Abstract
Observing and reporting clinical signs in laboratory animals is necessary for many reasons: the assessment of animal welfare, compliance with the principle of refinement (e.g. humane endpoints), regulatory compliance (e.g. reporting severity) and, importantly, as a scientific outcome, e.g. in animal models of disease or safety studies. Developments in the reporting of clinical signs will enhance the scientific value gained from animal experiments and further address the ethical cost. This paper discusses systematic approaches to the observation and reporting of clinical signs in animals (to be) used for research. Glossaries from public and corporate institutions have been consulted and a reference glossary has been set up, providing terminology to be tailored for institutional or project-specific use. The clinical examination of animals must be carried out by competent and specifically trained staff in a systematic way and repeated at adequate intervals and clinical observations must be registered effectively to allow this information to be used. The development of institutional or project-specific glossaries and the use of handwritten records or automated databases are discussed in detail. Among the users are animal care staff, veterinarians and researchers who will need to agree on a given set of clinical signs to be monitored routinely or as a scientific read-out and to train for the proper application. The paper introduces a long list of clinical signs with scientific terminology, descriptions and explanations as a reference glossary to be published and maintained online as a living document supported by the authors as an editorial committee.
Clinical signs are objective terms for describing abnormalities that are detected during a clinical observation or examination of an animal.
The reporting of clinical signs in laboratory animals is important for a number of reasons:
Retrospective reporting. In certain European countries, for example in The Netherlands, the gathering of such information serves to fulfil the requirements of retrospective reporting on severity. The Federation of European Laboratory Animal Science Associations (FELASA) supports the prospect of adopting retrospective reporting of scientific procedures on animals across Europe
1
and this issue is currently implemented as part of the new European Directive 2010/63.
2
Assessment of animal welfare. Data on clinical signs are instrumental for the recognition of pain, disease and distress in animals. Both specific and non-specific clinical signs can indicate severe discomfort to animals.
3
Carstens and Moberg
4
reviewed guidelines for the recognition and assessment of pain and distress in laboratory animals and published a table of indicators of pain that can be detected from the general behaviour, appearance and basic physiology of the animal. Effective communication between animal care staff, technicians, attending veterinarians and investigators. Clinical signs indicative of a health or welfare concern can be communicated objectively and consistently. Humane endpoints. Clinical signs are critical for the definition and application of the principles of humane endpoints. Ideal humane endpoints are those that can be used to end studies before the onset of pain and distress.
5
There are many excellent references on the use of clinical signs to define humane endpoints.6–13 Phenotyping. Reporting of clinical signs observed in animal models such as genetically-altered animals allows for improvements in phenotyping. This reporting should include the area of scientific interest, which is valuable for further characterizing the model, as well as those areas that may affect animal welfare.
14
Such information is essential for addressing the ethical cost–benefit analysis of the generation or use of such animal models. The endorsed Working Document on genetically-altered animals
15
drafted in the course of the implementation of Directive 2010/63 provides specific regulations and guidance on this. Safety/efficacy studies in pharmacology and toxicology. Reporting of clinical signs is routinely done in this field and aids the interpretation of clinical pathology in such studies. Scientific outcomes. Accurate data capturing should include any scheduled observations, as well as unscheduled observations, made during the in vivo phase of the research. This regards the phenotyping of models or any other scientific outcomes of research. These data will also contribute to serendipity, as unscheduled observations will be acquired systematically and may throw light on hypotheses under study or lead to novel ones. Also, unscheduled observations allow for timely expanding of specific observations, sample collection and analyses needed to complete the picture. Scientific publication. There is a general lack of reporting of the clinical signs seen in animal models in scientific publications, except when the clinical signs are an outcome parameter. The collection of standardized clinical data from laboratory animals may improve the quality of the scientific data processed by academics and may encourage the inclusion of such information in scientific publications. This, in turn, would potentially enhance animal welfare by providing input for project review and the refinement of procedures and models.
Historically in the laboratory animal field, clinical signs have been observed and recorded in toxicological research as part of the identification of dose-dependent adverse effects. Technicians responsible for the daily care of the animals carry out the observations and registrations. To this end, glossaries have been developed to standardize descriptive terminology. 16 Glossaries enable the data collected to be more accurate, favour descriptive terms, and avoid diagnostic terms (e.g. ‘distended abdomen’ is preferable to ‘pregnant’).
Many toxicology laboratories use automated systems to register observations on clinical signs as a part of the study outcomes. The use of standardized glossaries and automated systems is not as yet customary in most pharmaceutical companies or in academia but is clearly gaining ground.
The FELASA Working Group on ‘The reporting of clinical signs in laboratory animals’ was established to propose a glossary of clinical signs, after review of both public and privately owned (corporate) documents, that can model for a system for systematically observing and reporting clinical signs in animals used in research. The glossary would comprise a list of terms with definitions and background information that is scientifically precise and suitable for reference use by animal caretakers, technicians and investigators. The terms would only describe observations that can be made without the use of clinical instrumentation (e.g. a thermometer or weighing scales). The Working Group has chosen to restrict this task to mammalian laboratory species, with a particular bias to rodents.
This report aims to discuss the considerations relating to the reporting of clinical signs, which involves technical support, training and means of registration. The method of collection needs to be standardized in order to standardize the collection of data. This would include a method of examining the animal and examples of adaptable score sheets for recording observations.
The Working Group is mindful of the added bureaucratic burden that reporting of clinical signs will bring to an animal unit and will present the views of those in the field who are already practising such reporting.
Clinical observation and examination
Daily inspection
Daily inspection is to be done by persons who are familiar with the species and particularities that individual animals may have within normal limits. The room should be sufficiently lighted during observation. The observations should adhere to the following protocol:
When entering the animal room, be observant of any abnormal sounds (or lack of usual background noise), smells and other conditions. Check any notes on the management of the room or the animals. Observe the cages for any abnormal deposits, substrate disruption (or lack of maintenance of the nesting material by the animals), feed and drinking water (both supply and gradual consumption over time, in case of ad libitum administration, or leftovers, in case of meal feeding), etc. Observe each group for general activity level and any abnormal behaviour such as an animal dissociated from the group. Next, observe the animals one by one. Observe each animal from a distance for any abnormal behaviour, body contour, coat changes, etc. Next, when approaching the cage or pen, observe the response to external stimuli. In case the cage needs to be handled to view individual animals, as may be necessary when they are housed in containment such as an individually-ventilated cage (IVC) or an isolator, the response to stimuli is an integrated part of the initial observation (steps 2–4).
For daily inspection, the animals need not be handled, except where prior procedures or the observation of clinical signs require close attention. Only animals that need closer observation should be identified by reading their permanent individual mark (ear notch, tattoo, toe mark, transponder, or similar mark) or a temporary mark which can be applied to identify a single animal when group housed (for which there are many options, e.g. coat or skin coloration using a felt pen).
Clinical examination
The identification of the animal is read.
Clinical examination.

It is important to note whether a condition is generalized, symmetric or localized. Standardized anatomical nomenclature should be used to refer to specific parts or areas of the body (Figure 1).
Simple graphics indicating clinical terminology to map clinical signs onto the animal: (a) Ventral aspect of an animal indicating different parts of the body. (b) Ventral aspect of an animal indicating directional terms relative to the position in the body. (c) Dorsal aspect of an animal indicating different parts of the body (in lower case) directional terms relative to the position in the body (in upper case).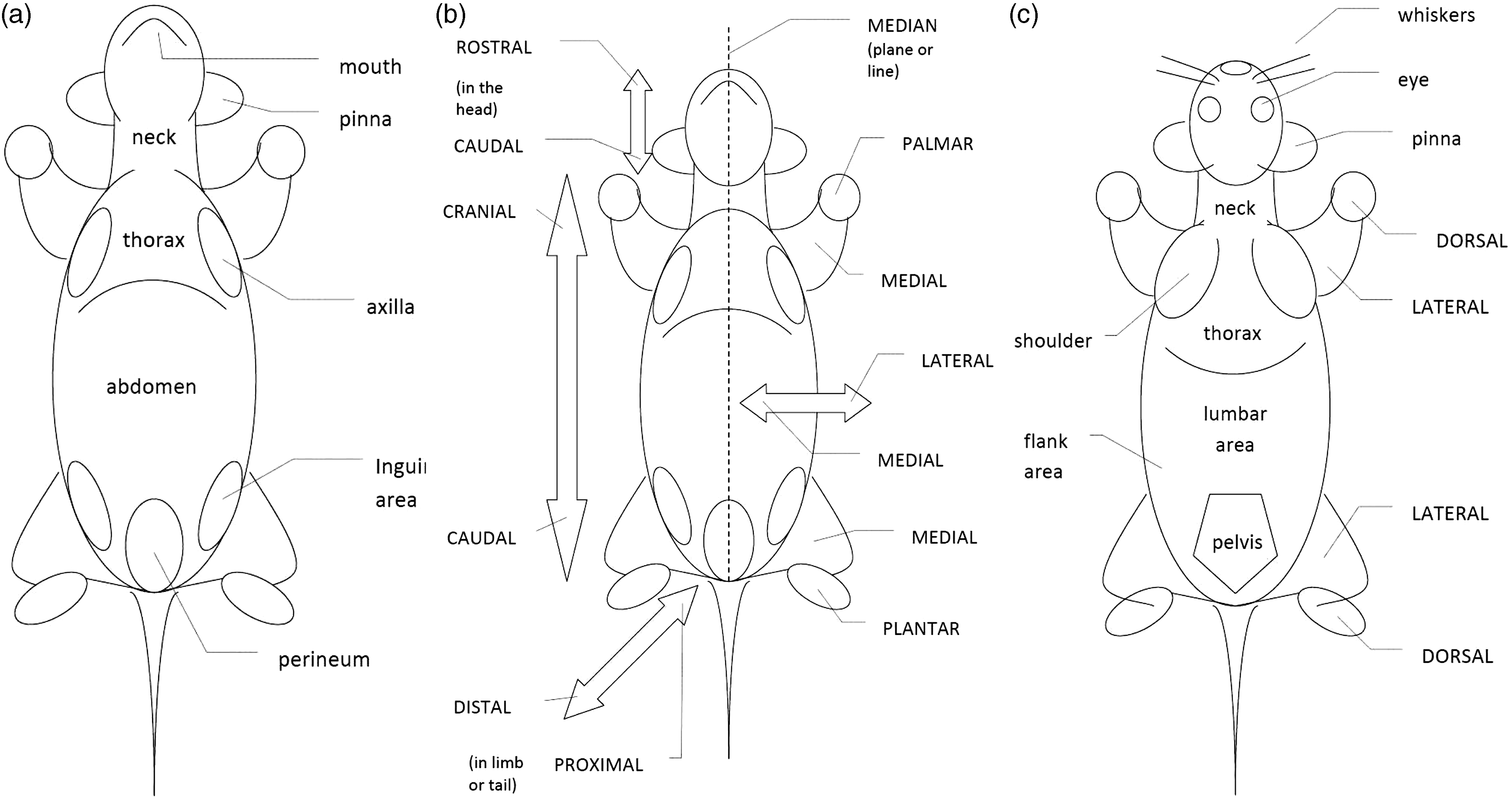
The behavioural response to capturing/handling should be noted, in particular abnormal passiveness or abnormal adverse reactions. Special conditions, such as aggressive animals or biocontainment conditions may prohibit the handling of animals for clinical examination. In such cases, the scheme should be followed by observation only, which may require more time than in a daily inspection and may be limited regarding some of the observations that can be made easily during handling (e.g. the ventral side of the body). When the animal is in hand, coat and skin texture and general muscle tone should be noted, as well as any behavioural responses such as withdrawal reactions. Observations such as skin temperature or moisture may be obstructed by wearing gloves but personal protective equipment must not be disregarded for routine clinical examination.
The scheme for clinical examination may appear elaborate but can be completed quickly by an experienced person. Only the definition and recording of a sign upon first observation will take some extra time.
Clinical examination may need to be repeated more frequently in a more focused way in the follow-up to a procedure or following up on the prior observation of a clinical sign until the conditions become stable or ameliorate.
The description of a clinical sign consists of the description of an abnormality, and its location, using anatomical terms. Anatomical terminology used by veterinarians can be very detailed. For common use, a simplified approach is preferred, as is outlined in Figure 1. It is important to indicate whether an abnormality occurs only on one side (left or right) or symmetrically. Reporting can be helped by using a simple graphic to indicate the location and size of a lesion, but this will mostly be done in a model- or species-specific setting.
The description of an abnormality should be given using terminology from a predefined glossary. Then, qualifiers can be added to indicate the size, extent (such as the amount of discharge), as well as chronicity (acute or sudden, subacute, chronic or permanent) which is reported as a repeated observation. Furthermore, day-to-day changes can be reported (increasing, decreasing, or healing). In the absence of a suitable predefined term, veterinary experts should be asked to provide one.
The reporting of clinical signs must not be confused with the reporting of diagnoses. A diagnosis is a conclusion to be made from clinical and additional diagnostic observations or measurements (e.g. blood analysis, microbiological testing). A clinical sign indicates some sort of abnormal condition but not its cause. An analysis of the cause will start with a differential diagnosis listing possible causes, taking into account their plausibility, in part based on other concomitant signs, the experimental use of the animal and epidemiological considerations. Veterinary textbooks 17 and other handbooks (such as Pritchett-Corning et al. 18 ) will show typical examples of conditions. Also, users of glossaries of clinical signs may have reference pictures showing typical examples of clinical signs listed. Such pictures are very useful as references supporting the description of clinical signs. However, to make a diagnosis from a picture alone would be inadequate and can often lead to false conclusions.
Users
Animal care staff
Animal care staff should be competent (educated and trained) to work with the type of animals in their care and to recognize normal and abnormal animal behaviours and external features. For breeding situations, this competence should include an understanding of normal reproduction and development. For animal research models, relevant information on the model should be readily available and consulted when needed.
Animal care staff should be competent to relate clinical signs to (affected) well-being, ill health, pain, and/or distress; and to report on this and to act on such observations in a responsible way. These reports should become more informative and objective by applying systematic approaches to observation and examination of animals and the use of standard terminology. Secondly, the reporting of clinical signs allows for better follow up (e.g. by a colleague on weekend duty or by consulting the veterinarian) and incorporation of these observations into the scientific outcome of the study. Animal care staff should use the reporting system in place and, if this is not a notification system, notify any senior staff, veterinarian and/or investigator depending on the local organizational structure and procedures.
Education for these functions should cover the biology (anatomy, physiology, ethology) of the species as well as health and welfare management, and breeding management as appropriate. Staff should be explicitly trained in the observation and examination of animals, as well as the use of terminology and registration systems used at the facility. The same requirements for competence should apply to persons conducting the procedures.
Investigator
The investigator is responsible for the selection of the model, the experimental design including the definition of humane endpoints, the acquisition of raw data and materials, and scientific reporting. A basic system for the reporting of clinical signs will cater for unscheduled observations to be reported in a systematic way, which will allow for a serendipitous approach to research outcomes and timely collection of data and samples when a humane endpoint is reached. Furthermore, standardized reporting of clinical signs will allow for good statistical evaluation and further interpretation of this information. An investigator may draft a model-specific score list for the systematic recording of clinical signs as a scientific outcome. Such a score list may contain standardized terminology from the glossary but may also include more specific or different observations to be made.
Veterinarian
Veterinarians attending to research animals should be trained in the laboratory animal medicine that applies to the species and research models in their care. The veterinarian will greatly benefit from good clinical assessments by technical staff and investigators who report on animals requiring closer attention. Also, the veterinarian can expand the clinical examination of the animal by including more specific diagnostic procedures. Finally, the veterinarian is responsible for translating clinical observations to diagnoses and for taking any measures needed. The overall outcomes of clinical reporting can be evaluated for any epidemiological trends in animal health and well-being, and any specific conditions that are relevant to the rest of the population. The veterinarian plays a crucial role in the education and training of technical and scientific staff, both on observational/examination skills and on the proper use of the institutional glossary and recording system.
Institutional veterinarians with a role in regulatory compliance who supervise the management of animal health and well-being, including the proper application of humane endpoints, should benefit from the reporting of clinical signs in terms of welfare (severity) evaluation.
Education and training
Present guidance documents on education and training (such as Weiss et al. 19 ) do not address skills in finding, examining and defining clinical signs specifically, indicating room for progress in this field. Furthermore, many scientists in biomedical science lack this type of training as part of their academic education, except for persons with clinical training in veterinary medicine, medicine or dentistry. Ethologists may be particularly skilled in noting behavioural signs but may not necessarily be skilled in their interpretation as a clinical condition. Thus, recognition and recording of clinical signs should receive due attention in laboratory animal science education. In addition to the acquisition of necessary skills, supplementary training is required in both clinical examination and the proper use of terminology in reporting. Members of the animal care staff interact with the animals on a daily basis and are familiar with the normal clinical presentation of the animals. Commonly occurring conditions are typically part of their professional experience. However, to apply terminology uniformly requires structuring and training. Well established institutional approaches can serve as the basis for tuition and education; but some practical training will be needed as well, in the acquisition of observation skills and in the proper use of terminology and means of registration. Typically, all direct users should be trained in the recognition and proper reporting of the signs listed for the institution or a specific study.
Means of registration
General sheets
The registration of clinical signs can be done using the daily log for an animal room or a specific group of animals (e.g. animals on a specific study). Each entry must be dated, and registration of the time of day may help to provide an estimate of the time of first occurrence/detection of the sign. This may also help to evaluate, for instance, behavioural features relative to the biological rhythm and the daily routines of the animals (e.g. before or after meal feeding). Any preferences for format will depend on the amount of data entry anticipated. For example, young healthy animals in stock will not generate many signs which need to be registered. For unscheduled observations, the institutional glossary needs to be locally available for reference purposes, and a format for registration needs to be provided, including all relevant items, e.g. the identification of an animal or a cage. In addition, any actions taken (such as reporting a case to the veterinarian) should be registered as well. It is important that any clinical cases that are brought to the attention of the investigator or the veterinarian are followed up. Any system used should inform any observer of significant observations made recently. This can be based on easy access to an automated database, printouts of the data on animals that have been scored recently, or the maintenance of the score sheets in the animal room. An example of a weekly general score sheet for one animal is presented in Figure 2a. This sheet can be started upon the first clinical remark on a particular animal. In addition, cage labels may be used to signal any animals with significant clinical conditions. New cases, when first observed, should be registered using approved terminology and reported as required by institutional organization and procedures. It is not unusual for laboratories or institutions to initiate a program for the systematic observation and recording of clinical signs by retrospectively analysing random comments from their own program. Typically, some of these entries will need some clarification. Next, they can be clustered to bring similar remarks together and bring them under a common denominator. These can then be renamed to what the staff agree upon, and a try-out phase can be started to see if the consistent application of this terminology is possible for all users, and determine how many cases cannot be described using the terms of the initial listing. The in-house glossary will still be completely flexible at this time, but training and communication will lead to improvements and finalization of this basic listing. This process is listed in Table 2.
. General score sheet. Quick start.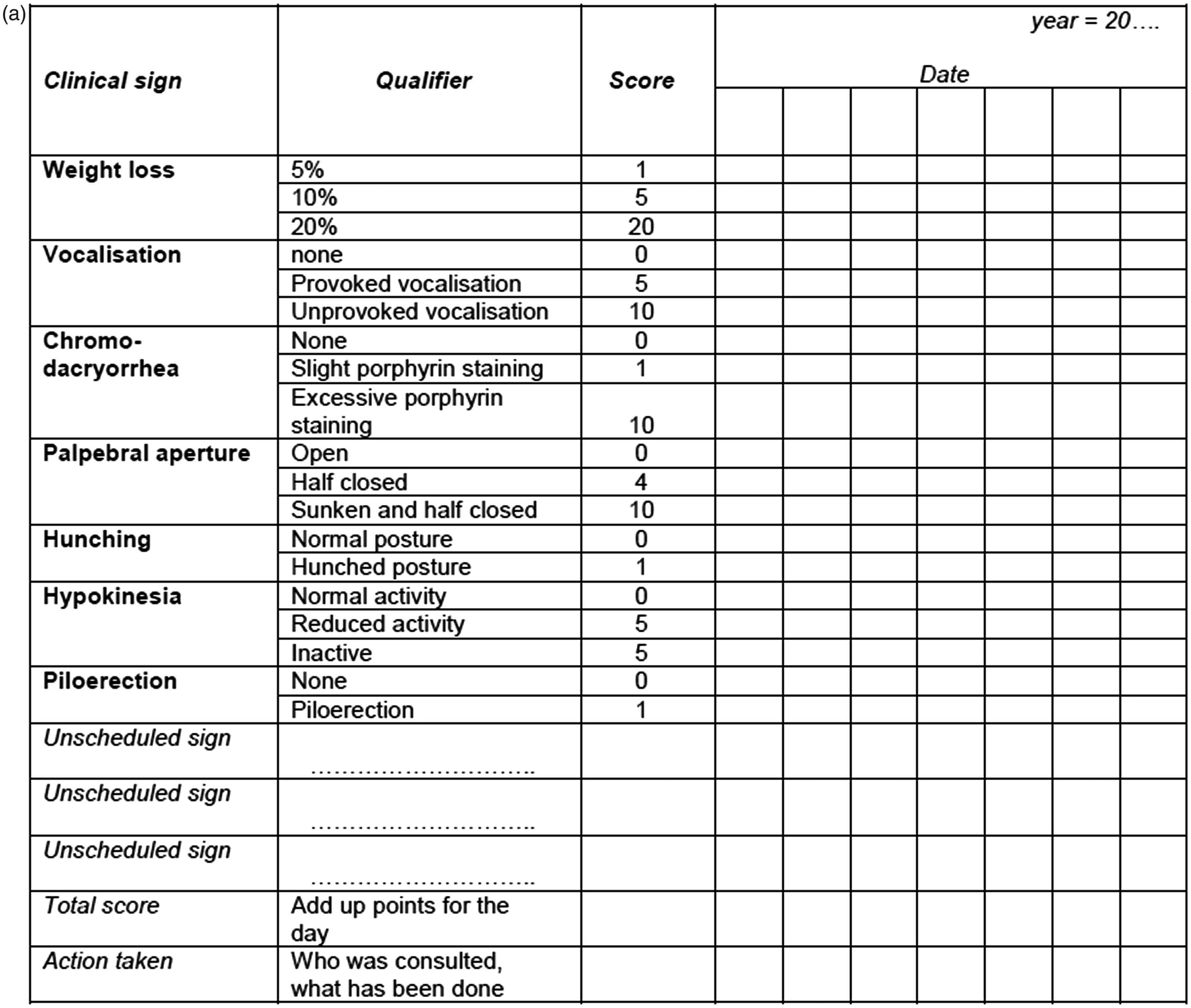

Score sheets – tailored for project
Clinical score sheets for specific models cater for the registration of scheduled observations in specific studies at the instruction of the investigator. Such records will typically list the observations that will contribute to the outcome of the study, with optional qualifiers (such as graded scores) or integrated scores that are derived from grading or combining of scores. The score sheets may indicate a humane endpoint and should leave room (open slots) for unscheduled observations. Such sheets can be designed to record data on one animal, for one observation, or observation days, or for a longer period of time. As an example of a model-specific form, a weekly score sheet for a mouse after the induction of experimental autoimmune encephalomyelitis (EAE) is shown in Figure 2b. Alternatively, forms can be designed to cover all animals on a specific study for single or multiple observations. When automated systems are used, data entry can be direct or the written data can be transferred to an automated system later on. Written reports should be managed in a traditional way, e.g. it needs to be clear who is to retain the record for further use of reference and how long it should be archived. This issue will be less complex in cases where automated records are made. Access by all users will be enhanced by keeping and updating documents in a shared folder, which will also facilitate the retention of records.
Project-specific lists will need to be first developed based on the clinical signs anticipated. In cases where painful conditions are likely to occur, typical species and model-specific clinical signs of pain should be included for systematic objective assessment and reporting.20–25
The form will evolve over time as some preprinted signs may never occur and others will need to be added based on their frequency.
Automated databases
Systems
Many automated systems for the management of colonies of laboratory animals or animal research have been developed in recent decades. Typical databases for animal management will use one record per animal containing fields for different types of data. In some systems, only animals in the study are registered; while in others, colony management, with or without breeding records and pedigrees, needs to be catered for. Not all such systems have modules for the registration of clinical signs, veterinary care and other items relevant to the management of animal health and welfare and data acquisition. It is beyond the scope of this report to elaborate on the types of automated animal management systems. In general, two fields of application have been developed and are in use in many institutions. Firstly, the organized acquisition of data on clinical signs in toxicology studies has been carried out for decades and has been automated from the 1980s onwards. Secondly, breeding records based on individual animals have always been relevant to systematic inbreeding and outbreeding, but the application of breeding records has become far more widespread after the technology for the genetic modification of animals was developed in the same period. This technology has also prompted the more systematic phenotyping of such models of which clinical signs are a significant part, in conjunction with physiological assessments, pathology, etc.
Thus, it can be safely assumed that increasing numbers of animals are being recorded in automated systems based on unique identification. Modules for the registration of clinical signs have been used routinely in toxicology, but less so in (academic) research institutions or breeders' establishments. Further development in the near future is anticipated, and therefore the time is ripe to discuss the preferred features of such systems. This discussion should not deal with obvious needs for continuity such as backups of data and database configurations but should focus on the types of information to be managed for the recording and reporting of clinical signs.
Animals
The unique record for each animal should contain specific information such as the species, strain, line, site and (approximate) date of birth, sex, etc. Any official or recommended nomenclature should be applied in the records, although this is not essential to the registration on the animal as such but to relate any data acquired to such information. The record number or animal number needs to be uniquely related to the animal identification attached to the animal or a defined small group of animals (such as animals in a litter), but these means of identification do not need to be the same. For instance a record number can point to a unique microchip number with a format that is entirely different. Also, records can be created and numbers can be assigned as animals are taken into a facility or into a specific study. It is necessary to consider which fields must be completed and which ones remain optional, and how rigid the system should be. For example in transgenics, several background strains are used frequently. However, crosses and backcrosses are made and it would be too rigid to allow for only one background strain or just an F1 hybrid. It may be sensible to include the option ‘mixed genetic background’ with or without more specific reference to the strains that are in the animal's pedigree.
Record keeping
It is important to consider how these records, and data entries to be added, will be managed. Which persons in the organization should have access to which records, with what type of authorization (read only, writing), and how will any changes be traceable? Should the system retain information on date and time when changes or additions have been made, and by which user? What should happen in cases where information is changed, e.g. should the older information be overwritten or should it be retained? How long and where should records be kept on animals that do not live anymore and studies become completed (please note, there are also legal requirements for this)? And who will need access to consult archived information, and will later additions or changes be allowed? In cases where the data management system is changed later, should these old data be readable using the new system or will the old management system be maintained for this purpose? Should the rights of a person who has left the organization be passed on to someone else, e.g. an investigator in the same research group? These are questions that need to be addressed upfront!
Writing and reading
Information on clinical signs should be entered by the persons who check animal health and well-being on a daily basis, typically involving animal care staff, but should not be restricted to them. Persons conducting procedures, animal welfare officers and veterinarians should be allowed to add information as well. Furthermore, mistakes can occur so there needs to be a limited time window to make corrections. Typical examples would be confusion about the identity of the animal reported or the need to correct the terminology used.
Automated systems offer the flexibility to be used by different users and to link to other locations or documents. To bridge the different needs of different users, glossaries can be predefined, e.g. by pre-programmed lists of conditions observed frequently. This terminology can be bilingual which can enable animal care personnel to use words in their native language, and which can then be read and used in English by the investigator.
Notifications
The system may or may not be used to notify specific persons of animals in need of attention. This option is intrinsic to the use of a centralized automated system rather than a local or paper-based system. Information of people involved, or even active notification, can be organized by shared access to the same database, by notifications by email (manually or automated), or be left to the discretion of persons who have actual access to the animals or who are responsible for daily inspections. It is important to restrict this type of notification to new cases and cases that deteriorate clinically, otherwise the information will soon fail to draw attention. Also, notifications may be restricted to clinical signs that may reflect affected health or well-being, or even be related to the severity expected in a specific protocol. An estimate of severity is to be based on procedures and observations on health and well-being. Clinical signs need to be assessed in an objective way, and the use of terminology such as ‘severe’ should be avoided in reporting. The duration of a condition will be reflected in the number of days that it lasts. However, in case a humane endpoint has been defined for a specific study or model, the nature and duration of typical clinical signs can help to define this endpoint more precisely and to ensure it is applied accordingly.
Recent history
Daily observations must be based on direct observations of the animals but also be related to observations made previously. As many observations will be made repeatedly over a longer time span (e.g. some sparsely haired areas), these should be copied easily for repeated reporting of the same observations. Defaults may be set to repeat those registrations until revoked. This will greatly simplify the registration process, thereby making it more time efficient and effective. Other types of observation are more likely to occur incidentally and may require repeated reporting.
Context
Apart from observations of clinical signs on one animal, other remarks may be given on individual animals or groups of animals because they are relevant to health or well-being. This should be set up in a most careful way. Examples would be the incidental deviations from set climatic conditions in a room, or an event in a particular cage (e.g. a drained water bottle). Some remarks might concern an animal with a behavioural condition that affects other animals clinically, e.g. aggression, or barbering (if one mouse in a cage still has its whiskers it is suspect to be the barber). Events to be reported should deal with deviations from the norm, e.g. food left would be the normal situation for animals fed ad libitum, in which case the absence of food is worth reporting. Contrarily, however, food left may indicate lack of appetite of an animal that is fed restrictedly (with meals). Thus, what and how to report should be worked out carefully to be relevant to the situation at hand, and personnel should be trained to make observations and report accordingly with sufficient knowledge of the biology and the normal husbandry of the species in mind.
The glossary
Terms, definitions, descriptions and qualifiers
Glossary of clinical signs.
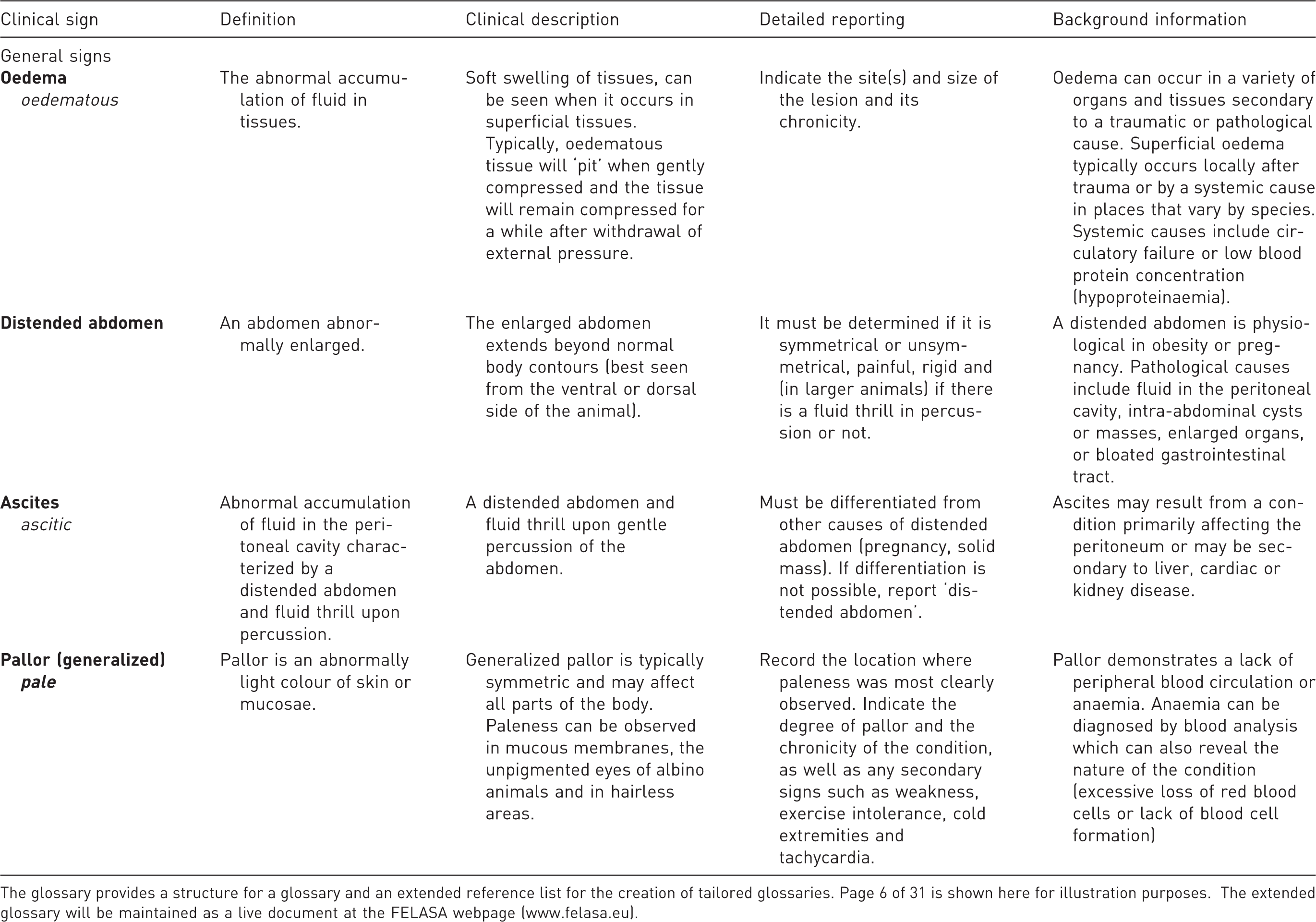
The glossary provides a structure for a glossary and an extended reference list for the creation of tailored glossaries. Page 6 of 31 is shown here for illustration purposes. The extended glossary will be maintained as a live document at the FELASA webpage (www.felasa.eu).
Clinical signs (alphabetical order).

The glossary is meant to be applicable to all mammalian species commonly used for research. The terminology can be adapted for use in birds (replacing ‘hair’ by ‘feathers’ and ‘parturition’ by ‘laying’, among other species-specific features). However, the glossary is not a veterinary dictionary nor does it offer information on the anatomy and physiology of the different species. We gladly refer to a wealth of information available on the biology of the various species (also handbooks which are easily accessible to hobbyists), and on the husbandry of the species in a research context. More specific information on the pathology, differential diagnosis, diagnostic confirmation and options for the management or treatment of conditions are found in general veterinary textbooks or specific references for laboratory animals.
Both a typical example from the glossary (Table 3) and the alphabetical listing (Table 4) are included in this report proper. The glossary will be managed as a live document on FELASA's website (http://www.felasa.eu) by the authors of this report as an editorial committee.
Translation of clinical signs to severity
There is no validated desktop approach to relating a randomly captured clinical sign, or a combination of signs, to the classification of severity of a condition. Clearly, some conditions are perceived to be more alarming than others, or justify immediate veterinary attention. The need to consult with a veterinarian upon first observation may be indicated on a score sheet or may result in automatic notification when a sophisticated database system is in place. However, to classify severity one needs to interpret the combined observations to make an overall assessment, and also the duration of the condition should be taken into account.
This situation is different for specific research models. For some the course of events can be predicted better and the pathology may be well documented. Thus, (sets of) clinical signs can be used to define scientific and humane endpoints based on onset, development and duration of the relevant sign(s), and a fair assessment of severity can be made. It is important to keep in mind that the clinical condition of the animal is one aspect of severity, and procedures are another. Typically, many procedures will not cause significant signs upon observations made the next day. The severity of a procedure should be assessed based on both experimental procedures and outcomes, and the experience of an animal should be evaluated over a longer period of time.
Footnotes
Acknowledgements
The Working Group wishes to acknowledge FELASA for venturing into this field and to thank the companies and academic institutions that have offered unpublished glossaries and score sheets for our consultation and use.
