Abstract
We have developed a new method for the measurement of subcutaneous tumour volume which consists in taking photographs of mice in their home cages, to refine the standard method of measurement with calipers. We consider this new method to be non-aversive, as it may be more compatible with mice behavioural preferences and, therefore, improve their welfare. Photographs are captured when mice voluntarily go into an acrylic tube containing graph paper that is later used as a scale. Tumour volumes measured with the caliper and the non-aversive photographic method were compared to those obtained by water displacement volume and weight. Behavioural and physiological changes were evaluated to assess animal welfare. Significant differences were found between measurements obtained with the caliper and the non-aversive photographic method, v. the reference volume acquired by water displacement (P < 0.001). Nevertheless, there was good consistency for these measurements when tumours were measured repeatedly, with all Intra-Class Correlation Coefficients above 0.95. Mice on which the non-aversive photographic method was employed were significantly less reluctant to establish contact with the experimenter (P < 0.001) and behaved less anxiously in a modified-Novelty Suppressed Feeding test. Particularly, statistically significant differences were found in connection with the latency to eat an almond piece (P < 0.05), the frequency of grooming (P < 0.001) and the frequency of defecation (P < 0.001). Corticosterone concentration in faeces and blood glucose were determined and no significant changes were found. Therefore, we propose the non-aversive photographic method to measure subcutaneous tumours as a way to refine methodologies in the field of experimental oncology.
Growing tumours in athymic (commonly referred to as ‘nude’) mice has been an important part of preclinical anticancer drug testing since they were first described in 1969. 1 Despite certain limitations, the growth of subcutaneous tumours in nude mice is still a commonly used tool in cancer-drug development. 1
Even though the standard method for subcutaneous tumour measurement is through the use of calipers, 2 which are inexpensive, fast and easy to use, these measurements have been described as inaccurate, as their variability can sometimes reach 25%. 3 New measurement techniques have been described, but these are laborious and dependent upon physical restraint or the use of anaesthetic agents.3,4,5,6
Beside animal welfare considerations, the stress produced by immobilization modifies both the animals’ behaviour and physiology 7 and, thus, experimental results can be affected. While the effect of stress over tumour volume is not clear yet, handling mice has been shown to increase tumour volume8,9 and metastases. 10 Controversially, in this last article, the authors also describe an increase of the natural killer (NK) cells in situ, which mediates the immune anti-tumour response. Increased activity of NK cells in animals reared in enriched environments, which are considered to be eustress, along with decreased tumour volume have also been reported. 11 The effect of stress over immunity and therefore over tumour development, seems to be affected by the perceived controllability and intensity of the stressor. 12
It is well known that mice prefer to have their bodies, and especially their vibrissae, in contact with another surface, a property called thigmotaxis. 13 Because of this property, the use of tunnels to handle this species minimizes anxiety responses, especially when they get used to the procedure. 7 This habituation is not detected when mice are manipulated by their tail, where the repetition of the procedure does not minimize negative responses to human contact. 7
We compared the accuracy of the measurements taken with a caliper to those obtained with a less stressful technique that we have called ‘non-aversive photographic measurement method’, which is described here for the first time. The effect of both methods on the mice physiology and behaviour were also evaluated in order to understand the impact that both methods have on their welfare.
Materials and methods
Animals and husbandry
Fifty-six, female and male, 4–6-week-old, specific pathogen free (SPF) nude mice (stock N:NIH(S)-Fox1nu) were acquired from the Animal Facility at the Faculty of Veterinary Sciences of La Plata National University, Argentina (LAE-FCV-UNLP). To avoid confounded effects of tail handling with caliper measurement, mice were cup handled 14 and manipulated by the same experimenter throughout the study. Animals were housed at the LAE facility, in Lab Products Super Mouse 750™ (480 cm2) individually ventilated cages (34 air changes per hour in positive pressure mode) with aspen shavings as bedding (Guillermo N Cabañas, Ciudad Autónoma de Buenos Aires, Argentina) and tissue paper as nesting material. Food pellets (Cooperación, Alicooper S.A., Santa Fe, Argentina) and water were administered ad libitum. All supplies were sterilized and cages were changed once a week. Animals were kept at a room temperature of 22 ± 2℃ and a 12:12 dark: light cycle (lights on at 7 a.m.).
This study was approved by the FCV-UNLP IACUC under protocol number 42-1-14T and conducted in compliance with the Guide for the Care and Use of Laboratory Animals 15 and the United Kingdom Co-ordinating Committee on Cancer Research (UKCCCR) Guidelines for the Welfare of Animals in Experimental Neoplasia (Second Edition). 16
Cell line and tumour transplant
A549 cells obtained from a solid tumour grown in different mice were used. Tumour pieces were fragmented into 2 mm3 chunks and transferred into a drop of Minimum Essential Medium (MEM) cell culture media before subcutaneous implantation in the right or left thoracic area of nude mice using a trocar. 17
For the inoculation, animals were anaesthetized with ketamine/xylazine (100–10 mg/kg, i.p., respectively) with no previous fast, and room temperature was kept at a minimum of 25℃ to prevent hypothermia. Intraoperative analgesia was provided by xylazine (administered for the anaesthesia) and 1% lidocaine at the site of the incision. As it was a minor procedure, a single dose of 10 mg/kg of tramadol was administered subcutaneously before the animals woke up as post-operative analgesia and they were closely monitored in order to avoid unnecessary pain. Surgery was aseptically performed under a clean bench with sterilized surgical instruments and materials and, as clipping was not needed for this strain, only povidone-iodine was employed as skin antiseptic. A small incision (approximately 0.4 cm) was made in the lateral abdominal area and a subcutaneous tunnel was produced with the trocar before implanting the tumour piece. Special attention was given in order to place the tumour in the middle of the lateral area of each mouse, as it is important for the non-aversive photographic method to have the whole surface of the tumour facing the camera. Because only a small incision was made, wound edges remained close together and no stitches were needed. The total length of the procedure for each animal was less than 5 min. After the surgery, mice were kept on a heating pillow until recovery, and oxygen was administrated when necessary. Animals were supervised every 8 hours for 5 days after the procedure and then daily until the end of the experiment. Observations included body condition scoring, surgical wound complications (presence of inflammation, burst releasing fluids, pus), mice general appearance and behaviour (ocular or nasal discharges due to lack of grooming, presence of mice sleeping outside the nest, nest quality, increased aggression, engaging in normal behavioural activities), behavioural changes due to tumour development (abnormal gait or impaired locomotion, increased grooming or nociception at the site of the tumour), skin general appearance and tumour characteristics and size. No deviations from normality were observed in any of the inoculated animals.
Experimental design
In order to avoid variability and reduce animal anxiety, the same experimenter (AR) carried out all the procedures so that complete blinding for treatment was not possible. To overcome this problem, tumour-volume recording was made by another researcher (ACCM) and AR did not have access to previously registered data on tumour volumes. Moreover, a blinded observer (ACCM) was designated to perform the behavioural observations.
Experiment 1
The aim of this experiment was to evaluate whether the new method would be valid to measure subcutaneous tumours. Thus, intra-observer variation with the caliper and the non-aversive photographic method was quantified as well as the time needed to take each measurement. In addition, the effect of each method on mice was evaluated by means of biochemical and behavioural parameters.
Twenty tumour-transplanted, female, nude mice were housed in groups of 5 and kept for 3 weeks to allow the tumours to grow. On the first day of the following 2 weeks, i.e. weeks 4 and 5, measurements were obtained with the caliper in half of the mice and with the non-aversive photographic method on the other half. Immediately after these measurements were taken, blood samples were collected on week 4 to analyse glucose levels, and, on week 5, the Novelty-Supressed Feeding (NFS) test was conducted. On week 6, intra-observer variation was evaluated measuring the tumours with both methods on 2 consecutive days (days 1 and 2 of week 6). Subsequently, mice were euthanized by cervical dislocation and tumours were dissected to be weighted and determine their volume by water displacement.
Experiment 2
Our purpose was to evaluate whether any of the methods induced variations in behaviour towards the experimenter over time, as well as in faecal corticosterone. Twenty female and 16 male mice were housed in groups of 2 animals per cage for 4 weeks. Tumour volume was measured with the caliper in half of the cages and with the non-aversive photographic method in the other half. To identify each mouse, the tumour transplant was made either on the left or on the right side. Every week, tumour measurements were performed on days 1 and 2. Behavioural observations were registered on day 2 immediately after tumour measurement. Cage change took place after behavioural observation. On day 3 of each week all faecal boli were collected from the cages.
Tumour-volume measurement
Tumour volume was calculated by the use of the modified ellipsoid formula (length x width 2 )/2. 18 Tumour measurements were made with a caliper or with the non-aversive photographic method, as needed. The time that was required to take each determination was recorded with a stopwatch. After euthanasia, tumour weight and water displacement data were registered. Water displacement was determined by the difference in distilled water volume at room temperature measured in a test tube after introducing the tumours. Results were expressed in cm3.
For the non-aversive photographic measurement method, a rectangular section of a transparent acrylic tube which was 12 cm long, 4 cm wide and 5 cm tall was used (Figure1(a)). To measure the tumours, graph paper, which occupied half the length of the tube, was used as a scale. The graph paper was inside of the tube, held in place in the middle of the width with an ‘L’ shape acrylic figure. This apparatus was put inside the animal’s home cage (experiment 2) or in another cage with the same characteristics (experiment 1). For experiment 1, animals were introduced one by one by cup handling them into the cage. Inside the cage, the tube was placed in one of the corners adjacent to its longest wall (Figure 1(a)). Mice voluntarily went into the tube through the opposite side of the graph paper as they explored the new object. Most of the times, the animals remained with their head and forelimbs behind the graph paper (Figure 1(b)). When mice had the cranial part of their body behind the graph paper, images were taken by the researcher with the multi-shot function of a NIKON DSLR D3100 camera. The camera was located perpendicular to the longest side of the cage at a distance of 15 cm. Images were then analysed with the Image J software,
19
using the graph paper as scale (Figure 1(c)). When setting the scale in the computer program, the distance taken as reference in the paper was located at the same height as the tumour localization. The scale was calibrated for each picture. Two measurements were taken in each photo: the greatest diameter of the tumour was considered the length and the shortest diameter was considered the width (Figure 1(d)).
Tumour measurement with the non-aversive method. (a) The apparatus, consisting of an acrylic tube with graph paper inside, is introduced into the mice home cage. (b) The mouse goes into the tube and pictures are taken when the tumour is located next to the graph paper. (c) The scale is set using the graph paper at the same height as the tumour is located. (d) Tumours are measured taking into account their greatest and shortest diameter.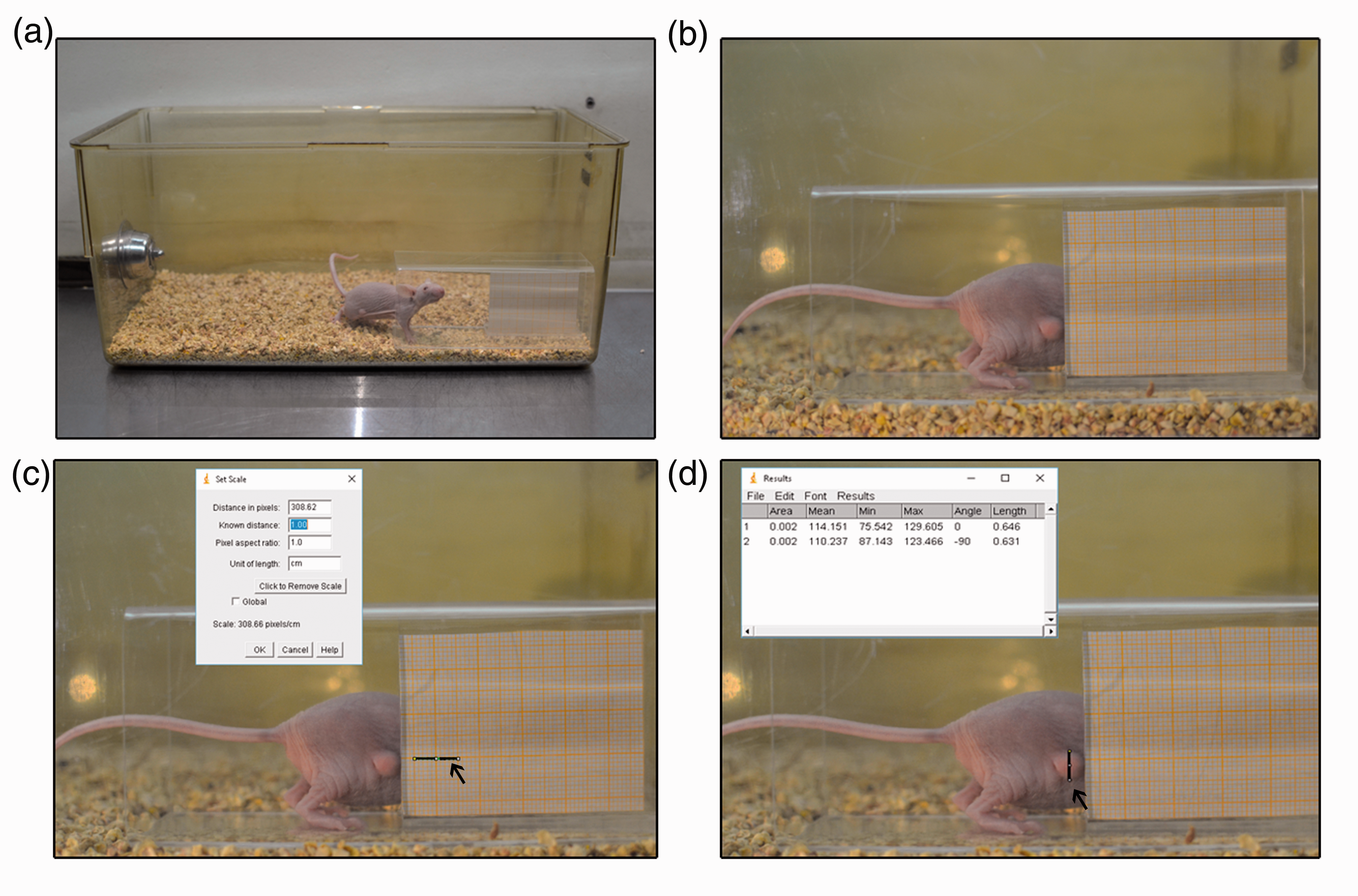
Blood collection and glucose determination
Blood samples were taken 5 min after measurements by means of submandibular bleeding using a 21 G needle. 20 A single drop was collected (approximately 100 µl) and blood glucose was immediately measured using a compact glucose analyser (Contour™TS, Bayer).
Faecal collection and corticosterone metabolite assay
Faeces were sampled from each cage once per week throughout the study. All faecal boli were collected from the cages 24 hours after the cleaning routine. Samples were frozen at −20℃ until processed. Faecal steroid metabolites were extracted according to the method described by Touma et al. 21 with slight modifications: faeces were homogenized and then 1 ml of 80% methanol was added to 0.1 g of humid faecal pellets. Samples were incubated at room temperature for 120 min in an orbital shaker, then shaken in a vortex for 10 s and finally, centrifuged at c. 500 g for 20 min. The supernatant was frozen at −20℃ until analysed.
Corticosterone metabolites were analysed by Enzyme Immunoassay (EIA) as described previously by Munro & Lasley 22 with polyclonal antisera and conjugated peroxidase for corticosterone detection (corticosterone CJM006, Department of Population Health and Reproduction, Coralie Munro, UC Davis, CA, USA).
Behavioural assays
Experiment 1
The NSF paradigm is a conflict test that elicits competing motivations: the drive to eat and the fear of venturing into the centre of a brightly lit arena. For this study, a modified NSF was used. 23 This version of the test does not require food deprivation, as it uses a preferred item (an almond piece) over the regular food chow. Mice were habituated to the almonds by introducing two almonds per cage in the cleaning cage routine. The day of the test, an almond piece (approximately 1 g) was placed in the centre of an open arena (30 x 40 x 16 cm). Each mouse was placed by cup handling in a corner of the box, and a stopwatch was immediately started. The latency to start eating (defined as the mouse sitting on its haunches and biting the almond with the use of its forepaws) was recorded for a 5-min period. Additionally, while each mouse was in the open field, the frequency of grooming and defecation were recorded. We hypothesized that by using a less aversive method of tumour measurement, mice would be less anxious, and therefore, more willing to venture into the centre of a brightly lit arena to eat a reward while displaying less anxiety-like behaviours.
Experiment 2
The frequency of the voluntary contact of mice with the experimenter was assessed. The evaluation was conducted in the home cage immediately after the second tumour measurement. Both mice from each cage were gently conducted to one side of the cage with a piece of cardboard. Due to cage design, mice were directed underneath the food hopper, so this side of the cage was the least exposed for them. Then, the experimenter placed her hand on the opposite side of the cage and the cardboard was removed. The number of contacts with their head or paws was registered for a 3-minute period. We inferred that by using a less aversive method of tumour measurement, mice would be less anxious and, therefore, more willing to leave the safer zone in their home cage to explore the experimenter’s hand.
Statistical analysis
Animals were randomly assigned to different cages upon arrival at the experimental room by means of a random number sequence generated in Microsoft Excel; in addition, cages were assigned to different treatments and selected for data collection at the different time points in the same modality. Statistical analyses were performed with R 3.3.0, R studio software (2016) and InfoStat (2015). Data was tested for normality using the Shapiro-Wilks normality test. P-values below 0.05 were considered significant for all analyses. Results were expressed by means ± SEM.
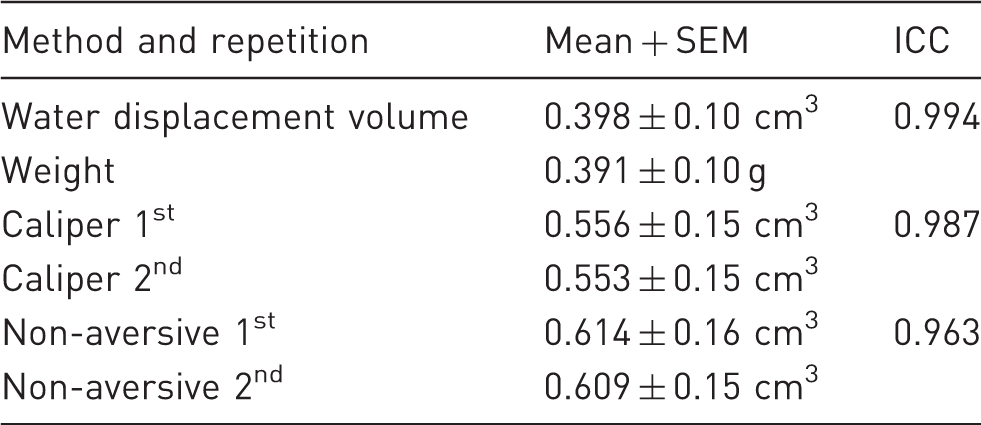
To measure intra-observer reliability in experiment 1, the Intra-Class Correlation Coefficient (ICC) was applied. We considered values greater than 0.75 as representative of excellent agreement between the first and the second repetition. 24 To compare across methods, the nonparametric Friedman test with Dunn’s post hoc test for multiple comparisons were used. Behavioural data was analysed with the non-parametric Kruskal Wallis test. Blood-glucose concentration was evaluated by Analysis of Variance (ANOVA).
In experiment 2, the differences in corticosterone concentration as well as the frequency of voluntary contacts to the experimenter between both methods were analysed on a weekly basis with 2-Way ANOVA. As we did not separate the animals to measure behaviour or to collect faecal samples, the cage was considered the experimental unit. To meet the test assumptions, corticosterone concentration in faeces was logarithmically transformed and the number of contacts with the experimenter was square root transformed. Results from treatments showing significant overall changes were subjected to post-hoc Tukey’s test.
Results
Experiment 1
Intra-Class Correlation Coefficients (ICC).
Latency to eat the almond piece in the modified version of the NSF test was significantly shorter for mice in the non-aversive photographic method group compared to those measured with the caliper (Figure. 2(a)). Similarly, frequency of grooming (Figure 2(b)) and of defecation (Figure 2(c)) were significantly lower in mice whose tumours were measured with the non-aversive photographic method. H values were 3.53 (P = 0.049) for the NSF test, 8.17 (P = 0.002) for the frequency of grooming, and 7.94 (P = 0.002) for the frequency of defecation.
Behavioural output observed immediately after both tumour measurement methods. Anxiety-like behaviour expressed as (a) the latency to eat the almond piece in the modified-novelty suppressed feeding (NSF) test, (b) frequency of grooming and (c) defecation during a 300-s period (n = 10 mice/group). Kruskal-Wallis test; *=P < 0.05; **P < 0.01.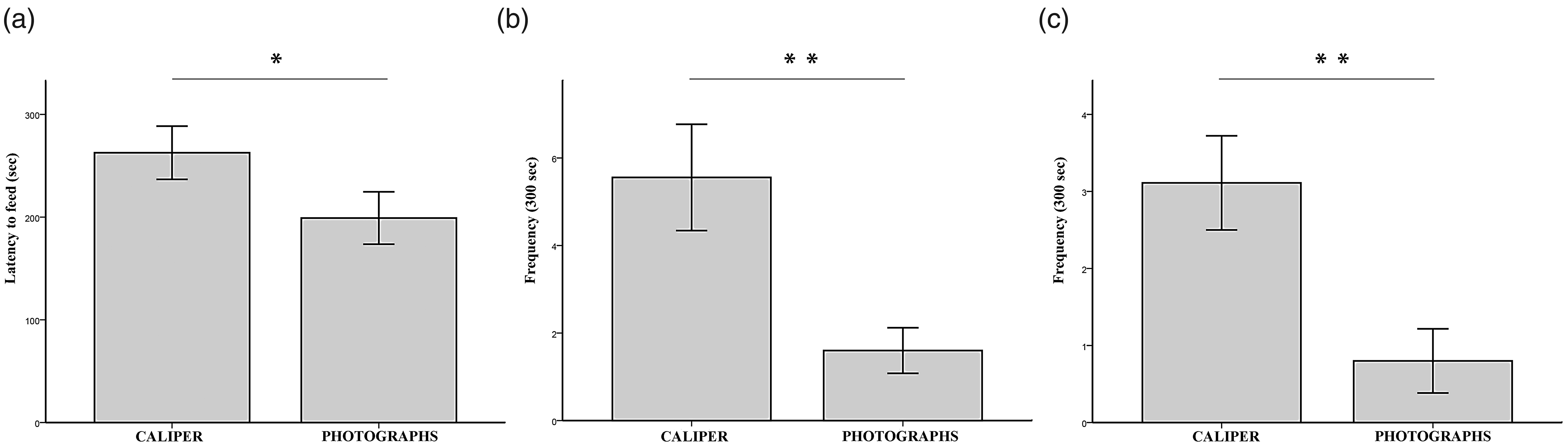
Blood-glucose concentration was slightly higher in the caliper group (142.33 ± 6.09 mg/dL) compared to the non-aversive photographic method group (130.70 ± 6.1 mg/dL) but these differences were not statistically significant (F1,18 = 1.81; P > 0.05). Finally, as expected, the non-aversive photographic method was more time consuming, given that it demanded 71.40 ± 10.73 s per animal v. 20.22 ± 1.62 s per animal when the caliper was used. In addition, image processing in the non-aversive photographic method took 98.10 ± 5.17 s per animal which included scale calibration per image, the measurement of the tumours, adding the data to a database and occasionally, enhancing the image contrast to have a better tumour delimitation.
Experiment 2
The number of voluntary contacts with the experimenter as well as faecal concentrations of corticosterone obtained with both methods are shown in Figure 3. No significant interaction was observed between factors. Voluntary contacts of mice with the experimenter were measured as shown in Figure 3(a). Significant differences were found between treatments (F1,64 = 17.53, P < 0.001) and among weeks (F3,64 = 7.21, P < 0.001). From week 2 and until the end of the study, mice that were exposed to the non-aversive photographic method were less reluctant to establish contact with the experimenter than those whose tumours were measured with the caliper. Significant differences in corticosterone concentration were observed across weeks (F3,64 = 6.13, P < 0.001) but not between methods (F1,64 = 0.69, P = 0.40) as shown in Figure 3(b).
Behavioural and physiological parameters over time in mice subjected to both tumour measurement methods. (a) Temporal sequence of mice–human interaction expressed as number of contacts in a 3-min period (n = 19 mice/group). (b) Temporal sequence of corticosterone concentration in faeces (n = 19 mice/group). Two-way Analysis of Variance; *=P < 0.05; **P < 0.01.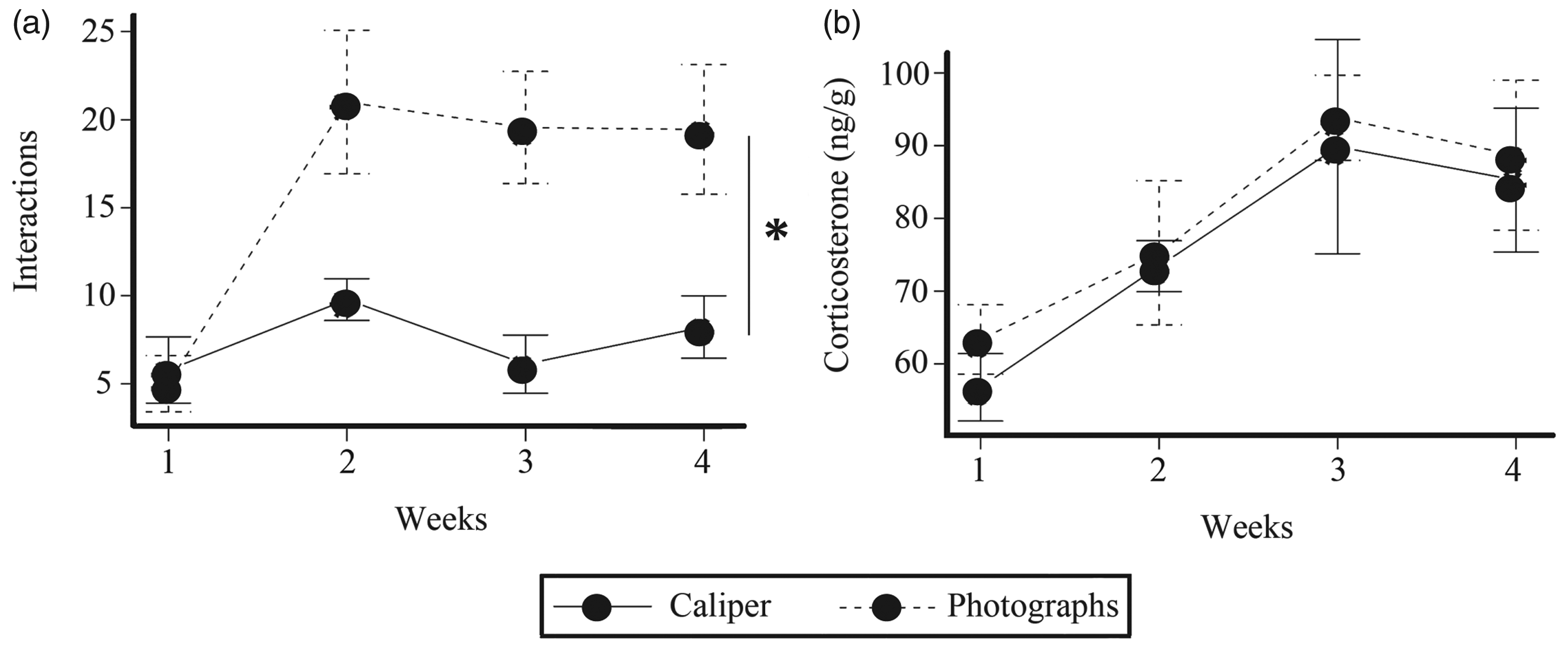
Discussion
To our knowledge, the non-aversive photographic method has been described here for the first time as a technique to measure subcutaneous tumours that does not require animal immobilization. The need for a method of these characteristics has been recently addressed by Delgado San Martin et al. 3 The technique described herein can be done in single- or paired-housed mice in their own home cage, without the need to manipulate animals to record tumour development over time. In this experiment, we identified each mouse by the side on which the tumour was implanted (left or right). Tumour measurement with the non-aversive photographic method in mice housed in groups larger than pairs would also be possible, as long as the mice are clearly identified. Moreover, even if measurement in the home cage is not possible, reduced anxiety was observed in the NSF test, after mice were cup-handled to another cage where they were presented to the non-aversive photographic method.
As it was expected for well-vascularized and non-calcified tumours, weight yielded the closest value to water displacement volume. 2 Both the caliper and the non-aversive photographic method produced measurements different from those obtained using the reference method, most likely because the former also considered the skin and subcutaneous tissue. 5 Even though caliper measurements were closest to water displacement volumes, there was good consistency between repetitions for both techniques, which would make the non-aversive photographic method acceptable in recording tumour development over time. As the non-aversive photographic method tends to overestimate the size of tumours, pilot studies prior to its implementation are recommended to determine which tumour size is acceptable when setting experimental endpoints. For this purpose, this new method should be compared with modern in vivo imaging techniques to overcome the issue of including additional tissues in the determination of tumour volumes. Indeed, this might also aid improving guidelines for mice welfare in cancer research as these do not specify which method they are based on and hence, can have profound implications when setting experimental endpoints.
By means of physiological and behavioural parameters, we evaluated the impact of both methods on mice welfare. The overall interpretation of animal welfare based on objective measurements is challenging given the intrinsic subjectivity of the nature of an animal's affective state. 25 Therefore, attention should be placed on the context and the behavioural responses that take place along with physiological data.
Although no significant differences were observed neither in faecal corticosterone nor in blood-glucose concentrations between both methods, the behavioural output for the non-aversive photographic method was drastically different, as mice were less reluctant to establish contact with the experimenter and behaved less anxiously in the NSF test. The short-term stress response to one brief event of restraint may not be visible in corticosterone metabolites over a 24-hour period and thus further studies should analyse long-term changes for this parameter. 26 However, the similarity with blood-glucose concentration might hint a similar physiological response between both methods, as sympathetic activation, which rapidly increases circulating glucose, anticipates the hypothalamic-pituitary-adrenal (HPA) response. 27 Nevertheless, by focusing on the behavioural data, improved welfare due to a reduction in anxiety for the new method can be hypothesized, as mice in the non-aversive photographic method group ate the almond piece significantly faster in the NSF test, were less reluctant to establish contact with the experimenter and displayed significant less grooming and defecation than mice in the caliper group. Both grooming and defecation are behavioural measures extensively used as anxiety traits.28,29,30 We infer that the non-aversive photographic method apparatus did not induce a highly-aversive response, as all the animals decided to enter into the tube. These results are in accordance with Hurst and West 31 , who established that picking up mice by the tail induced aversion and high anxiety, whereas the use of tunnels led to voluntary approach, low anxiety and acceptance of the physical restraint.
Overall, it can be predicted that it will be easier for mice to adapt to this new technique and hence, will produce less variability in experimental variables due to uncontrollable stress exposure. The non-aversive photographic method is also rather inexpensive, as only a transparent acrylic tube and a camera are needed. In our experiment, we used a NIKON DSLR D3100 camera but any good resolution camera can be employed. In addition, even though this technique is not time-consuming (approximately one minute per tumour), we infer that as mice get used to the procedure the time required will be reduced. Image processing time was also short for this new method as it took about 100 s to analyse each picture. We did not consider picture-selection time, but a proper training in image shooting can considerably reduce the amount of pictures taken per animal and hence minimize this aspect. Finally, this new technique can also be used in immunodeficient furred mice, provided the area surrounding the tumour is properly shaved.
Altogether, strong behavioural evidences of diminished anxiety responses were found for the non-aversive photographic method in this initial approach, which might help improve mice welfare in cancer research. Nevertheless, the lack of a physiological correlate for these findings highlights the importance of performing further research to characterize mice physiological profile. In particular, with regards to blood corticosterone concentration after the measurements, our experimental design only let us infer a similar association with blood glucose with no effect in the long-term release of this hormone.
Conclusion
As it has been mentioned before, we are describing here for the first time a technique to measure tumour volumes without the need for manipulating the animals and which can also be performed in the mouse’s home cage. We also conclude that the non-aversive photographic method is consistent across repetitions. This new technique could be helpful in cancer research, as it could reduce experimental variables related to stress, which negatively influence results. 32 As additional benefits, this method is economical and easy to conduct, so it can be easily carried out after a short training. Even though the non-aversive photographic method is slightly more time consuming, mice behaved less anxiously when this new technique was used, which leads us to the conclusion that its implementation can have a positive impact on their welfare. Altogether, the non-aversive photographic method might help to reduce unnecessary stress, particularly in studies that consider the influence of psychoneuroimmunology in cancer development.
Footnotes
Acknowledgements
We would like to thank Dr Vera Baumans and Dr S. Ivan Perez for their thoughtful comments on this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AR is funded by a PhD grant from CONICET. ACC received funding from a postdoctoral grant from CONICET. SLD is supported by an Independent Research Career Position from CONICET. CC and MAA have teaching positions at FCV-UNLP. This study was funded by a grant to CC from the Argentine Agency for Promotion of Science and Technology (ANPCyT - PPL2-2011-0008).
