Abstract
Postoperative ileus (POI) is a common complication after abdominal surgery characterized by motility disturbances leading to increased morbidity and mortality in surgical patients. Intestinal manipulation of the murine small bowel is an established animal model resulting in an increased postsurgical inflammation within the intestinal muscular externa and a delayed gastrointestinal transit. Some analgesics have been shown to affect inflammation. In this study, we compared the immunomodulatory effects of two different analgesics. Mice were treated with tramadol, metamizole or saline as a control in our established POI model. The postoperative inflammatory response was assessed by gene expression of pro-inflammatory cytokines at different time points and immunocytes extravasation into the muscularis externa. Functional motility analyses were performed by a gastrointestinal transit measurement. Metamizole application reduced the pro-inflammatory response after surgery and improved gastrointestinal motility, while tramadol showed no alteration in cytokine gene expression, influx of immunocytes and gastrointestinal transit compared with the controls. In conclusion. we suggest tramadol as analgesia in immunological studies on POI in mice as it does not affect the underlying inflammation of POI.
Introduction
Postoperative ileus (POI) is one of the most common complications after abdominal surgery. 1 Clinical symptoms include vomiting, nausea, pain, intolerance to food and delayed defecation. 2 The pathophysiology involves a neurogenic and an inflammatory phase after the surgical trauma.2,3 In the last years, multiple promising animal studies have addressed this clinically highly relevant topic.4–8 The surgical procedure of an intestinal manipulation (IM) has been used in all of these studies and is now accepted as a standardized and well-characterized model of POI in mice and rats. IM mimics the surgical handling of the bowel during abdominal surgery in patients and reproducibly induces a postoperative inflammation of the muscularis externa (ME) 9 and a delayed gastrointestinal (GI) transit , 10 the hallmark of POI. In human patients, analgesia non-steroidal anti-inflammatory drugs (NSAIDs) or opioids are routinely applied for treatment of postoperative pain. 11 Metamizole, is another but controversial analgesic to treat pain related to surgery. 12 All these analgesics are permitted for routine analgesia in research animals. However, in addition to their pain-relieving action, some analgesics have also been shown to modify inflammation and even GI motility and therefore they may be inappropriate for investigations on inflammation-driven diseases, for example POI or sepsis. Particularly in a POI-related study, Schwarz et al. showed that NSAID treatment ameliorated the inflammatory posttraumatic response by inhibition of COX-2 and thereby improved GI transit after IM. 13 An even more prominent example are opioids, e.g. buprenorphine, which are commonly used effective analgesics in laboratory rodents. However, opioids not only block pain sensation via spinal δ- and κ-opioid receptors, they also agonize peripheral µ-opioid receptors on enteric neurons. As a result, inhibition of gastric emptying, induction of stationary motor patterns and blockade of peristalsis and regular motility ensue.14,15 The corresponding clinical picture is an opioid-induced ileus, which is often also entitled POI leading to some confusion, but the more appropriate term ‘opioid-induced ileus’ continuously finds its way into the literature.16,17 Interference of analgesics in immune-mediated disease models such as endotoxemia, sepsis or POI are undesirable as it may hamper investigations on the underlying immune mechanism. For a long time, this problem was simply overcome by omission of perioperative analgesia. 18 However, increasing awareness of animal ethics and efforts to apply the 3R principle (replacement, reduction and refinement) requires alternative analgesia protocols. In order to fulfil these criteria, we performed this comparative study on the immune modulatory effects of metamizole, a painkiller from the ampyrone sulfonate family that has been described to exhibit marginal anti-inflammatory effects 19 and tramadol, a weak opioid with lower affinity to µ-opioid receptors than buprenorphine. 20
Material and methods
Animals
Forty-eight C57BL/6/J mice from Janvier (Saint-Berthevin Cedex, France) were enrolled in the study. All experiments were performed with male mice at an age of 8–12 weeks and were acclimatized for at least 5 days under specific pathogen-free conditions including 12/12 h light/dark cycle, 21℃ and 50–70% relative humidity in the animal housing facility of the University of Bonn (Germany). The mice were fed (V1534-300, SNIFF Soest, Germany) and watered ad libitum. The animals were housed in climatic rooms. Floor, ceiling and walls are covered with epoxy resin. The barriers consist of autoclave, material airlock and personnel airlock. Staff is only allowed with operation shoes, sterile aprons, operation caps, special masks and surgical gloves. Cages, bedding and water are autoclaved. Animals are transferred in clean cages only in transfer stations once per week. The health check is performed under FELASA-guidelines 21 using sentinel animals quarterly and a six-monthly health monitoring (currently GVG Diagnostics GmbH, Leipzig, Germany).
Morbidity and mortality were checked once daily during the study. The experiments were performed in accordance with federal law regarding animal protection, ARRIVE guidelines and were approved by the government (AZ: 84-02.04.2014.A509).
Operations
Surgery was performed under aseptic conditions. Anaesthesia was induced using isoflurane (Abbott, Wiesbaden, Germany) and oxygen as gas carrier (1.5 l/min). Induction of anaesthesia was achieved with 3 vol% of isoflurane. The mouse's face was placed into a suitable mask during the surgery (approximately 20–30 min), under 1.5–3% of isoflurane.
After laparotomy, the small intestine of mice was mechanically manipulated in a standardized manner, as described previously. 22 In brief, after laparotomy the small intestine was eventrated and smoothly rolled between two moist cotton tips from oral to anal. After IM, the intestine was placed back into the abdominal cavity. The peritoneum was closed with Vicryl 5-0 Ethicon and the skin was closed with Silkam 5-0. Operations and analgesic treatments were performed by a trained operator. All analyses were performed by a different person who was not blinded to the animal treatment.
Post-surgery care
Mice were treated with analgesia as indicated below after surgery. They were placed in cages that were placed under an infrared lamp during 2–3 h post-surgery. Afterwards, they were housed again under specific pathogen-free conditions including 12/12 h light/dark cycle, 21℃ and 50–70% relative humidity in the animal housing facility of the University of Bonn (Germany).
Animal euthanasia
Animals were sacrificed 6 or 24 h after surgery for further analysis. Euthanasia was performed by cervical dislocation.
Analgesia
The tramadol group was given tramadol (Tramal, Grünenthal GmbH, Aachen, Germany) 30 mg/kg of body weight (BW) 1 h before and 2 h after surgery subcutaneously. Afterwards, tramadol was dissolved within the drinking water (1mg/ml) and mice were watered ad libitum. The biological half-life of this drug is about 6 h. The metamizole group was treated with metamizole 20 mg/kg BW (Novaminsulfon, Ratiopharm, GmbH, Ulm, Germany) 1 h before and 2 h after surgery subcutaneously. Subsequently, they were given metamizole 200 mg/kg BW orally via gavage every 6 h. The biological half-life of this drug is about 3 h. The control group received 100 µl NaCl 0.9% (B. Braun Melsungen AG, Melsungen, Germany) 1 h before and 2 h after surgery (Figure 1).
Study protocol. Forty-eight mice were enrolled into the study. All mice were operated with intestinal manipulation (IM), the tramadol group received tramadol (30 mg/kg body weight (BW)) s.c. pre-and postoperative. Afterwards, tramadol was dissolved in drinking water and was offered to the animals ad libitum. The metamizole group was given metamizole (20 mg/kg BW) pre- and postoperative s.c.. Subsequently, animals were administered metamizole 200 mg/kg BW via gavage. The control group received NaCl as a vehicle s.c. pre- and postoperatively. In each group, eight animals were sacrificed for further analysis either 6 h or 24 h after surgery.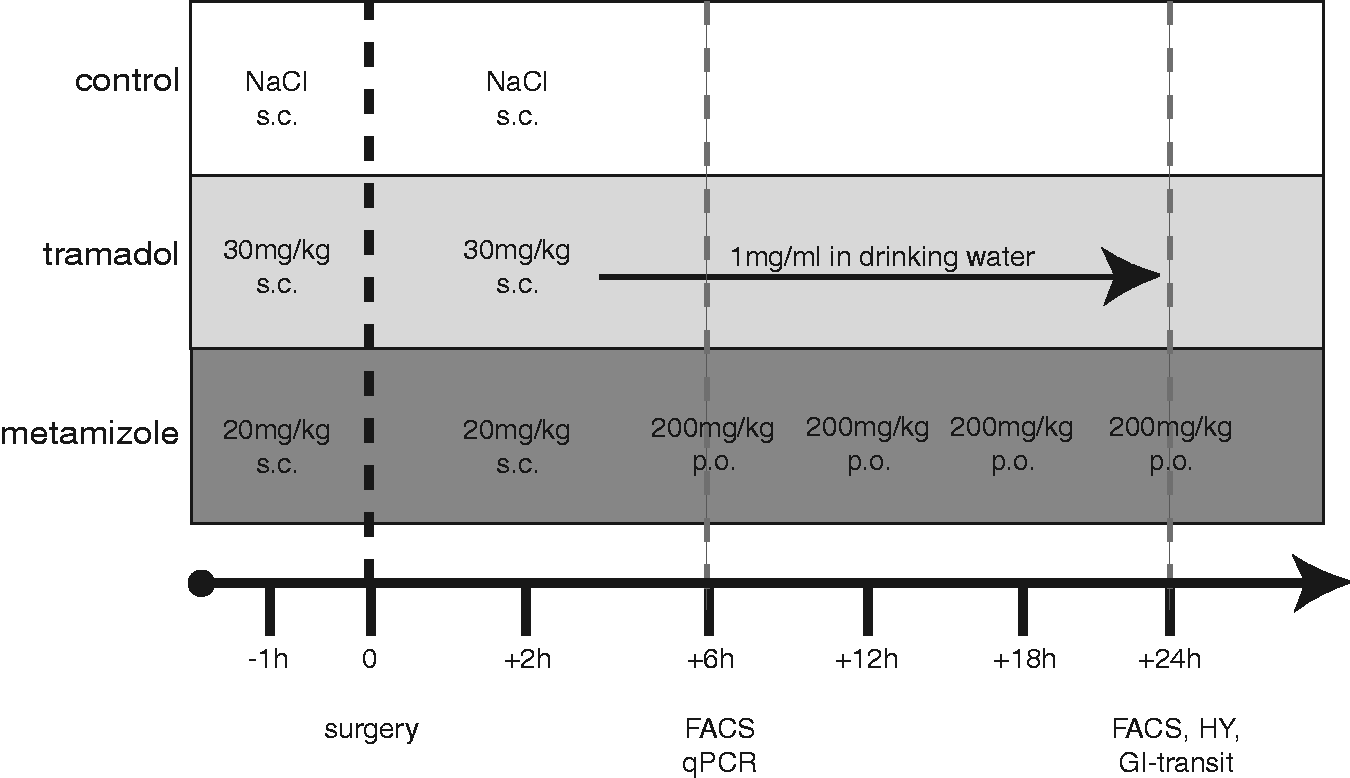
GI transit
GI transit was measured 24 h after IM by giving 100 ml fluorescein isothiocyanate (FITC)-labelled dextran (size 70 kDa; 6.25 mg/ml)/gavage within a fasting period of 90 min before organ harvest. The intestinal tract was divided into 15 segments (stomach–colon), and luminal contents were washed out to determine the absorbance of dextran. GI transit was calculated as the geometric centre of FITC-labelled dextran distribution, as described previously. 20
Histochemical analysis
Midjejunal, mucosal-free muscularis whole mounts were fixed in ethanol and used for staining of MPO+ monocytes and neutrophils. MPO+ cells were counted in 5 randomly chosen areas in each specimen at a magnification of 200 × and calculated as MPO+ cells/mm2.
Quantitative PCR
Gene expression of pro- and anti-inflammatory cytokines was analysed by qPCR. Reagents were from Life Technologies unless specified otherwise. Total RNA was extracted with Trizol® reagent using a tissue homogenizer (Precellys® 24; Peqlab, Erlangen, Germany) followed by DNase I treatment. cDNA was synthesized using a High Capacity cDNA rt kit. Expression of mRNA was quantified in triplicate by reverse transcription–PCR with specific probes/primers (see Table S1, supplementary material). The PCR was performed in Power SYBR® Green or Universal PCR Master Mix by amplification of 10 ng cDNA for 40 cycles (95℃ for 15 s, 60℃ for 1 min) on an AbiPrism® 7900HT (Life Technologies). Primers were designed for CXCL1 (5′- TCTCCGTTACTTGGGGACAC-3′; 5′-CCACACTCAAGAATGGTCGC-3′), CXCL2 (5′-TCCAGGTCAGTTAGCCTTGC-3′; 5-GGTCAAAAAGTTTGCCTTG-3′), IL-6 (5′- AAGTCGGAGGCTTAATTACACATGT-3′; 5′- CCATTGCACAACTCTTTTCTCATT-3′), MCP-1 (5′- GTCCTCAGGTATTGGCTGGA-3′; 5′- GGGTCAGCACAGATCTCCTT-3′) and IL-10 (5′-GATGCCCCAGGCAGAGAA-3′; 5′-CACCCAGGGAATTCAAATGC-3′). Gene expression of IL-1β was measured using a Tagman assay (Mm00434228_m1). Data quantification was performed by the ΔΔCT method and value normalized with respect to glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (4352339e, Tagman Assay) levels.
Flow cytometric analysis
Immunocytes were isolated after removal of the small intestine and separation of the mucosa from the muscularis layer from fine-cut pieces of ME. ME tissue was digested with a 0.1% collagenase type II (Worthington Biochemical, Lakewood, NJ, USA) enzyme mixture, diluted in HBSS, containing 0.1 mg/ml DNase I (La Roche, Mannheim, Germany), 2.4 mg/ml Dispase II (La Roche), 1 mg/ml BSA (Applichem), and 0.7 mg/ml trypsin inhibitor (Applichem) for 35 min in a 37℃ shaking water bath. Afterwards, the cell suspension was filtered through a 70 mm gauze and single cells processed further for FACS analysis. After use of Fc-blocking solution (clone 2.4G2 was a gift from the Institute of Molecular Medicine, Bonn, Germany), cells were stained for 30 min at 4℃ with fluorochrome-labelled Ab against CD45 (Pacific Blue, GK1.5; BioLegend, San Diego, CA, USA), F4/80 (Alexa488, BM8; BioLegend), Ly6C (PE-Cy7, HK1.4; BioLegend) and Ly6G (APC, 1A8; BioLegend). Flow cytometry analyses were performed on a FACS Canto II or LSRFortessa (BD Biosciences) and data were analysed with FlowJo software (Tree Star, Ashland, OR, USA). Hoechst 33342 (Life Technologies) was used to exclude dead cells.
Statistical analysis
Group sizes were established using data from a previous study where between five and eight mice per group achieved 80–85% power. All results are expressed as mean ± SD. Statistical analysis was performed using one-way analysis of variance (ANOVA) with Bonferroni's post-hoc test with Prism® 5.02 software (GraphPad, La Jolla, California, USA); p < 0.05 was considered statistically significant.
Results
The IM-induced inflammatory response after tramadol or metamizole treatment was examined in the early and the late postsurgical phase and compared with the control group. In addition, GI transit was determined 24 h after IM in each group.
Immune response in the early postsurgical phase
To explore the early phase immune modulation by tramadol and metamizole, infiltration of immunocytes was determined via flow cytometry and gene expression of pro- and anti-inflammatory cytokines was analysed by qPCR within ME 6 h after IM. The postoperative levels of Ly6G-Ly6C + monocytes were neither altered by tramadol (7.5 × 104 ± 4.6 × 104 cells/100 mg ME) nor by metamizole (8.1 × 104 ± 3.6 × 104) treatment compared with the control group (8.2 × 104 ± 5.1 × 104; F = 0.05, p = 0.9) (Figure 2(a)). In addition, the number of Ly6G+Ly6C + neutrophils did not change after administration of tramadol (3.2 × 104 ± 2.1 × 104) and metamizole (2.9 × 104 ± 2.1 × 2.24) compared with the control group (3.0 × 104 ± 2.2 × 104; F = 0.05, p = 0.9). Gene expression of pro-inflammatory cytokines IL-6 (0.75 ± 0.15; F = 1.7; p = 0.024) and CXCL2 (0.57 ± 0.17; F = 2.35; p = 0.015) was significantly reduced after metamizole treatment, but not after tramadol treatment (IL-6: 0.82 ± 0.35, F = 1.7, p = 0.4; CXCL2: 0.84 ± 0.44, F = 2.4, p = 0.56) compared with the control group (IL-6: 1.0 ± 0.1; CXCL2: 1.0 ± 0.24), (Figure 2(a) and (b)). The expression of other chemokines and cytokines was not altered after administration of tramadol (CXCL1: 0.94 ± 0.43; IL-1β: 1.2 ± 0.28; MCP-1: 1.0 ± 0.43) or metamizole (CXCL1: 0.77 ± 0.2; IL-1β: 1.20 ± 0.28; MCP-1: 1.01 ± 0.43) compared with the control (CXCL1: 1.0 ± 0.17, F = 0.75, p = 0.5; IL-1β: 1.05 ± 0.34, F = 0.74; p = 0.5; MCP: 1.0 ± 0.1, F = 0.09, p = 0.9) (Figure 2(c)–(f)). Finally, mRNA levels of the anti-inflammatory cytokine IL-10 were also not affected by tramadol (0.92 ± 0.29) or metamizole (1.04 ± 0.36) treatment compared with the control (1.0 ± 0.1, F = 0.23, p = 0.8) (Figure 2(g)).
Inflammatory response in the early phase of postoperative ileus (POI). Six hours after intestinal manipulation (IM) and analgesia treatment with tramadol, metamizole or saline (control), (a) infiltration of Ly6G+Ly6G+neutrophils and Ly6G−Ly6C+ monocytes into the muscularis externa (ME) was measured by flow cytometry. (b–g) Gene expression of IL6, CXCL1, CXCL2. IL-1β, MCP-1 and IL-10 was measured in ME samples and fold changes in mRNA levels were compared with the control group. Data are plotted as mean ± SD; n = 8 mice/group. Statistical analysis was done by a 2-way analysis of variance (ANOVA) (a) or a 1-way ANOVA (b–g) followed by Bonferroni post hoc test. *p < 0.05 versus control.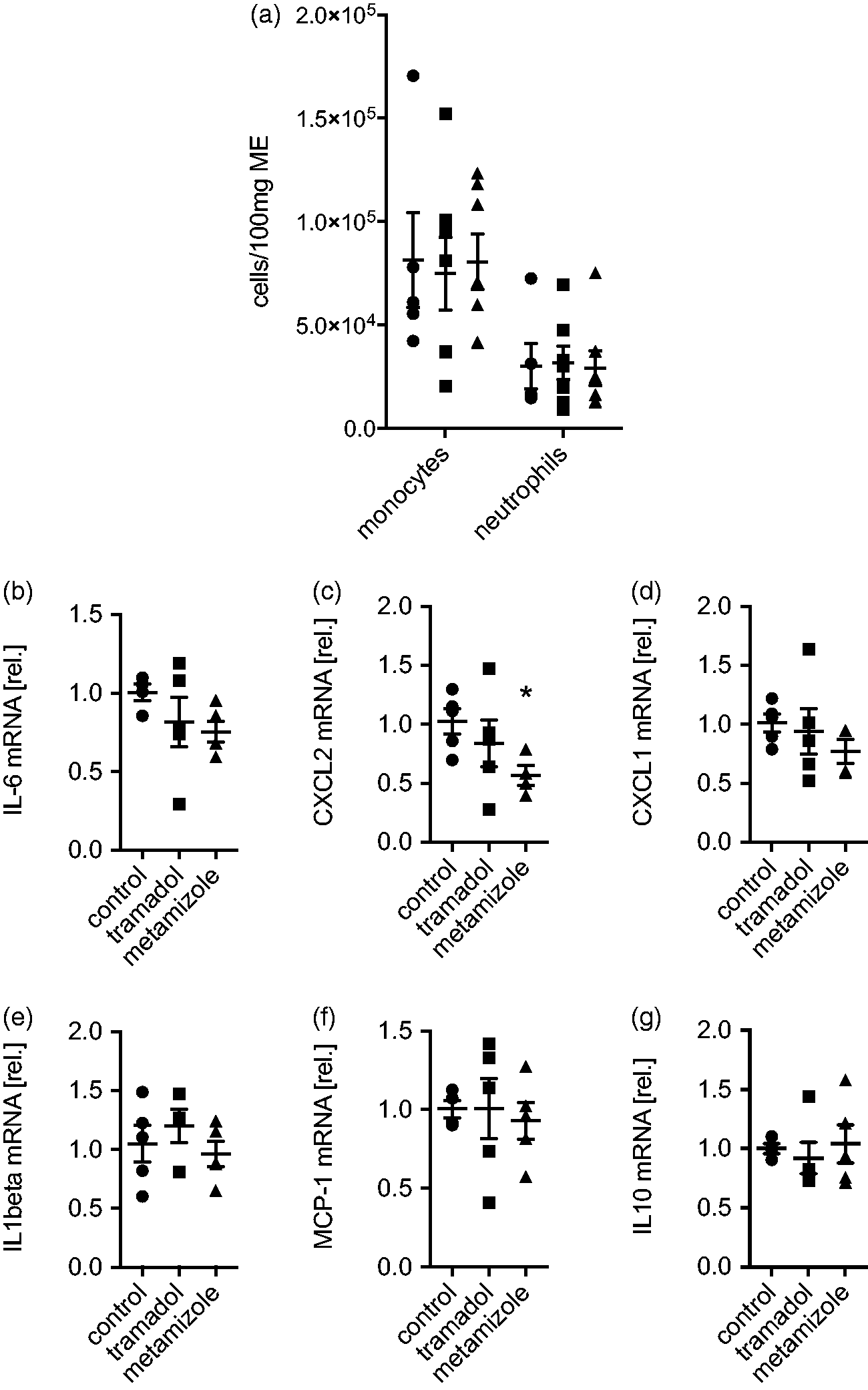
Immune response and motility alteration in the late postsurgical phase
IM-induced infiltration of immunocytes into the surgically manipulated ME is not an immediate event but starts several hours after surgery. In mice, infiltration levels peak around 24 h after IM. MPO+ leukocytes numbers were not affected by tramadol (502 ± 122 cells/mm2) or metamizole (534 ± 156) treatment compared with the control group (608 ± 144, F = 1.23, p = 0.3) (Figure 3(a)). A slight increase of Ly6G+Ly6C + blood-derived neutrophils was observed after tramadol treatment in the ME compared with the control group (231 ± 50 versus 168 ± 42; F = 5.78, p = 0.036) (Figure 3(b)). Metamizole administration did not alter infiltration of neutrophils (151 ± 37; F = 5.78, p = 0.98). Infiltration of the main immunocytes population, Ly6G-Ly6C + monocytes, was neither impaired by tramadol (1084 ± 263) nor by metamizole (940 ± 361) compared with the control group (980 ± 316, F = 0.36, p = 0.70) (Figure 3(b)).
Wild-type (WT) mice underwent intestinal manipulation (IM) and received analgesia with tramadol, metamizole or NaCl (control group). Animals were sacrificed 24 h after surgery and (a) MPO+ cells within the muscularis externa (ME) as well as (b) numbers of Ly6G+Ly6G+ neutrophils and Ly6G-Ly6C+ monocytes were determined. (c) Gastrointestinal (GI) transit was calculated by the geometric centre and plotted as mean ± SD. The GC was calculated by the distribution of a fluorescence marker along the complete GI tract (15 segments) within the same animals used in (a) and (b). st stomach; c: cecum. Data are displayed as mean ± SD; n = 8 mice/group. Statistical analysis was done by a 1-way analysis of variance (ANOVA) ((a) and (c)) or a 2-way ANOVA (b), followed by Bonferroni post hoc test. *p < 0.05; **p < 0.01 versus control.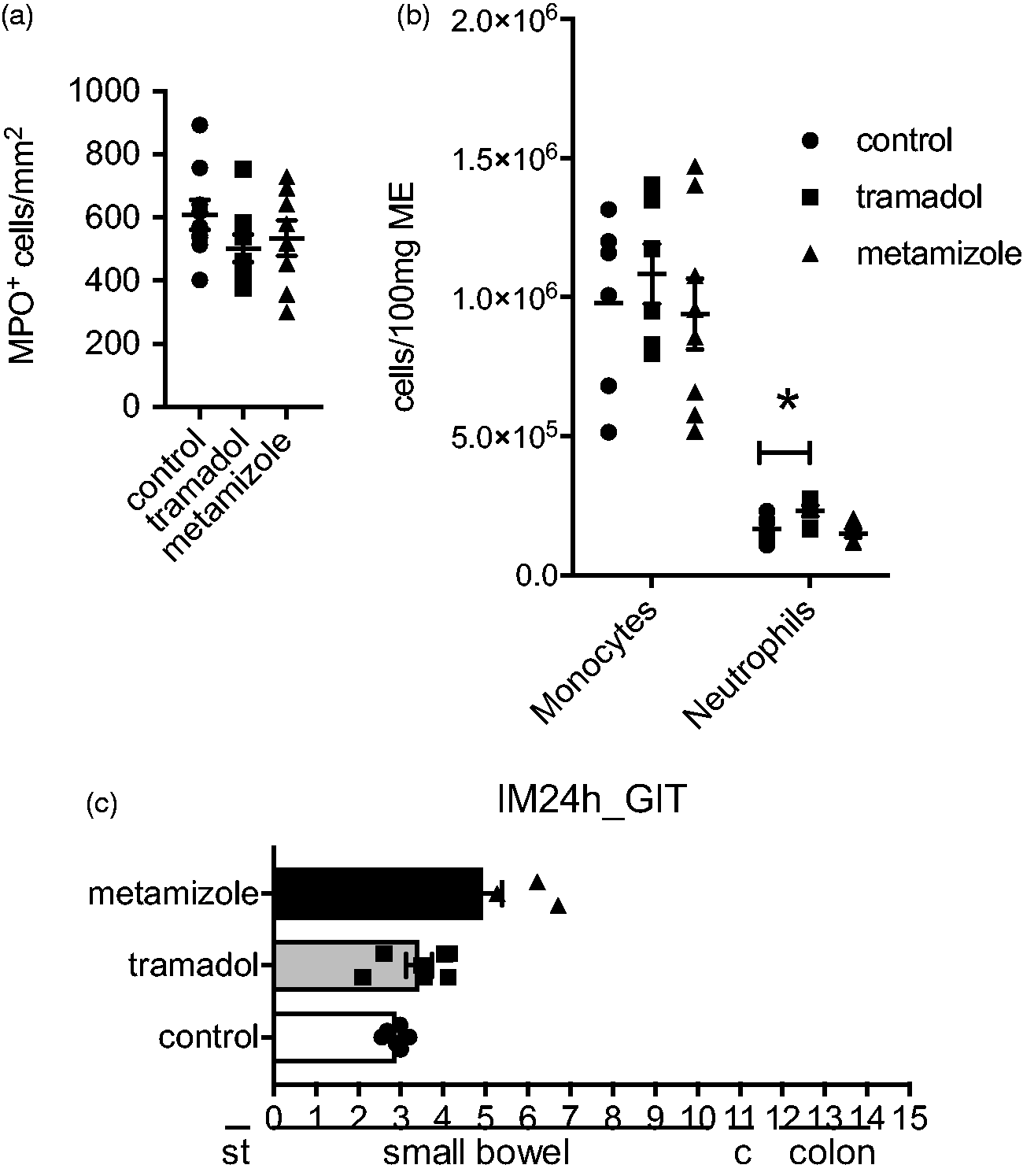
Finally, we evaluated the functional influence of tramadol or metamizole on the GI motility by determination of the GI transit 24 h after IM. Tramadol treatment did not modify GI transit compared with the control (3.43 ± 0.80 versus 2.88 ± 0.23 geometric center (GC), F = 9.51, p = 0.4) whereas metamizole administration (4.93 ± 1.24) significantly accelerated GI transit compared with the control (2.88 ± 0.23, F = 9.51, p = 0.0013) (Figure 3(c)).
Discussion
In the current study, we evaluated whether tramadol or metamizole may be appropriate analgesics for investigations related to the immune pathophysiology of POI in mice. Our results show that metamizole treatment is inappropriate as it inhibits the early inflammatory response, shown by reduced gene expression of IL-6 and CXCL-2. Even though the influx of immunocytes in the late phase of POI was not altered by metamizole, it improved the postoperative GI motility. The latter may not result from a direct effect linked to alterations in the postoperative immune response as the reduction in GI transit normally inversely correlates to the number of infiltrating immune cells. This finding is supported by another study wherein metamizole did not interfere with inflammatory response during pancreatitis in mice. 23 On the other hand, the reduced inflammatory gene expression in the early phase of POI may point to an immune-mediated effect. Another model of inflammation, using the carrageenan test, exhibited a reduction in the inflammatory response after scalpel incision in metamizole treated animals. 24 Nevertheless, the reduction of only two out of six immune mediators in our POI model indicate for a minor immune modulatory than a direct GI motility affecting role of metamizole. In contrast, tramadol did not alter the inflammatory response in the early phase after surgery, and also functional analyses revealed no influence in bowel function indicating that analgesia with tramadol may be used in the murine POI model. Tramadol is a racemic mixture consisting of two isomers with different spectrums of pharmacological activity. In the CNS, tramadol activates both opioid and non-opioid systems, which are mainly involved in pain inhibition.25,26 However, it has also been suggested to have local anti-inflammatory properties. Injection of tramadol into the temporomandibular joints did not only exhibit an analgesic effect but also reduced expression of pro-inflammatory cytokines within the tissue. However, the anti-inflammatory effect was after local administration and also dose depending, whereas nociceptive reaction was not dose dependant. 27 In our model, tramadol was not applied locally into the peritoneal cavity, but systematically, so that a local anti-inflammatory effect was avoided. Surprisingly, influx of neutrophils 24 h after IM was significantly increased after tramadol treatment compared with control, even though it was described vice versa before. 27 Nevertheless, the majority of infiltrating immunocytes in POI are monocytes whose numbers were not affected by the tramadol treatment. The slight effects on neutrophils may not be over interpreted particularly as none of the investigated immune mediators underwent any changes. Nevertheless, the slight increase in neutrophil influx should be kept in mind, particularly in studies in which neutrophils may play a more dominant role than in POI. Although tramadol delivery via drinking water is a more convenient and stressless way than an oral gavage, which is proven by the German Council of the Society for Laboratory Animals (GV-Solas), it might bear the risk of uptake of different amounts of tramadol which would impair comparability of the analgesic regimens. Furthermore, due to the high variance and the limited number of animals, we cannot totally exclude a false negative result. Intentionally, we did not include a control group that underwent laparotomy without IM, because pro-inflammatory cytokine expression and leukocyte influx in to the ME, both are hallmarks of POI development, are not induced by laparotomy alone. Inclusion of these groups would not be adequate for our scientific problem.
In conclusion, we demonstrated that tramadol hardly modulates the inflammatory response and the intestinal motility disturbances after IM, and therefore can be suggested for analgesia in POI research.
Footnotes
Acknowledgements
We thank Marie-Therese Puth of the Institute for Medical Biometry and Informatics of the university of Bonn for revising the statistical analysis of this study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: SW and JCK received royalties from Wolters Kluwer. SW and GH received grant support from Plantec Medical. SW received grant support from Thetis Pharmaceuticals Inc. KS and ML declare no conflict of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author contributions
Gun-Soo Hong: study concept and design; acquisition of data; analysis and interpretation of data; drafting of the manuscript. Kathy Stein: acquisition of data; analysis and interpretation of data; statistical analysis. Mariola Lysson: acquisition and analysis of data. Joerg C. Kalff: material support and critical revision of the manuscript for important intellectual content. Sven Wehner: study concept and design; preparation of the manuscript; technical and material support; study supervision. All authors approved the version to be published.
