Abstract
Animal models are in constant development to benefit scientific research. Rheumatoid arthritis (RA) is considered a very complex disease due to its complicated pathogenesis, and patients with rheumatic disease around the world are still unable to obtain effective, simple and curable treatment. In order to obtain a clear insight into the pathogenesis of RA, a rat model was established based on the concept of Bi syndrome in Traditional Chinese Medicine by simulating the conditions of RA as much as possible via the change in the physical conditions wind, damp, cold and heat (WDCH). For the first time, a new WDCH-induced RA model in female rats was successfully established and evaluated by body-weight change, paw swelling, blood cells analysis, spleen and thymus coefficients, autoantibodies and serum cytokine changes and histopathology. This model is characterised by its objectivity, no exogenous induction, short modelling time, extremely elevated expression level of autoantibodies and obvious histopathological change. The establishment of such a new model may provide more benefits in the research of the pathogenesis of RA.
Introduction
Arthritis is a common condition that causes acute or chronic joint inflammation. It includes rheumatoid arthritis (RA), which can recur and involve the heart. 1 It manifests clinically by warm, swollen and painful joints, in addition to abnormal levels of autoantibodies. RA is a chronic and systemic synovitis characterised by a symmetrical and aggressive inflammation of the joints, accompanied by positive serum rheumatoid factor, joint deformation and function loss. 2 While the cause of RA is not clear, it is believed that it is mainly characterised by the hyperplasia of the synovium, interstitial inflammatory cell infiltration, hyper-dense micro-vascularisation, pannus formation and the destruction of cartilage and bone.3–5
RA is mainly treated by drug administration, especially, non-steroidal anti-inflammatory drugs (NSAID; e.g. ibuprofen, aspirin), slow-acting anti-rheumatic drugs (SAARD; e.g. salazosulfapyridine, 6 chloroquine and leflunomide), cytotoxic drugs (methotrexate, 7 cyclophosphamide), glucocorticoids, biological agents (adalimumab, infliximab, etanercept) 8 or Chinese herbal medicine (Tripterygium wilfordii, sinomenine and total glucosides of paeonia).9–11 In addition, bone-marrow transplantation, immunoapheresis and surgery are often used in RA treatment. In the long term, 12 patients endure huge drug side effects, and the patient’s tolerance is obviously lowered, resulting in a lower clinical cure rate. The pathogenesis of RA is complicated and unclear, and existing classic animal models cannot truly reflect the pathological mechanism of arthritis, which is responsible for the difficulties in treating RA. 13
Arthritis animal models mainly include acute inflammatory models and chronic arthritis models. Acute models include the mouse ear-swelling model, the hen egg lysozyme-induced arthritis model, the Carrageenan-induced arthritis model and the rat subcutaneous air-pouch model.14,15 Chronic inflammatory models include two popular models of immune inflammatory reaction: the adjuvant-induced arthritis (AIA) model and the type II collagen-induced arthritis (CIA) model. Both of these models induce cellular and humoral immunity to cause the polyarthritis. Although they are the best models for simulating human RA pathology in clinical practice and are the predominant models used in RA research and drugs screening, they are induced by exogenous pathogens rather than originating from the body itself. In the last few years, models of arthritis in transgenic animals have also appeared, such as env-Px-Tg mice, human TNF-Tg mice, human CD4-Tg mice and human HLA B-27-Tg mouse models. Nevertheless, the use of the transgenic animal models still has some limitations such as the impossibility of exploring multiform disease due to one or several gene mutations affecting the disease. Hence, the pathogenesis in RA cannot be fully simulated. If the animal itself can objectively form a model of RA and induce cellular and humoral immunity in vivo, it will be more realistic, objective and valuable.
According to the ancient Chinese concept of yin and yang and the five elements, spring is mild, summer is hot, autumn is cool and winter is bitingly cold, which is the order of the four season. 16 The six natural factors of the climate are wind, damp, cold, heat, dryness and fire, and normal climate change favours the growth of all things. However, when the seasons change, if these six factors occur too much or too little, in discordance with the rules of nature, or if they change rapidly beyond the body’s capacity to adapt, human diseases occur. 17 In other situations, people can become ill, even under normal climate change, because of their poor adaptability. In these two situations, the six natural factors are called ‘the six evils; and exist in normal or abnormal climate change. The exogenous six evils generate the five evils in the body, causing a disorder in the internal yin and yang conversion and the imbalance of Zang Fu (viscera), 18 and producing internal wind, damp, cold, heat and dryness. Therefore, the body injury during the change of the four seasons can lead to different diseases. Exposure to the cold is known as the most evil spirit. In some situations, people immediately develop a disease, but in other situations, the cold will be stored and hidden in the skin, becomes deeper day by day, and will not disappear, even if the patient is exposed to the fever. If the two concepts of cold (exposure to cold and storing cold) are present in the same patient, he will be severely affected or will die (yang–yang symptoms). In the typhoid and miscellaneous diseases theory, 19 it was said that exposure to the cold was considered as a yang disease. Among yang diseases, zhi (spasm) and damp are two symptoms similar to exposure to the cold, but they are not actually the cold exposure disease. Rheumatism in Chinese feng shi is wind and shi (damp) and is considered as the most evil disease in the ‘six evils’. Wind is considered as a yang evil, and the disease is very fast but very easy to treat. However, damp is considered as a yin disease, and the disease is slow and difficult to treat. Sheng Qi Tong Tian Lun of Plain Question said, ‘in the presence of yang Qi, essence raise spirits, and the softness raise the tendons’. In the Nan Jing book, 20 it was said, ‘Qi (yang) warm the body, blood (yin) nourish the body’; when the tendons in the human body cannot get the warmth of yang and the nutrition from yin, the body will succumb to serious illness. In wind-cold-damp evil, there is a reciprocal pacing between the tendons and the muscle, which causes pain in the whole body, especially in a wet climate. The principle of damp syndrome was also discussed in the book Shanghan Lun 21 when describing arthralgia causes, indicating that this disease and the changes in natural conditions are inseparable.
Hence, inspired by this Chinese theory, a rat model of arthritis was established via wind, damp, cold and heat (WDCH) exposure. Body weight, paw swelling, spleen and thymus weight, blood phase change, serum autoantibodies and cytokine expression level and histopathology were evaluated and compared with those of AIA and CIA. The main feature of the new model is that the body catches a disease by stress, simulating a change in the natural environment. The establishment of this new model may have a great significance in the research of the pathogenesis of RA.
Materials and methods
Animals
The study protocol was approved by the Ethics Committee of China Pharmaceutical University (Permit Number: SYXK2012-0035, date of approval: 20 July 2012). The care and treatment of the animals were undertaken in accordance with the Provisions and General Recommendation of Chinese Experimental Animals Administration Legislation. The content and procedure of all animal experiments complied with the relevant laws and regulations, and the use of laboratory animals and management committee of IACUC (institutional animal care and use committee) regulations. There were no alternative methods in the experiments. All experiments were performed in compliance with the Guide for the Care and Use of Laboratory Animals. Adult female Sprague Dawley rats between seven and eight weeks old were obtained from the BK Experimental Animal Centre (Shanghai, China) and housed in a climate-controlled environment (22 ± 2℃, 50 ± 10% relative humidity) with 12-hour/12-hour light/dark cycles, six rats per cage, and supplied with adequate rat chow and water. Rats were randomly divided into five groups: the control group, the AIA group; the type II CIA group; the wind-damp-cold-heat-induced arthritis (WDCH) group; and the one week without WDCH stress (1W) group. Rats were euthanized with carbon dioxide at a flow rate of 10 L/min for three minutes and reduced to 0 for the maintenance phase for five minutes.
Reagents
Complete Freund’s adjuvant (heat-killed Myobacterium tuberculosis H37 RA, 4 mg/mL), incomplete Freund’s adjuvant and bovine type II collagen were obtained from Chondrex, Inc. (Redmond, WA). FITC anti-rat CD3, PE anti-rat CD4, APC anti-rat CD8a and PE anti- Rat CD45RA were obtained from BioLegend, Inc. (San Diego, CA), and RBC lysing solution (10×) was from FcMACS (Nanjing, China). rheumatoid factor (RF), anti-CCP antibody, anti-AFA antibody, anti-Col II antibody, tumour necrosis factor alpha (TNF-α) and interleukin (IL)-6 enzyme-linked immunosorbent assay (ELISA) kit were obtained from Enzyme-Linked Biotechnology (Shanghai, China). Anti-TNF-α monoclonal antibody (mAb) was obtained from Santa Cruz Biotechnology (Santa Cruz, CA), and anti-VEGFR2 mAb was from Abcam (Cambridge, UK).
Establishment of the arthritis model
For AIA, rats were subcutaneously injected in the hind paw palms with 0.1 mL of complete Freund’s adjuvant to induce arthritis for 21 days. 22 For CIA, collagen was dissolved at 2 mg/mL in 0.1 M acetic acid by gently stirring overnight at 4℃, and then an equal volume of complete Freund’s adjuvant was added at low speed in an ice-water bath until a stiff emulsion was generated. Rats were subcutaneously injected with 0.2 mL of the emulsion at the base of the tail, with a booster immunisation after 21 days to induce arthritis for 28 days. For WDCH, rats's hind paws were put into water at a constant temperature of 45 ℃ and 4 ℃ respectively for 10 minutes and then rats were blown with wind at a temperature of 20 ℃ for 10 minutes twice a day for 14 days. For 1 W, rats were treated with WDCH twice a day for 14 days and then observed for 1 W without WDCH stress (Figure 1).
Assessment of arthritis symptoms
Body weight, swelling of the hind paws and arthritis scores were measured every three days. Swelling of the hind paws was evaluated with a paw-volume measuring instrument (SANS Biotechnology, Nanjing, China) every three days. The arthritis scores were: 0, no arthritis symptom; 1, several toes are red and swollen; 2, most of the toes and soles are swollen; 3, ankle joints are swollen; 4, the whole paws are swollen and rats are hard to move.
Blood phase analysis in peripheral blood
Peripheral blood (20 µL) was collected from rats using EDTA-K2 embedded tubes and underwent a routine examination using a BC5390 analyser.
Autoantibodies and cytokine detection in blood serum
Peripheral blood (1 mL) was collected from rats, and serum was obtained by centrifugation at 3500 g for 10 minutes. Autoantibodies (RF, anti-CCP antibody, anti-AFA antibody, anti-Col II antibody) and cytokines (TNF-α and IL-6) were detected by double antibody sandwich ELISA kit.
CD4+ T-cell and CD8+ T-cell substrate and B-cell analysis in peripheral blood
Peripheral blood was collected from rats using EDTA-K2 embedded tubes and divided into two tubes, 100 µL of peripheral blood per tube. To one tube, 0.25 µg of FITC anti-rat CD3, PE anti-rat CD4 and APC anti-rat CD8a was added, and to the other tube, 0.25 µg of FITC anti-rat CD3, PE anti-rat CD45RA was added followed by incubation at room temperature for 20–30 minutes in the dark. After that, 2 mL of red blood cell lysate was added to the above tubes followed by incubation at room temperature for 15 minutes in the dark until the solution was clear and transparent. Phosphate-buffered saline (PBS; 2 mL) was used to rinse the cells, which then were suspended in 0.5 mL of PBS. CD4+ and CD8+ T-cell substrates and B cells were analysed by flow cytometry (MACSQuant™; Miltenyi Biotec, Bergisch Gladbach, Germany).
Histopathological analysis
The left hind paws were collected, fixed in 10% formaldehyde and decalcified with 5.0% ethylenediaminetetraacetic acid (EDTA) in 10% formaldehyde, which was changed twice a week for eight weeks at room temperature. The tissues were embedded in paraffin, and serial sections (5 µm) were cut, mounted on glass slides and stained with hematoxylin and eosin (H&E) for examination under a microscope. A histological assessment of arthritis severity was made by evaluation of ankle joint cavity in accordance with a scoring system. Synovial hyperplasia was scored as follows: 0 = normal, 1 = mild hyperplasia of the synovial lining layer and 2–4 = synovial lining hyperplasia and pannus formation. Inflammation was scored as follows: 0 = normal, 1=mild inflammation (one aggregate or little leucocyte infiltration), 2=moderate inflammation (≥2 leucocyte aggregates) and 3=marked inflammation (large leucocyte aggregates and significant leucocyte infiltration). Bone erosion was scored as follows: 0=normal; 1=minimal (one or two small, shallow sites), 2=mild (between one and four sites of medium size and depth), 3=moderate (five sites partially extending through the cortical bone), 4=marked (multiple foci partially or completely extending through the cortical bone) and 5=extensive (cortical penetration at 25% of bone length).
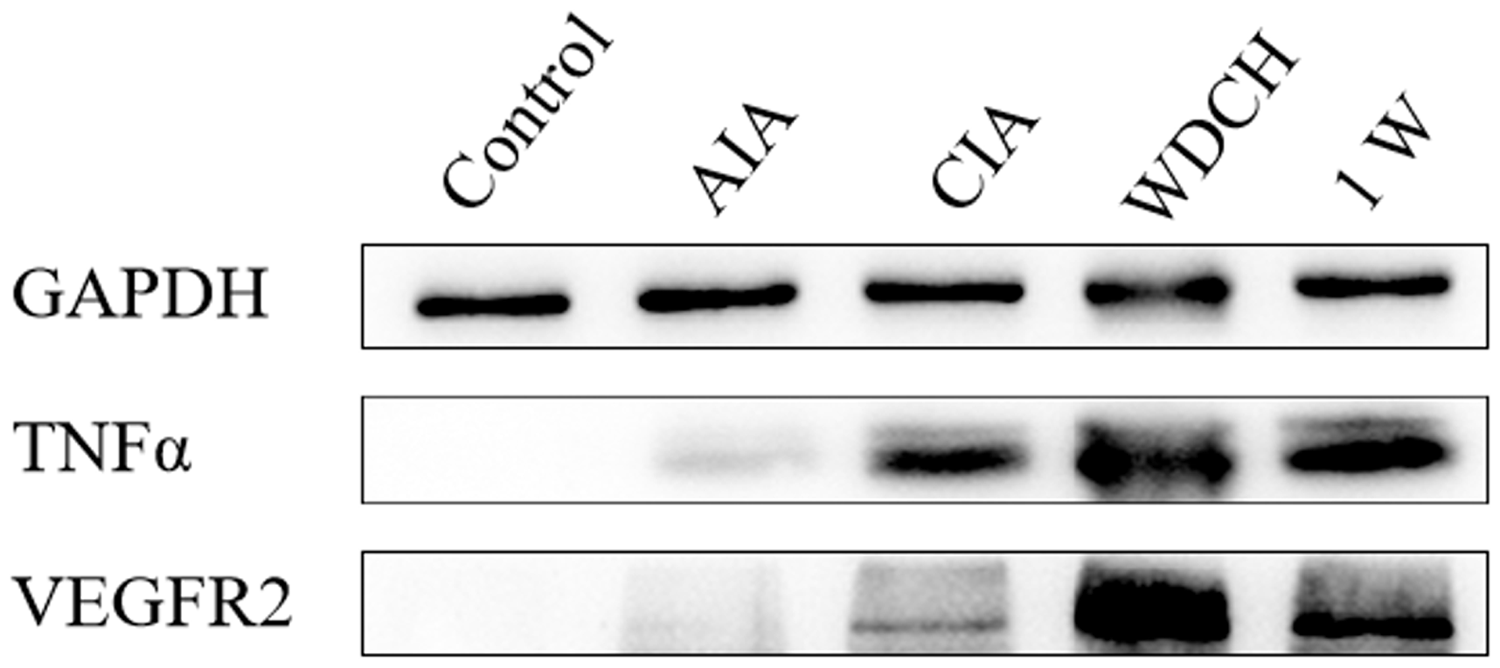
TNF-α and VEGFR2 expression in synovium by Western blotting
Synovium was extracted from ankle joint and lysed for 30 minutes in ice-cold lysis buffer (Wanleibio, Shengyang, PR China). Protein was obtained after centrifugation at 14,000 g for 10 minutes, mixed with 5 × loading buffer, placed into boiling water bath, loaded on 12% sodium dodecyl sulphate polyacrylamide gel, transferred to polyvinylidene difluoride membranes (Millipore, Billerica, MA) and blotted with anti-TNF-α mAb and anti-VEGFR2 mAb.
Statistical analysis
The data were analysed using IBM SPSS Statistics for Windows v19.0 (IBM Corp., Armonk, NY) and GraphPad v5.01 (GraphPad Software, Inc., La Jolla, CA) and are presented as means ± SD. The significance of any differences was determined using a two-tailed Student’s t-test, and was set at p < 0.05 or p < 0.01.
Results
Conspicuous arthritis symptom in the WDCH group
Body weight, paw swelling and arthritis scores were measured every three days for each group. The body weight of the WDCH group gradually decreased, despite a slight increase without stress (Figure 2(a)). Accordingly, paw swelling and arthritis scores gradually increased, despite a slight decrease without stress (Figure 2(b)–(d)).
A diagram of the experimental design. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress.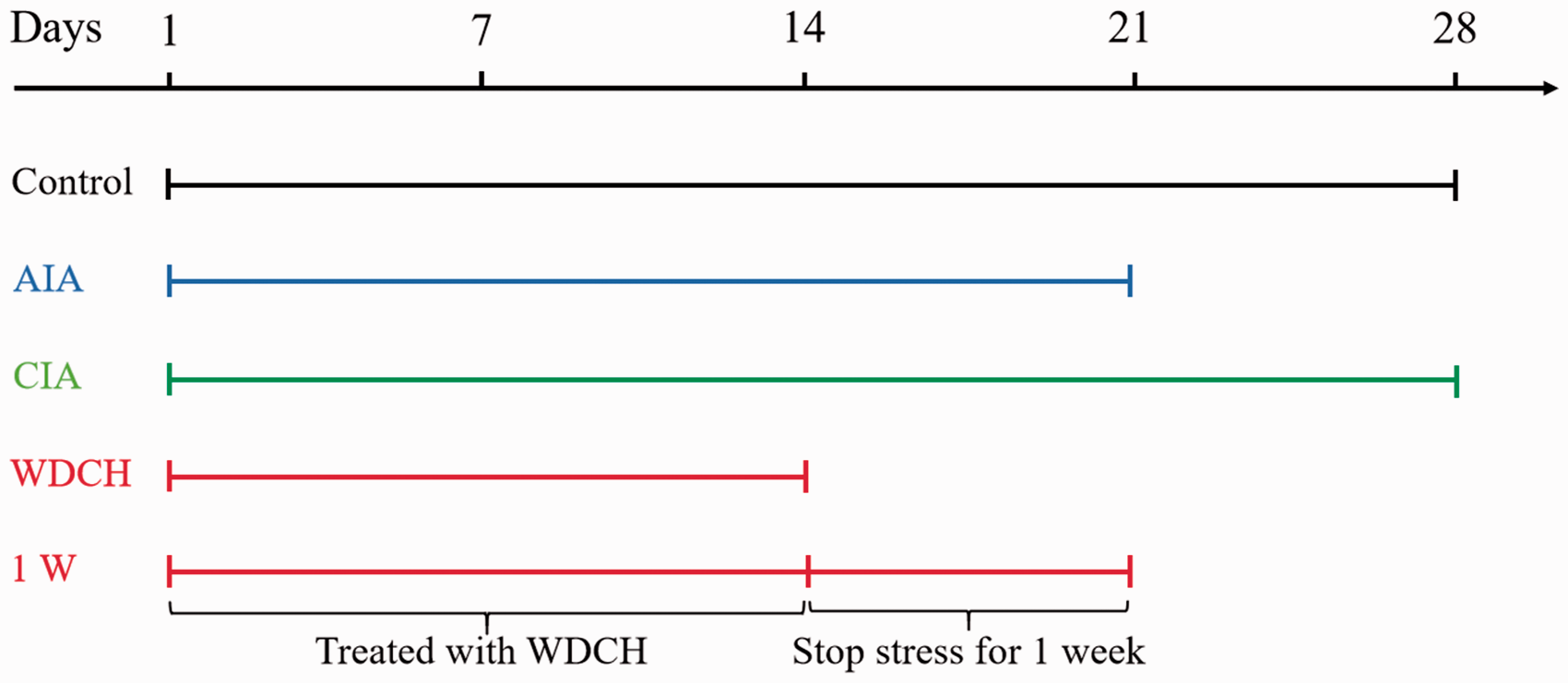
Significant rheumatoid changes induced by WDCH
Patients with RA often have elevated neutrophils, decreased red blood cells and haemoglobin, showing anaemia. The number of neutrophils increased significantly, and the number of red blood cells and the concentration of haemoglobin decreased significantly (Figure 3(a)–(c)), consistent with clinical RA symptoms.
Body weight, swelling of left and right hind paws and arthritis score. (a) body weight. (b) Joint scores. (c) Swelling of left hind paws. (d) Swelling of right hind paws. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. *p < 0.05; **p < 0.01 vs. control.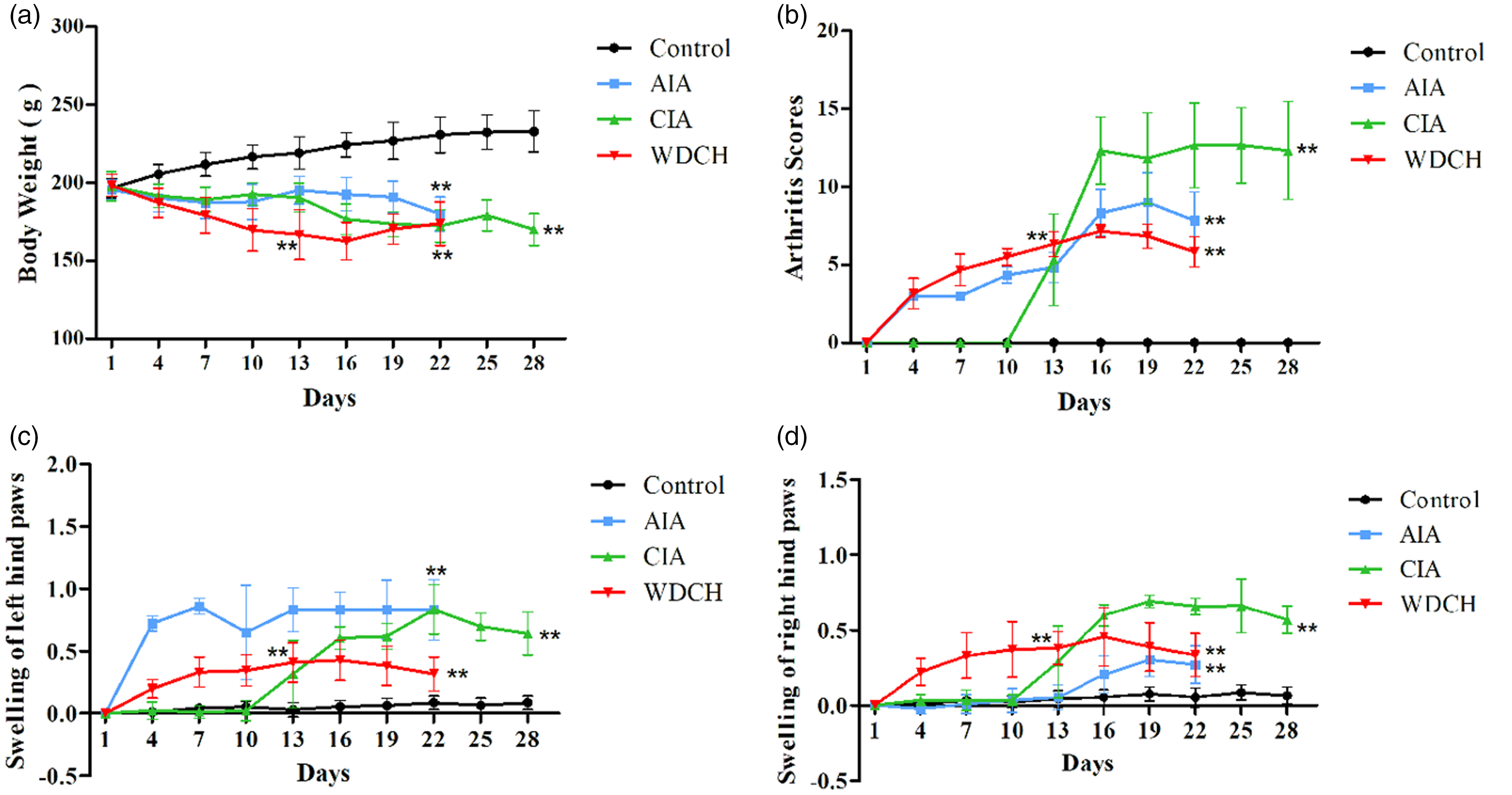
Autoantibodies, RF, anti-CCP antibody, anti-AFA antibody and anti-Col II antibody are important indicators for the diagnosis of RA. RF is one of the earliest indicators for the diagnosis of RA, and anti-CPP antibody is a highly specific indicator of RA. The above four autoantibodies were significantly elevated in the WDCH group (Figure 4(a)–(d)), which means that rats succumbed to RA.
Blood cells analysis of rats. (a) Neutrophil granulocytes. (b) Red blood cells. (c) Haemoglobin. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. *p < 0.05; **p < 0.01 vs. control. Expression level of autoantibodies and cytokines in peripheral blood. (a) Rheumatoid factor (RF) expression level. (b) Anti-CPP antibody expression level. (c) Anti-AFA antibody expression level. (d) Anti-Col II antibody expression level. (e) Tumour necrosis factor alpha (TNF-α) expression level. (f) Interleukin (IL)-6 expression level; Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. *p < 0.05; **p < 0.01 vs. control.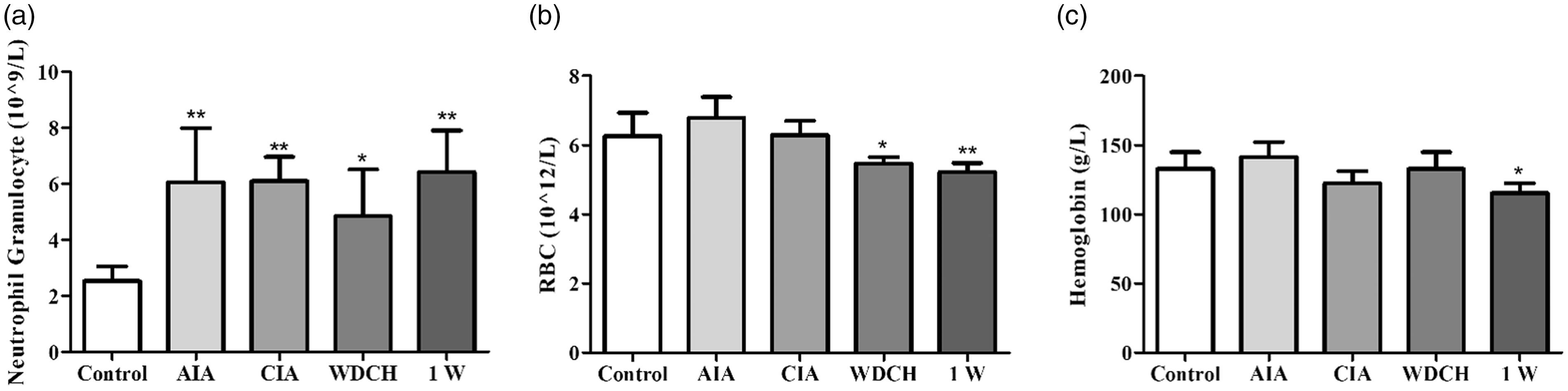
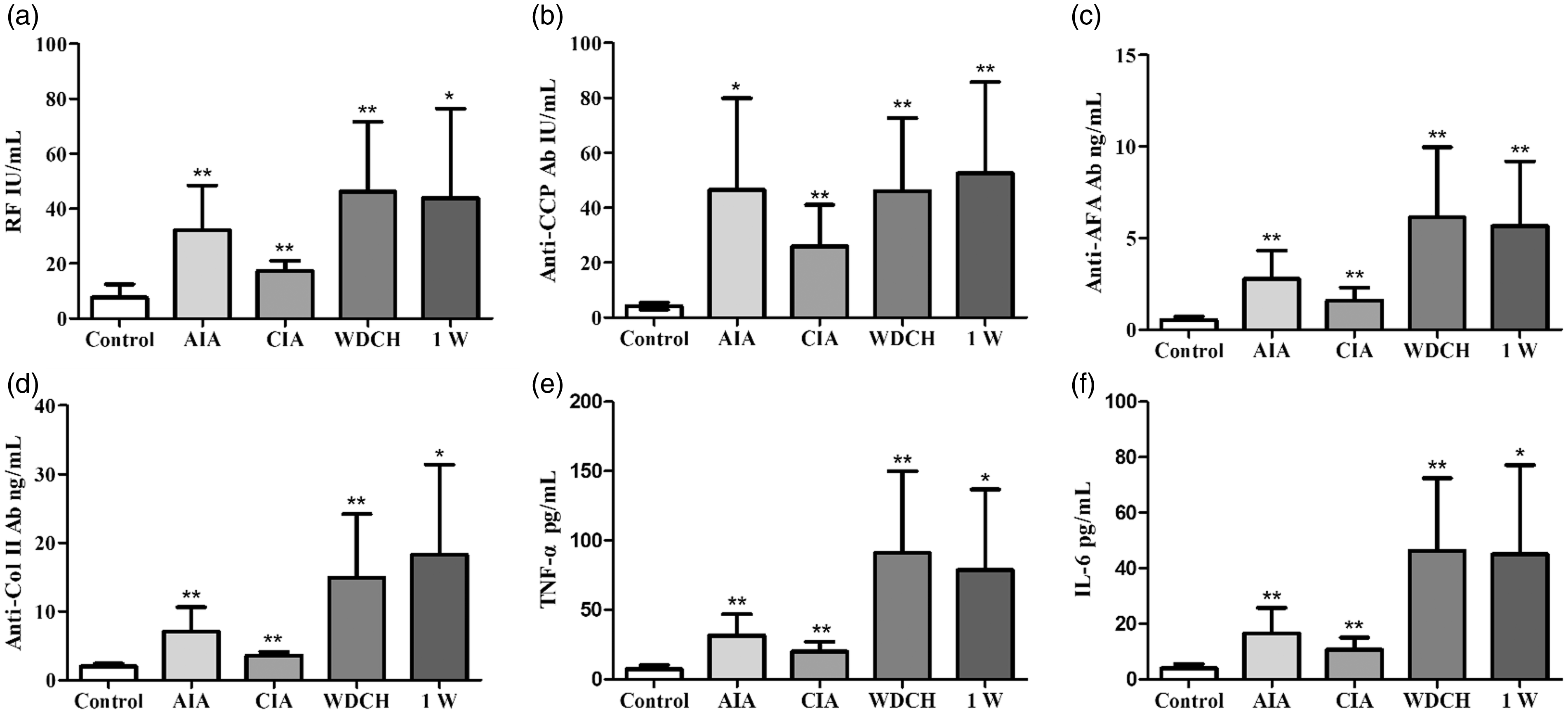
TNF-α and IL-6 are two important inflammatory cytokines. Their extremely elevated expression level indicated that the body was in a state of inflammation (Figure 4(e) and (f)).
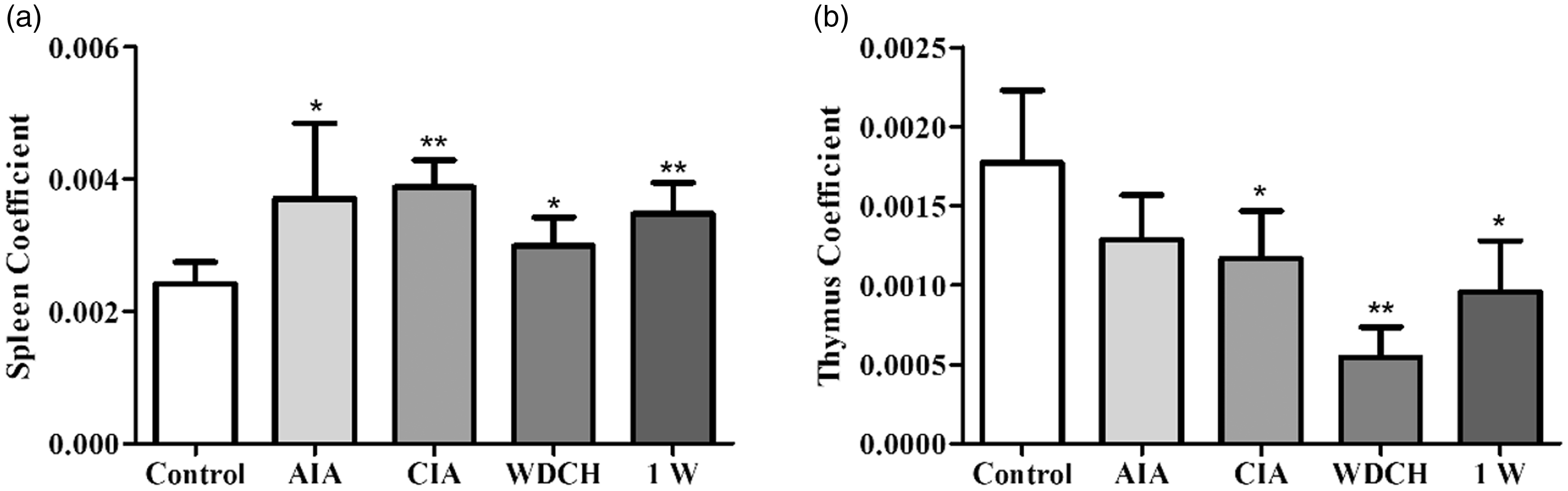
Compared with normal people, patients with RA have a significant increase in CD4+/CD8+ T-cell ratio in the peripheral blood. In accordance with that, the CD4+/CD8+ T-cell ratio was significantly increased in the WDCH group compared with the control group (Figure 5(a)). B cells, secreting antibodies and primarily mediating humoral immunity were significantly increased in the WDCH group compared with the control group (Figure 5(b)). The change in T and B cells displayed an immune imbalance in the rats, which was consistent with the result that the spleen and thymus weight to the body weight of rats in the WDCH group were different from those in the control group (Figure 6(a) and (b)).
Analysis of CD4+/CD8+ T and B cells in peripheral blood. (a) CD4+/CD8+ T-cell ratio. (b) B-cell percentage. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. *p < 0.05; **p < 0.01 vs. control. Spleen and thymus coefficients. (a) Spleen coefficient (b) thymus coefficient (spleen weight and thymus weight respective to body weight of rats). Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. *p < 0.05; **p < 0.01 vs. control.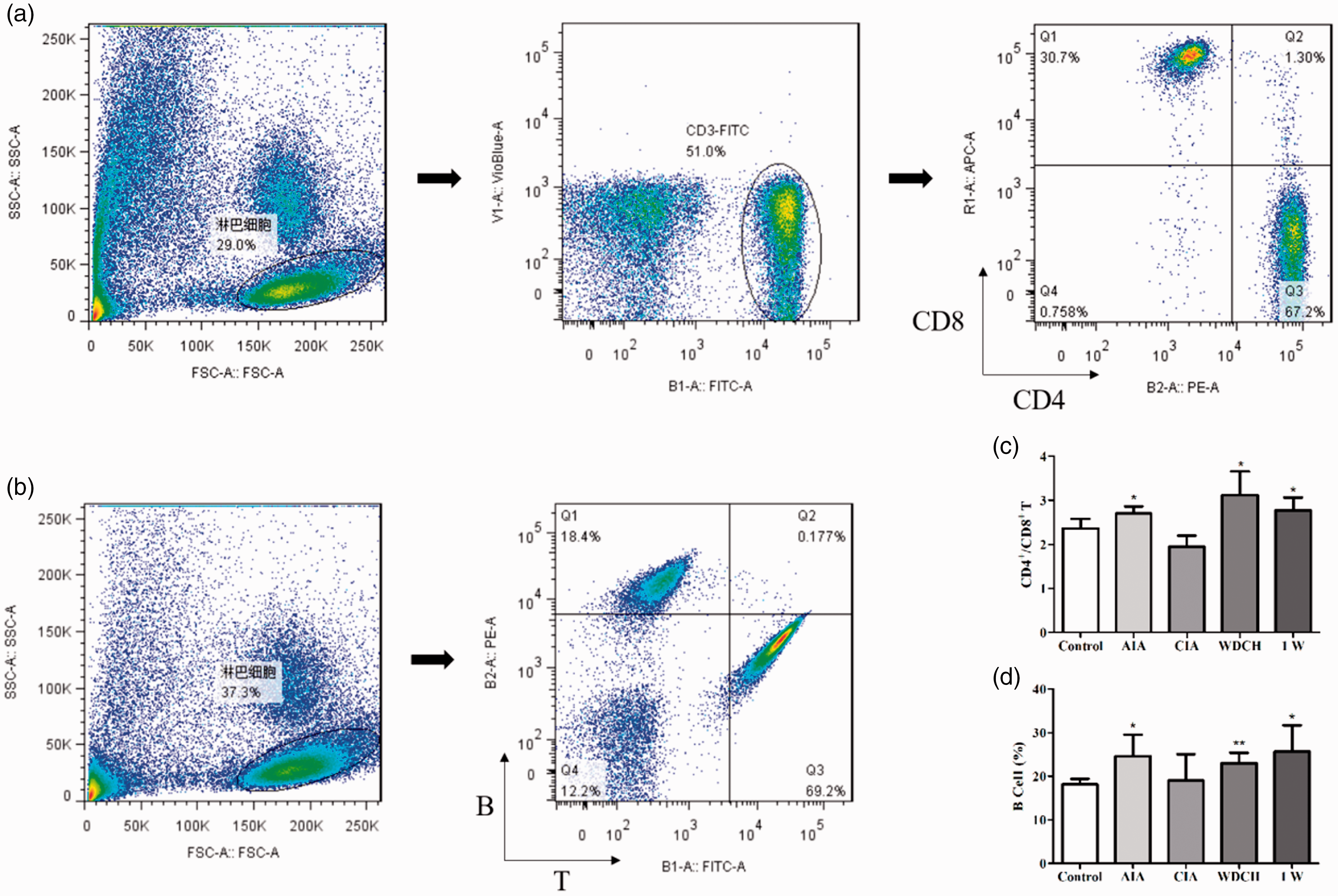
Joint lesion induced by WDCH
There was obvious synovial hyperplasia, inflammatory cell infiltration and pannus and bone erosion in the ankle joint of rats in the WDCH group with H&E staining (Figure 7), which indicated that joint lesions existed in rats induced by WDCH and which did not disappear, even without stress. The TNF-α expression level indicates inflammation in the synovium. VEGFR2 regulates lymphangiogenesis and angiogenesis, and promotes pannus formation. Inflammatory cytokine TNF-α and VEGFR2 expression was extremely elevated in the synovium (Figure 8), indicating the occurrence of inflammation and angiogenesis in the joint.
Hematoxylin and eosin staining of rats’ ankle joint. (a) Typical photo of hematoxylin and eosin staining (100 µm; synovial hyperplasia was annotated with pink quadrangular stars, pannus with red arrows, bone erosion with blue triangular stars and inflammation with black arrows). (b) Statistical analysis of synovial hyperplasia, pannus, bone erosion and inflammation. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress. Expression of TNF-α and VEGFR2 in the synovium by Western blotting. Control: normal rats untreated; AIA: adjuvant-induced arthritis; CIA: type II collagen-induced arthritis; WDCH: wind-damp-cold-heat induced arthritis; 1W: one week without WDCH stress.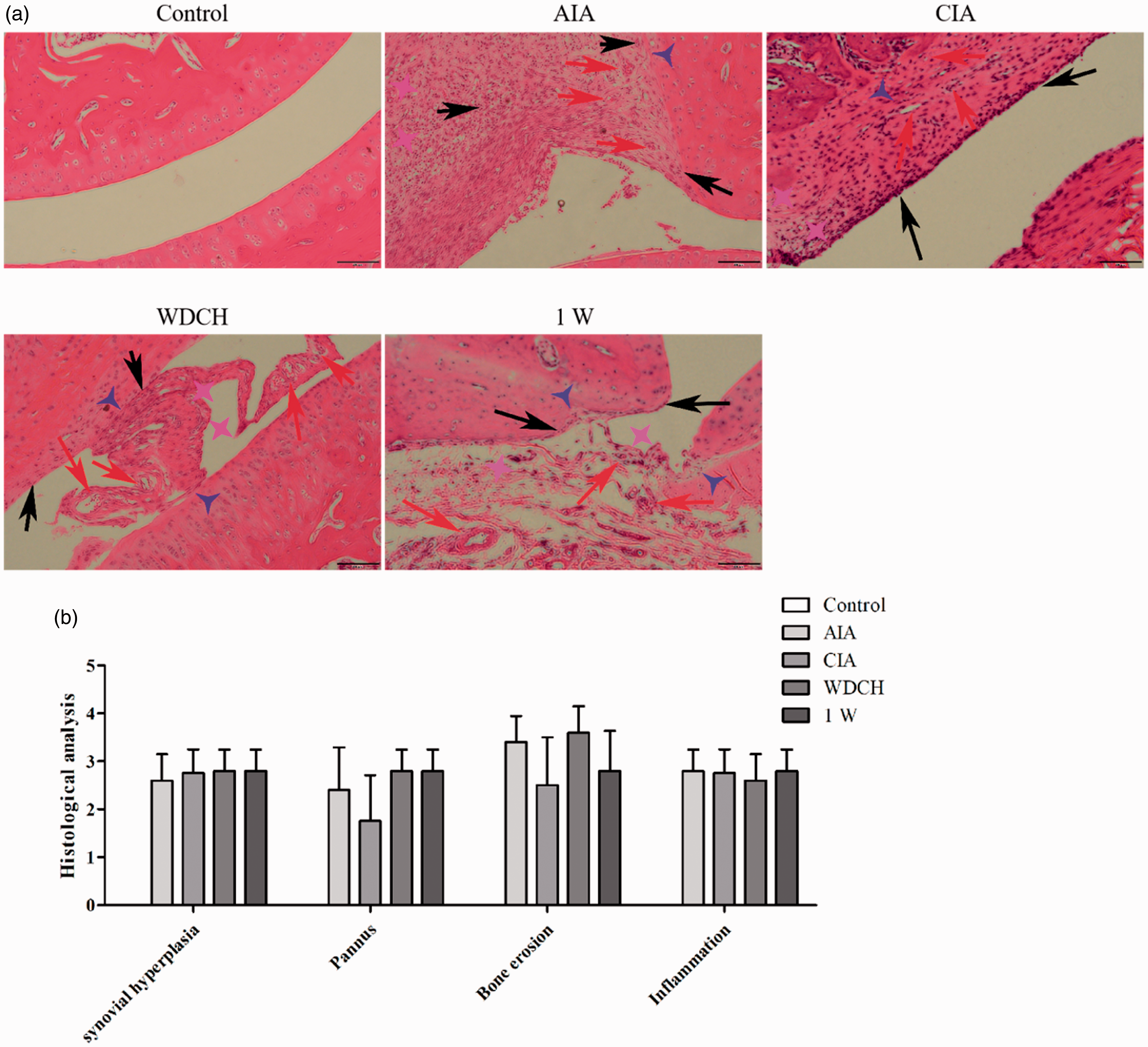
Discussion
RA is an autoimmune disease that does not have clear diagnostic criteria, and doctors usually need to refer to multiple indicators to make their diagnosis. Therefore, the evaluation of the established model was based on the morphological changes, haematological index and cytokine expression level. In this experiment, the body weight of rats in the AIA group, type II CIA group and WDCH group were significantly lower, and the swelling volume was significantly higher than those of the control group, indicating that the body had undergone illness. 23 A decrease in rat haemoglobin and number of RBC indicated slight anaemia, 24 and the number of neutrophils was significantly increased and the expression level of autoantibodies and inflammatory cytokines was significantly elevated, which is in accordance with the results of blood tests of patients with RA. 25 In addition, the CD4+/CD8+ T-cell ratio in the WDCH group was elevated, probably due to the immune dysfunction caused by the environmental stimuli. 26 Compared with the control group, there was obvious synovial hyperplasia, inflammatory cell infiltration and pannus and bone erosion in the ankle joint of rats in the arthritis group. Furthermore, Western blotting showed an elevated expression level of TNF-α and VEGFR2 in the synovium of rats in the arthritis group, indicating the occurrence of inflammation and angiogenesis in the joints.
The interaction between the environment and the body is like doing T’ai chi: ‘you are strong, I am weak; you are weak, I am strong’. When the environment changes suddenly, and the body cannot tolerate these changes, a disorder will occur in the body’s internal system, and a disease will appear. When the environment is not aberrantly changed and the resistance of the body itself is decreased, a disease could also appear. Traditional Chinese Medicine has been prevalent in the treatment of arthritis. Thousands of years ago, the treatment of arthritis and cold attacks was mentioned in the Guizhi Tang Fang and Synopsis Golden Grades, the trichosanthes Guizhi decoction, Dachengqi decoction and Ephedra soup, 27 but their mechanism of action is not clear. The treatment target and the effect on signalling pathways are unclear, although the disease is cured because Chinese medicines are mixed compounds. The bioactive molecule in traditional Chinese decoctions is not recognised and characterised, which impedes the expansion of the medicine in the world. At present, Chinese medicine theories can help in the study of disease pathogenesis.
To develop an effective anti-rheumatic treatment, it is very important to clarify the pathogenesis mechanism. This article revealed that the changes in WDCH could be key factors in RA pathogenesis. Therefore, it is highly recommended that resistance to the change in the environment and improvement in body immunity might help people to reduce the risks of RA. Only female rats were used in this study, considering that females have a higher incidence than males clinically. 28 This new model needs to be tested on male rats in further research. Arthritis mainly includes osteoarthritis and RA, which both have an unknown pathogenesis. Osteoarthritis is divided into primary osteoarthritis and secondary osteoarthritis; the causes may be related to age, sex, obesity, occupational overuse and other factors. 29 RA includes normal arthritis, autoimmune RA and ankylosing spondylitis (AS). 30 The clinical manifestation of these pathologies is mainly progressive joint pain, tenderness, stiffness, joint swelling, restricted activity and joint deformation. In addition, they have almost the same parameters of diagnosis, such as immune cell number change, autoantibodies and inflammatory cytokine change in serum, X-ray, computed tomography, magnetic resonance imaging osteoporosis and joint deformation. We hope to continue exploring the established model with biomarkers to provide inspiration for RA treatment.
Research Data
Research Data for A novel arthritis model induced by wind, damp, cold and heat in female rats
Research Data for A novel arthritis model induced by wind, damp, cold and heat in female rats by Chuang Ge, Jingchao Hao, Xiaodong Wu, Chencheng Li, Rui Zhi, Pengcheng Yu, Xinmin Wang, Jialiang Hu and Hanmei Xu: For the WRIST Group in Laboratory Animals
Footnotes
Acknowledgements
We thank Jiangsu Province Hospital on integration of Chinese and Western Medicine, Nanjing, China, for assistance with this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Project Program of State Key Laboratory of Natural Medicines (No. SKLNMZZCX201821) and the National Science and Technology Major Projects of New Drugs (2018ZX09301053-001, 2018 ZX09301039-002,2018ZX09201001-004-001) in China. This project was also funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD) and Natural Science Foundation of Jiangsu Province (BK20160757) and by ‘Double First-Class’ University project of China Pharmaceutical University (NO. CPU2018GY13 and CPU2018PZH03).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
