Abstract
The body’s motion and function are all in part effected by a vital tissue, the tendon. Tendon injury often results in limited functioning after postoperative procedures and even for a long time after rehabilitation. Although numerous studies have reported surgical procedures using animal models which have contributed to both basic and clinical research, modeling of tendon sutures or postoperative immobilizations has not been performed on small experimental animals, such as mice. In this study we have developed an easy Achilles tendon suture and postoperative ankle fixation model in a mouse. Right Achilles tendons were incised and 10-0 nylons were passed through the proximal and distal ends using a modified Kessler method. Subsequently, the right ankle was immobilized in a plantarflexed position with novel splints, which were made from readily available extension tubes. Restriction of the tendon using handmade splints reduced swelling, as opposed to fixating with the usual plaster of Paris. Using this method, the usage of the right Achilles tendons began on postoperative days 13.5 ± 4.6, which indicated healing within two weeks. Therefore our simple short-term murine Achilles tendon suture procedure is useful for studying immediate tendon repair mechanisms in various models, including genetically-modified mice.
Prolonged misuse of an area affected by injury not only frustrates patients, but also has potential risks of tendon adhesions with the surrounding tissues, contracture deformities in the joints, and re-ruptures after primary repair. To address these problems, postoperative therapies such as immobilization using casts for three weeks, modified Kleinert protocols (active extension, rubber-band passive flexion),1,2 and early mobilization including Duran techniques (passive extension, passive flexion) 3 are currently the standard early programs for rehabilitation of flexor tendon injuries. Although these protocols have been well developed, prolonged healing compromises the function of tendons. Since there is no standard protocol to follow for mice, let alone for humans, we developed an easy Achilles tendon suture and postoperative ankle fixation in mice, which has advantages in genetic engineering and handling.
Female C57BL/6 strain mice (Japan SLC, Inc., Shizuoka, Japan), aged 14–18 weeks and weighing between 19 and 22 g were used. Animal care and treatment were provided in accordance with institutional guidelines. All procedures were performed in accordance with the guidelines of our institutional animal care and use committee (Approval Number 2013-186).
A 15 mm long splint, constructed from sagittal split pieces of extension tubes (SF-ET2527; Terumo Corporation, Tokyo, Japan) was used in the treatment. To prevent pressure sores, corners with sharp edges were removed by cutting (Figures 1a–c).
Ankle fixation using extension tubes and cyanoacrylate adhesives. (a), (b), (c) Side view of a separated extension tube. Sharp angles (circles in a) and edges (arrows in b) were trimmed. (c) Trimmed splint. (d) Immobilized right leg using handmade splints. Arrows: nylon sutures. (e) The removed splints on postoperative day (POD) 7. The nylon sutures (arrows) were also removed with the splints.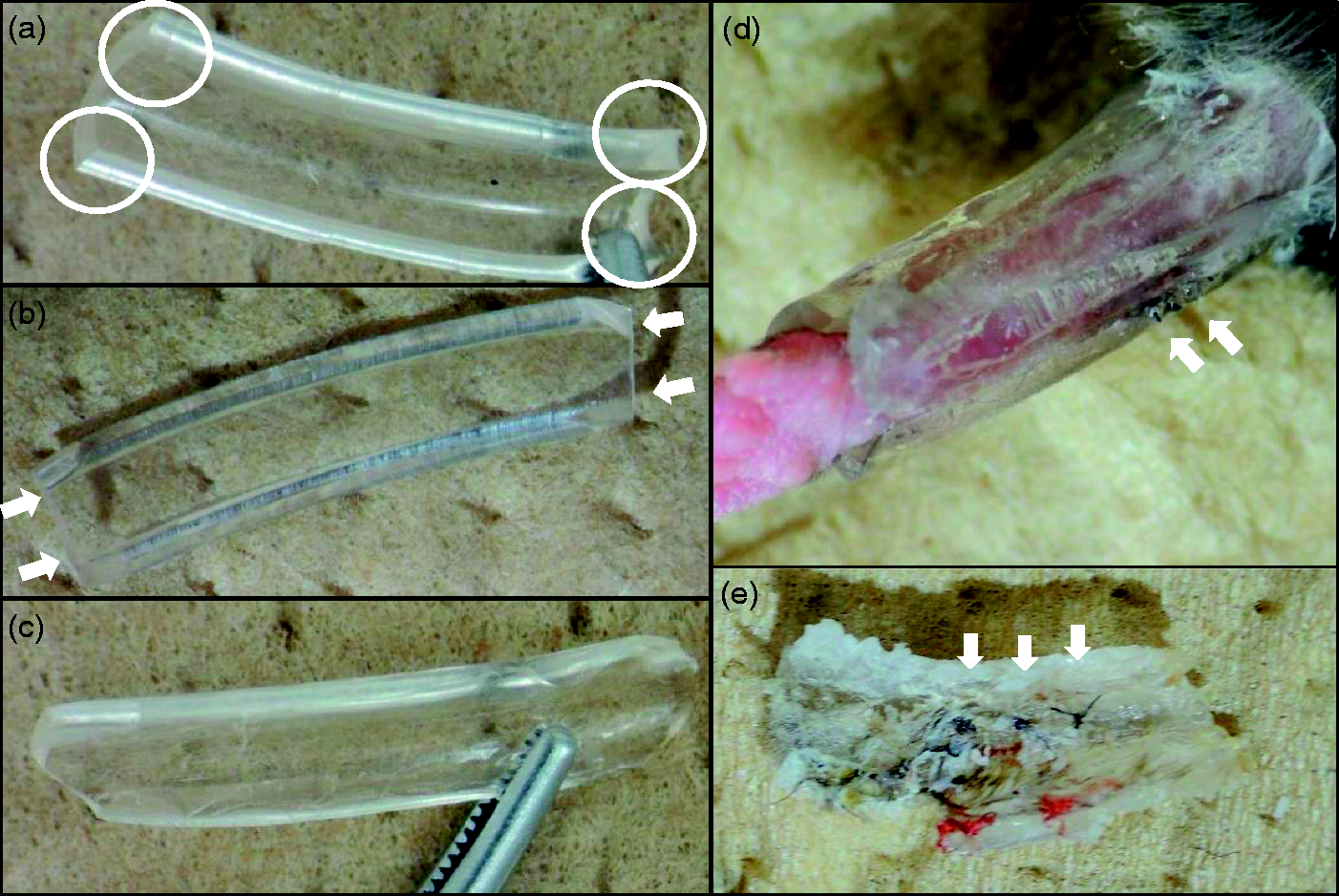
For surgery, general anesthesia was administered to the mice by inhalation of 4% isoflurane with a rodent ventilator (Narcobit K-100RG, Natsume Seisakusho Co., Ltd., Tokyo, Japan) in an induction box. Anesthesia was maintained by the inhalation of isoflurane vaporized in fresh 96–100% air at 500 mL/min through a handmade facemask, which was composed of a latex glove thumb. The isoflurane gas concentration was adjusted to 2–4% with visual respiratory monitoring including signs of chest and abdominal motions. Before proceeding with surgery, the anesthesia depth was checked by testing the pedal reflex followed by pinching of the feet with forceps.
The right Achilles tendons were exposed for incision using straight Iris fine scissors (13-183; BRC Bioresearch Center Co Ltd, Nagoya, Japan). After the tendon was ruptured with the scissors, 10-0 nylon sutures were passed through the proximal and distal ends using a modified Kessler’s method. The sutures were then tied up, and the skin was closed with 7-0 nylon sutures. Matrigel was used (354262, matrix high concentration [HC], phenol-red free and lactose dehydrogenase elevating virus [LDEV]-free; Corning, Bedford, MA, USA) as a carrier. The matrigel injection solution (25 uL) was formulated with cold matrigel and equal amounts of Dulbecco’s modified Eagle’s medium, containing 20% fetal bovine serum, 1% penicillin–streptomycin solution, and 1% glutamax (35050-061; Life Technologies, Grand Island, NY, USA). Injections were applied among the tendon stumps before the sutures were tied up. Soon after skin closure, the right ankles were immobilized with two or three pieces of the prepared splints and an alpha cyanoacrylate adhesive (Aron Alpha, GEL-EX, #05274; Toagosei Co Ltd, Tokyo, Japan) for seven days (Figure 1d). Careful observations were made by monitoring the circulation in the right foot, and also by looking for extreme swelling, or painful behaviors such as avoidance of using the right leg, and by looking for pressure sores on postoperative days (POD) 2 or 3. On POD 7, the splints and the nylon sutures in the skin were carefully removed under anesthesia (Figure 1e).
To determine the effect of the surgical operation, the mice were monitored carefully to see whether they used contraction of their right triceps surae muscles and whether they plantarflexed their ankles bearing their full weight. The maximum plantarflexed angle was also measured (Figures 2a–c). A paired two-tailed t-test for statistical analysis was used, and data were shown as mean ± standard deviation. Indeed, the mice started to use the operated right Achilles tendons after 13.5 ± 4.6 POD (Figure 2a, n = 6), indicating that the healing period in this model to the extent of the use of the leg was approximately two weeks. The range of motion (ROM) was also evaluated by measuring the angle between the fibula and fifth metatarsal bones under deep anesthesia just before sacrificing, as seen in the photographs (Figure 2b). The maximum plantarflexion angle of the operated right leg was 143.3 ± 13.7° (Figure 2c). As the angle degree of the non-operated leg was 183.3 ± 4.08° (Figure 2c, n = 6, P < 0.005), the operated legs did not recover fully with this procedure (Figure 2b). The significant ROM differences between the right and left legs were considered to be caused by absence of passive exercises in this study.
Postoperative assessment. (a) We monitored use of the Achilles tendon when a mouse loaded its weight on the right Achilles tendon and plantarflexed (circle). (b) Measurement of the maximum plantarflexed angle between the longitudinal axes of the left and right fibulae and the 5th metatarsal bone. (c) Average degrees of maximum plantarflexion of the right and left ankles. There were significant differences between them (P < 0.005). (d) Histological appearance around the sutured stump (dashed line) of the Achilles tendon in sagittal sections. Hematoxylin–eosin stains done on postoperative day (POD) 14. Arrows: chondrocyte-like polygonal-shaped cells. Arrow heads: tenocyte-like spindle-shaped cells. (e) Masson’s trichrome staining indicated that chondrocyte-like polygonal-shaped cells were expanded (white line) at POD 21 around the stump.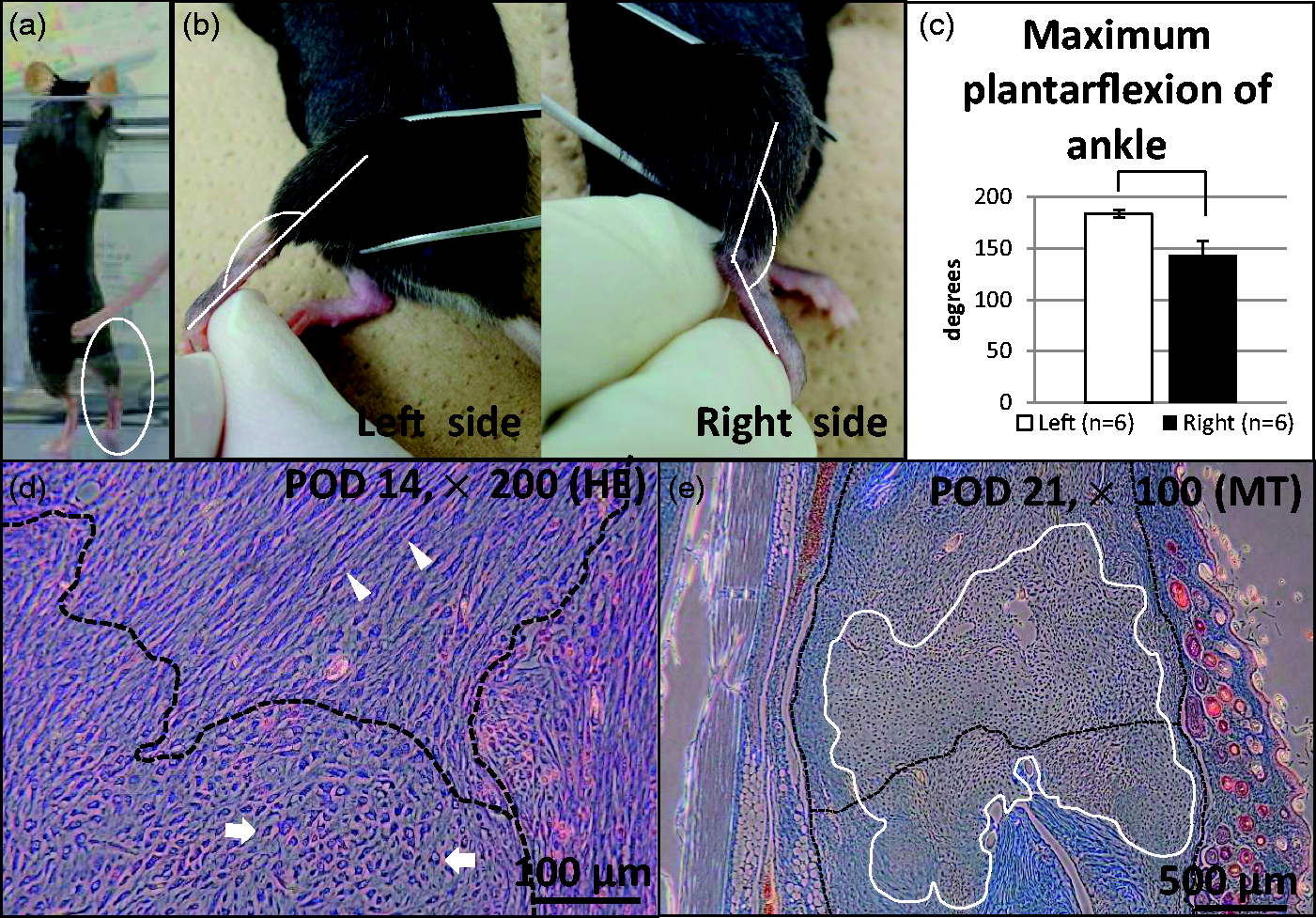
In the postoperative treatment of human flexor tendon injuries, early passive movement is important for triggering ‘intrinsic’ tendon healing.
To examine whether ectopic chondro-ossification occurred in the mice, 4 µm tissue sections were cut in a sagittal plane. These sections were stained with conventional hematoxylin and eosin and Masson’s trichrome, and then mounted on glass slides.
Fourteen days after surgery, proliferated fibroblast-like spindle-shaped cells (Figure 2d, arrow heads) were observed around the tendon stump (Figure 2d, dashed line) at an early stage. However, polygonal-shaped chondrocyte-like cells also appeared (Figure 2d, arrows) followed by expansion (Figure 2e, white line). Observations of these chondrocyte-like cells in human samples4,5 have been reported; but a novel model would be more suitable for examining tendon regeneration and improving tendon suture methods and postoperative therapies.
The surgical method demonstrated here is easy and reliable, facilitating basic research in tendon tissue regeneration such as in drug therapy and cell transplantation. We conclude that our new murine Achilles tendon suture model is useful with respect to the suture itself and immobilization of the tendon. However, there are still limitations, as differences exist with active and passive exercises. Future improvements may help understand rehabilitation and its effects more fully, and in ways which are closer to human clinical procedures.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partly supported by JSPS KAKENHI Grant Number 25670747.
