Abstract
Hypothermia is a treatment strategy for different clinical conditions and an essential part of cardiopulmonary bypass in complex cardiac procedures. Clinically, cooling patients is achieved via a mattress and heat exchanger integrated into a membrane oxygenator connected to a waterbed using a refrigerator system based on volatile and toxic liquids. Peltier elements are known as environmentally friendly thermoelectric generators that enable rapid warming and cooling. In this paper, we describe the construction of a novel device for rapid and precise control of mouse warming and cooling using thermoelectric Peltier elements. Six male BALB/c mice were subjected to deep hypothermia and were rewarmed under full physiological monitoring. After rewarming, all animals were observed for two hours, and pathology was evaluated in several organs. All animals tolerated the rapid cooling process well and remained active after rewarming. Temperature-relevant changes were seen via electrocardiography, with heart-rate patterns showing a strong linear correlation to body temperature. No myocardial ischaemia was seen. However, two animals experienced bradycardic atrial fibrillation which spontaneously converted to normal sinus rhythm during rewarming. No histological damage was seen in the heart, liver, kidney or lungs. Our device can effectively be used for heat shock and hypothermia studies in mice, and we foresee no obstacles for its application to other small rodents such as hamsters and young rats. In comparison to known experimental and clinical methods of hypothermia, our device is environmentally friendly, cost-effective and easy to handle, allowing precise control and maintenance of body temperatures ranging from 18℃ to 42℃.
Introduction
Small-animal surgical research is important for understanding various pathophysiological processes and for the development of innovative surgical procedures. During experimental surgery, animals are prone to hypothermia in both thoracic and abdominal procedures due to their high surface area to mass ratio.1,2 Hypothermia may affect various physiological processes such as blood clotting,3,4 haemodynamics5,6 and pharmacological availability of various substances.7,8 Therefore, intraoperative temperature management of the animal plays a pivotal role in postoperative outcome. 9 On the other hand, certain small-animal surgeries require, test or model therapeutic hypothermia (TH; also known as targeted temperature management or protective hypothermia). TH is used clinically in cardiac surgery and is a neuroprotective strategy with many clinical indications, including the treatment of cardiac arrest and stroke survivors, traumatic head injuries, newborn hypoxic–ischaemic encephalopathy and neurosurgery, and has been applied in cases of acute respiratory distress syndrome (ARDS). 10 During human procedures, cooling is achieved via a mattress and heat exchanger integrated into the membrane oxygenator connected to a waterbed. Such systems use chlorofluorocarbons, also known as ‘vapour’ or R-12 in the refrigerator manufacturing industry. These substances are classified as volatile, toxic and ozone-depleting, and most of them are now forbidden or severely regulated since the adoption of the Montreal Protocol in 1987.
We recently developed a model of cardiopulmonary bypass (CPB) in mice whereby moderate hypothermia was achieved by topically cooling the animals with gauze soaked in a cold saline solution. 11 This commonly used method lacks precise temperature control. To optimise experimental CPB procedures with extracorporeal circulation and hypothermic phases in small animals, precise temperature modulation is needed. The use of clinically used cooling systems in this context is limited in its application due to its size, costs and complicated maintenance. Therefore, an environmentally friendly, cost-effective and easy-to-handle device for temperature management in small animals is needed. In this paper, we describe the construction of a novel device for rapid and precise control of mouse warming and cooling using thermoelectric Peltier elements.
Materials and methods
Animals
Six male BALB/c mice were obtained from Charles River. Animals weighing between 25 and 35 g were utilised for the experiments. This study was conducted in compliance with the German Animal Protection Law. The animal protection committee of the local authorities (Lower Saxony State Department for Food Safety and Animal Welfare (LAVES)) approved all experiments (Approval: 33.12-42502-04-14/1556).
Device construction
A square aluminium plate (20 cm × 30 cm × 0.7 mm) was used as the main board for cooling and warming the animal. A 12 V/60 W thermoelectric Peltier cooler/heater (TEC1-12706; Hebei IT Co. Ltd) was glued to the backside of the plate using thermic glue (Thermal Adhesive; MG Chemicals). Adjacent to the thermal element, seven copper plates equal in size to the aluminium board were adhered. The copper plates were joined together using small round-shaped aluminium pads (30 mm × 1 mm) that allowed enough space between the metal plates for proper heat dissipation. Usually, as one side of the Peltier element heats, the other side cools and vice versa. Therefore, it was important to avoid backside overheating of the thermal element during cooling of the animal. Therefore, to increase heat dissipation further, all copper plates were connected to an aluminium heat sink (Xilence; AM2.HP) using thermic glue and were ventilated by three cooling fans. This allowed for effective and stable deep cooling of the main aluminium board for several hours. For temperature regulation, an NTC 1 K temperature sensor (Vishay BC Components, Vishay Intertechnology, Inc.) was connected between a temperature controller (ITC-1000; Inkbird Tech. Co. Ltd) and the aluminium board. A second temperature sensor was coated with a polymerised resin and connected to a second temperature controller (ITC-1000; Inkbird Tech. Co. Ltd). This was used as a rectal probe. A reversible switch was placed between the power supply and the Peltier element for convenient switching from cooling to heating. For an illustration of the heating/cooling pad, see Figure 1. For detailed specifications of device components, see Supplemental Table S1.
Schematic design of (a) the heating/cooling small-animal pad and (b) the final device.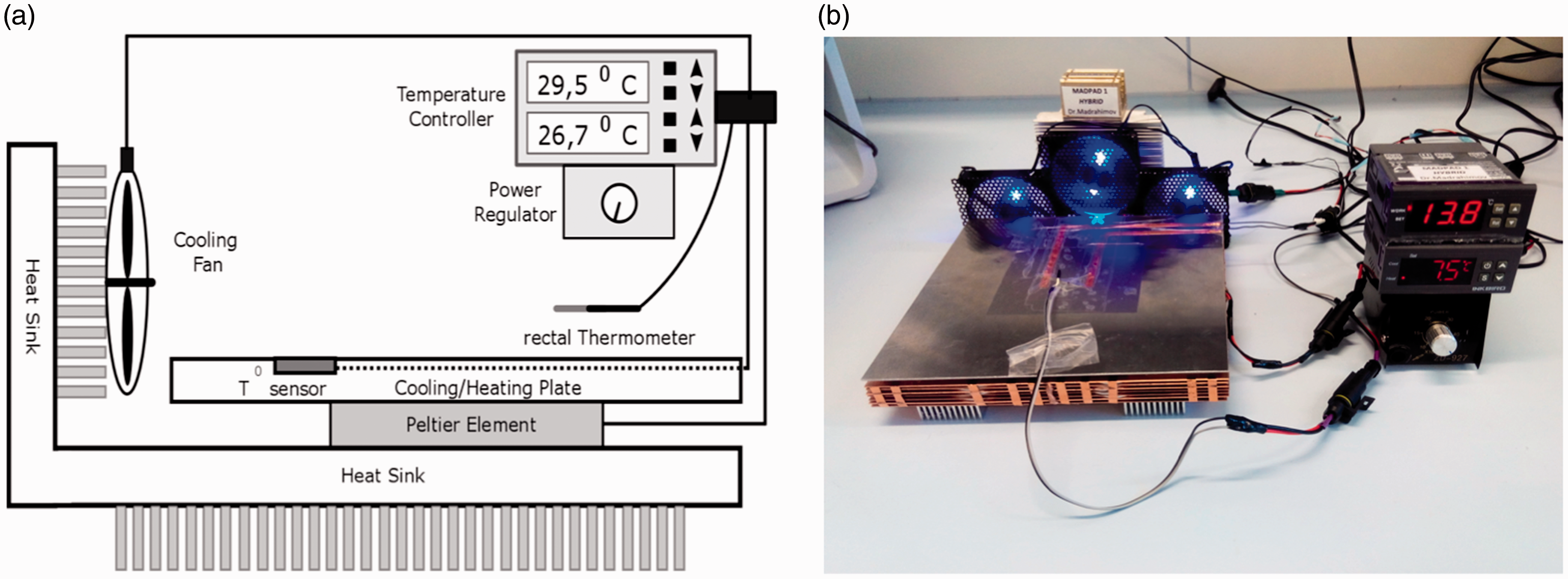
Anaesthesia
Animals were anaesthetised with isoflurane in an induction chamber. After successful anaesthesia was achieved, animals were placed onto the thermal plate. Further maintenance of anaesthesia throughout the experiment was achieved using mask inhalation of vaporised isoflurane under control of spontaneous breathing. The concentration of isoflurane anaesthesia was maintained between 1.3% and 2.5% in oxygen, depending on the stage of the procedure, with a constant flow rate of 0.5 L/min.
Experimental set-up
After induction of anaesthesia, animals were placed on the thermal plate and initially kept at normothermia. Electrocardiography (ECG), rectal temperature and invasive pressure were monitored using a Multichannel Data Acquisition System (Hugo Sachs Elektronik GmbH). ECG needle electrodes were placed subcutaneously in the right and left forelimb and in the left thoracic wall. Afterwards, a 5 mm incision was made in the left inguinal area, and the femoral artery was separated from the femoral nerve and vein in a sharp and blunt manner under a microscope at 16–25 × magnification. The distal part of the artery was ligated, and the proximal part was temporarily ligated using a slip knot with an 8-0 silk suture. After incision of the anterior wall of the artery, a 1 Fr polyurethane cannula (Instechlabs, Inc.) was inserted and moved towards the iliac artery. Afterwards, the invasive pressure tubing was then connected to a pressure transducer. Visualisation of the surgical set-up is displayed in Figure 2. Cooling of the animal was initiated by setting the heating/cooling pad to 15℃. Under stable haemodynamic conditions, the temperature of the animal was lowered to deep hypothermia (down to 20℃). This temperature was kept for a further 10 minutes. Next, the animal was rewarmed to normal core body temperature. During rewarming, the difference between the plate and rectal temperature was kept <6–8℃ (similar to the clinical guidelines of <10℃ for CPB) to avoid air micro-embolisation of the animal.
12
After the animal regained normal body temperature with stable haemodynamic conditions, anaesthesia was terminated, and the animal was allowed to wake up.
Surgical set-up using real-time electrocardiography, invasive arterial pressure and temperature control.
Clinical observation
Postoperative observation included assessment of the behaviour, activity and examination of the body of the animal for two hours after waking up. 13
Organ harvesting and histology
Two hours after waking up, all animals were subjected to isoflurane euthanasia followed by exsanguination and organ harvesting. To assess potential organ damage, the heart, lung, liver and kidneys were harvested for routine histology. All organs were fixed in 4% formalin and were subjected to paraffinisation and hematoxylin and eosin (H&E) staining. Organs were evaluated for signs of acute cold or heat damage in the form of cell death, inflammatory cell infiltration, thrombosis or oedema.
Statistical analysis
All data obtained using ISOHEART Software (Harvard Apparatus) were analysed using GraphPad Prism 5.0 (GraphPad Software, Inc.), and the correlation between parameters was calculated after Pearson analysis.
Results
Animal rectal temperature and plate temperature
During the experiments, the temperature of both the animal and the thermal pad was monitored continuously in real time (Figure 3). The average initial animal temperature was 35.52 ± 0.51℃ and fell to 20.35 ± 1.05℃ within 40 minutes of the initiation of cooling. The maximal cooling rate was 0.4℃/min. The rewarming of the animal lasted more than 65 minutes, with animals reaching 35.05 ± 0.18℃ during this time. It should be noted that the increase in plate temperature was performed in a stepwise manner, thus keeping the difference in plate and body temperature well below 10℃ (6–8℃). During rapid cooling, the maximal temperature difference was 14.79℃, while during rewarming it did not exceed 6.73℃. Stepwise warming of the animal was easily managed by changing the setting temperature on the controller every 10 minutes.
Animal and pad temperature (±SD) over time during the cooling and rewarming period of the experiment. Animals were rewarmed in a gradual manner to prevent air micro-embolisation.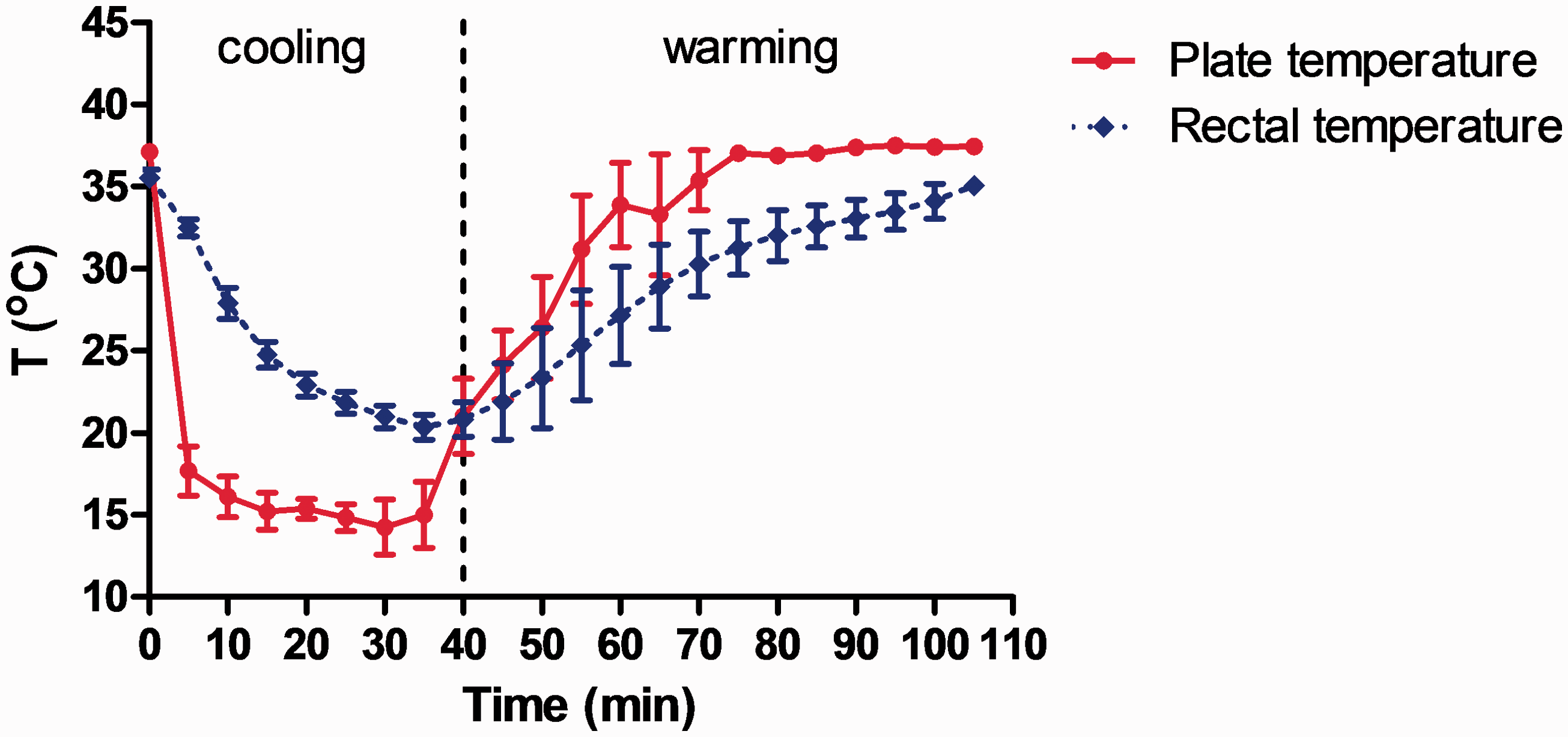
Haemodynamic monitoring
Haemodynamics were monitored in real time during the course of experiments. At the start of the experiment, the average heart rate was 612.83 ± 89.24 bpm and dropped during deep hypothermia to 232.33 ± 33.69 bpm (Figure 4(a)). After rewarming, the average heart rate was 692.33 ± 72.11 bpm (Figure 4(a)). No myocardial ischaemia was seen on ECG during the experiments. Nevertheless, in two of six animals, bradycardic atrial fibrillation was observed at the deepest point of cooling, which spontaneously converted to a normal rhythm after rewarming the animal. Heart-rate patterns showed a strong linear correlation to body temperature by Pearson analysis (r = 0.857, p < 0.0001; Figure 4(b)). Invasive blood-pressure measurements showed a stable mean arterial pressure (MAP) throughout the whole experiment. During normothermic conditions, deepest hypothermia and at the end of the rewarming period, MAP averaged 85.75 ± 3.64, 84.35 ± 2.35 and 85.7 ± 2.86 mmHg, respectively (Figure 4(c)). Figure 4(d) shows the data normalised to initial measurements (time = 0 minutes).
Haemodynamic monitoring during hypothermia and rewarming. Values are expressed as means ± SD. (a) Heart rate over time, (b) the relationship between heart rate and rectal temperature, (c) mean arterial pressure (MAP) and (d) variables normalised to initial measurements (time = 0 min). n = 6. Pearson analysis showed a correlation between heart rate and temperature (r = 0.0857, p < 0.0001).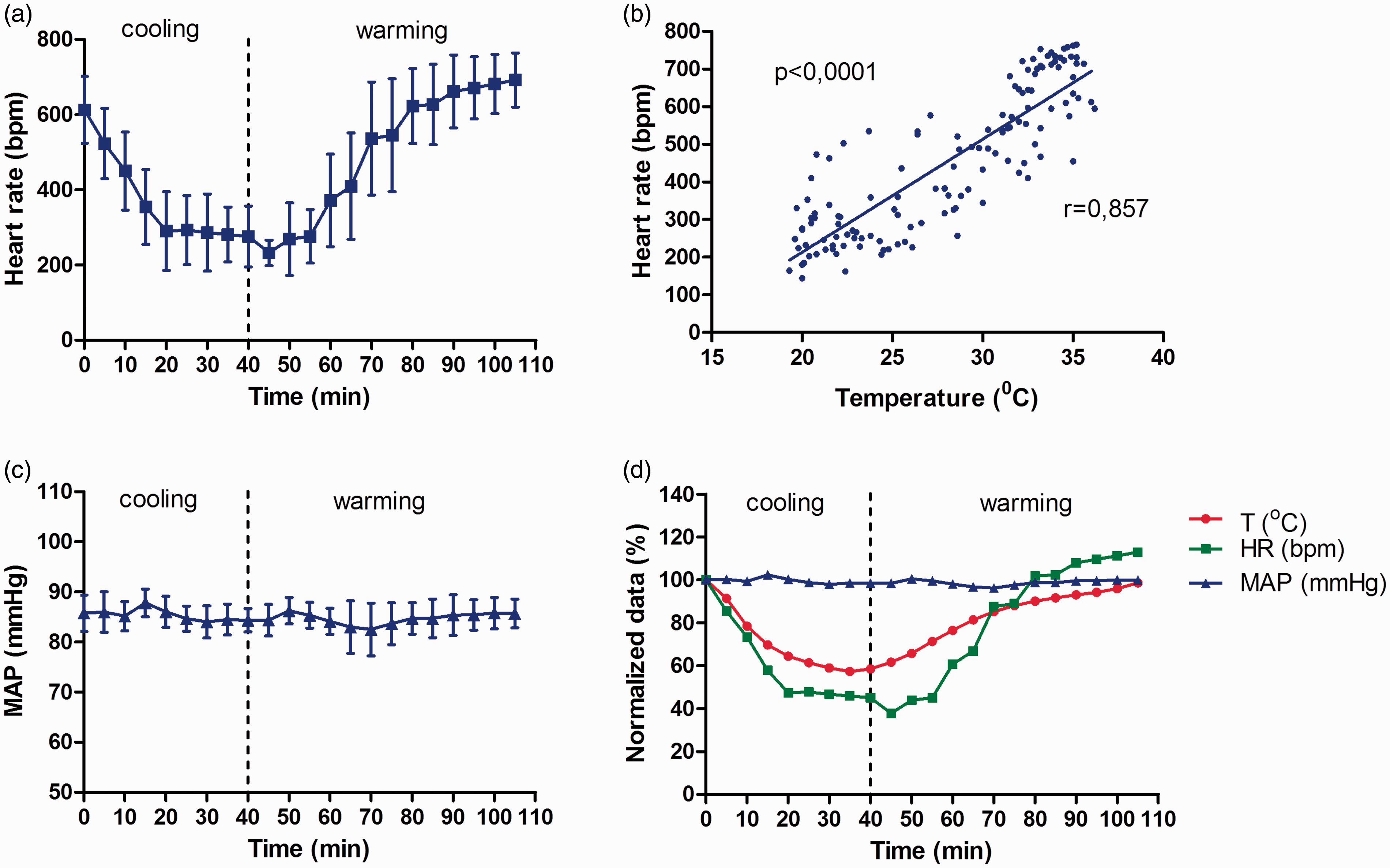
Clinical condition after waking up
All animals fully recovered 30 minutes after narcosis was terminated. No changes in activity, including drinking and eating behaviour, were observed during the two-hour observation time. Upon investigation of the body, special attention was given to the assessment of the backside of the animal that had a large surface contact to the cooling/heating board. No lesions or changes in the fur pattern or skin were detected in any of the animals.
Histological evaluation
Histological investigation of H&E sections revealed no detectable changes to the heart, lung, liver or kidney. There were no signs of organ oedema, necrosis, micro-thrombosis or cellular infiltration which would be expected upon organ damage (Figure 5).
Hematoxylin and eosin staining of (a) murine myocardium, (b) lung tissue, (c) kidney and (d) liver after hypothermia and rewarming. 200 × magnification.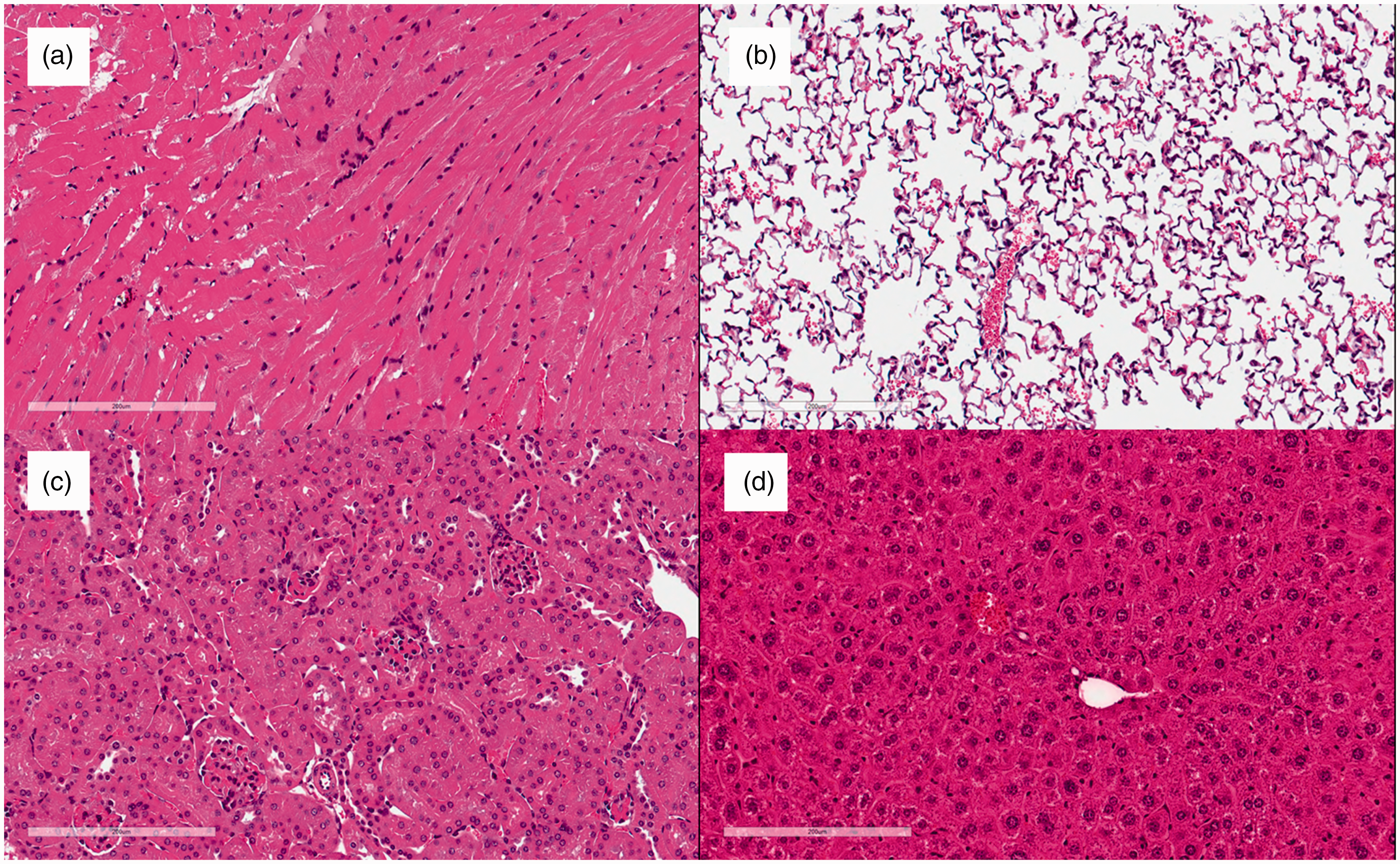
Discussion
We have developed and tested a novel temperature-controlled heating and cooling plate for preclinical murine research. It allows for precise thermoregulation and was proven safe for animals undergoing deep hypothermia and rewarming. Tight control of the heating/cooling plate is crucial, as seen by Roehl et al. 14 who observed renal injury in rats undergoing surgery due to accidental overheating of a thermal heating plate. Although only tested on male BALB/c mice, we foresee no obstacles for its application to other strains of mice, female mice or other small rodents such as hamsters and young rats. In addition, the plate could easily be made bigger to accommodate larger rodents such as adult rats and rabbits.
To date, methods to achieve small-animal hypothermia have required surgery or intervention
15
(Figure 6), and in all cases, precise thermoregulation was not possible. Furthermore, hypothermia requiring surgical intervention makes it less suitable for immunological research, as any kind of surgical procedure initiates cytokine production and possibly a humoral response.16,17 In the study by Leker et al.,
18
the use of cannabinoid agonist HU-210 to induce hypothermia showed significant side effects. There are also safety concerns in the model used by Florian et al.
19
where a hydrogen sulphite/air mixture was applied for inhalation in rats. Xue et al.
20
used a neurotensin receptor agonist to develop therapeutic hypothermia in a rat stroke model. However, only mild hypothermia was achieved. They used an ice pad to achieve mild hypothermia for 10 hours in rats, and the rewarming process lasted more than two hours. We would argue, however, that it is still difficult to control body temperature using precooled pads. In contrast, using our device, temperature changes in the animals can be adjusted individually, depending on their size, weight, age and strain.
Overview of the available methods of small-animal hypothermia induction.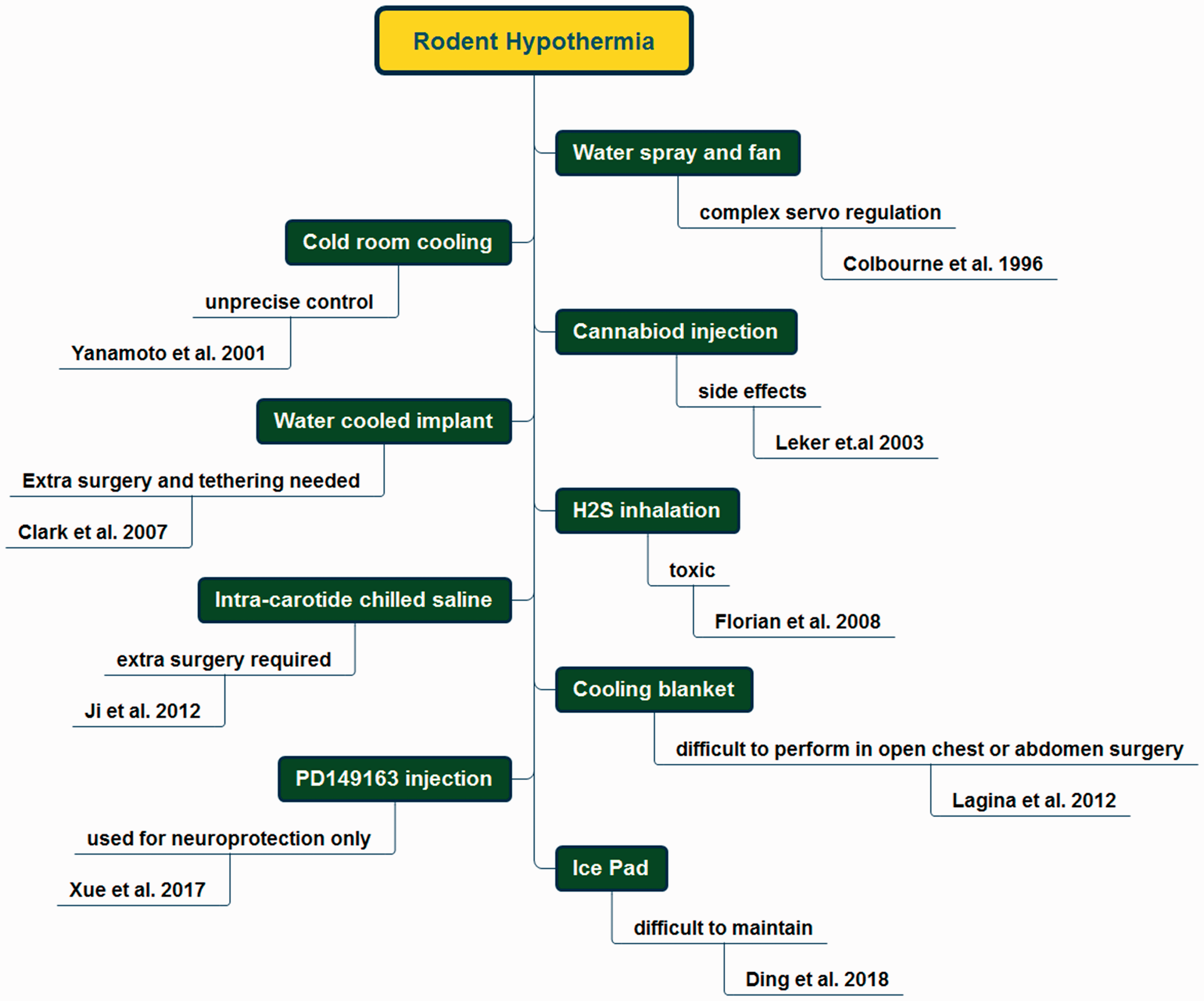
All cooling devices used in clinical practice are based on standard refrigerating systems connected to a water bath. Powerful pumps maintain circulation of the toxic, volatile liquids through a cooling bed with the help of a tubing system. Such devices are big, consume a lot of energy and have the problem of excessive heat production. The thermoelectric effect was first described by French physicist Jean Charles Athanase Peltier in 1834 who discovered heat or cold could be generated between two different materials under an electric charge current.21–23 Recently, the production of thermoelectric coolers or freezers has gained popularity, and thermoelectric elements are common in laboratory polymerase chain reaction thermocyclers where quick changes in temperature are required. We based our murine heating/cooling device on thermoelectric Peltier elements, as they are cost and energy efficient, versatile, reliable, durable and of minimal size. Having non-moving parts, they require minimal maintenance, are completely noise and vibration-free and contain no flammable materials. Importantly, reversal from cooling to heating is achieved by simply changing the polarity of the current. 24 Prior research showed effective thermoregulation of brain tissue using Peltier elements. 25
Our device has numerous other benefits. The construction of our murine heating/cooling plate used minimal resources, with all required parts easily available and inexpensive. It was designed as an operating table that facilitates performance of any surgical intervention with thermoregulation between 18℃ and 42℃. Moreover, it can be successfully applied in heat shock models. Unlike other models of hypothermia, it does not require any surgical intervention, and therefore no tissue damage occurs prior to initiating the experiment. Hence, all animals subjected to thermoelectric hypothermia tolerated the procedure well and recovered fully after rewarming. Mice cooled below 20℃ risk both cardiac and respiratory arrest. 26 As our animals did not have extracorporeal support, animals were kept no longer than 10 minutes after achieving our goal temperature of 20℃. Usually, patients on cardiopulmonary bypass can be cooled down to 18℃ or even 16℃ for several hours, depending on the type of surgery. In the future, we will use this device to test the effect of therapeutic hypothermia (<20℃ for up to one hour) on the prevention of multi-organ damage in a murine model of extracorporeal circulation.11,27
Supplemental Material
LAN873687 Supplemental Material - Supplemental material for Warming and cooling device using thermoelectric Peltier elements tested on male mice
Supplemental material, LAN873687 Supplemental Material for Warming and cooling device using thermoelectric Peltier elements tested on male mice by Nodir Madrahimov, Ruslan Natanov, Abdurasul Khalikov, Erin C Boyle, Danny Jonigk, Ann-Katrin Knoefel, Thierry Siemeni and Axel Haverich in Laboratory Animals
Footnotes
Acknowledgements
We thank Slavica Schümann for her excellent technical assistance in this project.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This research was internally funded by the Department of Cardiothoracic, Transplantation, and Vascular Surgery at the Hannover Medical School.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
