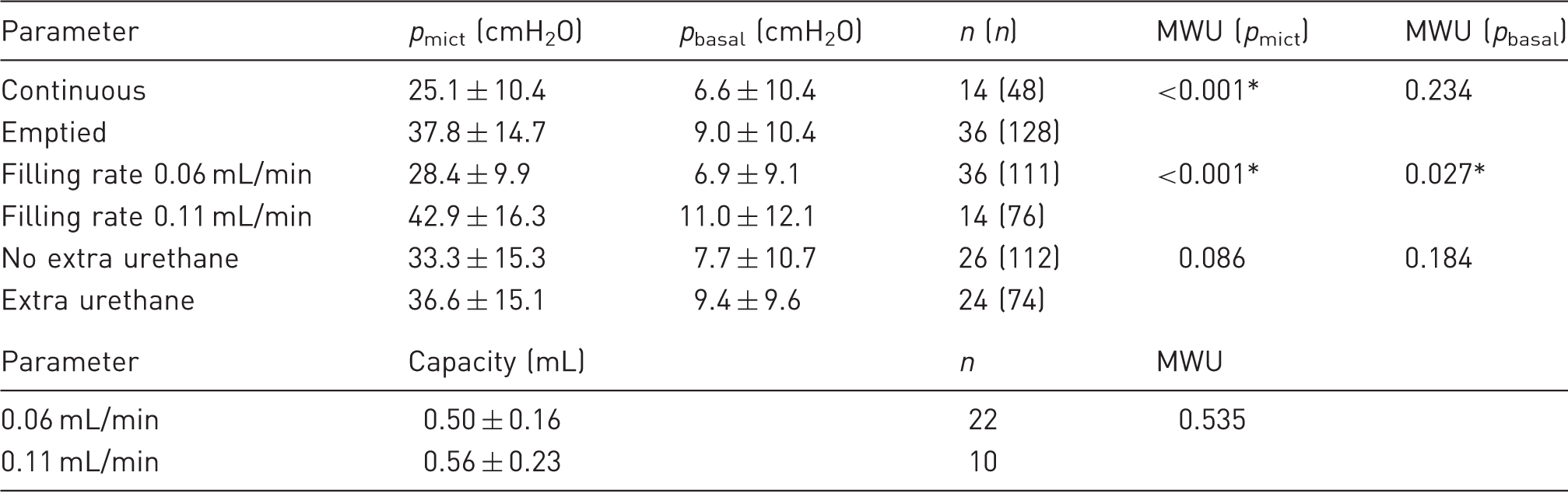
There was overlap between categories (n = number of animals, (n) = number of contractions). The non-parametric Mann–Whitney U-test (MWU) was used to compare data. *P < 0.05 was significant.
In the 32 animals in which the bladder was emptied, the capacity was determined: 0.52 ± 0.18 mL. There was no relationship between animal weight and mean capacity or total amount of urethane and mean capacity (Spearman’s correlation coefficient respectively 0.009 and 0.136, P = 0.961 and P = 0.458). Comparison showed that the mean capacity of consecutive fillings did not change over time (ANOVA, P = 0.890). Neither was there a relationship between the amount of urethane given and the total duration of the experiment. In all animals in which voiding contractions could be evoked, post void residual volumes remained. In 14 contractions out of five animals, both filled volume and post void residual volume were determined. The mean residual volume was 0.25 ± 0.23 mL, which was 44 ± 29% of the filled volume.
Filling rate
Comparison of micturition pressure (pmict) and basal pressure (pbasal) related to filling rate, extra urethane and continuous filling vs emptied bladders.
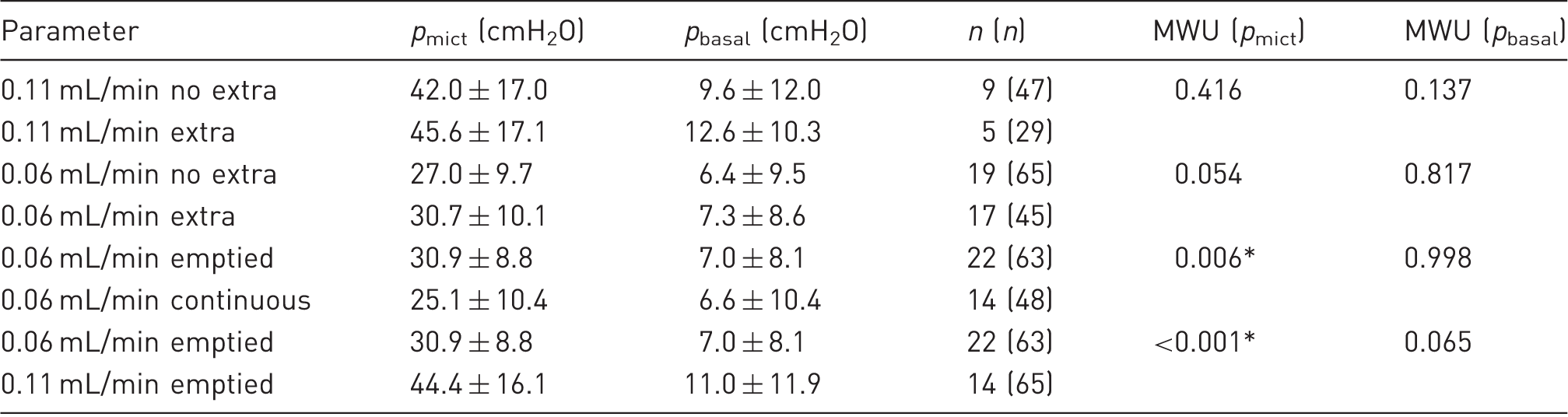
There was overlap between categories (n = number of animals, (n) = number of contractions). The non-parametric Mann–Whitney U-test (MWU) was used to compare data. *P < 0.05 was significant.
No contractions
In 12 of the 113 animals no bladder contractions could be evoked by filling. When comparing animals with and without contractions, the total amount of urethane given was 0.97 ± 0.14 mL vs 0.91 ± 0.11 mL, the extra amount of urethane was 0.13 ± 0.09 mL vs 0.09 ± 0.06 mL; the ratio (s.c./total i.p.) was 0.60 ± 0.20 vs 0.59 ± 0.25 and body weight was 411 ± 46 g vs 413 ± 52 g. None of these comparisons showed significant differences.
Discussion
The effect of several types of anesthetic (e.g. inhalation agents, barbiturates and opioids) on the micturition of rats and mice has been studied before.4,12,13 When anesthetized rats are compared to conscious rats, differences are found in bladder and external urethral sphincter (EUS) functions.12,14,15 In anesthetized animals, residual volumes and a smaller capacity have frequently been described.2,4,16–18 Comparison of data is difficult since different strains, male and female animals and animals with different body weights have been used. Overall, in conscious animals, pbasal ranged from 3 to 11 cmH2O and pmict from 24 to 60 cmH2O.19–23 There was also a large range in micturition volumes (0.17–1.2 mL) but residual volumes were low (0.06–0.08 mL).19–23 The main difference therefore appears to be in the voiding efficiency, which was much higher in conscious animals.
However, when anesthesia is unavoidable, the preferred agent is urethane, because although there are some changes in bladder and EUS functions, the micturition reflex is not suppressed. 1 To obtain good anesthesia in a rat, a concentration of 1 g urethane/L blood is necessary, and this can be accomplished by giving 1 g/kg i.p. 24 The literature shows that a dose of 1.2 g/kg is commonly used.16,25
In our hands, the dissection could only commence ∼3 h after the administration of urethane because only then was the animal sufficiently anesthetized. Findings in the literature are very diverse, Cheng and de Groat 17 have stated that urodynamic examination usually begins 3–4 h after the induction of anesthesia (1.2 g/kg i.p.), cystometry is started one hour after injection of urethane (1.0 g/kg i.p.) according to Yokoyama et al., 25 and Field et al. 5 have stated that the induction time for urethane (1.2 and 1.5 g/kg i.p.) is less than 5 min. It must be kept in mind that in these studies different animals and different models were used (f.i. spinalized and infarcted).
Since the dose of urethane was calculated from the animal’s weight, the correlation coefficient between both should be 1. That the coefficient of the total amount given differs from 1 (Figure 2, R = 0.63) can be explained by the fact that some animals received more and some less than the calculated amount based on 1.2 g/kg. Animals are not exactly the same and may respond somewhat differently to injected urethane – those with more fat could ‘store’ urethane. Animals need more urethane when it is applied i.p. only. This can be explained by the finding that substances administered i.p. are more readily absorbed in the circulation and metabolized than those administered s.c. 26
The tracheotomy caused more problems than it prevented, hence it was omitted from the dissection procedure. In practice, the trachea can be opened within a few minutes in cases where breathing problems arise.
Unfortunately, in 12 animals no bladder contractions could be evoked by filling. A possible explanation for this absence of contractions is the filling grade of the bladder at the time of opening the abdomen and thereafter. When the bladder has been (too) full for (too) long, overdistension of the bladder wall may occur which eventually results in a non-contractile bladder. It is therefore important to check the bladder volume immediately after opening of the abdomen. To minimize the risk of overdistension, one could decide to always insert the catheter just after opening the abdominal space, even when the bladder is not yet full.
With continuous filling, pmict was lower than when the bladder was emptied in between fillings. A lower filling rate also resulted in lower pbasal and pmict. Extra urethane had no effect on pbasal or pmict.
One of the disadvantages of urethane is that it is known to produce side-effects (Sigma Product Information U2500). Intraperitoneal administration has been shown to produce leakage of plasma into the peritoneal cavity, which can induce hemodynamic instability and alter levels of renin and aldosterone.2,24 A number of the severe effects of urethane on the reno/vascular system and direct damage to abdominal organs are not seen when routes of administration other than i.p. are used. This indicates that systemically administered urethane produces much less long-term damage.
In summary, we can recommend urethane as an anesthetic for electrophysiological experiments on the lower urinary tract in rats when conscious animals cannot be used. We advise starting with 1 g/kg (∼2/3 i.p. and ∼1/3 s.c.) and adding extra urethane only when necessary and only in very small doses. We also recommend only opening the trachea when breathing problems arise. Furthermore we suggest inserting a catheter into the bladder immediately after opening the abdomen, to relieve or prevent an overdistended bladder. Overall, we conclude that urethane is a well suited anesthetic for acute lower urinary tract physiological research in the intact rat. The anesthesia lasts long and allows the recording of bladder (voiding) contractions and peripheral nerve activity.
Footnotes
Acknowledgements
All authors were responsible for conception and design of the experiments. Handling of animal and surgical procedures were done by EvA. EvA, MC and FC were also responsible for collection, analysis and interpretation of data. EvA drafted the manuscript and all authors were responsible for revising it.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was partly funded by Marie Curie actions FP7 of the European Union, TRUST (training of urology scientists to develop new treatments) program under Grant 238541.
