Abstract
Pulmonary arterial hypertension is a life-threatening disease with a poor prognosis. Oral treatment with vasodilators is often limited by systemic hypotension. Inhalation of vasodilators offers the opportunity for selective pulmonary vasodilation. Testing selective pulmonary vasodilation by inhaled nitric oxide or alternative substances in animal models requires an increased pulmonary vascular tone. The aim of this study was to identify animal models that are suitable for investigating selective pulmonary vasodilation. To do so, a haemodynamic stable pulmonary hypertension was initiated, with a 30 min duration deemed to be a sufficient time interval before and after a possible intervention. In anaesthetized and mechanically-ventilated Sprague–Dawley rats pulmonary hypertension was induced either by acute hypoxia due to reduction of the inspired oxygen fraction from 0.21 to 0.1 (n = 6), a fixed infusion rate of the thromboxane analogue U46619 (240 ng/min; n = 6) or a monocrotaline injection (MCT; 60 mg/kg applied 23 days before the investigation; n = 7). The animals were instrumented to measure right ventricular and systemic arterial pressures. Acute hypoxia caused a short, and only transient, increase of pulmonary artery pressure as well as profound systemic hypotension which suggested haemodynamic instability. U46619 infusion induced variable changes in the pulmonary and systemic vascular tone without sufficient stabilization within 30 min. MCT provoked sustained pulmonary hypertension with normal systemic pressure values and inhalation of nitric oxide caused selective pulmonary vasodilation. In conclusion, out of the three examined rat animal models only MCT-induced pulmonary hypertension is a solid and reliable model for investigating selective pulmonary vasodilation.
Pulmonary arterial hypertension (PAH) is defined as an increase in mean pulmonary arterial pressure≥ 25 mmHg at rest. 1 The prognosis of PAH is poor, and significantly worsens in patients with advanced disease.2,3 Early specific treatment of PAH seems to delay clinical deterioration and to improve long-term outcomes.4–6 However, specific oral therapy for PAH is often limited by systemic hypotension and peripheral oedema.
To optimize therapy for PAH, vasoreactivity is tested during cardiac catheterization. 1 As a standard procedure, pulmonary vascular reactivity is examined by the inhalation of nitric oxide (NO).1,7 The evaluation takes approximately 30 min and includes the following three steps: stabilization, inhalation of NO at a dose of 10–20 parts per million (ppm), and a NO washout. 8 The fast onset of its effects and short half-life establish NO as a valuable testing substance, but as a treatment option the use of inhaled NO is limited by the need for continuous application. 7
The development of new inhaled vasodilators with a longer half-life time than NO represents a promising treatment choice for patients with PAH since it includes the possibility of selective pulmonary vasodilation. Successful selective pulmonary vasodilation reduces the pulmonary arterial pressure without adverse systemic effects such as systemic hypotension. 9 To investigate new compounds as potential selective vasodilators in animal models, two requirements are desirable: first, the simultaneous measurement of pulmonary and systemic haemodynamics; second, at least 30 min of haemodynamic stability with a solid, increased pulmonary vascular tone to compare the effects with NO as the reference substance.
Hypoxic pulmonary vasoconstriction is provoked by an alveolar oxygen tension below 70 mmHg. 10 Reduction of the inspired oxygen tension seems to be an easy and straightforward approach for inducing pulmonary hypertension without further pharmacological intervention. Accordingly, various experimental studies applied hypoxic inspiratory gas mixtures to investigate acute pulmonary hypertension.11–18 However, it remains unclear whether acute hypoxia is an appropriate animal model for investigating selective pulmonary vasodilation in rats.
Several investigators have used a continuous infusion of the thromboxane A2 receptor agonist U46619 to induce PAH.19–21 U46619 induces marked pulmonary vasoconstriction. 22 Albeit, the selective effects of U46619 on the pulmonary compared with its effects on the systemic circulation and the haemodynamic stability in rats make its use less reliable.
Monocrotaline (MCT)-induced pulmonary hypertension represents a reliable and reproducible animal model of PAH. In rats a single subcutaneous injection of MCT results in pulmonary hypertension within three weeks and an increased mortality rate within six to eight weeks.23,24 However, use of the MCT model is time-consuming and staff-intensive. In addition, after MCT challenge some animals present with signs and symptoms of increased stress, justifying the search for an alternative animal model of pulmonary hypertension.
The aim of this study was to examine the suitability of acute hypoxia, thromboxane or MCT-induced pulmonary hypertension of animal models for investigating selective pulmonary vasodilation in rats.
Materials and methods
Animals and housing
Our experiments were planned as a pilot study to identify a suitable, small animal model of pulmonary hypertension to investigate selective pulmonary vasodilation. Since our investigation was a pilot study, a small sample size of six to seven animals per group was chosen.
We investigated the feasibility of acute hypoxia, the thromboxane analogue U46619, as well as MCT-induced pulmonary hypertension. Acute hypoxia induces hypoxic pulmonary vasoconstriction, which varies in magnitude among species. 10 The most commonly used small species in laboratory research are rats and mice. Because the extent of hypoxic pulmonary vasoconstriction is small in mice we decided to apply it on rats. 25 Since pulmonary artery vasoreactivity and, in particular, hypoxic pulmonary vasoconstriction are more pronounced in male than female Sprague–Dawley rats, only male rats were used to exclude gender bias in our pilot study. 26
All animal experiments were approved by the local animal welfare committee (Federal State Administration of Saxony, Leipzig, Germany: reference numbers TVV 28/09 and TVV 33/10). The care and handling of the animals were in accordance with the European Community Laboratory Animal Directive 2010/63 and with the German Animal Welfare Act. Male Sprague–Dawley rats were purchased from Charles River Laboratories (Sulzfeld, Germany) and were kept at the Max Bürger Research Centre (Leipzig, Germany). Two rats were housed in clear Euro-standard cages type R2, 18 cm height (Bioscape GmbH, Castrop-Rauxel, Germany) to allow social interaction and welfare. For environmental enrichment the cages were provided with paper tunnels (125 ×90 mm; Bioscape). Standard housing conditions were achieved with a temperature of 22.5℃, a relative air humidity of 45 ± 10%, and a circadian rhythm of 12/12 h light/dark cycle. The rats had free access to standard rodent food and water (ssniff-Spezialitäten GmbH, Soest, Germany). The studies were performed at the Central Animal Facility, University of Leipzig, Germany.
Anaesthesia and surgical procedure for haemodynamic measurements
On the day of the experiment, the mean body weights of the rats were 347 ± 21 g in the protocol group with acute hypoxia (n = 6); 275 ± 40 g in the protocol group with thromboxane analogue infusion (n = 6); 323 ± 20 g in the group after MCT challenge (n = 7); and 360 ± 21 g in the saline control group (n = 7). The rats were anaesthetized with an intraperitoneal bolus of ketamine (100 mg/kg; WDT, Garbsen, Germany), xylazine (15 mg/kg; Pfizer GmbH, Berlin, Germany), and atropine (0.1 mg/kg; Braun, Melsungen, Germany) using a stock solution containing 51.28 mg/mL of ketamine, 7.69 mg/mL of xylazine, and 0.05 mg/mL of atropine. The body weights ranged from 251 to 394 g, therefore the injected volumes ranged from 0.49–0.77 mL. An adequate depth of anaesthesia was indicated by the lack of motoric response to a toe pinch. After achieving a sufficient level of anaesthesia the rats were put into a supine position and fixed onto a heating pad (FHC Inc, Bowdoin, ME, USA). The heating pad was connected to a temperature control system via a rectal thermometer to secure a body temperature of between 36℃ and 37℃ (FHC Inc). A tracheostomy was performed and a metal cannula was placed into the trachea. The animals were mechanically ventilated with a respiratory frequency of 70–75 breaths per minute, a tidal volume of 8–10 mL/kg body weight, and a positive end-expiratory pressure of 4 cmH2O at an inspired oxygen fraction of 1.0 (Biological Research Apparatus, Comerio, VA, Italy). The fraction of inspired oxygen (FiO2) was maintained at 1.0 during the whole protocol of the U46619 infusion and the MCT challenge. A polyethylene catheter was placed in the left external jugular vein for continuous infusion of 6% hydroxyethyl starch with an infusion rate of 5 mL/h (Smith Medical International Ltd, Ashford, UK). At the right carotid artery an ultra-miniature pressure catheter was positioned for continuous recordings of heart rate and systemic arterial pressure (Millar Instruments, Houston, TX, USA). To obtain the right ventricular systolic pressure (RVSP), a Codman catheter was advanced from the right external jugular vein into the right ventricle (Johnson & Johnson, Raynham, MA, USA). The tracings of the arterial line during the positioning of the Codman catheter indicated arrhythmias. Throughout the stabilization period and the experimental protocols themselves, the arterial tracings were uniform, suggesting a sinus rhythm. Heart rate, mean systemic arterial pressure, and RVSP were continuously measured and recorded (ADInstruments, Bella Vista, NSW, Australia). An intraperitoneal catheter was placed for the maintenance of anaesthesia. Anaesthesia was maintained at one-third of the initial dosage. Because of the variations in body weight, the infusion rate ranged from 0.16 to 0.26 mL/h, which equated to 33.3 mg/kg/h of ketamine, 5 mg/kg/h of xylazine, and 0.03 mg/kg/h of atropine, respectively. Every 20 min a toe pinch test was performed. Since no motoric motion was observed in response to the test, the infusion rate was maintained during the protocols.
Exclusion criteria
Two rats both assigned to the acute hypoxia protocol were excluded from further interventions because the preparation lasted longer than 40 min or an obvious blood loss occurred.
Study protocols
Protocol of acute hypoxia
For the acute hypoxia protocol, an additional polyethylene tube was inserted into the left carotid artery for arterial blood sampling (Smith Medical International Ltd). After surgical preparation the rats were ventilated with an FiO2 of 0.21 for 30 ± 9 min by using room air to ensure an equilibrium of the oxygen tension (n = 6). The baseline values for the RVSP, systemic arterial pressure, and heart rate were documented. Arterial blood was sampled for blood gas analysis using a Radiometer ABL 800 Basic (Radiometer GmbH, Willich, Germany). To investigate the effects of acute hypoxia, the ventilator gas inlet was connected to a gas tank containing 90% nitrogen and 10% oxygen (Air Liquide GmbH, Düsseldorf, Germany). The haemodynamic parameters were noted after 10 and 30 min of acute hypoxia. At the end of the protocol a second arterial blood gas analysis was scheduled and the experiment was terminated by exsanguination of the animals.
Protocol of U46619 infusion
In the second set of experiments the rats were instrumented as described above. An additional catheter was placed into the left external jugular vein for the infusion of the thromboxane A2 receptor agonist U46619 (Sigma-Aldrich Chemie GmbH, Steinheim, Germany). A stock solution was prepared by diluting 1 ml of U46619 in 9 mL of normal saline solution (stock solution concentration: 1 mg/mL U46619). The final concentration for the experiment was achieved by adding 144 µL of stock solution to 9.86 mL of saline solution. After 21 ± 6 min of stabilization and documentation of the baseline values, RVSP, systolic arterial pressure, and heart rate, a continuous infusion of U46610 was started with a dose of 240 ng/min which was equal to 1.0 mL/h and this was left unchanged during the experiments (n = 6). RVSP, systolic arterial pressure, and heart rate were documented and analysed after 10 and 30 min of continuous infusion of U46619.
Protocol of MCT challenge
For the MCT-induced PAH protocol, the rats were challenged with a single subcutaneous injection of 60 mg/kg of MCT dissolved in 0.5 mL of normal saline solution (MCT, Sigma Aldrich GmbH; n = 7). The animals in the saline control group received an equal volume of normal saline solution (n = 7). The rats were weighed daily and evaluated using a score sheet to determine potential humane endpoints.
Determination of humane endpoints
To determine humane endpoints, a daily evaluation was performed using a scoring system for the following characteristics: activity (0 = normal, 1 = slow, 2 = hyperactive); physical appearance (0 = normal, 1 = dull fur, 2 = chromodacryorrhoea, 3 = maiming); vocalization (0 = none, 1 = provoked by light tough, 2 = unprovoked); eating behaviour (0 = normal, 1 =reduced, 2 = increased or suspended); breathing frequency (0 = normal, 1 = reduced, 2 = increased); and body temperature (0 = normal, 1 = reduced, 2 = increased). The score sheet included an open slot for unscheduled observations. The scoring system was adapted from the recommendation by Carstens and Moberg. 27
Haemodynamic measurements after MCT challenge
After MCT challenge the rats were anaesthetized and underwent the surgical preparation described above. After 17 ± 1 min of stabilization, RVSP, systolic systemic pressure, and heart rate were obtained as baseline values before the inhalation of NO was started (pre-NO). To estimate the vasoreactivity 20 ppm of NO were inhaled over 10 min and the haemodynamic parameters were documented (NO 20 ppm). After 10 min of NO washout, the haemodynamic values were ascertained (post-NO). The haemodynamic protocol after the MCT challenge allowed each animal to be its own control. Therefore, no haemodynamic measurements were performed in the rats of the saline control group. The animals in the saline control group were anaesthetized and mechanically ventilated at an FiO2 of 1.0, and scheduled for heart removal and lung perfusion.
Thickness of the arterial walls
The animals in the MCT and saline control groups were sacrificed via exsanguination. For histological examination of pulmonary artery thickness, the lungs were perfused according to a protocol adapted from the procedure described by Jones and co-workers. 28 The procedure was modified by positioning the perfusion catheter directly into the pulmonary artery and the removal of the heart before lung perfusion was started. Standard haematoxylin and eosin staining was performed using 4 µm thick lung sections. Images were obtained using a light microscope (Leica, Wetzlar, Germany) at 40 × magnification. To evaluate vascular hypertrophy, the luminal-to-vessel area ratio of five intrapulmonary arteries with an outer diameter of between 50 and 100 µm was calculated from each rat using ImageJ software (National Institutes of Health, Bethesda, MD, USA).
Ratio of the right ventricle weight to left ventricle plus septum weight
The weights of the right and left ventricles plus septum were determined and the ratio of the right ventricle weight to left ventricle plus septum weight (RV/LV + S) was calculated to estimate the effects of MCT-induced pulmonary hypertension on right ventricular hypertrophy.
Statistical analysis
Data are expressed as mean ± standard deviation (SD). Two-sided P values of less than 0.05 were considered to be statistically significant. Statistical analyses were performed using SPSS 20.0 (IBM Corp, Armonk, NY, USA). Normal distribution of the data was confirmed using the Kolmogorov–Smirnov test. Intra-group comparisons were performed by analysis of variance (ANOVA) for repeated measures with post hoc testing according to Bonferroni. Differences between groups were analysed with the Student’s t-test for unpaired samples. A post hoc correlation analysis was performed using Pearson’s correlation coefficient.
Results
Protocol of acute hypoxia
Haemodynamic results
The reduction of inspired oxygen (FiO2) from 0.21 to 0.1 induced a transient increase of RVSP from 30.7 ± 2.0 mmHg to 34.7 ±2.0 mmHg (P < 0.05 vs FiO2 0.21). However, after 30 min of acute hypoxia, the RVSP values decreased to 31.2 ± 3.5 mmHg, and hence, to comparable values at an FiO2 of 0.21 (not significant vs FiO2 0.21 or 10 min at FiO2 0.1; Figure 1a). Hypoxia caused a profound decrease in systolic arterial blood pressure after 10 and 30 min compared with normoxia (FiO2 0.21: 90 ± 10 mmHg, 10 min at FiO2 0.1: 81 ± 9 mmHg, 30 min at FiO2 0.1: 74 ± 8 mmHg; P < 0.05 for both vs FiO2 0.21; Figure 1b). The heart rate remained stable when the FiO2 was reduced from 0.21 to 0.1 (FiO2 0.21: 307 ± 38 bpm, 10 min at FiO2 0.1: 316 ±28 bpm, 30 min at FiO2 0.1: 314 ± 19 bpm; Figure 1c).
Protocol of acute hypoxia. Acute hypoxia was induced by the reduction of the inspired oxygen tension (FiO2) from 0.21 to 0.1. Presented are the right ventricular systolic pressure as an estimation of the pulmonary arterial systolic pressure (a), the systolic arterial pressure of the systemic circulation (b), and the heart rate (c). Each animal is depicted as a single line, n = 6; *P < 0.05 vs FiO2 0.21.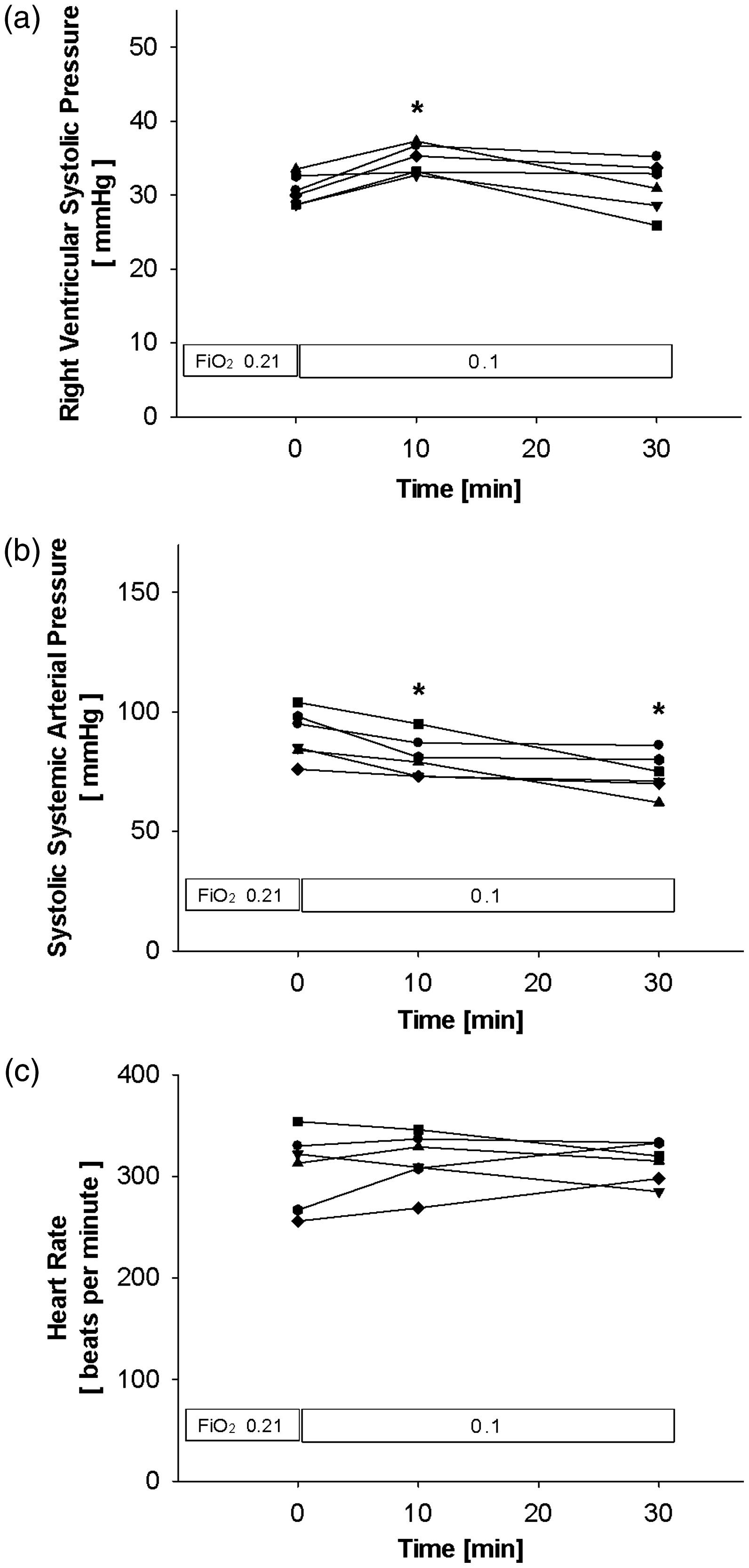
Arterial blood gas tensions
To confirm the concentration of inspired oxygen, arterial blood gas analyses were performed at an FiO2 of 0.21 and after 30 min at an FiO2 of 0.1. As expected, the reduction of the FiO2 from room air to hypoxic gas mixture decreased the arterial oxygen tension from 97 ± 7 mmHg to 34 ±3 mmHg (P < 0.05) corresponding to arterial oxygen saturation values of 95 ± 1% and 43 ± 7%, respectively (P < 0.05). The arterial tension of carbon dioxide did not differ at the two inspired oxygen tensions (FiO2 0.21: 33.9 ± 2.0 mmHg; FiO2 0.1: 30.2 ± 3.1 mmHg). The pH values decreased when the FiO2 was reduced from 0.21 to 0.1 (FiO2 0.21: 7.45 ± 0.01; FiO2 0.1: 7.39 ± 0.06; P < 0.05). The concentrations of haemoglobin were comparable during ventilation with room air and the hypoxic gas mixture (FiO2 0.21: 12.0 ± 0.6 g/dL; FiO2 0.1: 11.7 ± 0.9 g/dL). The haematocrit values were similar at both concentrations of inspired oxygen (FiO2 0.21: 37.0 ± 1.9%, FiO2 0.1: 35.8 ± 2.1%).
Protocol of U46619 infusion
Haemodynamic results
Infusion of the thromboxane receptor agonist U46619 only tended to increase the RVSP from 36.1 ± 4.6 mmHg at baseline to 40.0 ±5.2 mmHg after 10 min of U46619 infusion corresponding to a percentage increase of 9.7 ± 6.0% (n = 6; P > 0.05). One rat out of the six died after 17 min of U46619 infusion when the RVSP rapidly increased to a maximum value of 55.6 mmHg. After 30 min of U46619 infusion the RVSP values of 41.9 ± 4.2 mmHg did not differ from values at baseline and after 10 min infusion of the thromboxane analogue (n = 5; P > 0.05 for both). After 30 min of U46619 infusion the mean percentage change of RVSP further increased compared with baseline (14.5 ± 18.6%) and 10 min of thromboxane analogue infusion (4.3 ± 13.9%). Remarkably, the responses to the continuous infusion of U46619 varied between the individual animals (Figure 2a).
Protocol of U46619 infusion. To increase pulmonary vascular resistance, the thromboxane A2 receptor agonist U46619 was continuously infused. The right ventricular systolic pressure (a), the systolic arterial blood pressure (b), and the heart rate (c) were measured before the start of the U46619 infusion (baseline) as well as during 10 and 30 min of U46619 infusion. Each animal is depicted as a single line, n = 6 at baseline and after 10 min of U46619 infusion, n = 5 after 30 min of U46619 infusion; *P < 0.05 vs baseline, §P < 0.05 vs 10 min of U46619 infusion.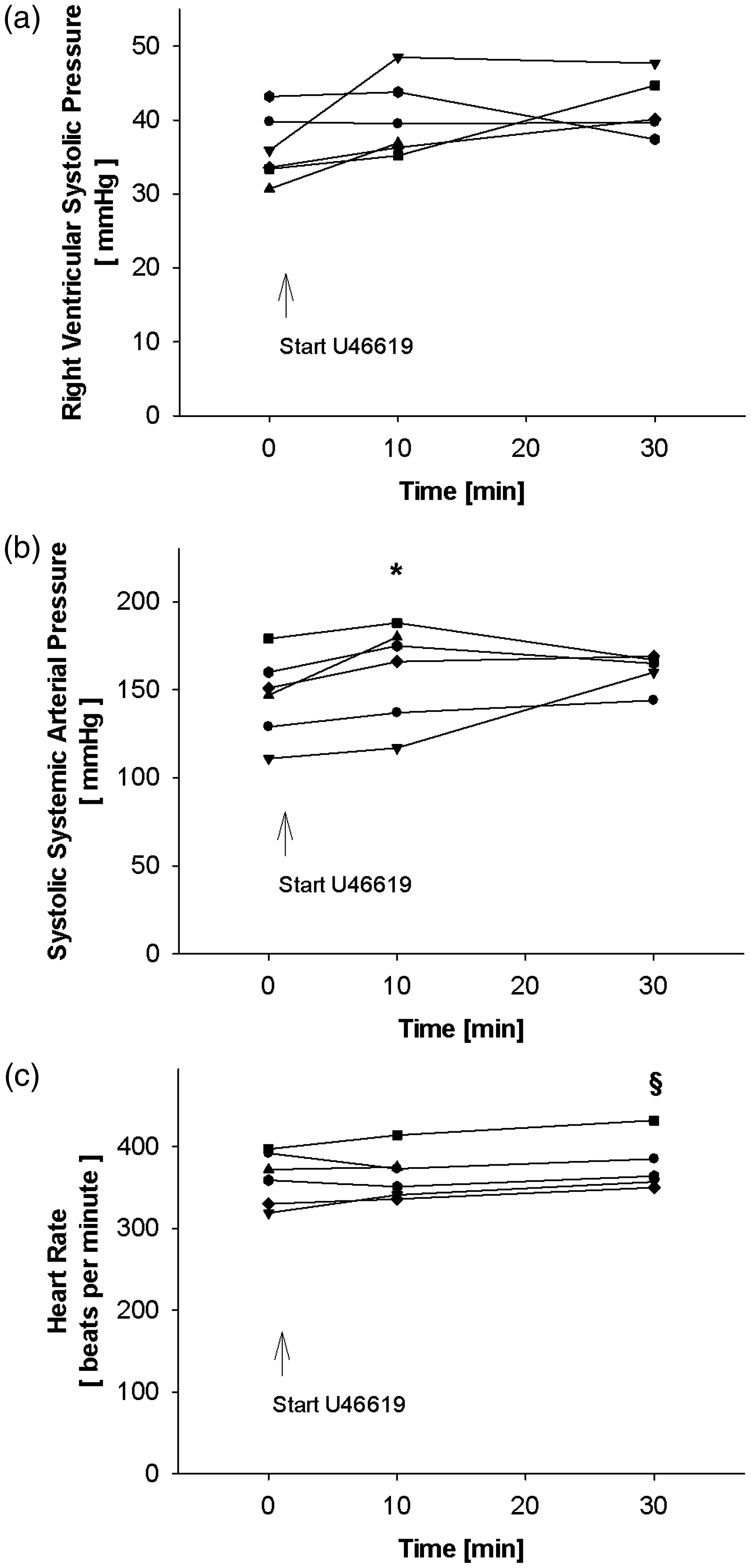
The systolic arterial pressure was enhanced from 146 ± 24 mmHg at baseline to 161 ± 28 mmHg after 10 min of U46619 infusion, equivalent to a percentage increase of 9.7 ± 6.0% (n = 6; P < 0.05). The systolic arterial pressure remained elevated after 30 min of thromboxane analogue infusion (161 ± 10 mmHg), corresponding to a percentage increase of systolic arterial pressure of 12.8 ± 17.1% compared with baseline, and of 5.4 ± 16.7% when compared with 10 min infusion of U46619, respectively. Because of the reduced number of animals the systolic arterial blood pressure values did not differ from the values at baseline or after 10 min of U46619 infusion (n = 5; P > 0.05 for both; Figure 2b).
There were no differences between the heart rate values at baseline (362 ± 32 bpm) or after 10 min of U46619 infusion (365 ± 29 bpm; P > 0.05). The heart rate values were higher after 30 min (378 ± 33 bpm) than after 10 min of thromboxane analogue infusion (P < 0.05, Figure 2c).
In the one rat that died after 17 min of U46619 infusion a post-mortem thoracotomy was performed without showing any macroscopic pathologies of the heart or lungs. We assume that the animal died because of acute heart failure due to a tremendous increase of pulmonary and/or systemic vascular resistance.
Protocol of MCT challenge
Scoring humane endpoints
The MCT-challenged rats presented with dull fur from day 13 until the end of the observation period, equivalent to a humane endpoint of 1. Furthermore, one rat had a minor nasal scab from day 21 until day 23 after the MCT challenge. This rat was carefully observed but presented no other signs or symptoms of pain, distress or suffering and was therefore included in the study. The seven rats in the saline control group appeared without any signs or symptoms of stress, corresponding to a humane endpoint of 0 from day 1 until day 23 of the protocol.
Body weights
On day 1, the rats showed similar body weights (MCT group: 213 ± 13 g, saline control group: 221 ± 40 g). In both protocols the body weights increased during the experimental study. As expected, the gain in body weight was less after the MCT than after the saline challenge on day 9, and from day 12 until the end of the observation period on day 23 (P < 0.05 for day 9, days 12–23; Figure 3). On day 23 the average body weight was 323 ± 20 g in the MCT group (n = 7) and 360 ± 21 g in the saline control group (n = 7), corresponding to an average percentage body weight gain of 52 ± 24% and 69 ± 31%, respectively.
Protocol of monocrotaline (MCT) challenge: body weights. Rats were challenged with a single subcutaneous injection of either 60 mg/kg of MCT or a corresponding volume of saline solution (NaCl). The animals were weighed daily. Data are presented as mean ± SD, n = 7 in each group; *P < 0.05 vs NaCl on the respective day.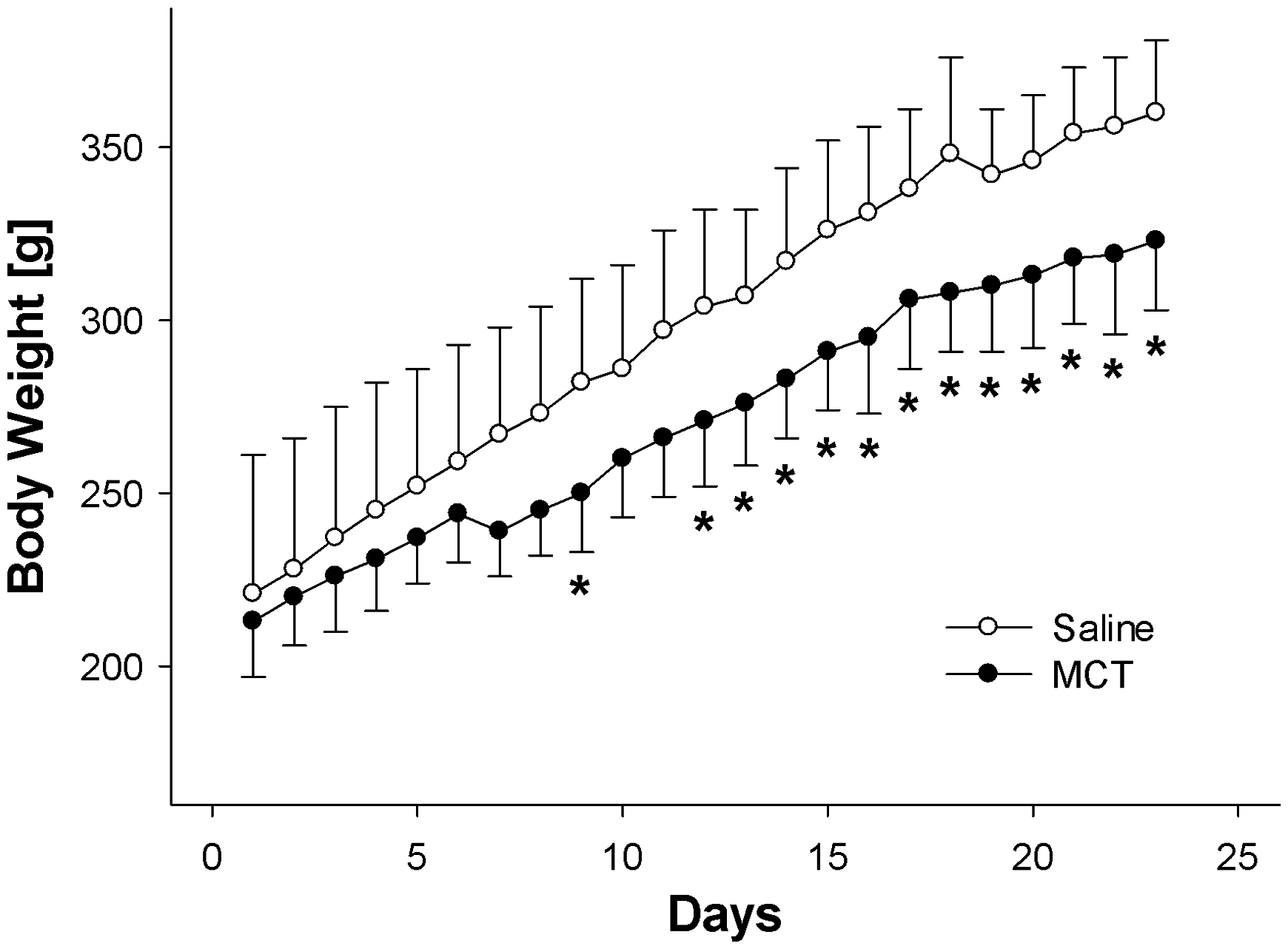
Haemodynamic results of the MCT-challenged rats
NO caused a decrease in the RVSP compared with both pre-NO and post-NO values (pre-NO: 74.5 ± 10.8 mmHg, NO 20 ppm: 65.1 ± 9.3 mmHg, and post-NO: 75.2 ±9.5 mmHg, P < 0.05 vs NO 20 ppm for both). Expressed as a percentage change, inhalation of NO provoked a decrease in the RVSP by 12.4 ± 5.8%. By contrast the systolic arterial pressure remained stable during the inhalation of NO, suggesting selective pulmonary vasodilation (pre-NO: 123 ± 33 mmHg, NO 20 ppm: 111 ± 23 mmHg, and post-NO: 107 ± 21 mmHg). The heart rate was unaltered by the NO inhalation (pre-NO: 350 ± 23 bpm, NO 20 ppm: 350 ± 15 bpm, post-NO: 349 ± 31 bpm). Haemodynamic results of the MCT protocol are presented in Figures 4a–c.
Protocol of monocrotaline (MCT) challenge: haemodynamic parameters. To test the vasoreactivity to inhaled nitric oxide (NO) 23 days after MCT challenge, the right ventricular systolic pressure (a), the systolic arterial blood pressure (b), and the heart rate (c) were measured at baseline, during inhalation of 20 ppm NO with a 10 min duration, and 10 min after cessation of the NO inhalation. Each animal is depicted as a single line, n = 7; *P < 0.05 vs pre-NO, #P < 0.05 vs post-NO.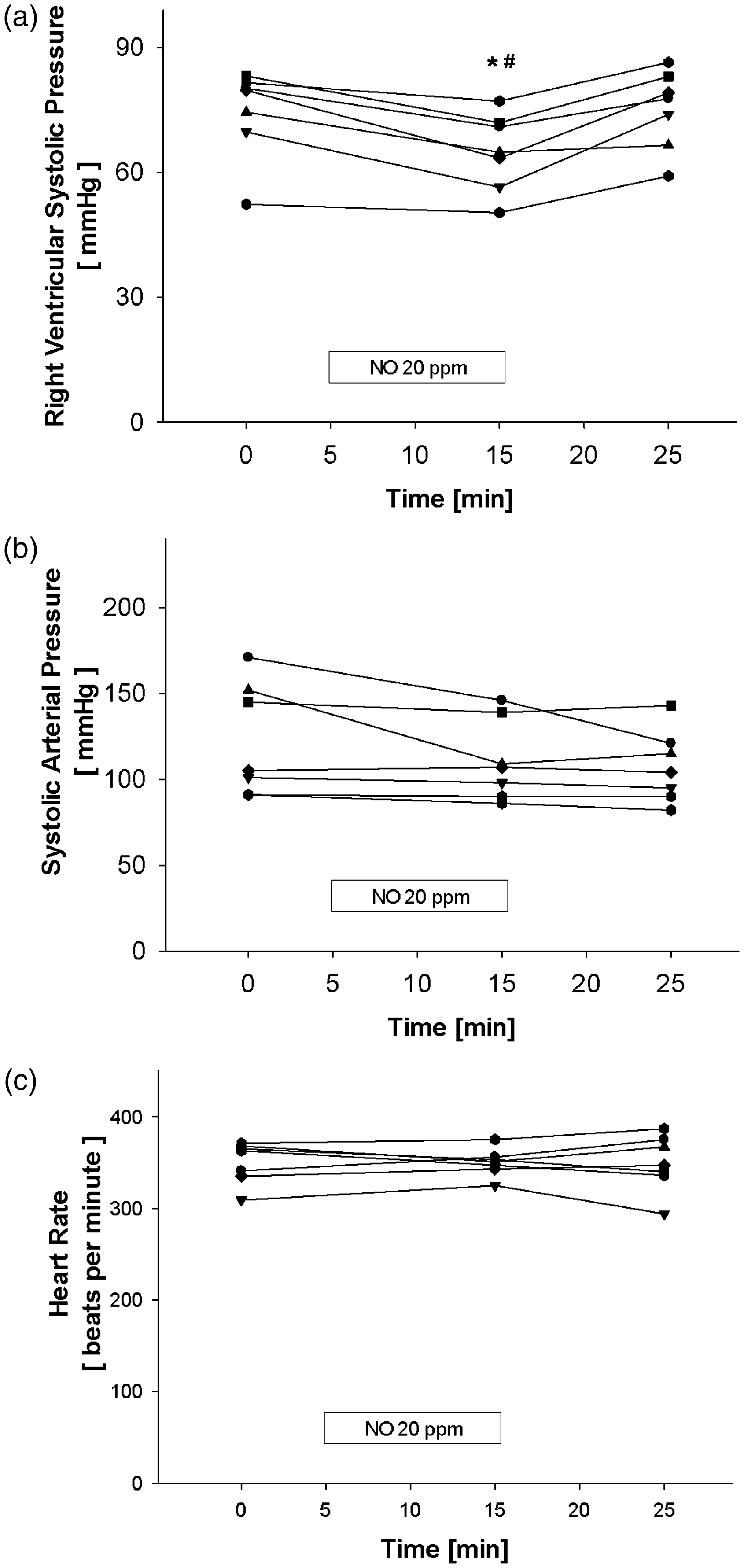
Thickness of the pulmonary arterial walls
To evaluate both the right ventricular and the left ventricular weight index as well as the wall thickness of the pulmonary vessels, each heart was dissected before the perfusion was started. After removal of the heart each catheter with the fixatives displaced easily and it was difficult to get a closed perfusion system. Accordingly, two of the rats, one after MCT and one after saline challenge, were excluded from the histological evaluation because of inadequate perfusion. For better quality of lung perfusion and histology we recommend strictly following the protocol by Jones and colleagues. 28 The luminal-to-vessel area ratio was significantly lower after MCT challenge compared with saline challenge, suggesting an increase of pulmonary arterial wall thickness (MCT group: 0.18 ± 0.05, n = 6; saline control group: 0.50 ± 0.09, n = 6; P < 0.05)
Right ventricular and the left ventricular weight index
As an index of right ventricular hypertrophy, the ratio of the right ventricle weight to left ventricle plus septum weight (RV/LV + S) was calculated. The challenge with MCT caused an increased RV/LV + S ratio compared with the saline application (MCT group: 0.51 ± 0.12, saline control group: 0.35 ± 0.05; P < 0.05).
Discussion
In our study we investigated the animal models of acute hypoxia, thromboxane-induced or MCT-induced pulmonary hypertension. Our study aimed at identifying an experimental setting that would allow the investigation of selective pulmonary vasodilation. We found that acute hypoxia induced a profound decrease in systemic arterial pressure and only a transient increase in RVSP that returned to normoxic values within 30 min of hypoxia. Continuous infusion of the thromboxane analogue U46619 caused a variable and unstable increase in both RVSP and systemic arterial pressure in our experiments. Therefore, both acute hypoxia and the continuous infusion of the thromboxane analogue U46619 induced pulmonary hypertension, but were inappropriate for investigating selective pulmonary vasodilation. By contrast, a single MCT injection resulted in pulmonary hypertension after 23 days with a solid and substantial increase in RVSP while systemic pressure values remained normotensive. Inhalation of NO reversibly lowered RVSP without systemic effects, suggesting a successful selective pulmonary vasodilation.
The examination of pulmonary selectivity requires a parallel measurement of systemic and pulmonary blood pressures. Even if our study did not aim for a comparison of the three protocols, it is apparent that mean baseline systemic arterial pressures and body weights were different among the groups. In a post hoc analysis we could confirm significantly lower baseline systemic arterial values in the acute hypoxia protocol compared with either the U46619 infusion or the MCT challenge protocol. In parallel, the mean body weights were significantly different among the three groups, with the lowest values in the U46619 infusion group, higher values in the MCT challenge group and the highest values in the acute hypoxia group. The significantly different body weights can be explained by the consecutive conductance of the experiments and, thereby, the lack of randomization. When we summarized the values of all the animals, the body weight and the baseline systemic arterial pressure were significantly inversely correlated (R = −0.568, P < 0.05). Since the anaesthesia was adapted to each body weight, the level of anaesthesia did not seem to be a causal factor for the systemic arterial blood pressure. A possible explanation might be the constant dosage of hydroxyethyl starch that might have induced a stronger effect on the systemic arterial blood pressure in the lighter animals of the thromboxane protocol when compared with the heavier animals of either the acute hypoxia or MCT protocol. An adjustment of the hydroxyethyl starch infusion rates to body weights might improve future experimental settings.
We used the RVSP as a measure of the pulmonary arterial systolic pressure. RVSP is often used to characterize pulmonary circulation since invasive haemodynamic measurements have shown that advancing a pressure catheter from the right ventricle into the pulmonary artery causes a stepwise increase in diastolic pressure, whereas the systolic pressure remains unchanged.29–34
In the present study, acute hypoxia caused a transient increase in RVSP. After 10 min of hypoxia, the RVSP returned to normoxic values. Our results are in accordance with previous findings which have shown a peak in the mean pulmonary arterial pressure at one minute of acute hypoxia followed by a decline to baseline values after 10 min of inhaling a hypoxic gas mixture. 14 Taken together, the results suggest that acute hypoxia provokes only a short-term elevated pulmonary arterial pressure in rats. In addition, we found that the systemic blood pressure profoundly declined during hypoxia, which is in accordance with other studies.11,14 We conclude that acute hypoxia briefly triggers hypoxic pulmonary vasoconstriction in Sprague–Dawley rats and also induces profound systemic vasodilation. Our study was undertaken to identify a stable animal model of pulmonary hypertension that would permit the investigation of selective pulmonary vasodilation. However, in anaesthetized rats, haemodynamic instability reveals acute hypoxia as an inappropriate animal model for examining selective pulmonary vasodilation with inhaled NO as a reference substance.
In order to use hypoxic pulmonary vasoconstriction as a model of pulmonary hypertension, modifications of the experimental protocol are necessary. For example, in the studies by Badejo et al. 20 and Schwenke et al. 17 ventilation with a hypoxic gas mixture was restricted to 4–6 min thereby using the initial hypoxic increase of pulmonary pressure in anaesthetized rats. Alternatively, a successfully extended hypoxic exposure of the rat seems possible in awake, freely moving animals that were instrumented two days prior to the hypoxic exposure.12,13,15 The observation period ranged between 90 and 180 min showing an increase in mean pulmonary arterial pressure and a decrease in mean systemic arterial pressure. However, instrumentation of the animals, recovery from the anaesthesia, and chronic instrumentation characterize this approach as demanding where animal welfare is concerned.
The thromboxane analogue U46619 is a potent vasoconstrictor that is frequently used to induce pulmonary hypertension. Some studies suggest that U46619 reveals greater effects on pulmonary than on systemic circulation.20,22,35,36 In our investigation, the infusion of the thromboxane analogue increased both RVSP and systemic arterial pressure. It might be possible that the applied dosage of U46619 per body weight contributed to the differences. In our experimental setting we chose a constant infusion rate of 240 ng/min of U46619 that caused variable responses in RVSP. Out of the six animals, one rat died after a rapid increase in RVSP and in two rats the RVSP did not increase at all. For the latter two animals the dosage of U46619 might have been too small, since other investigators have used either a higher steady infusion rate such as 374–506 ng/min of U46619 or priming doses followed by continuous infusion of 150–250 ng/min of U46619.20,35,36 The above-quoted studies adjusted the infusion rate of U46619 to achieve a mean pulmonary arterial pressure of approximately 30 mmHg.20,35,36 Retrospectively, it might have been better to have had set a target value for the RVSP even though the range of vascular preconstriction might differ among the animals. Within a predefined range of pulmonary artery pressure Aranda and colleagues have successfully investigated the effects of 100 ppm inhaled NO and other vasodilators on the pulmonary and systemic circulation in thromboxane-challenged rats. They found a significant decrease in pulmonary arterial pressure, whereas systemic pressure remained stable, suggesting selective pulmonary vasodilation by inhaled NO. 35 In addition, it has been suggested that thromboxane may induce bronchoconstriction and platelet aggregation.37–40 Our animal ventilator did not measure airway pressure, actual expiratory tidal volume or lung compliance as valuable parameters of bronchoconstriction. Furthermore, no blood analysis of platelet function was conducted. Therefore, we cannot exclude that bronchoconstriction or thromboembolic complications contributed to the instability of the thromboxane protocol in our study. Collectively, the thromboxane model seems to offer the opportunity for investigating pulmonary hypertension. However, the dosage, and thereby experimental success rate, seems to depend considerably on the investigator’s experience. Further studies may clarify whether bronchoconstriction or thromboembolic complication are relevant side-effects of U46619-induced PAH.
MCT-induced pulmonary hypertension has been subjected to criticism because of the lack of plexiform lesions, concomitant myocarditis in some animals, damage to the airways, and toxicological side-effects.24,41 In the liver, MCT is converted to its active metabolite dehydromonocrotaline. Dehydromonocrotaline contributes to the development of portal hypertension; it accumulates in erythrocytes, inducing platelet-mediated thrombosis and fibrin activation. 24 Despite its limitations, the MCT model mimics many components of human PAH such as pulmonary arterial and right ventricular hypertrophy. 24 Accordingly, we found pulmonary arterial hypertrophy, an increased right-to-left heart weight ratio, and enhanced values of RVSP in MCT-challenged rats. The inhalation of NO reduced RVSP without any decline in systemic arterial pressure, indicating selective pulmonary vasodilation. Therefore, the MCT model of PAH is a solid and reliable experimental setting for investigating selective pulmonary vasodilation.
The use of inhaled NO is often recognized as the gold standard, and is thus the reference substance for selective pulmonary vasodilation. While planning an animal study, biometric as well as statistical calculations are mandatory to avoid unnecessary animal experiments, and should be performed in accordance with the 3 R principle of replacement, reduction, and refinement. In our investigation, the MCT protocol was the most effective animal model for demonstrating selective pulmonary vasodilation. However, it should be pointed out that, based on our data, the sample size for detecting the NO effect on pulmonary arterial pressure as significant with a statistical power of 0.8, would require at least n = 15 evaluable cases per group in a controlled study. The corresponding calculation could be performed with the knowledge of standard deviation and the amount of the expected effect using standard formulas. For a selective pulmonary vasodilator with a lower effectiveness than inhaled NO, the sample size is expected to be considerably higher. For example, an effect in the magnitude of 80% compared with that of inhaled NO in our study would require a sample size of n = 23 animals in the intervention and control groups.
In summary, we examined the feasibility of acute hypoxia, infusion of the thromboxane analogue U46619 or MCT-induced pulmonary hypertension as rat animal models to investigate selective pulmonary vasodilation. We aimed for an experimental period of 30 min that would allow the use of NO as the reference substance for selective pulmonary vasodilation. We found that acute hypoxia causes only a transient increase in RVSP, and was too short for the examination of selective pulmonary vasodilation by NO. In addition, during acute hypoxia, the systemic arterial pressures declined, suggesting haemodynamic instability. A defined infusion of the thromboxane analogue with a fixed dosage caused variable responses to the pulmonary and systemic vascular tone in our study. The correct dosage of thromboxane and thereby successful implementation of sustained pulmonary hypertension seems to depend on the investigator’s experience. MCT induced a stable and reliable PAH. The inhalation of NO reduced RVSP without a decline in systemic arterial pressure, indicating selective pulmonary vasodilation. Therefore, the MCT model of PAH is a useful experimental setting for investigating selective pulmonary vasodilation. The present study may help researchers in choosing the appropriate animal model for investigating selective pulmonary vasodilation in rats, thereby avoiding unnecessary experiments and animal deaths.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The project was supported with funding by the Department of Anaesthesia and Intensive Care Medicine, University of Leipzig, Medical Faculty.
