Abstract
The turquoise killifish Nothobranchius furzeri is an increasingly popular model species for comparative vertebrate research, and the basic physiology including responses to stressful stimuli are of primary interest. We exposed adult killifish to a single or repeated periods of acute confinement followed by analysis of tissue cortisol and plasma cortisol concentrations. Individuals were also sampled for messenger RNA (mRNA) expression of corticotropin-releasing hormone (CRH), mineralocorticoid receptor (MR), and glucocorticoid receptor (GR) in the brain to examine the effects of repeated stress events on constitutive expression of these important stress axis components. Following a single 30-minute confinement stress, male plasma cortisol significantly differed from baseline (p = 0.04). Both male and female whole-body cortisol were significantly increased (p = 0.004 and p = 0.04, respectively) at 15 and 30 minutes poststress. Despite obvious dimorphic behavior and morphology, cortisol concentrations did not differ between the sexes. Exposure to daily repeated confinement for one week altered the cortisol response in both sexes. Time 0, 15, and 60 minutes poststress cortisol concentrations were depressed in repeatedly stressed males (p ≤ 0.05), and times 0, 30 and 120 minutes poststress cortisol concentrations were depressed in repeatedly stressed females (p ≤ 0.05). Constitutive expression of CRH, MR, and GR mRNA in the brain following one week of repeated stress events did not differ among treatments or sexes. This study introduces the first description of hypothalamic-pituitary-interrenal axis activity in this important model species. Reduced cortisol production in repeatedly stressed adult killifish suggests acclimation to repeated stressors. Furthermore, acclimation was rapid, and plasma cortisol concentrations altered significantly in as little as one week.
Introduction
The turquoise killifish Nothobranchius furzeri is endemic to semi-arid regions of southeastern Africa. There, rain is restricted to just a few months per year, and N. furzeri inhabit ephemeral pools created as water drains to lowland plains. The nature of this habitat has led to a unique adaptation in which the process of development is extremely rapid. As the temporary pools dry, eggs become encased in dry soil, where they can remain in diapause for more than a year. Developed embryos are triggered to hatch by accumulation of moisture as the next rainy season proceeds. Newly hatched fry then rapidly advance to maturity, with males showing nuptial coloration in as few as 28 days posthatch.1–3
Turquoise killifish belonging to the gona-rhe-zhou (GRZ) strain currently have the second shortest lifespan of any known vertebrate. 4 Compared to more prevalent and similarly sized model teleosts, like the medaka Oryzias latipes and zebrafish Danio rerio, turquoise killifish develop to maturity in a fraction of the time, and after only three months from hatching, adult killifish show accumulation of aging biomarkers leading to death by multiple organ failure. 5 Rapid development, symptoms of aging similar to longer-lived vertebrates, and facultative diapause observed in developing embryos, are a few reasons GRZ N. furzeri constitute an opportune model organism for comparative studies. The increasing use of N. furzeri in scientific research calls for a better understanding of their physiology, including the effects of stress.
The hypothalamic-pituitary-interrenal (HPI) axis is a widely accepted indicator of stress in fish due to consistent increases in the teleost glucocorticoid (GC), cortisol, following stimuli that threaten homeostasis.6,7 Once released into circulation, cortisol binds intracellular glucocorticoid receptors (GRs) and mineralocorticoid receptor (MRs) expressed in many tissues. The receptor complexes then bind to DNA response elements to alter transcription.8–13 Perhaps the most studied aspect of GC function is the effect on metabolism. High levels of GCs seen during a stress response actively inhibit glucose uptake while stimulating gluconeogenesis in the liver. In teleosts, another important function of cortisol is hydromineral balance. Increased blood circulation induced by stressors often results in distension and recruitment of gill lamellae, thereby increasing the surface area subject to osmotic and ionic regulation. Cortisol is believed to act at the gills to promote chloride cell proliferation and ion-transporting enzyme production to combat flux during stress.13,14 Relative to the immediate actions of the catecholamines and glucagon, GCs work to enhance and prolong increases in blood glucose needed to fuel increased metabolic demand.15,16
Both laboratory and husbandry practices subject fish to frequent stressors. To make efficient use of available space, species are often kept in unnaturally high densities, causing social stress and concentrating waste products. The accumulation of biological waste may make maintaining stable water parameters difficult. Furthermore, people often net, transport, handle, or otherwise interact with captive fish in ways that elicit stress.17–19 Chronic or repeated exposure may alter HPI axis responsiveness to future stressors, as changes in GC production and receptor density have been reported.14,17,20–22
Chronic stress may also manifest deleterious effects on other physiological systems. For example, the reallocation of energetic substrate initiated by cortisol reduces investment in growth, reproduction, and immunity.16,17 Elevated GCs may also disrupt function of the hypothalamic-pituitary-gonadal (HPG) axis and decrease lymphocyte and cytokine populations, whereas increased corticotropic-releasing hormone (CRH) suppresses appetite and feeding. 16 Furthermore, chronically elevated cortisol can have a number of adverse effects on signaling within the brain and is often implicated in psychiatric disorders in higher-order vertebrates. 23 For these reasons, the HPI axis is intimately correlated with general health and well-being of laboratory fish. Therefore, an understanding of how laboratory and husbandry practices affect the HPI axis and downstream physiology of captive fish species should be a priority, especially for species used as models in biomedical research.
Turquoise killifish not only mature and age rapidly, but the sexes are highly dimorphic both in morphology and behavior.3,24 Males often reach sizes of 6–8 cm compared to females averaging around 5 cm. 3 Females develop little to no coloration, but upon sexual maturation males take on vibrant nuptial coloration and elongated dorsal and anal fins. Maturation of males is also accompanied by aggression toward conspecific males and pursuit of females, while females remain docile in comparison. 1 Together with reports that sex and reproductive status affect the stress response in mammals,25–27 these differences suggest turquoise killifish sexes might also exhibit different physiological responses to stress. Little research has been conducted in this regard in fish. 28 We describe sex-specific HPI axis activity of adult turquoise killifish before and after introduction to a novel confinement stressor, including alterations in tissue and plasma cortisol as well as expression of stress axis genes (CRH, MR, and GR) in the brain, following repeated stress exposure. N. furzeri plasma and whole-body cortisol concentrations are reported for the first time.
Material and methods
Killifish husbandry
Eye-up embryos were received from Stanford University and incubated in coconut fiber dampened with Yamamoto’s ringer. Upon reaching the golden-eye stage, embryos were transferred to approximately 200 ml tubs filled with hatching media (1 g/l humic acid in system water). Fry were raised in accordance with previously outlined protocols.1,29
At approximately one-day posthatch (dph), twice-daily feedings of Artemia nauplii commenced. At 10 dph, growing fry were transitioned to Otohime dry feeds (Marubeni Nisshin Feed Co, Tokyo, Japan). By three weeks posthatch (wph), the subadult killifish were randomly assigned (five or six fish per tank) to 10-l glass aquaria connected to a recirculating aquaculture system (RAS) with a 200-gallon sump, 2-inch Bio-Barrels (Pentair Aquatic Eco-Systems Inc, Apopka, FL), and Aqua Ultima II Pressurized Bead Filter (Aqua Ultraviolet, Temecula, CA). System water was maintained at 25° ± 1℃, alkalinity of 100–150 parts per million (ppm), hardness of 100–150 ppm, and a salinity of<0.5 parts per thousand. Ammonia and nitrite were consistently <0.01 ppm and nitrates <100 ppm. At around four dph, males began to develop nuptial coloration and spawning groups were formed by placing each male with four or five females. Groups were provided spawning substrate in the form of fine glass beads and maintained until experimental manipulations at about 10 wph. Fish numbers used in this experiment were chosen to reduce aggression and meet minimum numbers for detecting statistical differences. All fish husbandry was performed in the Aquatic Research Laboratory (not Association for Assessment and Accreditation of Laboratory Animal Care International accredited) at Southern Illinois University. Animal numbers and procedures were approved by the Institutional Animal Care and Use Committee of Southern Illinois University (Protocol #15-022).
Stress test
Two weeks prior to sampling, males (1.25 ± 0.07 g; mean ± SEM) and females (0.48 ± 0.03 g) were moved in groups of six to sex-specific tanks (12 10-l glass aquaria; six per sex) filled to 2.5 l of water on the RAS system. Four distinct treatments were randomly assigned to three tanks per treatment: single-stress (SS) males, SS females, repeated-stress (RS) males, and RS females. One week prior to sampling, RS treatments were exposed to a daily confinement stressor for seven consecutive days. The stressor consisted of reducing the volume of each aquarium to 0.78 l for males and 0.3 l for females, to maintain similar densities because of size differences, and using a perforated crowder to confine all the fish to one end of the tank, yielding a stocking density of approximately 100 mg/ml. The fish remained confined for 30 minutes, followed by removing the crowder and refilling each tank to its original volume. During the stressor, tanks were aerated but off the RAS system until refilled.
On the day of sampling, all treatments were exposed to the confinement stressor. Prior to confinement one fish was removed from each tank for prestress cortisol and constitutive gene expression. Another fish was removed from each tank at 15, 30, 60, and 120 minutes after confinement was initiated. Blood was collected from the caudal vein of large males into heparinized capillary tubes for plasma cortisol quantification. Sufficient blood collection was not possible from the smaller females. Fish destined for whole-body cortisol extraction were placed into 2-ml vials, while excised tissues were immediately placed in 1 ml of Tri Reagent® RT (Molecular Research Center, Ann Arbor, MI). All sampled fish were quickly euthanized using a buffered solution of MS-222 (Western Chemical Inc, Ferndale, WA). Whole fish and tissues were flash-frozen by immersion in liquid nitrogen and stored at –80℃ until further analysis.
Whole-body cortisol extraction
Each fish was partially thawed, patted dry, and weighed. Females were placed individually into 12 ml glass vials with 0.5 ml of 1× phosphate-buffered saline (PBS) (4℃). The much larger males had to be split into two vials that were recombined at a later step. The combination of tissue and PBS was then homogenized using a rotor-stator homogenizer for one minute. The blade was then rinsed with another 0.5 ml of 1× PBS (4℃), which was added to the homogenate.
Diethyl ether (5 ml) was then added to each homogenate vial, vortexed for one minute, then centrifuged for five minutes at 3000 rpm at 4℃. After storage at –80℃ for two hours, the ether was decanted into a separate vial. At this point, the cortisol extracted from each male was recombined by pouring the ether extract from both homogenate vials into one new vial. The freezing and decanting process was repeated using another 2 ml of diethyl ether. After overnight ether evaporation, the cortisol fraction was reconstituted by adding 4 ml of 1× PBS to each vial and incubating at 4℃ for approximately 24 hours. Finally, samples were moved to –80℃ for storage preceding further analysis.
Cortisol enzyme immunoassay (EIA)
Both plasma and whole-body cortisol concentration was measured using cortisol EIA kit # 500360 (Cayman Chemical, Ann Arbor, MI). Following the recommended protocol, each sample was run in duplicate and compared to an intraassay standard curve. The EIA was validated for this species using a parallelism and spike and recovery method on pooled tissues from four additional male killifish. Half of the pooled tissue was processed as previously described, and the remainder was spiked with an additional 500 pg/ml of cortisol standard prior to homogenization. Recovery was >90% and serial dilution of pooled samples was parallel to the standard curve (p > 0.05). Cortisol concentrations were calculated relative to the standard curve.
Gene selection and primer design
Oligo-primers for Nothobranchius furzeri target and reference gene amplification using reverse transcription-quantitative polymerase chain reaction analysis.
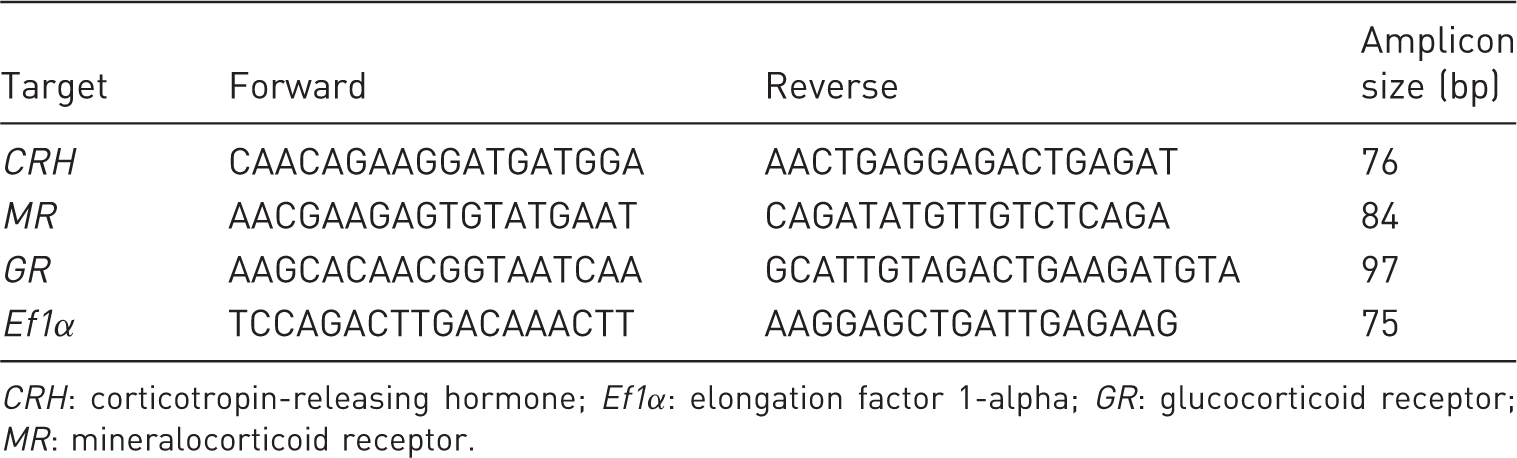
CRH: corticotropin-releasing hormone; Ef1α: elongation factor 1-alpha; GR: glucocorticoid receptor; MR: mineralocorticoid receptor.
RNA isolation, quantification, and complementary DNA (cDNA) conversion
Cranial samples including intact brain and minimal surrounding tissues were collected from euthanized fish with a scalpel. Total RNA was isolated from the cranial brain samples in Tri Reagent® RT (Molecular Research Center Inc). Total RNA concentration and sample purity were assessed by measuring the absorbance at 260 nm and 280 nm with a NanoDrop® 2000 spectrophotometer (Thermo Scientific, Wilmington, DE). All samples analyzed had a 260/280 ratio≥1.80. One microgram of total RNA was used to produce cDNA using a High-Capacity cDNA Reverse Transcription Kit (Applied Biosystems, Foster City, CA).
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Relative quantification of messenger RNA (mRNA) expression was performed by real-time qPCR using the SsoFast™ EvaGreen® Supermix protocol on a CFX-96 real-time thermocycler (BioRad Laboratories, Hercules, CA). Gene-specific forward and reverse primers (0.5 µl at 10 µM each), SsoFast™ EvaGreen® Supermix (5 µl per reaction), and RNase/DNase-free water (3 µl per reaction) were added to 1 µL of cDNA. Cycling parameters consisted of: 95℃ for three minutes, followed by 40 cycles of 95℃ for 15 seconds and 55℃ for 15 seconds. Melt curves were performed following amplification for data validation. For this experiment, EF1α served as the reference gene and relative expression was quantified using the 2–ΔΔCt mathematical model, relative to prestress (time 0) expression. 30
Statistical analyses
Datasets were first tested for homogeneity of variance using Spearman rank-order correlation coefficient (p > 0.05), and normality using the Shapiro-Wilk test (p > 0.05). When necessary, datasets were transformed to meet these assumptions for the analysis of variance (ANOVA). Changes in cortisol concentration over time within each treatment were analyzed by one-way ANOVAs followed by testing using the Fisher least-significant-difference method. Differences between treatments at each time point were analyzed via pairwise student t tests. All statistical tests were performed using SAS 9.4 software (SAS Institute Inc, Cary, NC). With respect to whole-body cortisol and gene expression, a probability of p ≤ 0.05 was considered significant. A probability of p < 0.1 was considered significant for differences in plasma cortisol since analytical dilutions likely increased sample variation and a biologically relevant cortisol stress response was observed.
Results
Plasma cortisol
There was an overall effect of time on male plasma cortisol concentration resulting from exposure to a crowding stress (F4,10 = 3.29; p = 0.06). Plasma cortisol concentration was significantly (p = 0.04) elevated at 30 minutes compared to that of prestress (0 minutes) fish. At 60 minutes, plasma cortisol concentration had declined and was no longer significantly different (p > 0.1) from prestress values (Figure 1).
Plasma cortisol content (mean ± SEM, n = 3) of adult male turquoise killifish following a single acute stressor. Different letters indicate significant differences across time (p < 0.1).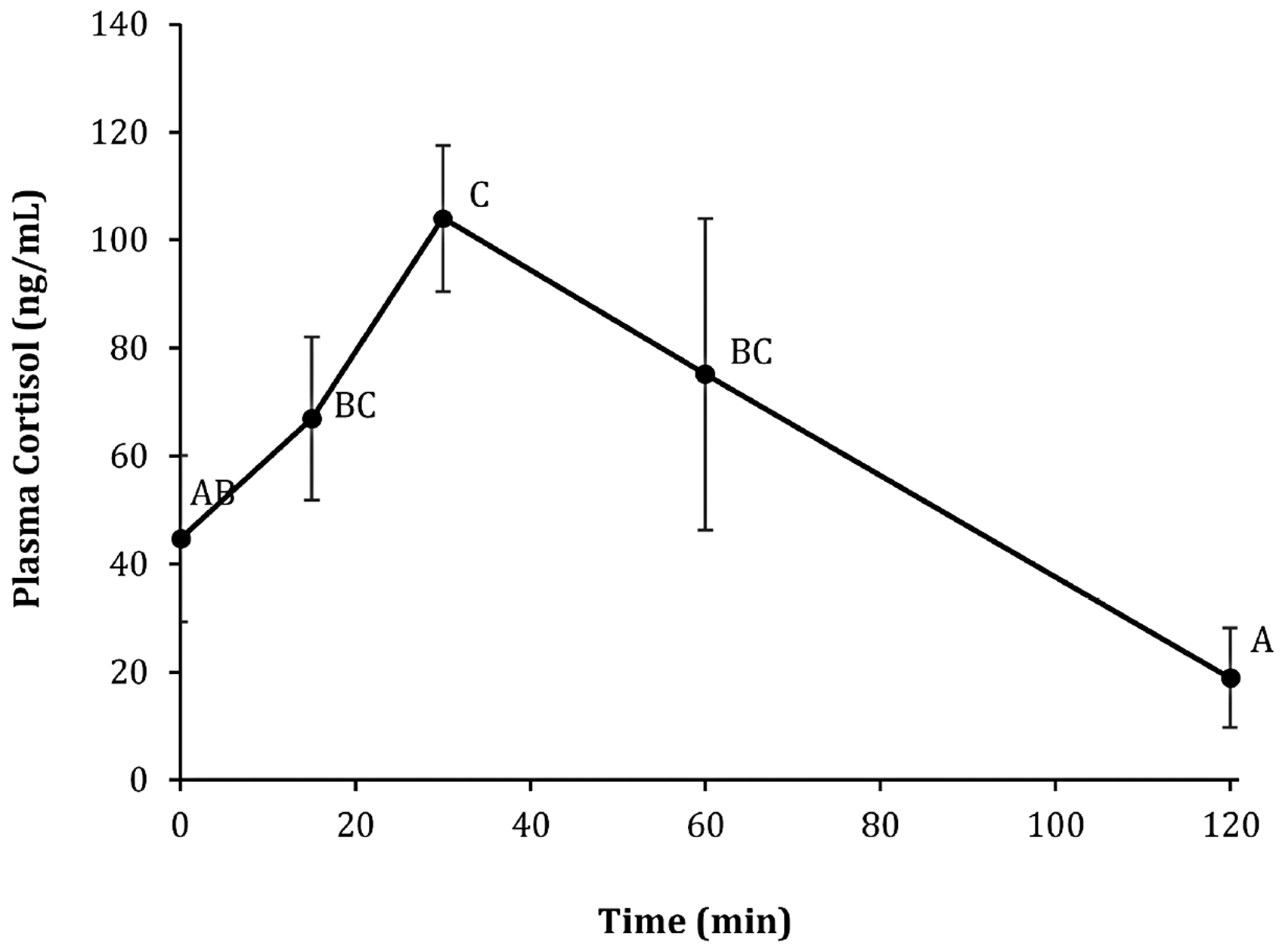
Whole-body cortisol
There was a significant effect of time both in SS males and females (F4,10 = 7.85; p = 0.004 and F4,10 = 3.66; p = 0.04, respectively). By 15 minutes, whole-body cortisol was significantly elevated above time 0 (prestress) concentrations and remained elevated at 30 minutes in both sexes. Whole-body cortisol concentrations declined and were no longer different from prestress values at both 60 and 120 minutes poststress. There were no significant differences in cortisol concentrations between the sexes at any time point measured before or after stress exposure (Figure 2).
Whole-body cortisol content of adult male (♂) verses female (♀) turquoise killifish (mean ± SEM, n = 3) following a single acute stressor. Different letters indicate significant differences (p ≤ 0.05) across time within treatments. There are no significant differences between sexes at any time (p > 0.05).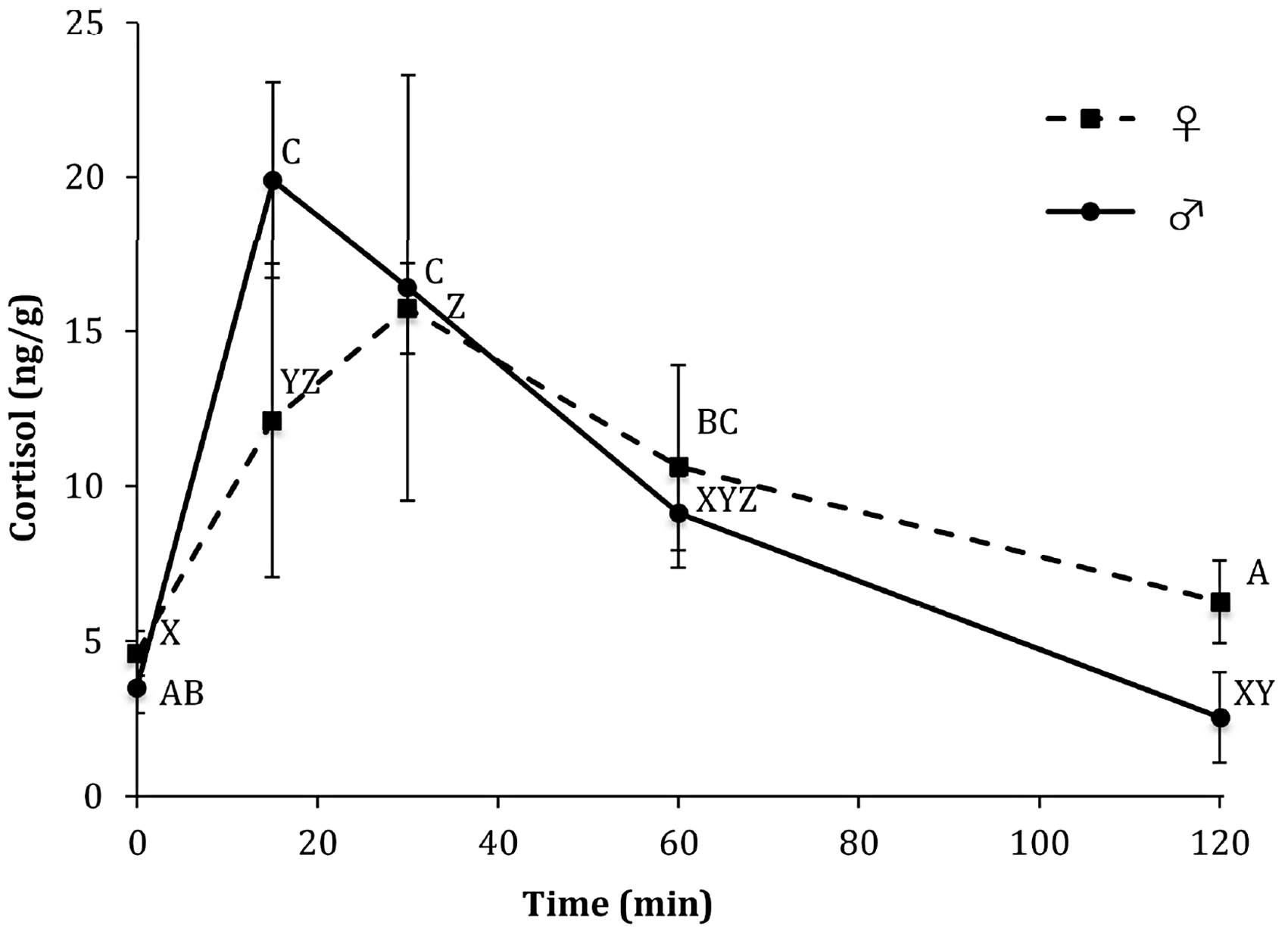
Both RS treatments in adult male and female fish had significant cortisol increases following confinement exposure on the sampling date (F4,10 = 78.27; p < 0.001 and F4,10 = 4.8 7; p = 0.02, respectively). However, RS treatments differed from their SS counterparts. There was a significant difference in cortisol content between the SS and RS treatments in males at time 0 (F1,4 = 9.25; p = 0.04), 15 minutes (F1,4 = 3.21, p = 0.05), and 60 minutes (F1,4 = 28.7; p = 0.006) after stress exposure, with the RS treatment having lower cortisol concentrations at these time points (Figure 3(a)). Similarly, there was a significant difference in cortisol content between the two female treatments, SS and RS, at time 0 (F1,4 = 0.41; p = 0.05), 30 minutes (F1,4 = 9.19; p = 0.04), and 120 min (F1,4 = 9.15; p = 0.04) after stress exposure. As in males, female cortisol content was lower at these time points in the RS treatment (Figure 3(b)).
Whole-body cortisol content (mean ± SEM, n = 3) of adult (a) male (♂) and (b) female (♀) turquoise killifish following single-stress (SS) and repeated-stress (RS) events. Different letters indicate significant differences (p ≤ 0.05) across time within treatments. Asterisks indicate significant differences between treatments at a given time point (p ≤ 0.05).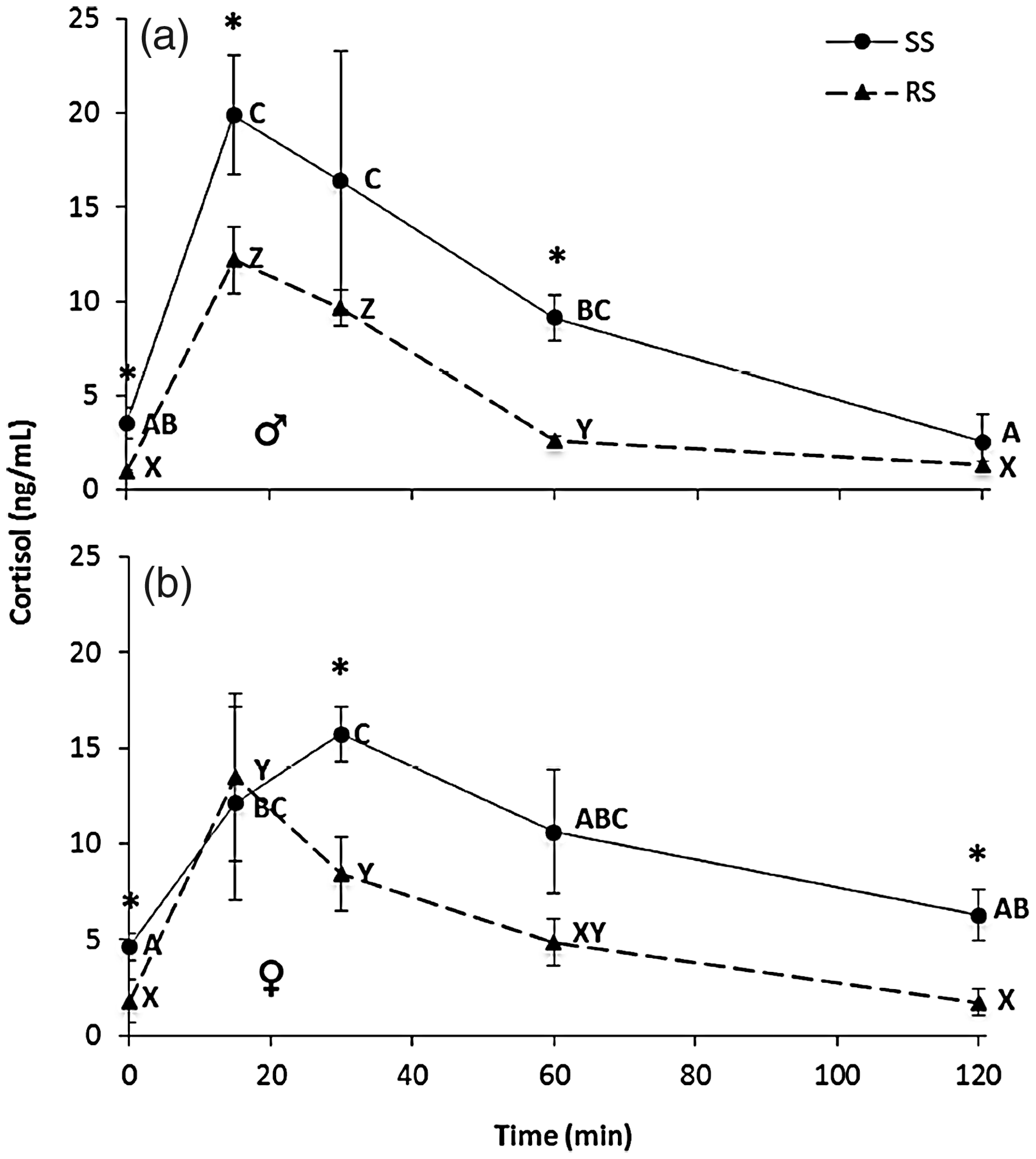
Gene expression
There were no significant differences in constitutive expression of brain CRH, GR, or MR mRNA in male or female killifish previously exposed to RS events compared to nonstressed fish (Figure 4).
Brain corticotropin-releasing hormone (CRH), mineralocorticoid receptor (MR), and glucocorticoid receptor (GR) expression (mean ± SEM, n = 3) in male (♂) and female (♀) killifish following repeated-stress (RS) events for one week compared to nonstressed (NS) fish. No significant differences were reported (p > 0.05).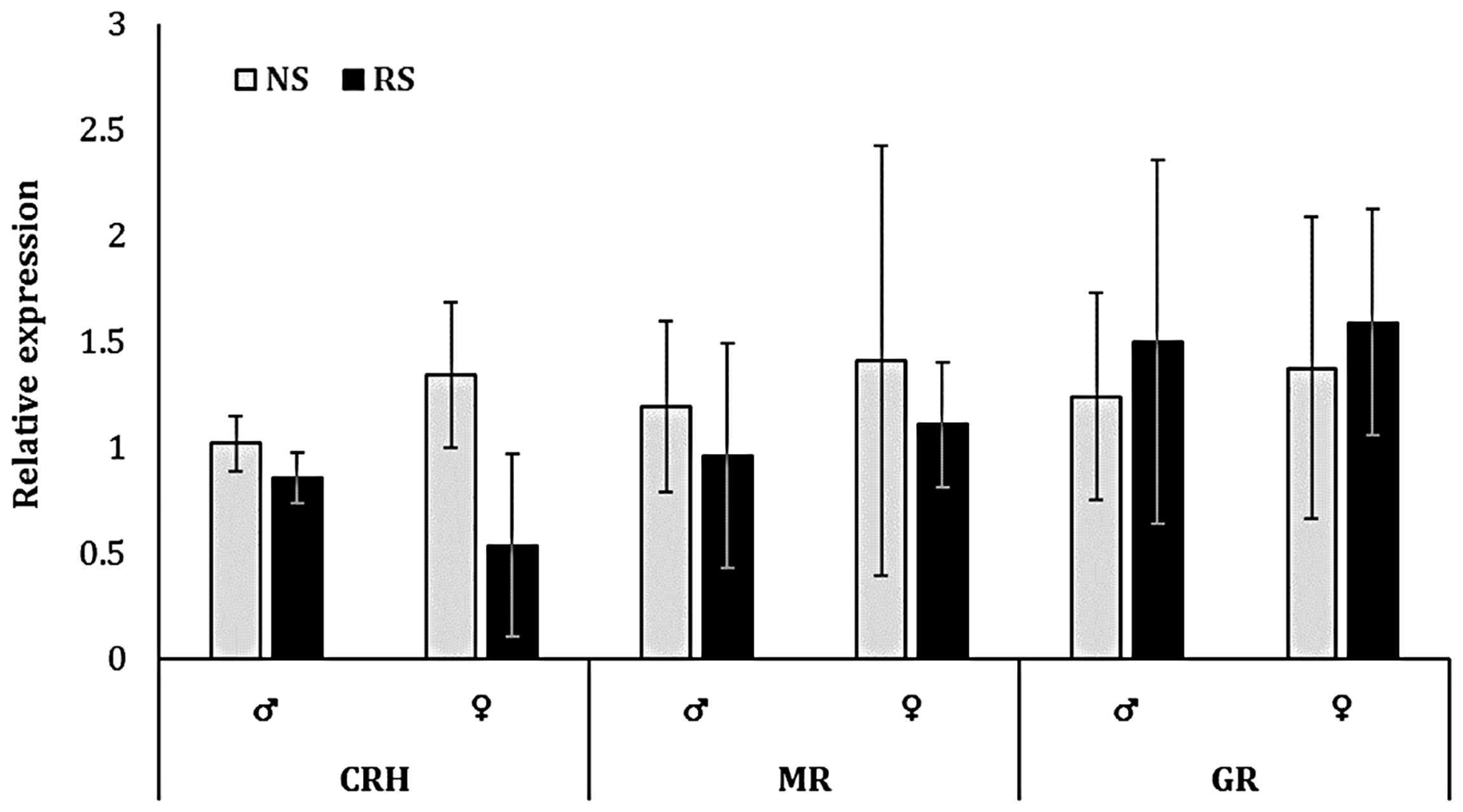
Discussion
Our study reports the first known description of a cortisol response in the turquoise killifish. Prestress (time 0) plasma cortisol values averaged almost 45 ng/ml, markedly higher than reported values for many teleosts. For example, bull trout Salvelinus confluentus prestress cortisol was reported at 8.1 ng/ml, while other trout species including rainbow trout Oncorhynchus mykiss and brown trout Salmo trutta had prestress values well below 5 ng/ml.14,31,32 Additional studies also report much lower basal cortisol values for species of sturgeon, paddlefish, walleye, and perch.33–36 Relatively high prestress plasma cortisol could be due to the initial difficulty in collecting enough blood from this small fish, with recovery values averaging 9 ng/ml at 120 minutes poststress likely reflecting more accurate basal plasma cortisol concentrations.
Following confinement stress, N. furzeri plasma cortisol concentrations peaked at around 100 ng/ml. Despite considerable variability both within and between species, these results are congruent with reports for well-studied salmonids, having peak poststress cortisol concentrations between 40 and 150 ng/ml.14,32,36 Plasma in some species, however, may reach a much greater maximum at nearly 200 ng/ml and 400 ng/ml for Eurasian perch Perca fluviatilis and adult jundia Rhamdia quelen, respectively.10,36
The whole-body cortisol values we have reported for N. furzeri are perhaps most similar to those reported for adult zebrafish Danio rerio before and after induced stressors. Prestress whole-body cortisol averaged 4 ng/g in N. furzeri compared to 2–4 ng/g in zebrafish. 37 Both turquoise killifish and zebrafish show similar peak concentrations between 20 and 30 minutes poststress with averages around 15–16 ng/g.37,38
With respect to HPI axis activity, adult turquoise killifish do not appear to be sexually dimorphic. While a 30-minute confinement stress induces a robust cortisol stress response, adult male and female cortisol production seems to reach a comparable maximum and decrease over time. Similarly, the finding of no differences in receptor expression between the sexes runs contrary to our hypothesis, given the breadth of obvious morphological and behavioral distinctions.1,3 Life span is also not different between the two sexes despite marked sexual dimorphism in behavior and morphology.
Sex steroids, estradiol and 11-ketotestosterone, are known modulators of stress in mammals,25–27 and potential modulators in fish. However, little research has been conducted on the interactions of the HPG and HPI axes with fishes, and those that have been reported are often equivocal. 28 In two species of sexually mature salmonids, a study reports significantly higher sensitivity of female interrenal cells to exogenous adrenocorticotropic hormone (ACTH) and a greater ability to utilize pregnenalone as a substrate, suggesting the potential for greater cortisol production in mature females vs males. 39 Similarly, maturing female rainbow trout Oncorhynchus mykiss exhibited prolonged elevations in plasma cortisol while confined, compared to maturing males. 40 In contrast, while basal cortisol was higher in juvenile Chinook salmon females than males, when both were exposed to a chemical stressor only male cortisol significantly increased. 41 This agrees with a previous study using sockeye salmon Oncorhynchus nerka, in which basal cortisol was greater in females than males, but confinement induced an increase in cortisol only in males.42,43 In the zebrafish, no evidence of a sexually dimorphic cortisol response was observed. 38
Relevant literature reports contrasting results with regards to the effects of chronic or RS on activity of the HPI axis in teleosts. In the present study, the magnitude of the cortisol response did not differ between RS and SS killifish, but prestress (time 0) concentrations differed between male treatments, and recovery from stress exposure was different both for males and females. Several analogous studies of repeated acute exposure on teleosts yielded comparable cortisol results. Eurasian perch greatly decreased peak cortisol response to acute handling and returned to prestress concentrations more rapidly after eight weeks of repeated exposure. 36 Rainbow trout also responded with a marginal decrease in peak cortisol after acute handling, and a faster return to baseline. 36 Similarly, juvenile rainbow trout exposed to acute stress daily for four weeks resulted in a decreased cortisol response following an additional acute stressor. 44 Similar changes have been observed in other teleosts chronically stressed by high stocking density. For example, densely stocked brook charr Salvelinus fontinalis had significantly decreased plasma cortisol relative to lower-density groups after 30 days. 45 Stocking density also blunted responses to subsequent stimuli in gilthead sea bream Sparus aurata. 46 Although supportive of our results, these studies demonstrate the effects of longer-term habituation to stressors than the one-week repeated stressor administered in the present study. It is possible that acclimation to stress may scale with species life span; however, more research is needed to address this possibility.
In contrast to the studies discussed above, multiple studies report different responses in cortisol production due to high stocking density in fish. In gilthead sea bream, red porgy Pagfrus pagrus, zebrafish, Senegalese sole Solea senegalensis, and various salmonids, high stocking density was concurrent with increases in cortisol content.37,47–50 Furthermore, studies investigating the effects of stocking density on European sea bass Dicentrarchus labrax found that high-density treatments had significantly greater cortisol responses to additional acute stressors than low-density counterparts. The disparate results reported in the literature clearly indicate differences in physiology among fish species and populations. Inconsistencies in the cortisol stress response are also likely related to differences in experimental procedures and use of stressors that are not equal in longevity, intensity, or homeostatic disturbance.51,52
Decreased HPI axis activity elicited by chronic stressors is often a result of desensitization, potentially through prolonged increases in CRH expression and desensitization of the CRH receptor. 53 Prolonged stress exposure may also result in decreased sensitivity of adrenal or interrenal tissues to ACTH stimulation, blunting future cortisol production. 1 Desensitization may also constitute a decreased in expression of MR and GR at target tissues to mitigate negative effects of chronic hypercortisolism; however, reported increases in receptor densities are likely linked to stress-induced pathologies.22,54,55 Moreover, the density of receptors within the brain increases under chronic stress, thereby increasing the capacity for negative feedback and HPI axis downregulation. 56 Interestingly, multiple studies report decreases in MR and/or GR expression following prolonged stress exposure.56–60 Such changes may reflect negative effects of excessive GC binding within the brain. Previous research suggests imbalances in receptor signaling may alter production of neurotransmitters and neuropeptides, leading to dysfunction and potentially deleterious effects on hippocampal cells. 61
Results at hand are consistent with habituation or acclimation. Both terms have been used synonymously to describe decreased GC responses to similar stimuli and involve changes in perception of the stressor over time.54,62,63 After continued exposure, the same or similar mild stimuli may no longer be perceived as threatening. Other studies note habituation as the most likely cause for decreased GC responsiveness when similar stimuli are repeated.36,44 There were no correlative changes in brain CRH, GR, or MR mRNA expression in the present study, suggesting potential mechanisms beyond gene transcription are responsible for the observed differences in circulating cortisol.
Our study reports the first known characterization of the cortisol stress response in turquoise killifish, a model laboratory fish species. We demonstrated that just one week of daily repeated acute stress can significantly alter the cortisol stress response in male and female adult turquoise killifish, without altering brain CRH, GR or MR expression. Future research should seek to expand our collective knowledge of this species’ basic physiology. With respect to the HPI axis, a greater understanding of how stress is induced by laboratory procedures is directly relevant to the development and fitness of this short-lived model species, improving its utility as an animal model and informing practices to improve animal welfare for this species.
Supplemental Material
Supplemental material for Rapid acclimation of the cortisol stress response in adult turquoise killifish Nothobranchius furzeri
Supplemental material for Rapid acclimation of the cortisol stress response in adult turquoise killifish Nothobranchius furzeri by Dallas W Henderson and Brian C Small in Laboratory Animals
Footnotes
Acknowledgments
The authors would like to extend thanks to Dr Chi-Kuo Hu (Stanford) for providing N. furzeri embryos for this research, as well as Dr Tyrone Genade (Iowa College) and Dr Jason Podrabsky (Portland State) for correspondence and advice regarding breeding and husbandry. Lastly, thanks to the Department of Animal Science, Food, and Nutrition, and the Center for Fisheries, Aquaculture, and Aquatic Sciences at Southern Illinois University for providing funding, facilities, and equipment for this research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
