Abstract
Representative urine collection that respects the standards of animal welfare is still an issue in experimental nephrology. The commonly used metabolic cages induce stress in rodents. In mice, the volume of collected urine is sometimes insufficient for further analysis. The aim of this experiment was to analyse the effects of time of day, temperature and 2%, 5% or 10% sucrose solutions on diuresis, weight change and liquid intake of adult mice placed in metabolic cages for urine collection. Mice were placed in metabolic cages for 12 h during the day or night at standard ambient (22℃) and thermoneutral (28℃) temperatures. To determine the effect of acclimatisation, mice were placed in metabolic cages for five consecutive days. Diuresis increased with concentrations of sucrose. Body weight reduction was most rapid in the group given tap water and decreased with increasing sucrose concentrations. A drastic drop in body weight was observed in mice placed in metabolic cages for four consecutive days with access to tap water and food, indicating that time spent in metabolic cages should be kept to a minimum, as prolonged confinement in metabolic cages can be harmful to mice. The administration of concentrated sucrose solutions can potentially aid in mouse urine collection by reducing the time spent in metabolic cages. Sucrose supplementation increased the albumin/creatinine ratio. However, without showing estimates of glomerular filtration rate, renal haemodynamics, plasma electrolytes and urinary electrolyte excretions, the results of this study do not provide any conclusion about the effect of sucrose on renal function.
Introduction
In experimental nephrology, urine collection is a crucial and irreplaceable tool for assessing renal function. Collecting spot urine is difficult, even impossible, in some experimental settings, and/or insufficient in terms of volume for further qualitative or quantitative analyses. Housing mice in metabolic cages is a widespread and commonly used method for urine collection. 1 The use of metabolic cages is, however, problematic and can stress experimental animals. In mice, as well as in rats, solitary housing in a new environment introduces so-called isolation stress. These cages do not contain any enrichment, and the mice must move on wire mesh without any nesting material. This discomfort might affect physiological variables, such as glucose metabolism, reactivity to stressors, animal behaviour, as well as the production of reactive oxygen species, leading to oxidative stress.2–6
Ideally, the urine collected from experimental animals should be of sufficient volume, free of contamination, collected within the shortest time possible, and obtained without pain or distress. Several factors are known to influence urine production, including metabolic activity, liquid composition, temperature, and time spent in metabolic cages. Stechman et al. showed that weight, diuresis, food, and water intake were stabilised after spending 3–4 days in metabolic cages. 7 On the other hand, Kalliokoski et al. showed that mice do not adapt to metabolic cages. 8 Indicators of distress such as activation of the hypothalamic–pituitary–adrenal axis, oxidative stress, and increased overall metabolism persist even after 3 weeks in a metabolic cage. 8 In light of these contradictory results, a better understanding of murine response to housing in metabolic cages is needed for optimisation of the protocol for urine collection while limiting stress on the animal. Furthermore, metabolic activity can influence urine production. Metabolic activity is affected by internal as well as external factors, such as day/night cycle and ambient temperature. However, these factors are often not considered in studies where metabolic cages are used for urine collection and analysis.
Several studies have shown that the administration of a 5% sucrose solution results in a multiple-fold increase in urine output compared to water administration.9–11 In addition, in these studies, short-term sucrose administration had no effect on body weight and serum glucose concentrations. Thus, replacing drinking water with a sucrose solution could solve some of the issues surrounding urine collection, such as obtaining sufficient amounts of urine and minimising the distress of the animals. However, this might be associated with changes in urine content, including markers of renal function. Arikawe et al. showed that rats fed a diet containing 25% or 50% sucrose for 12 weeks had increased plasma and urinary creatinine and urea. 12 On the other hand, Kobayashi et al. showed that the administration of sucrose at a dose of 1.5 g/kg to healthy males did not affect creatinine clearance. 13 The same study showed that sucrose enhances purine degradation, which results in an increased plasma concentration of uric acid, while urinary excretion remains unaffected. 13 However, whether the short-term administration of sucrose solutions instead of water in metabolic cages affects markers of renal function is unknown.
To our knowledge, a systematic analysis of the effects of sucrose on urinary output and proteinuria in mice housed in metabolic cages has not yet been published. The aim of this experiment was to analyse the effect of sucrose solutions on diuresis, weight change and liquid intake of adult mice placed in metabolic cages for urine collection. Pilot experiments testing time of day and ambient temperature were performed at the same time to make the protocol more practical and animal-friendly. The purpose of these preliminary experiments was to determine the effect of time of day and temperature on urine output in order to minimise time spent in metabolic cages.
Methods
Animals
The investigation comprised 20 male and 20 female mice aged 15 months., A strain of mice that is commonly used for the induction of kidney diseases and, thus, for monitoring metabolic and renal markers using urine collection in metabolic cages, 129Sv4 (Anlab, Prague Czech Republic), was used.14,15 In addition, this strain is typically used for the generation of gene-targeted mice. 16 The age of 15 months was chosen to mimic the onset and pathology of human metabolic disorders and chronic kidney disease, both of which increase in frequency with age. The total number of animals required for the main experiment was determined according to G power analysis using G power software (G-Power 3.1.9.2., Kiel, Germany) from the preliminary results of our previous experiments (F3,32 = 2.90, effect size f = 0.75, number of groups = 4, total samples size = 36). Animals were housed in groups of five in standard type III cages (Trigon plus, Czech Republic) and were placed in one room. Mice had wood bedding and a 12/12 h light/dark cycle at an ambient temperature of 22℃ and a humidity of 40–50%. All animals had free access to tap water and a commercial pellet diet (Standard 19% protein, 4% fat rodent diet, Velaz, Czech Republic). All experiments were performed in the same room and by the same researchers in order to standardise the effects of surrounding factors such as human contact, smell, and fluctuations of light, humidity and temperature. All but experimental conditions were kept equal for all groups of mice. The experiment was approved by the ethical committee of the Institute of Molecular Biomedicine, Faculty of Medicine, Comenius University, Slovakia and complies with the animal welfare and ethical guidelines of the European Convention for the Protection of Vertebrate Animals used for Experimental and Other Scientific Purposes.
Experimental design
The same 40 mice were used in all experiments. The order of the experiments is as follows: (1) effect of light/dark cycle; (2) effect of temperature; (3) effect of sucrose; and (4) effect of acclimatisation. The allocation of mice to specific groups was randomised, and a recovery period of 7 days was used between experiments. The first session included male mice only, and the last session included only half of the mice of both sexes. Thus, the total number of housings of a single mouse in a metabolic cage was 3 or 4 for female mice and 5 or 6 for male mice. After the completion of all planned experiments, the mice were anaesthetised using ketamine and xylazine (100 mg/kg and 10 mg/kg of the bodyweight, in ratio 3:1) and then humanely killed by cardiac puncture and exsanguination. Samples of liver, kidney, brain and heart were taken, snap-frozen in liquid nitrogen and kept for further analysis.
Effect of light/dark cycle
Male mice (n = 20) were individually placed in metabolic cages (Tecniplast, Buguggiate, Italy) with a floor area of 200 cm2. All animals had free access to tap water but no access to chow. The mice were housed in metabolic cages for 12 h during the day or night (n = 10 for each group). Subsequently, the groups were given a recovery period of 36 h and then swapped. Diuresis was measured as the volume of urine collected in the metabolic cages. The urine collected in the tubes of metabolic cages was measured by pipetting the urine from the collection tubes using micropipettes. Body weight loss during the 12 h spent in the metabolic cages was also measured.
Effect of temperature
Mice of both sexes (n = 40) were individually placed in the metabolic cages for 12 h during the day with free access to tap water, no access to chow, and at a temperature of either 22℃ or 28℃. All mice were used in turn for both temperatures (n = 10 for each turn) with a recovery period of 36 h between turns. Diuresis and body weight loss were assessed.
Effect of sucrose
To analyse the effect of sucrose on diuresis, concentrations of 2%, 5% and 10% sucrose in tap water were provided as a drinking liquid, with tap water used as a control. With a 7-day recovery period after the previous experiment, mice of both sexes were individually placed in metabolic cages for 12 h (5 male and 5 female mice for the water group, 5 male and 5 female mice for each of the three different sucrose concentration groups, total n = 40 mice, 20 male and 20 female mice). The mice had free access to the corresponding liquid and no access to chow. Animals were weighed before and after 12 h in the metabolic cages. Liquid intake and diuresis were measured after 12 h in the metabolic cages, and urine samples were stored at −20℃ for subsequent analysis.
Effect of acclimatisation
Determination of the effect of acclimatisation and a reduced-stress environment on diuresis, water intake, and weight reduction was performed in accordance with Stechman et al., 7 with several minor differences including the total time spent in the cages (5 instead of 7 days), the age of mice (15 month instead of 6–8 month old mice), food availability (4 out of 5 days instead of full), and cage enrichments (missing instead of present). Mice of both sexes were individually placed in metabolic cages for 5 days (n = 10 for each sex) after a 7 day recovery period from the previous experiment. Additionally, mice had free access to water during the entire experiment. Animal weight, diuresis and food and water intake were measured daily. To obtain urine without contamination, food was removed after the fourth day. Urine collected during the fifth day was stored at −20℃ for analysis.
Urine sample analysis
Creatinine was measured using the Jaffe method. 17 This method is based on the reaction of creatinine with picric acid under alkaline conditions, which results in a yellow-orange complex. Absorbance was measured at 492 nm using the Olympus AU400 analyser (Beckman Coulter, California, USA). The concentration of proteins in urine was measured using the pyrogallol red-molybdate method, which is characterised by the binding of proteins on pyrogallol red in the presence of molybdate. 18 The coloured complex was read at 592 nm using COBAS 6000 (Roche, Basel, Switzerland). In both methods, 10 µl of the samples was used and diluted fivefold with distilled water. The albumin/creatinine ratio expressed in mg/mmol was calculated from urinary protein and creatinine concentrations.
Statistical analysis
GraphPad Prism 5.0 (GraphPad Software, San Diego, California, USA) was used for statistical analysis. To evaluate the effect of the dark/light cycle on diuresis and body weight, a paired t-test was used for comparison. To analyse the effect of temperature in both sexes on diuresis and body weight, two-way analysis of variance (ANOVA) was used with temperature and sex as the tested factors. To assess the effect of acclimatisation and different concentrations of sucrose in drinking water on diuresis, liquid intake, and other parameters, repeated measures (RM) one-way ANOVA with subsequent Bonferroni post-hoc tests and Pearson’s correlation analysis was performed. The data sets of urine output were log-transformed for the analysis. The results are reported as the mean ± standard deviation; p-values below 0.05 were considered statistically significant.
Results
In a preliminary experiment, male mice were housed in metabolic cages for 12 h during the day or during the night (Figure 1). The mean value of diuresis during the day was 55 μl (SD = 84), whereas the mean value of diuresis during the night was 90 μl (SD = 125). The difference was not found to be significant (t18 = 1.19, p = 0.25). The mice lost 1.8 g (SD = 0.27) of their body weight in the group that spent 12 h in metabolic cages during the day and 2.6 g (SD = 0.49) in the group that spent 12 h in the cages at night (t19 = 6.35, p < 0.001). Afterwards, mice of both sexes were housed in metabolic cages during daylight hours at a standard ambient temperature of 22℃ or at a thermoneutral temperature of 28℃. No differences in body weight loss (F1,22 = 3.70, p = 0.08) or collected urine volume (F1,22 = 2.58, p = 0.12) were observed between these temperatures. However, a significant gender effect of temperature on diuresis was found (F1,22 = 17.37, p < 0.001). Thus, further experiments were conducted during the day and at the standard temperature of 22℃.
Effect of day and night on diuresis. Using paired t-test no significant differences were found (p = 0.25).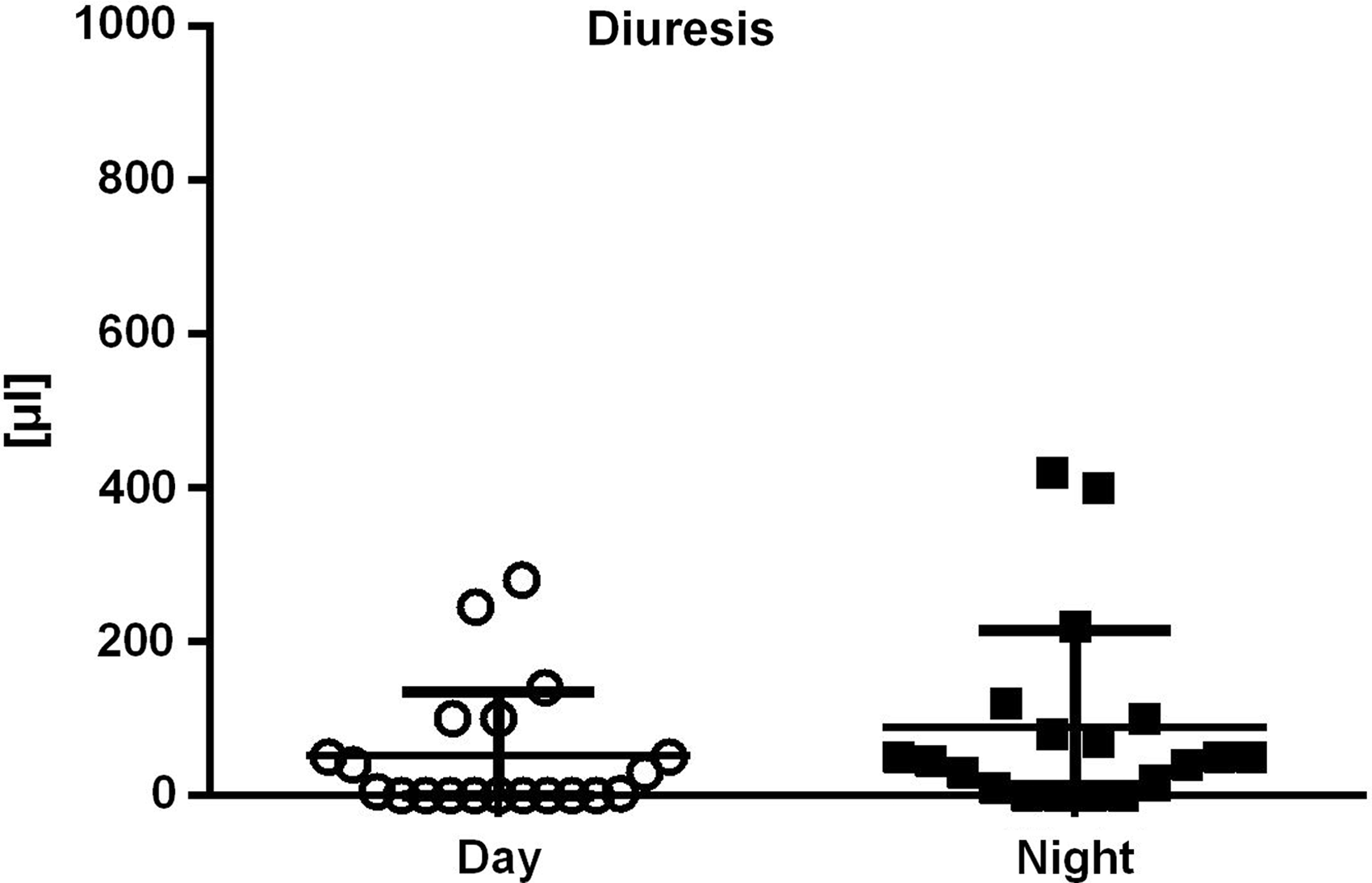
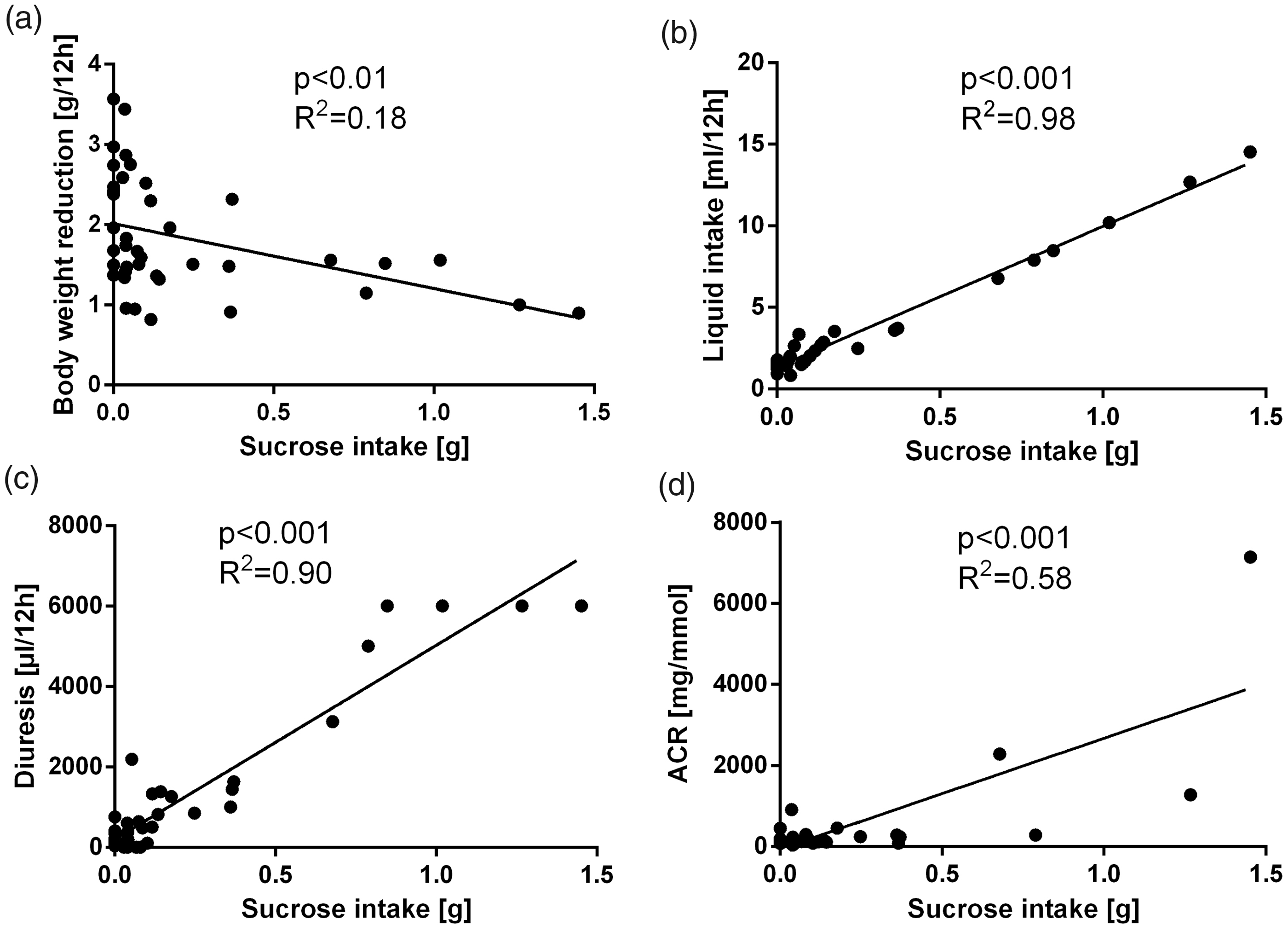
In the experiments with sucrose solutions, a significant association between sucrose intake and measured physiological parameters was found. Pearson’s correlation analysis showed a negative correlation of sucrose intake with body weight (r = −0.42, r2 = 0.18, p < 0.01 for females; r = −0.56, r2 = 0.32, p < 0.001 for males) and a positive correlation with liquid intake (r = 0.99, r2 = 0.88, p < 0.001 for females; r = 0.95, r2 = 0.91, p < 0.001 for males), diuresis (r = 0.95, r2 = 0.90, p < 0.001 for females; r = 0.78, r2 = 0.61, p < 0.001 for males) and albumin/creatinine ratio (r = 0.76, r2 = 0.58, p < 0.001 for females; r = 0.41, r2 = 0.16, p < 0.05 for males). These correlations were found in both sexes (Figures 2 and 3).
Linear regression showing the correlation of sucrose and (a) bodyweight reduction, (b) liquid intake, (c) diuresis and (d) albumin/creatinine ratio (ACR) in males. Linear regression showing the correlation of sucrose and (a) bodyweight reduction, (b) liquid intake, (c) diuresis and (d) albumin/creatinine ratio (ACR) in females.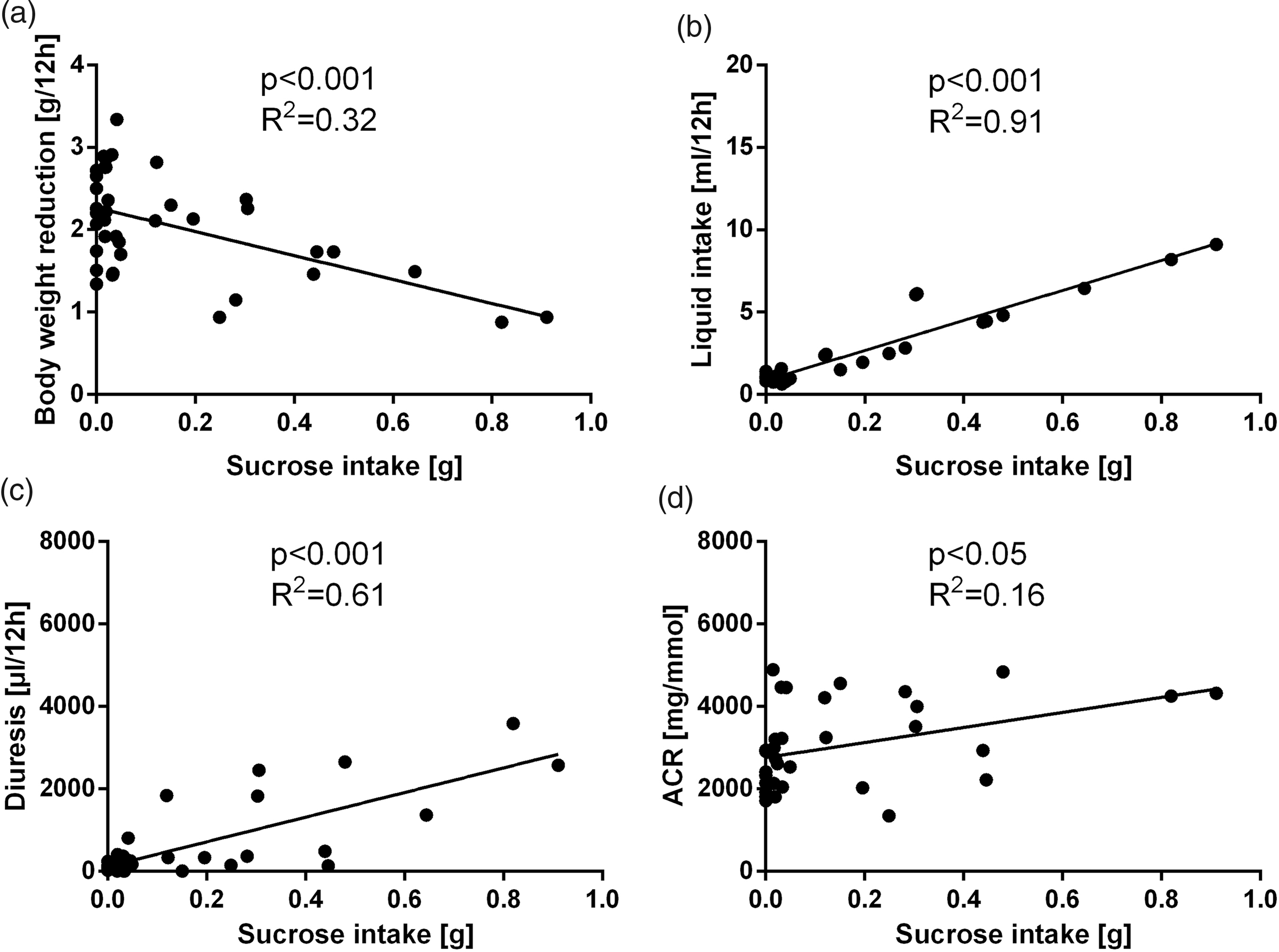
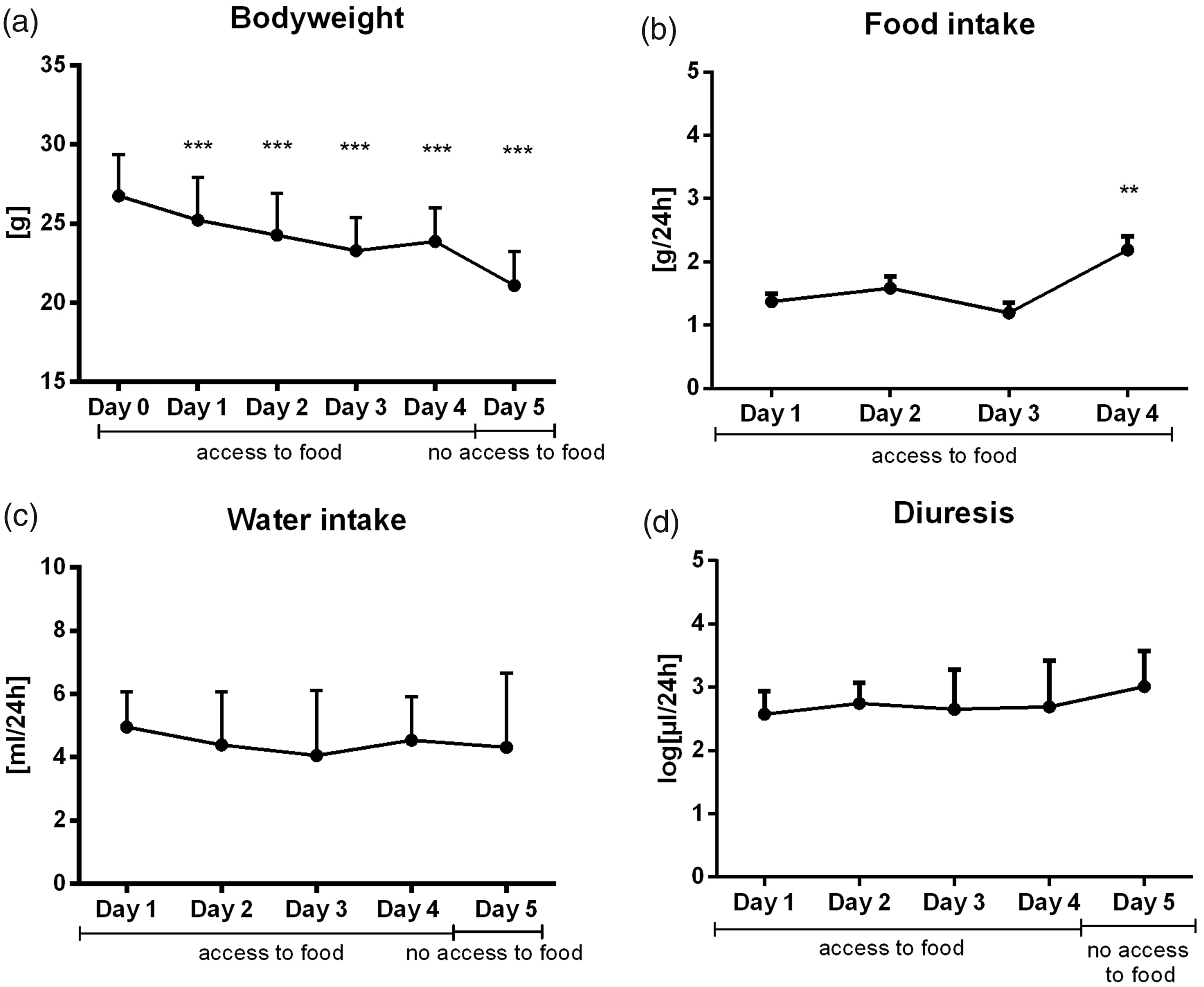
In the acclimatisation experiment, a significant increase in urine output was observed during the monitored 5 day period in males (F4,55 = 8.61, p < 0.001, Figure 4(d)) but not in females (F4,53 = 1.07, p = ns, Figure 5(d)). Similarly, liquid intake was only increased in male mice (in males F4,44 = 34.4, p < 0.001, Figure 4(c); in females F4,44 = 0.72, p = ns, Figure 5(c)). Food intake was highest on the fourth day in both sexes (mean = 3.19, SD = 0.65 g per 24 h in males, F3,33 = 101, p < 0.001, Figure 4(b); mean =2.19, SD = 0.75 g per 24 h in females, F3,33 = 8.09, p < 0.01, Figure 5(b)). Before the fourth day, a drastic loss in body weight was observed (in males F5,55 =156.7, p < 0.001, Figure 4(a); in females F5,55 = 97.0, p < 0.001, Figure 5(a)). Body weight loss reached a peak on the third day (mean = 5.55, SD = 1.25 g loss in males and mean = 3.48, SD = 0.92 g loss in females compared to baseline) and stabilised on the fourth day, but it did not reach the baseline values.
Effect of acclimatisation on males: (a) bodyweight, (b) food intake, (c) water intake and (d) diuresis during 5 days in metabolic cages. All animals had free access to food for 4 days. On the fifth day food was removed to collect uncontaminated urine. Results are expressed as mean + SD. *** denotes p < 0.001; ** denotes p < 0.01 in comparison to day 0 or 1, respectively. Effect of acclimatisation on females: (a) bodyweight, (b) food intake, (c) water intake and (d) diuresis during 5 days in metabolic cages. All animals had free access to food for 4 days. On the fifth day food was removed to collect uncontaminated urine. Results are expressed as mean + SD. *** denotes p < 0.001; ** denotes p < 0.01 in comparison to day 0 or 1, respectively.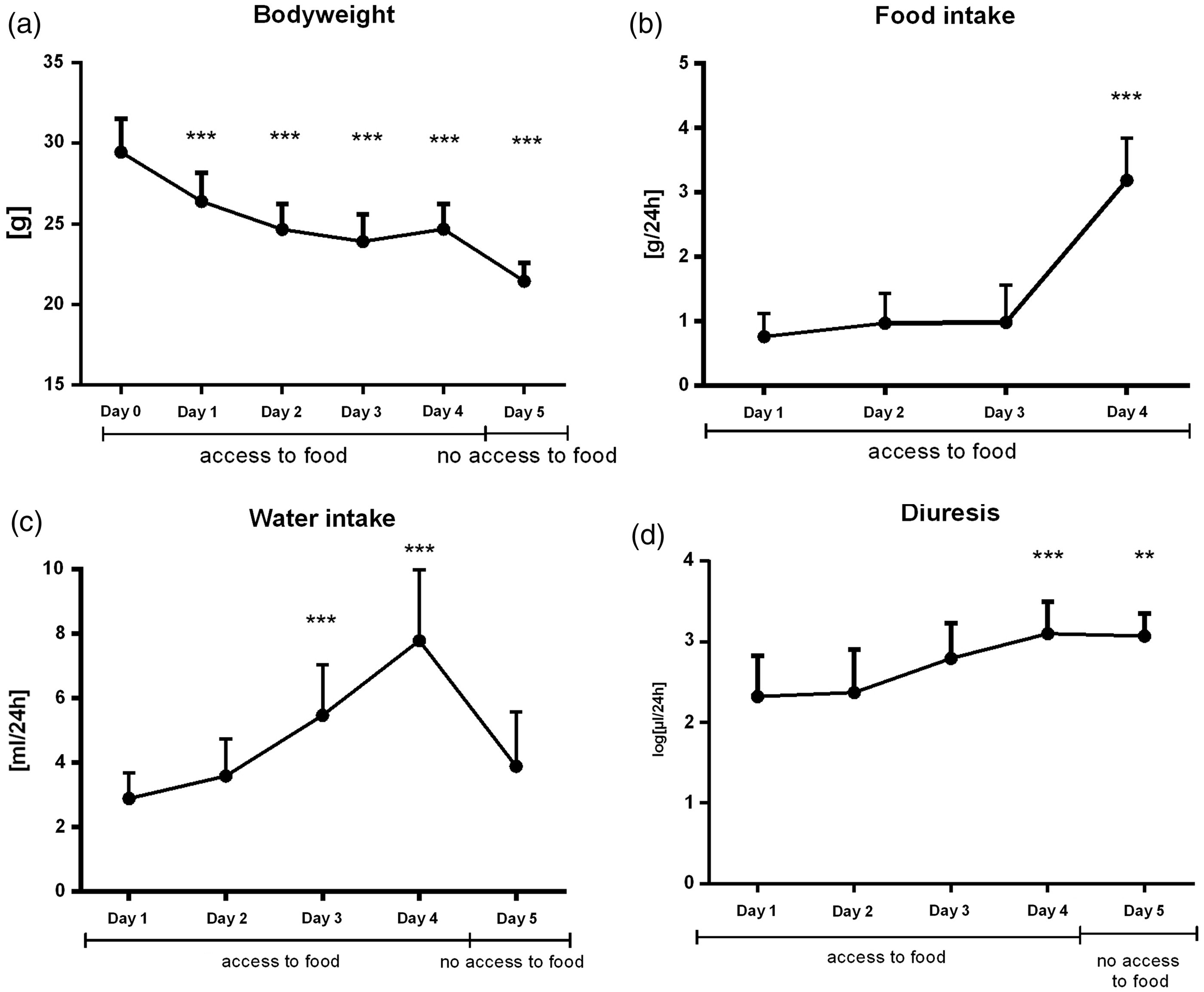
Discussion
This study focused on some of the shortcomings of the widely established method of urine collection in mice using metabolic cages, while also pointing to possibilities for its optimisation. We found no effect of the day/night cycle or increased ambient temperature to thermoneutrality on diuresis. Studies suggest that there is a circadian rhythm of diuresis in rodents, with urine output being increased during the active phase of the animal. 19 The effect of the day/night cycle was tested on male mice only, as gender differences were not expected in this rhythm. On the other hand, the fact that only male mice were used in the first round might represent a limitation of the study, since the male mice were exposed to a higher number of experimental sessions in total. The apparent absence of a circadian rhythm could be attributed to the fact that the passive phase of laboratory rodents is sometimes disrupted (by the animal house staff, who can make noise in the corridors or enter their housing area briefly). This could lead to increased daytime activity and diuresis.
Mice are nocturnal animals, and the thermoneutral temperature of 28℃ has been suggested as being the ideal temperature for testing metabolism in mice. 20 A decreased loss of body weight was observed in mice housed in metabolic cages during the day. This could be explained by the fact that mice are active during the night and probably burn fewer calories during the day while sleeping. 21
Several studies show that mice are under constant cold stress when housed at 22℃ and that this temperature could lead to bias due to increased metabolism. However, mice might thrive at 22℃ if nesting material is provided or if they are able to behaviourally thermoregulate by huddling together with other mice.22–25 Diuresis was assessed as the volume of collected urine, and it was similar at both tested temperatures. Since the collected urine was not chilled or collected under mineral oil during the collection in metabolic cages, an evaporation effect cannot be ruled out; thus, the results could be biased. From a practical point of view, however, it was determined that urine collection in metabolic cages should continue during daylight and at the standard ambient temperature of 22℃.
In the main part of the study, different concentrations of sucrose solutions were compared to tap water as the only liquid provided for drinking in the metabolic cages. Positive correlations between sucrose intake, liquid intake and diuresis in both genders were found. Moreover, sucrose intake was negatively correlated with body weight. The results of Pearson’s correlation analysis indicate that liquid intake and diuresis were lowest in the group that received only water and gradually and significantly increased with the increasing concentrations of sucrose. Body weight reduction was the most rapid in the group with tap water and gradually decreased with increasing sucrose concentration. We assume that the decrease in body weight reduction with increasing concentrations of sucrose is not attributed to a different water intake/diuresis ratio, but rather likely to the effect of increased caloric feeding due to the greater sucrose consumption. However, other potentially explanatory parameters such as specific caloric contribution and plasma osmolality were not measured in this study.
The same mice were used in all of the described experiments. This could have influenced the behaviour of the animals in the metabolic cages. In addition, male mice might have been more profoundly affected because they were housed more in metabolic cages than the female mice. To minimise this bias, recovery periods were introduced between the experiments. This way of testing could have led to a slower acclimatisation to the stressful environment of a metabolic cage, and the reduced stress could have, in turn, affected the urinary output of the animals. However, the groups that received tap water were exposed to the same conditions, and urinary output did not increase significantly with time in those groups.
After measuring urinary albumin and creatinine and calculating their ratio, a positive correlation between sucrose intake and albumin/creatinine ratio was found. Linear regression analysis showed a slight increase in albumin/creatinine ratios with increasing concentrations of sucrose, indicating a dose response. However, it remains unknown whether this is a relevant effect. A 1% sucrose (hypotonic) solution has been shown to inhibit antidiuretic hormones and reduce urinary ion concentrations and osmolarity in rats. 26 Inhibition of antidiuretic hormones might result from a slight reduction in plasma osmolarity or increased blood volume. On the other hand, an isotonic solution resulted in urinary salt and osmotic concentrations higher than the control. It is not clear how changes in albuminuria in healthy mice in response to sucrose administration relate to the pathophysiology of albuminuria in disease models. The mechanisms of the effect of sucrose intake on albuminuria are largely unknown, with little data available from the literature. Kubota et al. found an increased albumin/creatinine ratio in rats on a high-sucrose diet. 27 Overall, changes in the albumin/creatinine ratio induced by sucrose solutions have not yet been studied. Depending on the research questions being investigated, this slight effect on albumin/creatinine ratio should be taken into consideration when considering sucrose administration as a way to decrease the time the animals are housed in metabolic cages.
Next, the experiment conducted previously by Stechman et al. was reproduced with subtle differences in methodology. 7 Mice were acclimatised to housing in metabolic cages for 5 days. An increase in liquid intake in males and food intake in both genders was found on the fourth day, similar to the previously reported results. On the last and fifth day, diuresis increased in both genders with the removal of food. However, throughout the 5 days in the metabolic cages, a considerable body weight decrease was observed in both sexes (mean 27% with SD 3.18 for males and mean 21% with SD 2.89 for females). The welfare of the mice was clearly affected. The harmful effects of stress in metabolic cages are well described.8,28 Some studies have shown that mice do not habituate to this kind of confinement and even claim that the environment does not meet the common standards of animal welfare. The results of this study indicate that the physiological variables affected by metabolic cages were partially stabilised after 3 days of housing (their weight loss stabilised, food and water intake increased, diuresis increased), even if stress persisted, as highlighted by the fact that mice did not reach their original weight. We propose that it is preferable to house mice in metabolic cages for short periods because of the stress and significant weight loss present during the first 3 days, which might affect other physiological parameters. Although we did not follow any other stress-related parameters other than weight changes, significant weight reduction was present throughout the 5-day period in metabolic cages. A standard period for the collection of urine for the assessment of renal parameters is 24 h. However, this study has shown a significant weight loss in mice during the initial 24 h in metabolic cages (more than 10% weight loss, which is considered severe). This indicates that all concurrent and subsequent metabolic analyses might be affected by this obvious discomfort. Thus, we consider the initial 24 h housing a factor that should be addressed in order to minimise the adverse outcomes in metabolic experiments while preserving the animal’s wellbeing.
In summary, this study found no differences in urinary output between mice housed in metabolic cages during the day or during the night and between mice housed at standard or thermoneutral temperatures. Sucrose in drinking water resulted in increased liquid intake and diuresis in both genders. Acclimatisation only increased liquid intake and diuresis in male mice. However, housing in metabolic cages resulted in a dramatic body weight loss in both genders even after 1 day. Weight loss continued in animals until the third day, when it partially stabilised, but did not reach basal values (Figures 4(a) and 5(a)). Increased sucrose concentrations partially prevented weight loss, at least for 12 h (Figures 2a and 3a), but the slightly higher urinary albumin/creatinine ratio in mice drinking sucrose solutions should be taken into account. However, this does not prevent the use of sucrose solutions, especially if all groups in an experiment are housed the same way. Furthermore, if one takes into consideration the stress and harm imposed on the animals by housing them in metabolic cages, the administration of a sucrose solution seems even more reasonable.
It is expected that sucrose will induce osmotic diuresis and thus can change renal haemodynamics and tubular electrolyte handling. In order to confirm the usefulness of sucrose administration as a tool to decrease the time spent in metabolic changes, it is necessary to perform additional measurements, including systemic and renal haemodynamic parameters, such as glomerular filtration rate and urinary electrolyte excretion, and the histological evaluation of the kidney. Therefore, without showing estimates of glomerular filtration rate, renal haemodynamics, plasma electrolytes and urinary electrolyte excretions, the results of this study do not provide any conclusion about the effect of sucrose on renal functions. Moreover, the possibility of using spot urine normalised by creatinine is worth considering, since it has been previously used with success. 29 Further studies should also be aimed at shortening the time spent in metabolic cages with sucrose solutions in animal models of renal injury and at discovering alternatives to sucrose in the stimulation urine production in experiments where sucrose is not suitable.
Footnotes
Acknowledgements
The authors are grateful to Teri Hreha, PhD (Washington University in St. Louis School of Medicine, St. Louis, MO, USA) for language editing. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted. This article does not contain any studies with human participants performed by any of the authors.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is the result of the project implementation (ITMS 26240120027) supported by the OPRaD funded by the ERDF.
