Abstract
The aim of this study was to evaluate a total intravenous anaesthesia (TIVA) protocol using propofol and sufentanil without neuromuscular blocking agents (NBAs) for a non-recovery lung pathology study in rabbits including 10 h of pressure-controlled ventilation. TIVA was started with 20 mg/kg/h propofol and 0.5 µg/kg/h sufentanil. The depth of anaesthesia was assessed by reflex testing and monitoring of spontaneous movements or respiratory efforts. Vital parameters were monitored to assess the effects of the TIVA protocol. The infusion rates were increased whenever reflex testing indicated inadequate depth of anaesthesia, and were reduced when vital parameters indicated unnecessarily deep levels. Median infusion rates of 35 mg/kg/h propofol and 2.0 µg/kg/h sufentanil were needed to ensure an adequate depth of anaesthesia. This protocol suppressed spontaneous movements, breathing and palpebral reflexes, but was unable to suppress corneal and pedal withdrawal reflexes. Since significant drops in arterial blood pressure (ABP) were observed and the animals were not exposed to painful procedures, positive corneal and pedal withdrawal reflexes were tolerated. In conclusion, propofol and sufentanil is a suitable combination for long-term anaesthesia in non-recovery lung pathology models in rabbits without painful procedures. ABP must be monitored carefully because of the circulatory side-effects, but it is an inappropriate surrogate marker for depth of anaesthesia. Due to the lack of neuromuscular blockade this TIVA protocol allows the adjustment of infusion rates based on reflex testing. The resulting decreased risk of unnoticed awareness is a decisive refinement in anaesthesia for similar studies including long-term mechanical ventilation in rabbits.
Rabbits are frequently used as animal models for research on lung pathologies such as ventilator-induced lung injury (VILI), acute respiratory distress syndrome (ARDS) or ventilator-associated pneumonia (VAP).1–3 Anaesthetic regimens used for such models are commonly a combination of intravenous (IV) hypnotic, analgetic and neuromuscular blocking agents (NBAs). Combinations of (1) pentobarbital and pancuronium, (2) propofol, diazepam and pancuronium, (3) pentobarbital, urethane and pancuronium, (4) pentobarbital and pipecuronium, and (5) propofol, fentanyl and vecuronium have been used by authors studying lung pathologies.2–6
Depth of anaesthesia in rabbits can be assessed by testing palpebral, corneal and pedal withdrawal reflexes.7,8 However, this is not possible with anaesthesia protocols that include NBAs, and animals are therefore at risk of unnoticed awareness due to inadequate depth of anaesthesia. 9 Hedenqvist et al. introduced a total intravenous anaesthesia (TIVA) protocol using sufentanil and midazolam but without any NBAs. 10 However, the animals in their study were anaesthetized for only one hour for a surgical procedure and spontaneous breathing was tolerated.
It was the aim of this study to assess the suitability of an anaesthesia protocol without employing an NBA for a non-recovery experimental model of controlled, long-term ventilation in rabbits. TIVA with propofol and sufentanil was administered to five New Zealand White rabbits. All animals were orally intubated and mechanically ventilated for 10 h. During anaesthesia, reflex testing was performed and the animals were continuously monitored for signs of spontaneous breathing and movements to assess the level of anaesthesia. Vital signs, blood gases and several laboratory parameters were monitored during the experiments to register possible changes in cardiopulmonary function and potential adverse effects of the suggested anaesthetic regimen.
Animals
All experiments were performed according to the German Animal Welfare Act and were approved by the local authorities (Niedersächsisches Landesamt für Verbraucherschutz und Lebensmittelsicherheit LAVES, Oldenburg, Germany; AZ: 33.4-42502-04-13/1251). Evaluation of the anaesthesia protocol was part of a non-recovery study on volatile bacterial metabolites using an experimental model of ventilator-associated pneumonia. Local authorities had requested an evaluation of the anaesthetic regimen in preparation for the main study.
Five female New Zealand White rabbits (New Zealand White; Harlan Laboratories Inc, Indianapolis, IN, USA) with body weights of 3.0–3.2 kg were included in this evaluation study. The rabbits were housed in groups of 4–5 animals in pens of 8 m2 with hiding shelters. They were kept on straw bedding and fed a standardized rabbit diet (ssniff® K–H Alleinfuttermittel für die Haltung von Kaninchen; ssniff Spezialdiäten GmbH, Soest, Germany). Hay and drinking water were provided ad libitum. Room temperature was kept constant at 18–20℃ with a relative humidity of 40–60%. Light cycle was maintained by a constant change of 12 h of light and 12 h of darkness. The animals were kept under these conditions for at least 14 days prior to the experiments. They were examined and confirmed to be clinically healthy the day before the experiments. At the beginning of the experiment (one hour after anaesthesia induction) arterial blood samples from the auricular artery were taken to determine haematocrit (HCT), white blood cell count (WBC), gamma-glutamyl transpeptidase (GGT), glutamate dehydrogenase (GLDH), blood urea nitrogen (BUN) and serum creatinine (sCr) in order to exclude any subclinical disease.
Materials and methods
Each rabbit was administered 30 mg/kg of ketamine (Ketamin Inresa; Inresa Arzneimittel GmbH, Freiburg, Germany) and 1.5 mg/kg of midazolam (Midazolam Ratiopharm®; Ratiopharm GmbH, Ulm, Germany) as an intramuscular (IM) injection to induce anaesthesia together with a subcutaneous (SC) depot of 0.06 mg/kg of glycopyrrolate (Robinul®; Riemser Arzneimittel AG, Greifswald, Germany) to reduce salivation and bronchial secretions. A 22-gauge IV catheter (Vasofix® Braunüle®; B Braun Melsungen AG, Melsungen, Germany) was placed percutaneously in an auricular vein for drug administration and a 24-gauge catheter (Vasofix® Braunüle®; B Braun Melsungen AG) was placed percutaneously in the opposite auricular artery for invasive blood pressure monitoring.
Approximately 10–15 min after the administration of ketamine and midazolam the animals had lost their righting reflex and were considered to be unconscious. Orotracheal intubation was then performed without direct visual control under acoustical guidance (spontaneous breathing sounds) using a cuffed, 3.0 mm ID endotracheal tube (ETT) (Microcuff®; Kimberly-Clark Global Sales LLC, Roswell, GA, USA), and the tube position was confirmed by capnography. Anaesthesia was then deepened with a bolus injection of 7 mg/kg of propofol (Propofol Lipuro®; B Braun Melsungen AG) and 0.75 µg/kg of sufentanil (Sufenta®; Janssen-Cilag GmbH, Neuss, Germany). The animal was connected to a paediatric intensive care respirator (Babylog®; Drägerwerk AG & Co KGaA, Lübeck, Germany), and pressure-controlled ventilation (PCV) with a constant positive end-expiratory pressure (PEEP) of 3 mbar and a maximum peak inspiratory pressure of 20 mbar was started. The initial tidal volume was 6 mL/kg, with a respiratory rate of 45 breaths/min and an inspiration–expiration ratio of 1:1.5. In order to ensure normoventilation the respirator settings were adjusted according to the results of the blood gas analyses that were performed throughout each experiment. The initial inspiratory oxygen concentration (FiO2) was 100%, which was reduced to 30% when peripheral oxygen saturation (SpO2) was at least 96% in order to minimize oxidative stress.
Anaesthesia was maintained with continuous IV infusions of propofol and sufentanil using two infusion pumps (Perfusor FM®; B Braun Melsungen AG). Starting rates were 20 mg/kg/h for propofol and 0.5 µg/kg/h for sufentanil. In addition, a continuous IV infusion of balanced electrolyte solution (Sterofundin®; B Braun Melsungen AG) was administered at a rate of 35 mL/h. The animal was placed under a convective warming blanket connected to a forced-air warming device (Mistral-Air®; The Surgical Company International BV, Amersfoort, The Netherlands) in order to maintain a rectal temperature of 38–38.5℃.
Palpebral, corneal and pedal withdrawal reflexes were tested every 15 min, and the animals were observed for spontaneous movements of the eyes (e.g. nystagmus and blinking) and the jaw as well as for signs of spontaneous breathing (e.g. decreased tidal volumes and/or increased inspiratory pressure). Palpebral reflexes were tested by touching the medial canthus with a cotton swab. Single drops of sterile saline were used to test the corneal reflexes. Pedal withdrawal reflexes were tested by pinching the skin between the toes of the hind paws. Reflex testing was performed by the same investigator (VR) to avoid inter-investigator bias. The administration rate of sufentanil was increased by 0.25 µg/kg/h if palpebral reflexes were positive and/or if spontaneous breathing and/or spontaneous movements occurred. The administration rate of propofol was additionally increased by 5 mg/kg/h in case of palpebral reflexes remaining positive and/or spontaneous breathing and/or spontaneous movements.
Vital parameters (invasive blood pressure [IBP], pulse rate [PR], SpO2 and rectal temperature) were monitored continuously (Dash 2000®; GE Healthcare GmbH, Solingen, Germany). The monitor was connected to a notebook (ThinkPad R50e; IBM, Armonk, NY, USA) using its AUX-output connector and a set of all vital parameters was automatically transferred into an Excel file (Microsoft Excel®; Microsoft Corp, Redmond, MA, USA) every 7 s.
Blood samples for arterial blood gas analyses (GEM Premier 3000®; Instrumentation Laboratory GmbH, Kirchheim bei München, Germany) were taken one hour after anaesthesia induction and then every 3 h, up to 10 h after the beginning of each experiment. Sets of blood samples for laboratory examinations were taken at the beginning (one hour after anaesthesia induction) and at the end (10 h after anaesthesia induction) of each experiment. Laboratory examinations were performed at Laboklin (Labor für Klinische Diagnostik GmbH und Co KG, Bad Kissingen, Germany) and included HCT, WBC, GGT, GLDH, BUN and sCr. The total blood volume withdrawn from each rabbit for blood gas and laboratory analyses was in accordance with recommendations for blood sampling of the German Society of Laboratory Animals.
As part of the main study investigating volatile bacterial metabolites in a model of ventilator-associated pneumonia, computer tomography (CT) scans of the thorax of each animal were performed at 1, 4, 7 and 10 h after induction. Mechanical ventilation and TIVA were interrupted temporarily in order to place the animals in the CT scanner. Mechanical ventilation was continued directly after the animal was correctly positioned. Administration of TIVA had to be interrupted during the duration of the CT scan lasting approximately 5 min. The animals were given bolus injections of 7 mg/kg of propofol and 0.75 µg/kg of sufentanil before and after each CT scan in order to maintain adequate levels of anaesthesia for these procedures. Figure 1 gives an overview of all procedures and measurements performed during the experiments.
Timeline showing all procedures and measurements performed during the experiments. SpO2: peripheral oxygen saturation, CT: computed tomography.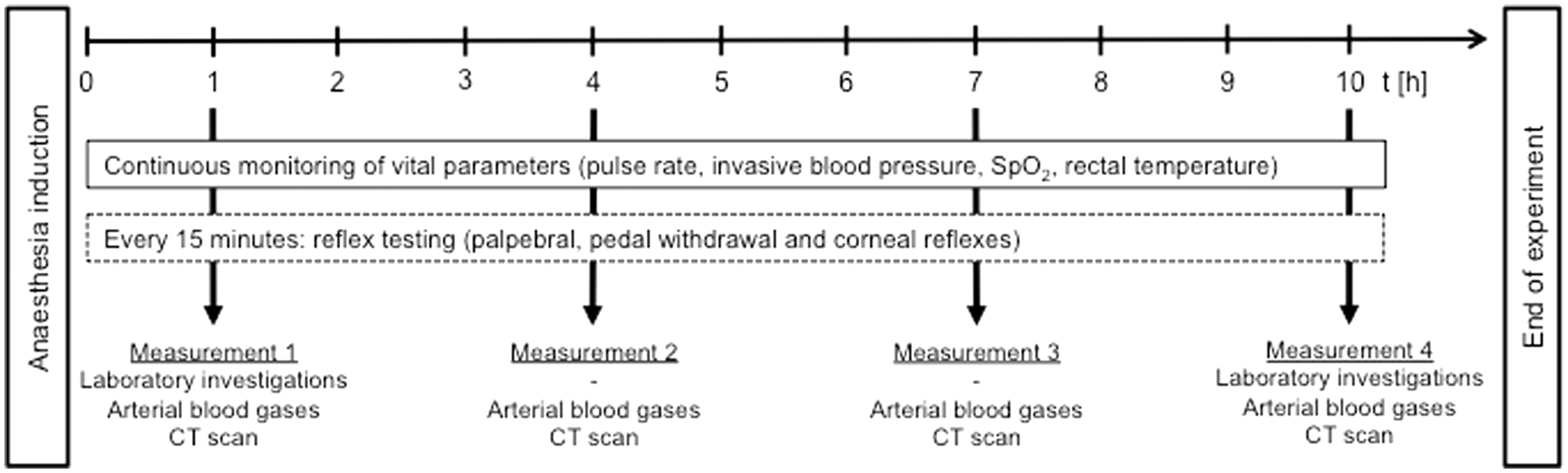
After 10 h of mechanical ventilation TIVA was deepened with propofol and sufentanil until reflexes disappeared and circulatory arrest occurred as shown by the loss of pulsatile signal in arterial blood pressure (ABP) measurement. Cardiotomy was then performed and the lungs were removed for histopathological examination.
Results
Results of the blood examinations one hour and 10 h after anaesthesia induction in New Zealand White rabbits (n = 5).
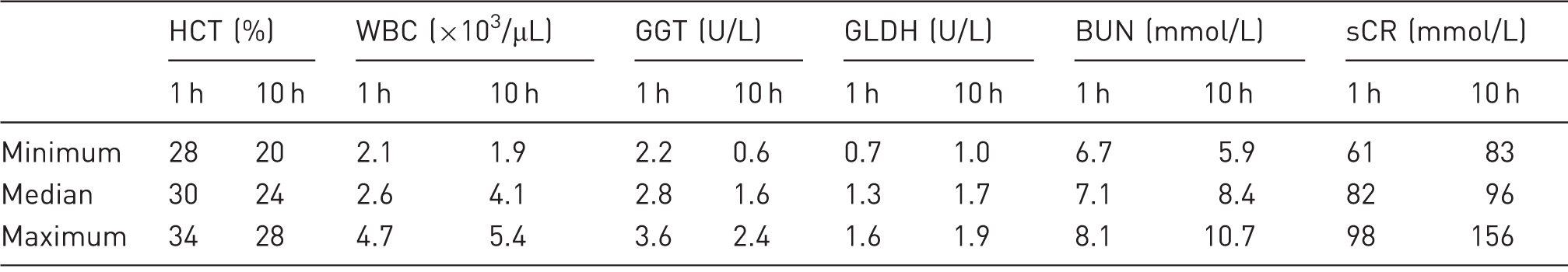
HCT: haematocrit, WBC: white blood cell count, GGT: gamma-glutamyl transpeptidase, GLDH: glutamate dehydrogenase, BUN: blood urea nitrogen and sCr: serum creatinine.
Administration rates of propofol and sufentanil, vital parameters and blood gases measured during propofol and sufentanil anaesthesia in New Zealand White rabbits (n = 5).

Values are presented as median and range. MAP: mean arterial pressure, PaCO2: partial pressure of carbon dioxide, PaO2: partial pressure of oxygen, FiO2: fraction of inspired oxygen.
Coughing on introduction of the ETT into the trachea was suppressed by the induction doses of propofol and sufentanil. All animals tolerated the onset of PCV.
Infusion rates of both propofol and sufentanil had to be increased during the experiments following positive palpebral reflexes, and/or spontaneous movements and/or respiratory efforts. Table 2 shows the adjusted infusion rates during the experiments. Corneal and pedal withdrawal reflexes could not be suppressed by these infusion rates.
In addition, the bolus administrations of propofol and sufentanil before and after every CT scan were necessary to suppress responses to adverse stimuli such as movements of the ETT in the trachea during positioning of the animal as well as to maintain adequate levels of anaesthesia for the scanning procedure.
Discussion
It was the aim of this study to assess the suitability of an anaesthesia protocol which did not include NBAs for an experimental model of controlled long-term ventilation in rabbits in a non-recovery setting. To this end, five New Zealand White rabbits were given a TIVA using propofol and sufentanil. Depth of anaesthesia was assessed by reflex testing and monitoring of spontaneous breathing and movements. Vital parameters were monitored to detect any side-effects of the TIVA regimen.
Propofol is a hypnotic agent with a rapid onset and a short duration of action. It therefore allows rapid adjustment of anaesthesia levels by adapting its rate of infusion. The combination of sufentanil, a highly potent opioid agent, and propofol effectively suppresses spontaneous breathing, as desired for studies on lung pathologies. Sufentanil is 500 times more potent than morphine and has a favourable context-sensitive half-time after continuous administration for several hours. Propofol and sufentanil were both used in recently published anaesthesia protocols for rabbits. De Mulder et al. used propofol in combination with fentanyl, and Hedenqvist et al. administered sufentanil and midazolam for anaesthesia in rabbits.4,10 In the present study, sufentanil was used instead of fentanyl because of its favourable pharmacological characteristics (e.g. half-time).
Midazolam has only a weak hypnotic effect. Moreover, in the study by Hedenqvist et al. the rabbits were allowed to breathe spontaneously with the midazolam and sufentanil combination. 10 A combination of propofol and sufentanil was therefore considered best for the purposes of the present study. As this combination is also commonly used in humans requiring long-term ventilation, the results of translational research using this TIVA protocol are potentially more comparable than animal experimental studies using other anaesthetic protocols. 11
Several other combinations have been proposed for long-term anaesthesia in rabbits, but most used substances that are restricted to veterinary medicine. Anaesthesia protocols incorporating xylazine or alfaxalone are commonly used in veterinary anaesthesia, but as both substances are not used in humans, the study results may be of limited value from a translational point of view. In addition to these limitations, the two substances suppress spontaneous breathing insufficiently and are therefore not considered suitable for the present study. 12
The results of this study show that a combination of propofol and sufentanil provides adequate anaesthesia over several hours when the infusion rates are adjusted according to the actual depth of anaesthesia. With the employed anaesthesia induction sequence, all animals tolerated the ETT and the onset of PCV. However, during the ongoing experiments all animals showed movements of the eyes (nystagmus and blinking) and the jaw. The administration rates of sufentanil and propofol had to be increased to achieve suppression of palpebral reflexes, spontaneous movements and respiratory efforts. Using reflex testing or the occurrence of spontaneous movements or breathing to adjust infusion rates is not possible when NBAs are part of the anaesthetic protocol, and inadequate depths of anaesthesia or even awareness may go unnoticed.
Although no surgical stimulus was applied during this study, the infusion rates had to be adjusted during the 10 h experiments to ensure adequate levels of anaesthesia. Rabbits undergoing surgery as part of a study protocol are exposed to higher levels of pain and stress. In such models NBAs are often used to provide optimal surgical conditions, and dosage adjustment is not necessarily part of the anaesthesia protocols, not even in open-chest models of lung research. In 2005, D’Angelo et al. used pentobarbital sodium and urethane for anaesthesia in their study on respiratory mechanics. Pancuronium bromide was administered at anaesthesia induction, which made assessment of anaesthetic depth impossible before the initiation of invasive procedures such as tracheal and vascular cannulation. 5 In 1997, De Mulder et al. introduced an open-thorax rabbit model and used a combination of propofol and fentanyl. In their study, NBAs were only given after the individual adjustment of anaesthesia levels that were considered to be necessary for the surgical procedures. 4 Vecuronium bromide was then administered after reflexes were sufficiently suppressed and there was no reaction to painful stimuli. However, once vecuronium bromide had been given, it was no longer possible to continuously ensure an adequate depth of anaesthesia. Heart rate and/or blood pressure can be used to assess depth of anaesthesia, since insufficient anaesthesia can be associated with their increase due to (surgical) stress. In our study, positive reflex testing and spontaneous movements revealed inadequate levels of anaesthesia with no concomitant increases in heart rate or blood pressure. Heart rate and blood pressure must therefore be considered to be inappropriate surrogate parameters for assessing the depth of anaesthesia in experimental settings without surgical stress.
In our study, the heart rate decreased with the increasing administration rates of propofol and sufentanil during the first 2 h but then remained stable for the rest of the duration. Mean arterial pressure (MAP) also showed an initial downward trend in all rabbits before stabilizing at 40–45 mmHg for the remaining study time. Compared with normal values in awake rabbits this must be regarded as marked hypotension. 13 But since oxygenation, decarboxylation, serum lactate and markers of organ function remained stable, no animal was considered to be in a critical circulatory state and treatment was not considered necessary. To avoid a further drop in ABP, the infusion rates of propofol and sufentanil were not increased and positive pedal withdrawal reflexes were tolerated throughout the study. And since the aim of this study was to evaluate the effects of the propofol and sufentanil combination and its suitability for long-term anaesthesia in rabbits, the use of additional drugs was not taken into consideration.
Heart rates and MAP in the present study are similar to values measured in rabbits under isoflurane and ketamine/medetomidine anaesthesia.10,14 Authors who used NBAs in their anaesthesia protocols reported less hypotension, but it remains unclear whether inadequate levels of anaesthesia were a possible explanation for this observation.2,4 For this study, ABP was measured in the auricular artery, which may also lead to lower values compared with measurements in more central arteries (e.g. femoral artery). 15
In conclusion, TIVA with propofol and sufentanil provides adequate anaesthesia for non-recovery studies including long-term mechanical ventilation in rabbits. By reflex testing and monitoring of spontaneous movements (including respiratory efforts) an appropriate anaesthesia level can be achieved. If NBAs are required as part of the anaesthesia protocol, the depth of anaesthesia should be monitored by additional parameters independent of muscular activity (e.g. bispectral index). In these studies the dosage of the anaesthetic drugs should be liberally increased during the experiments to avoid the risk of unnoticed awareness. Hypotension is a side-effect of general anaesthesia and therefore vital parameters need to be monitored very carefully. IBP monitoring and blood gas analyses should be considered for longer lasting experiments to enable advanced interpretation of organ and tissue perfusion.
Although all animals in the present study tolerated the ETT and mechanical ventilation, it was not possible to completely suppress all reflexes. Pedal withdrawal reflexes and corneal reflexes were not suppressed even at the highest infusion rates of propofol and sufentanil used in this study, indicating that anaesthesia levels were not deep enough for painful surgical procedures. However, as significant reductions in pulse rates and MAP values were observed, these reflexes were tolerated to avoid a further decrease in MAP. The evaluated TIVA protocol enabled tolerance of the minor distressful procedures needed for this study protocol. In conclusion, NBAs are not necessary for anaesthesia in rabbit studies of lung pathologies and increase the risk of awareness. The combination of propofol and sufentanil without NBAs therefore might represent a significant refinement of general anaesthesia in rabbit models of lung pathologies including long-term mechanical ventilation without major painful surgical procedures.
Footnotes
Acknowledgements
The author(s) would like to thank Claudia Ottersbach, Jutta Sichelstiel and Hartmut Danneberg for their support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a grant from the Faculty of Medicine of the University of Göttingen.
