Abstract
Neuromuscular-blocking agents are commonly used in laboratory animal research settings. Due to actions of cholinergic receptors at locations other than the motor end-plate, these agents have a strong propensity to modulate autonomic outflow and may therefore not be desirable in studies examining autonomic function. This study aimed to compare the effect of two non-depolarizing neuromuscular-blocking agents, pancuronium and cisatracurium, on blood pressure, heart rate and non-invasive indices of autonomic function (heart rate variability, systolic blood pressure variability and baroreflex sensitivity) under two different types of anaesthesia in Lewis rats. Pancuronium produced a profound vagolytic response characterized by tachycardia, reduction in heart rate variability and baroreflex sensitivity under urethane anaesthesia, and with minimal effect under isoflurane anaesthesia. Conversely, cisatracurium produced no evidence of vagolytic action under either urethane or isoflurane anaesthesia. Therefore, for studies interested in examining autonomic function, particularly baroreflex or vagal function, neuromuscular blockade would be best achieved using cisatracurium.
Neuromuscular blockers inhibit nicotinic acetylcholine receptors (nAChRs) at the skeletal neuromuscular junction and are used clinically to facilitate tracheal intubation, mechanical ventilation and the application of continuous paralysis. In the laboratory animal research setting, neuromuscular blockers are frequently used in conjunction with general anaesthesia to facilitate surgical procedures and in vivo studies of physiological parameters, with the non-depolarizing neuromuscular blocker pancuronium commonly used in studies that examine cardiorespiratory and autonomic functions (see examples1–3).
The effects of neuromuscular blockers on nAChRs at the motor end-plate are well established; however, neuromuscular blockers also have the propensity to bind to cholinergic receptors located at other sites, e.g. nAChRs present at autonomic ganglia, muscarinic receptors located within the autonomic nervous system or prejunctional nicotinic or muscarinic receptors. As such, neuromuscular blockers can cause haemodynamic changes through autonomic ganglion blockade and both vagolytic and sympathomimetic actions, potentially resulting in hypertension and tachycardia.4–6 Nevertheless, different neuromuscular blocking agents can impact the cardiovascular system with varying potencies,4,6 and clinically, the newer generation non-depolarizing neuromuscular blockers (e.g. rocuronium, vecuronium and cisatracurium) are reported to have fewer cardiovascular side-effects. 7
Cardiovascular regulation, particularly by the autonomic nervous system, is also impacted by general anaesthetics, with both inhalational and intravenous anaesthetics having the potential to modify cardiac output, blood pressure (BP) and autonomic nerve activity. Urethane is one of the most widely used anaesthetics in laboratory animal studies, either alone or in combination with other drugs. Its reported minimal impact on the cardiovascular and respiratory systems combined with provision of deep anaesthesia for extended periods make it suitable for surgical procedures using laboratory animals. 6 The inhalational anaesthetic isoflurane is also used widely in laboratory animal medicine, particularly in recovery surgeries, due to its rapid induction and recovery periods, 8 but it also has the potential to impact autonomic function and regulatory reflexes. 9
Autonomic regulation of cardiovascular function can be assessed in a variety of manners. Beat-to-beat variations in heart rate (HR), termed ‘heart rate variability’ (HRV), provide a strong indication of the capacity of the parasympathetic nervous system to regulate HR both tonically and in synchrony with the respiratory cycle, as well as in response to cardiovascular reflexes. 10 Conversely, beat-to-beat variations in systolic blood pressure (SBPV) can provide information regarding the degree of sympathetic vasomotor tone, while the ability of the autonomic nervous system to respond to ongoing changes in BP and produce counteractive changes in HR can be examined non-invasively through the determination of spontaneous baroreflex sensitivity (BRS). Measurement of these indices in response to different challenges (e.g. administration of a vasoactive agent) or experimental conditions (e.g. response to anaesthesia) can provide insight into autonomic nervous system functions. 11
For research studies examining cardiovascular and autonomic functions that require the use of anaesthetized animals, the impact of anaesthesia and the use of neuromuscular blockers is therefore a potentially confounding variable when interpreting cardiovascular physiological variables. Consistent with the guiding 3R (replacement, reduction and refinement) principles, refinement of anaesthesia should therefore act not only as a critical parameter in the promotion of animal welfare and alleviation of pain, but also to ensure that the choice of anaesthetic drug combinations has minimal impact on the data collected. The aim of this study was therefore to examine two different neuromuscular blockers, pancuronium and cisatracurium, under different anaesthetic conditions, urethane and isoflurane, focusing on cardiac vagal control, given its primary role in HR regulation. Pancuronium was chosen, given its widespread usage in laboratory animal research, and cisatracurium as representative of the newer generation of non-depolarizing neuromuscular blockers, specifically from the atracurium group. Alongside the vecuronium group, these represent the most commonly used classes of neuromuscular blockers. 12 Furthermore, despite its frequent usage, limited information is available regarding the effects on cisatracurium on autonomic function compared with vecuronium or rocuronium (see example 13 ). It also has the advantage of undergoing the Hofmann elimination, and is therefore a useful consideration in studies involving animal models with kidney or liver failure. 12 Our hypothesis for these studies was that cisatracurium would provide a more stable autonomic, and hence cardiovascular, environment under both anaesthetic conditions tested.
Materials and methods
All experiments were performed on 12–14-week-old mixed-sex Lewis rats (n = 26 total), a normotensive control strain of rodent used in cardiovascular and autonomic physiology studies2,11 that shows comparable levels of cardiovascular autonomic control as other commonly used normotensive rat strains such as the Sprague Dawley and Wistar Kyoto rats. 14 All experiments were approved by the Animal Ethics Committee of Macquarie University, and were conducted in accordance with the Australian code of practice for the care and use of animals for scientific purposes. All animals were obtained from the Animal Resources Centre (ARC) in Perth, Australia and were group-housed at the Central Animal Facility of Macquarie University on a 12 h light–dark cycle (lights on 06:00 h) at 22 ± 2℃ with access to food and water ad libitum. Experiments were performed during the light period.
Experimental procedures
Animals were anaesthetized with either urethane (1.3 g/kg intraperitoneally (IP) for induction and 0.13 g/kg intravenously (IV) for maintenance as required) or isoflurane (5% for induction and 1–3% for maintenance) in 100% oxygen. Following induction, a tracheotomy was performed to permit artificial ventilation, and the right femoral artery and left and right femoral veins were cannulated for the recording of arterial BP and drug administration, respectively. Animals received intravenous fluids (saline; 1 mL/kg/h) and core body temperature was maintained at 37℃ using a rectal probe and thermostatically-controlled electric heating blanket. At the end of the procedure, the animals were euthanized with sodium pentobarbital (60 mg/kg IV; Virbac®, Milperra, Australia) or 3 M potassium chloride.
Study design
Animals were randomly allocated to one of the following groups for anaesthesia and neuromuscular blockade:
Group I: Urethane with pancuronium (2 mg/kg IV bolus for induction; 1 mg/kg/h continuous IV infusion for maintenance, n = 7) Group II: Urethane with cisatracurium (6 mg/kg IV bolus for induction; 6 mg/kg/h continuous IV infusion for maintenance, n = 8) Group III: Isoflurane with pancuronium (2 mg/kg IV bolus for induction; 1 mg/kg/h continuous IV infusion for maintenance, n = 5) Group IV: Isoflurane with cisatracurium (6 mg/kg IV bolus for induction; 6 mg/kg/h continuous IV infusion for maintenance, n = 6)
Then each animal underwent the following protocols, consecutively:
Collection of baseline HR and mean arterial pressure (MAP) under continuous saline infusion (1 mL/kg/h IV) and examination of baroreflex function. Administration of the sympathetic ß-blocker atenolol (1 mg/kg IV; to eliminate cardiac sympathetic regulation of HR and isolate vagal control of HR), measurement of HR, MAP, HRV and SBPV, and examination of baroreflex function. Administration of neuromuscular blockade, measurement of HR, MAP, HRV and SBPV, and examination of baroreflex function.
The measurements of HR, MAP, HRV and SBPV were taken from the arterial pressure waveform. Baroreflex function was examined using both pharmacological and cross-spectral analysis. Pharmacologically, phenylephrine (50 µg/kg IV) was administered, and the resulting increase in MAP and consequent decrease in HR were measured. Phenylephrine was administered immediately prior to administration of atenolol, 30 min following administration of atenolol and 30 min following administration of the neuromuscular blockade. Cross-spectral determinants were obtained from the HRV and SBPV measurements obtained as described below.
Data analysis
All data were acquired using a CED1401 (Cambridge Electronic Design Ltd, Cambridge, UK) and analyses were performed offline using Spike2 (Cambridge Electronic Design Ltd).
From the arterial BP recording, pulse interval was derived to calculate HR and the arterial pressure waveform smoothed with a one second constant to obtain MAP. Both HR and MAP were averaged across an 80 s segment immediately prior to administration of atenolol, and one segment following administration of atenolol. A period of 10 min was required for HR and MAP to stabilize following administration of either cisatracurium or pancuronium. Consequently, data were not analysed during this initial 10 min period, but were analysed over an 80 s period at 10–15, 15–20, 20–25 and 25–30 min following administration of the neuromuscular blocker. All analyses were performed on the same 80 s segments to allow for correlation between the changes in HR and MAP observed and the associated changes in HRV, SBPV and BRS.
Heart rate variability
From the 80 s segments of arterial pressure used to analyse HR and MAP, HRV was estimated. For each 80 s segment, the pulse interval was derived and uniformly resampled at 10 Hz. Power spectrums were then generated using fast Fourier transformation (256 bin Hanning window, 0.04 Hz resolution) and the total (TP; 0–3 Hz), low (0.25–0.75 Hz, low frequency [LF]; indicator of tonic and ongoing reflex sympathetic and vagal modulation of HR) and high (1–3 Hz, high frequency [HF]; predominantly vagal indice of HR control) power calculated. 11 Data were only analysed if the power spectrum clearly showed peaks that were centred on the LF and HF bands. A Grubbs outlier test was used to identify any erroneously high estimates and if so, data were excluded from further analysis. The minimum numbers of animals used for analysis are provided in the tables and figures.
Systolic blood pressure variability
Using the same 80 s segments analysed for HRV, SBPV was estimated. For each 80 s segment, systolic BP was derived and the waveform was processed as described for the estimation of HRV and the total (TP; 0–3 Hz), low (0.25–0.75 Hz, LF; a measure of sympathetic modulation of BP) and high (1–3 Hz, HF; reflective of the influences of respiratory and the cardiac cycle on BP) power calculated. 11
Pharmacological estimation of baroreflex function
From the induced increase in MAP and the resulting decrease in HR, BRS was determined by expressing the decrease in HR relative to the increase in MAP obtained (BRS = HR/MAP [bpm/mmHg]) to provide an indication of the ability of the baroreflex to respond to large changes in BP.
Cross-spectral estimates of baroreflex function
From the estimates of HRV and SBPV, BRS was estimated by cross-spectral analysis of the amount of LF power (αLF) and HF power (αHF) present in both the HRV and SBPV power spectra as described previously, 11 providing an indication of ongoing baroreflex function at resting BP.
Statistical analysis
The minimum number of animals used per group was determined using statistical power analysis (GPower v3.1) 15 based on data from our previous studies2,11 with α = 0.05 and power = 0.80. All data were expressed as means ± standard error (SEM) and presented in one of two ways: (1) time-course responses to administration of pancuronium versus cisatracurium in the presence of urethane or isoflurane anaesthesia or (2) the effect of either atenolol, pancuronium or cisatracurium on each variable measured at 30 min post-administration versus baseline between the two anaesthetic conditions. All data-sets were analysed using a two-way analysis of variance (ANOVA) with repeated-measures and Bonferroni’s post hoc analysis (GraphPad v6; GraphPad Software Inc, San Diego, CA, USA), with the choice of neuromuscular blocker or anaesthetic examined as the independent variables, respectively. P ≤ 0.05 was considered to be significant.
Results
Baseline cardiovascular parameters under urethane versus isoflurane anaesthesia
Comparative effect of atenolol on heart rate (HR), mean arterial pressure (MAP), heart rate variability (HRV), systolic blood pressure variability (SBPV) and baroreflex function under urethane versus isoflurane anaesthesia.
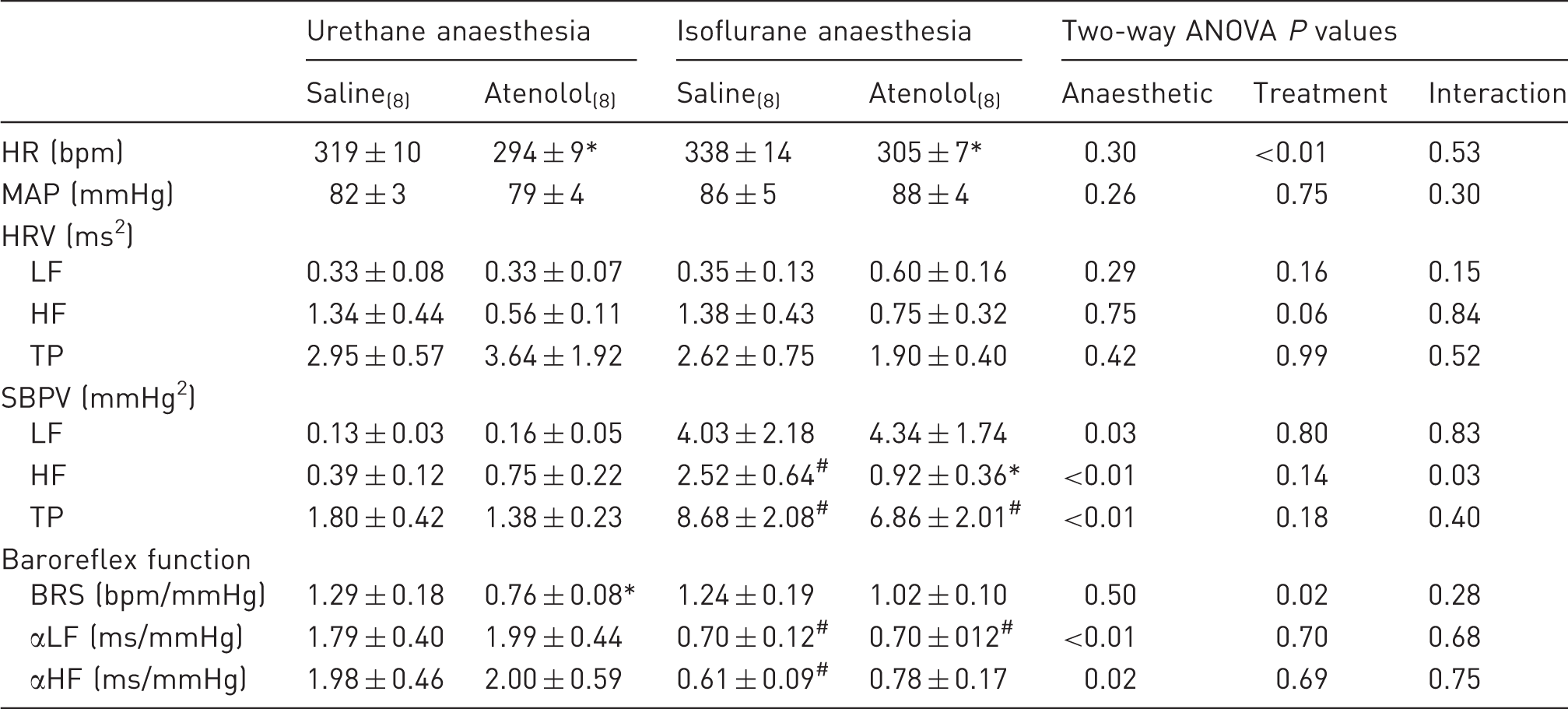
*P ≤ 0.05 saline versus atenolol under the same anaesthetic condition; #P ≤ 0.05 urethane versus isoflurane under the same treatment condition. Minimum n value for all data in the indicated column is denoted in subscripts. Baroreflex sensitivity (BRS) was determined using the pharmacological method while αLF and αHF were derived from cross-spectral analysis. ANOVA: analysis of variance; HF: high frequency; LF: low frequency; TP: total power.
Effect of sympathetic blockade on cardiovascular parameters
Atenolol was administered to eliminate sympathetic control over HR. The effects on HR, MAP, HRV, SBPV and baroreflex function are detailed in Table 1. Atenolol produced an expected decrease in HR under both urethane and isoflurane anaesthesia (P < 0.05); however, this was without significant effect on the HF component of HRV (P = 0.06). Under urethane anaesthesia, this was associated with a reduction in BRS estimated using the pharmacological approach only (P < 0.05). Under isoflurane anaesthesia, however, atenolol produced a reduction in the HF component of SBPV, such that this parameter was no longer different to estimates obtained under urethane (P < 0.05). No effect of atenolol on baroreflex function was observed under isoflurane.
Effect of pancuronium on cardiovascular parameters under urethane versus isoflurane anaesthesia
Comparative effect of pancuronium on heart rate (HR), mean arterial pressure (MAP), heart rate variability (HRV), systolic blood pressure variability (SBPV) and baroreflex function under urethane versus isoflurane anaesthesia.
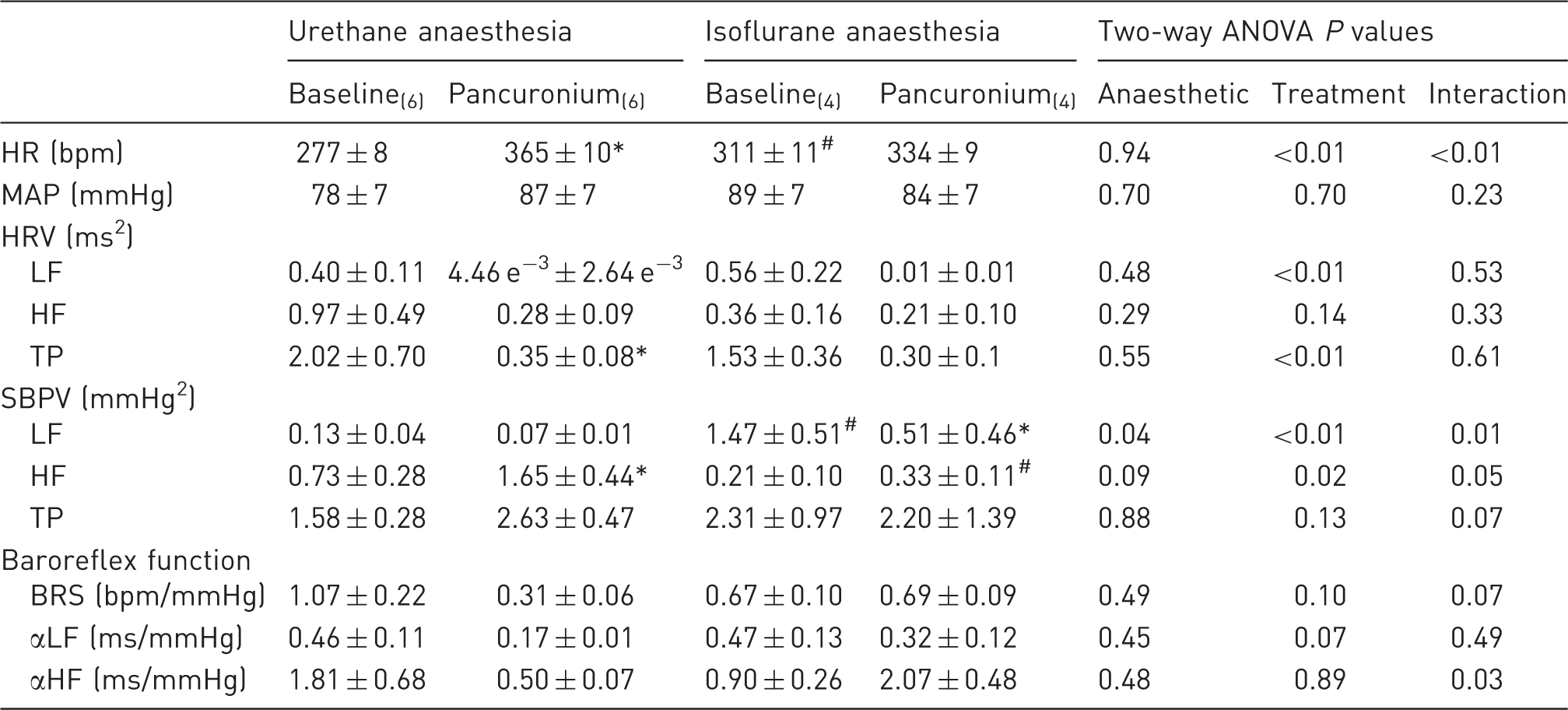
Measurements were taken immediately preceding and 30 min following administration of pancuronium. *P ≤ 0.05 baseline versus pancuronium under the same anaesthetic condition; #P ≤ 0.05 urethane versus isoflurane under the same treatment condition. Minimum n value for all data in the indicated column is denoted in subscripts. Baroreflex sensitivity (BRS) was determined using the pharmacological method while αLF and αHF were derived from cross-spectral analysis. ANOVA: analysis of variance; HF: high frequency; LF: low frequency; TP: total power.
Effect of cisatracurium on cardiovascular function under urethane versus isoflurane anaesthesia
Comparative effect of cisatracurium on heart rate (HR), mean arterial pressure (MAP), heart rate variability (HRV), systolic blood pressure variability (SBPV) and baroreflex function under urethane versus isoflurane anaesthesia.
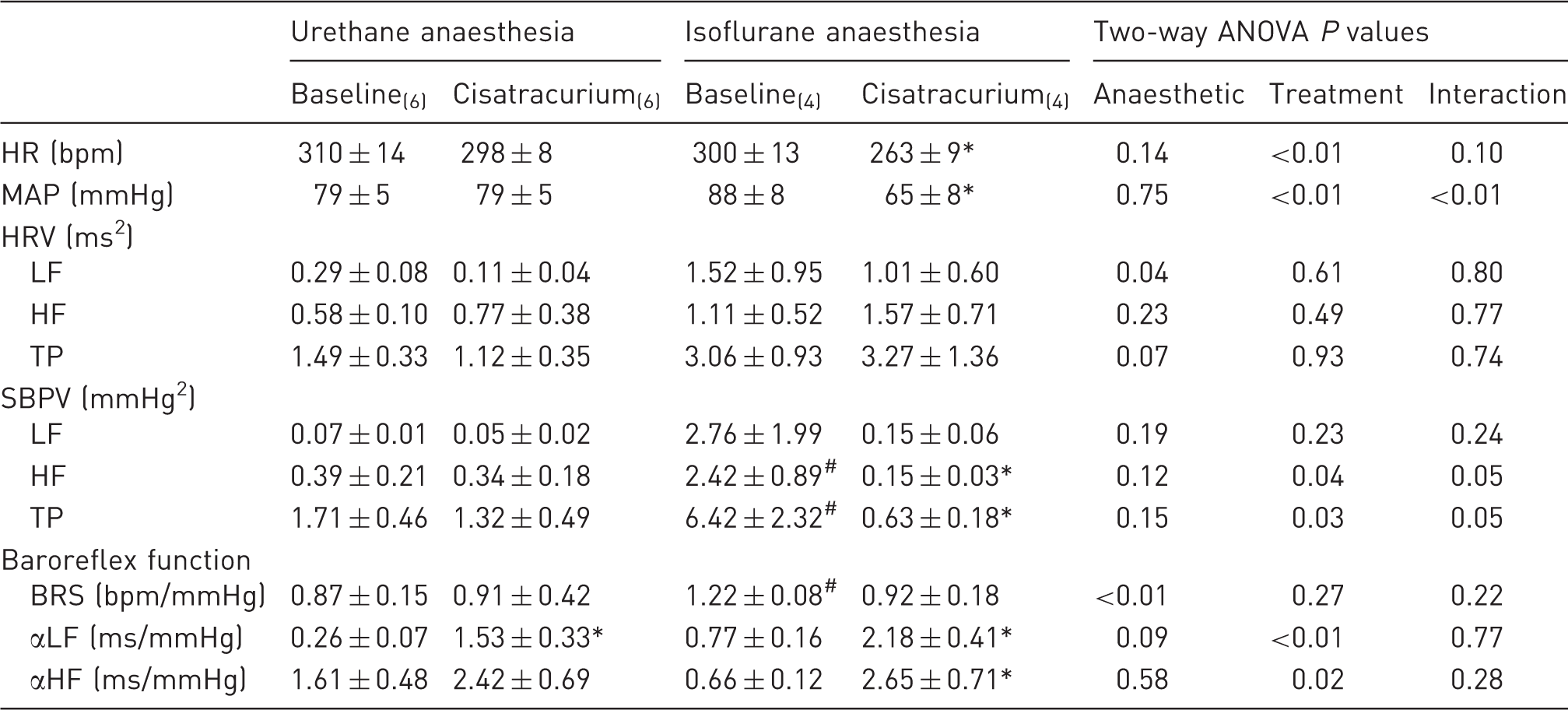
Measurements were taken immediately preceding and 30 min following administration of cisatracurium. *P ≤ 0.05 baseline versus cisatracurium under the same anaesthetic condition; #P ≤ 0.05 urethane versus isoflurane under the same treatment condition. Minimum n value for all data in the indicated column is denoted in subscripts. Baroreflex sensitivity (BRS) was determined using the pharmacological method while αLF and αHF were derived from cross-spectral analysis. ANOVA: analysis of variance; HF: high frequency; LF: low frequency; TP: total power.
Comparison of the effect of pancuronium and cisatracurium on cardiovascular parameters under either urethane or isoflurane anaesthesia
The level of HR following administration of pancuronium was greater than that following administration of cisatracurium under both urethane and isoflurane anaesthesia (both P < 0.001). However, the underlying cause for these differences in HR varied depending on the anaesthetic used. Under urethane, but not isoflurane, anaesthesia, administration of pancuronium increased HR (Table 2), while under isoflurane anaesthesia, cisatracurium reduced HR (Table 3). Such divergence of effect on MAP was not apparent. When the level of MAP at 30 min post-administration of either cisatracurium or pancuronium was compared with baseline under the same anaesthetic (i.e. anaesthetic was not the variable), the analysis revealed that choice of neuromuscular blocker was not a significant variable when urethane was the chosen anaesthetic (P = 0.65). When the same analyses were performed under isoflurane anaesthesia, the choice of neuromuscular blocker was again not a significant variable (P = 0.32); however, time was (P = 0.04), reflecting the fact that cisatracurium produces a reduction in MAP when administered under isoflurane.
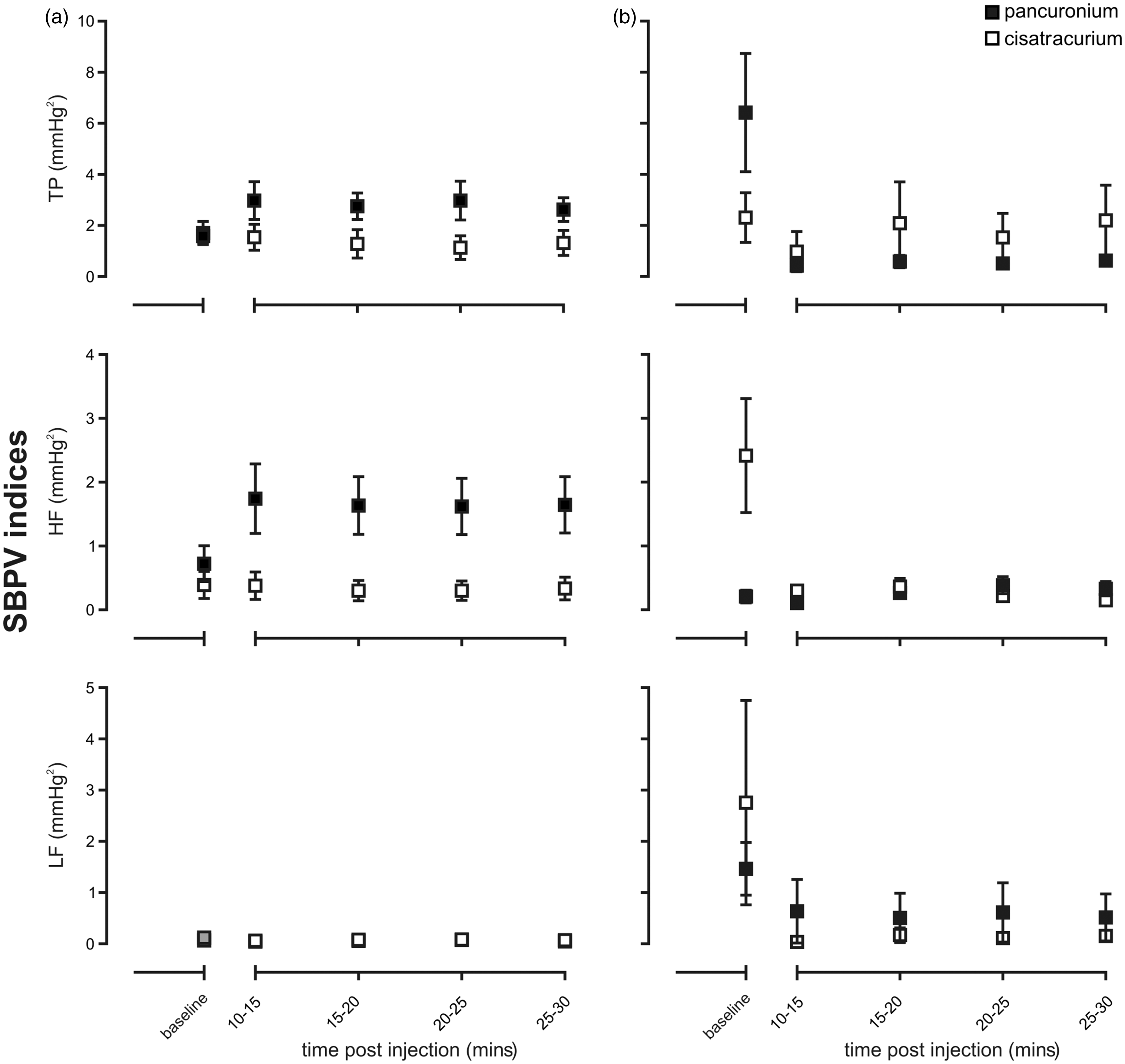
Overall, when administered under urethane anaesthesia, pancuronium and cisatracurium produced comparable effects on HRV (Figure 1). Under isoflurane anaesthesia, however, pancuronium was observed to produce a greater attenuation in HRV than cisatracurium when estimated as either TP or LF power (P < 0.05). When considering SBPV, despite the differing effects of pancuronium and cisatracurium under urethane or isoflurane anaesthesia (see Tables 2 and 3), both neuromuscular blocking agents produced comparable changes in all components of SBPV when compared under the same anaesthetic condition, except for the HF component which was greater in the presence of the pancuronium/urethane anaesthesia (P = 0.05, Figure 2a).
Time-course effect of pancuronium versus cisatracrium administered under urethane (a) or isoflurane (b) anaesthesia on total (TP), high (HF) and low (LF) frequency power spectral indices of heart rate variability (HRV). n ≥ 5 per group; *P ≤ 0.05. Time-course effect of pancuronium versus cisatracrium administered under urethane (a) or isoflurane (b) anaesthesia on total (TP), high (HF) and low (LF) frequency power spectral indices of systolic blood pressure variability (SBPV). n ≥ 4 per group; *P ≤ 0.05.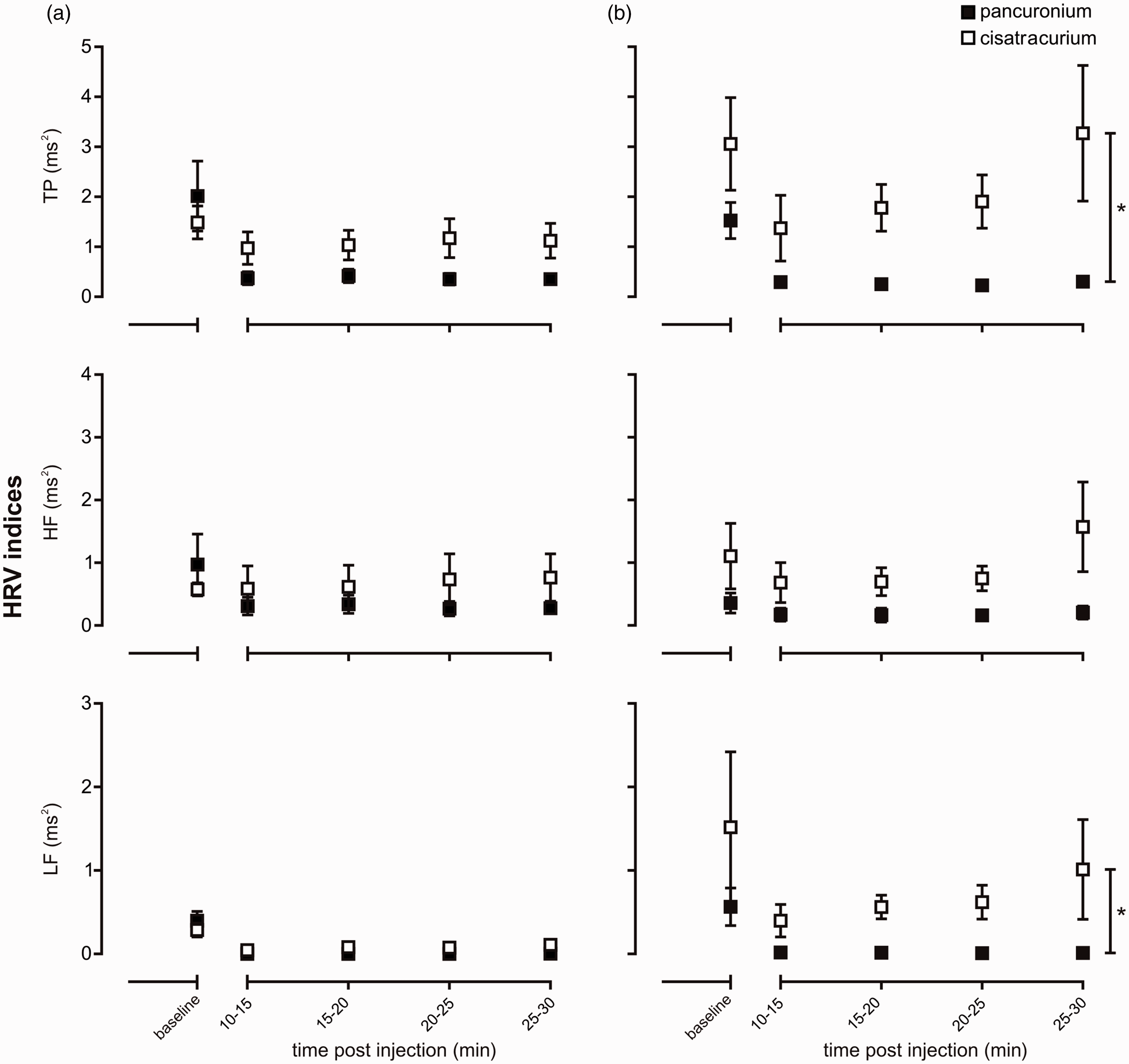
All estimates of baroreflex function were influenced by the two neuromuscular blocking agents, though to varying extents. Under urethane anaesthesia, the effect of neuromuscular blockade on BRS as measured by pharmacological estimates differed (P < 0.05), due to a suppression of baroreflex function following pancuronium administration (P < 0.05) that was not evident after cisatracurium (P > 0.99). Cross-spectral estimates of baroreflex function also differed following administration of either of the neuromuscular blocking agents under urethane (P < 0.05, Figure 3a), due to an increase in αLF following cisatracurium administration and a reduction in the αHF component following pancuronium administration under urethane. Under isoflurane anaesthesia, the effects of the neuromuscular agents on baroreflex function estimated using pharmacological means (P = 0.55) or via the αHF component (Figure 3b) were similar. The effect on the αLF component differed (P < 0.05, Figure 3b), due to an increase in αLF following administration of cisatracurium.
Time-course effect of pancuronium versus cisatracrium administered under urethane (a) or isoflurane (b) anaesthesia on baroreflex function examined using cross-spectral analysis of low frequency (αLF) or high frequency (αHF) estimates of heart rate variability and systolic blood pressure variability. n ≥ 4 per group; *P ≤ 0.05.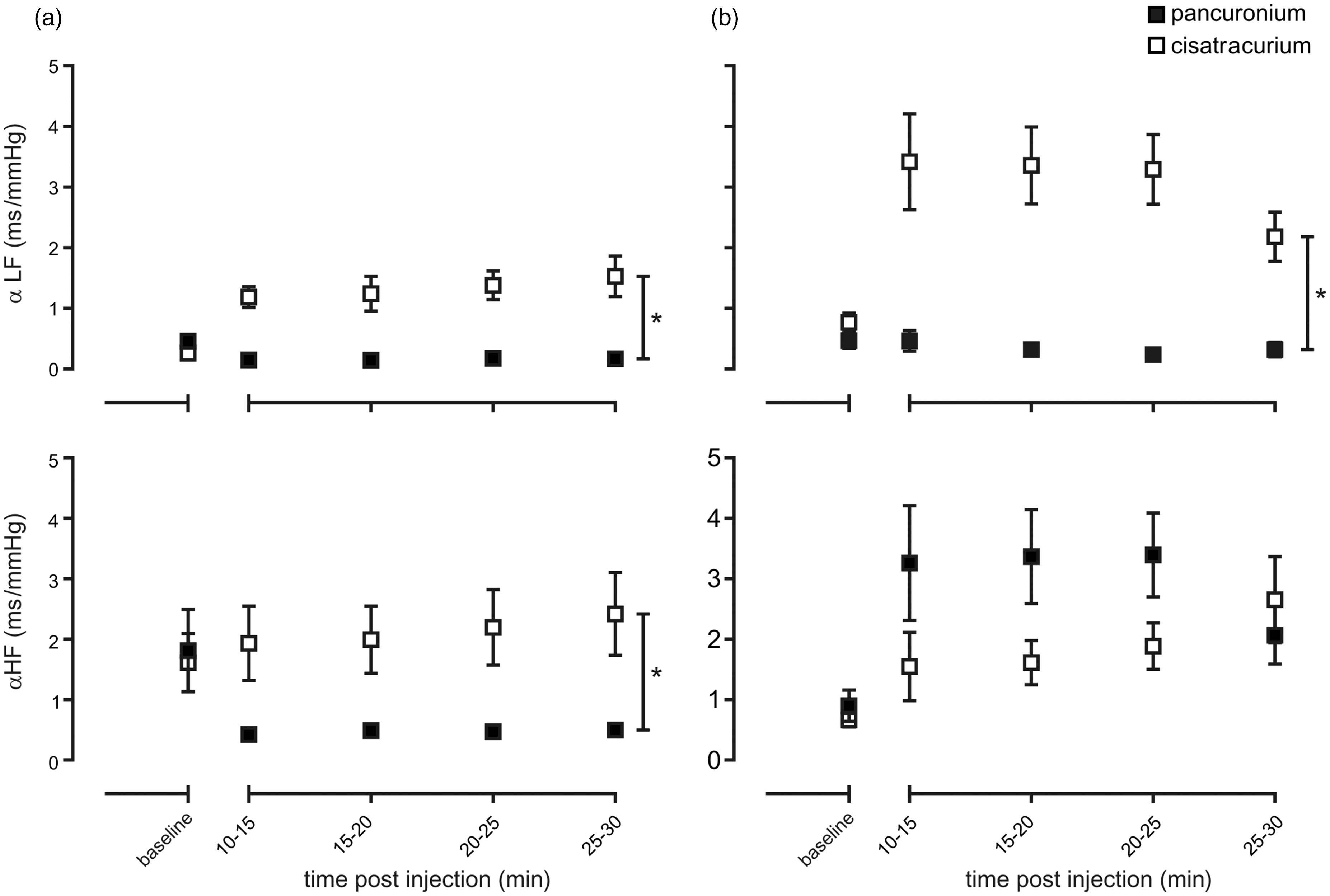
Discussion
The major findings of the present study are: (1) pancuronium has profound vagolytic effects when administered under urethane anaesthesia that are not evident under isoflurane anaesthesia; (2) cisatracurium produces bradycardia and hypotension when administered under isoflurane anaesthesia which is not evident when administered under urethane anaesthesia; (3) cisatracurium promotes baroreflex control of HR regardless of the anaesthetic agent used. Together, these findings indicate that when undertaking experiments aimed at examining autonomic control of cardiovascular function in anaesthetized laboratory animals the choice of neuromuscular blockade needs to be carefully considered. Cisatracurium, and likely the other new generation neuromuscular blockers, provide more favourable conditions regardless of the type of anaesthetic used.
Administration of the β1-adrenoceptor antagonist atenolol was used to eliminate sympathetic input to the heart so that subsequent changes in HR would be vagally mediated. Atenolol resulted in a predictable decrease in HR, consistent with the removal of a sympathetic drive to the heart, the degree of which was not affected by the type of anaesthetic used. Atenolol also produced an effect on HRV, showing a trend towards reducing HF that similarly was not impacted by the type of anaesthetic. Typically, the HF component of HRV is thought to reflect vagally-mediated regulation of HR and is abolished following administration of atropine. 10 Nevertheless, administration of atenolol has been shown to alter HF power with variable responses observed that appear to be age-, sex- or disease-dependent or relate to the duration of atenolol administration.16–19 The exact mechanism by which atenolol changes HF power remains unclear, but may relate to the sympathovagal interactions that are present at the level of the sinoatrial (SA) node. MAP and SBPV were unchanged by atenolol, indicating appropriate compensatory neurohormonal mechanisms were subsequently able to maintain normal BP in the face of a decrease in HR and loss of any β1-adrenoceptor-mediated vasodilatory actions. 20 Surprisingly, BRS as assessed via pharmacological means was reduced following the administration of atenolol under urethane anaesthesia. Here we measured the reflex bradycardia as a proportion of the pharmacologically induced increase in BP. Previous work has demonstrated that under urethane anaesthesia in rodents, administration of atenolol has no impact on baroreflex-mediated bradycardia, suggesting that this aspect of reflex control is entirely mediated via the parasympathetic nervous system. 21 Of note, however, this change in BRS following atenolol administration was not supported by our cross-spectral estimates of baroreflex function (e.g. αLF and αHF), which were both unchanged by the administration of atenolol, and which may rather be an anomalous finding.
Another important observation from our study was the differing effects produced by the two anaesthetic agents on autonomic regulation of the cardiovascular system. Most notably, baroreflex control of HR was less under isoflurane compared with urethane anaesthesia. Typically, urethane anaesthesia does not affect BRS in normal laboratory rodents, such as the Lewis rat used in the present study; 11 however, isoflurane has been well documented to attenuate baroreflex function in a variety of species, including rodents.9,22,23 Therefore, the use of isoflurane should be cautioned. Furthermore, in studies examining baroreflex function under adverse conditions such as hypertension (e.g. spontaneously hypertensive rat) or kidney disease (e.g. Lewis polycystic kidney rat), this function is suppressed by urethane anaesthesia, 11 and our current results suggest it would be further suppressed by isoflurane anaesthesia. In association with the reduction in baroreflex function, an increase in SBPV was also observed under isoflurane anaesthesia. This increase in BP lability is not surprising given that baroreflex function was suppressed and abolition of this reflex pathway is known to result in an increase in SBPV. 24
Interestingly, while isoflurane anaesthesia was associated with some deficit in cardiovascular autonomic control this did not exacerbate the inhibitory effect of pancuronium on autonomic function. When administered under urethane anaesthesia, pancuronium produced an expected tachycardia.25,26 In association with this there was a marked reduction in HRV, reflective of the reported vagolytic actions of this agent associated with blockade of cardiac muscarinic receptors and loss of both vagal modulation of SA node activity and atrioventricular (AV) conduction,22,27,28 which likely contributed to the strong tendency for baroreflex control of HR to reduce and of SBPV to increase. When administered under isoflurane anaesthesia, although HRV was suppressed following pancuronium administration, suggestive of some withdrawal of cardiac vagal tone, this did not translate into an increase in HR or a reduction in BRS, implying that any withdrawal of vagal tone that may have occurred was not profound. The underlying reason for the differing effect of pancuronium under urethane versus isoflurane anaesthesia cannot be elucidated from our current data-set, but it is possible that this occurred due to the fact that isoflurane itself is a vagolytic agent, 9 and so the available cardiac vagal tone for pancuronium to inhibit was markedly reduced.
In contrast to the profound effects of pancuronium on cardiovascular autonomic control, administration of cisatracurium had minimal effect when administered under urethane anaesthesia, with the only notable change observed being an increase in BRS. Thus, cisatracurium would appear to be the optimal neuromuscular blocking agent for use in studies examining the autonomic nervous system, especially those that examine baroreflex function due to the preservation of this, and presumably other, cardiovascular reflexes. Moreover, our results suggest that this neuromuscular blocker is cardioprotective given the known association between a reduction in baroreflex function and cardiovascular risk. 29 This widens the applicability of our findings to both research studies necessitating neuromuscular blockade in animal models of disease which are susceptible to cardiovascular death, as well as to the use of neuromuscular blockers in the human population, for example to their use in patients undergoing cardiac surgery where haemodynamic stability is critical. 5 Nevertheless, a degree of caution is needed when using this agent alongside volatile anaesthetics. When administered under isoflurane anaesthesia, cisatracurium produced bradycardia and hypotension. Importantly, however, the HF component of HRV, which reflects respiratory modulation of HR, was not impacted by cisatracurium, indicating that there was likely no change in the mechanical ventilation of the animal following administration of cisatracurium, which if inhibited could result in hypoxia and a reduction in BP and HR. Furthermore, positive effects on SBPV and BRS were observed, with an improvement in BRS and a reduction in SBPV being noted. From our study it is not possible to determine whether these changes in SBPV and BRS were a direct consequence of cisatracurium, and therefore a reflection of its effect on autonomic transmission, or if these changes were associated with the reduction in BP and HR observed.
Conclusion
Under both urethane and isoflurane anaesthetic conditions, cisatracurium shows reduced vagolytic properties and, as such, is better placed to provide optimal conditions by which to assess the autonomic nervous system function in health and disease.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Health and Medical Research Council of Australia Project grant GNT1030301. The authors received no other financial support for the research, authorship, and/or publication of this article.
