Abstract
Clinical chemistry is an essential analytical tool in many areas of research, drug assessment and development, and in the evaluation of general health. A certain amount of blood is required to evaluate all blood analytes. Experiments where mice are used, it is difficult to measure all analytes due to the small amount of blood that can be obtained from a single animal. To overcome this problem, separate cohorts of animals are used in toxicity studies for hematology and biochemistry analysis. This requires the use of extra animals and additional resources. Hence interpretation of results derived from using these different animals can be unreliable. This study was undertaken to explore the possibility of using diluted plasma for measuring various biochemistry analytes. Plasma from mice was diluted to 3, 5 and 10-fold with Water for Injection, and various biochemistry analytes were analyzed using an automated analyzer. Results of diluted and undiluted plasma from the same mouse were compared. Most of the analytes from the diluted plasma were found to be well within the ranges of the undiluted plasma except for sodium, potassium and chloride. Diluting plasma to analyze some analytes also freed up undiluted plasma for analyzing electrolytes. In conclusion, in order to obtain reliable and interpretable data from a single mouse it is worthwhile considering diluting the plasma, which should reduce the number of animals used in an experiment.
Clinical pathology testing of laboratory animals in toxicity and safety studies is an important part of safety assessment for food additives, drugs, and chemicals. The primary purpose for evaluating clinical chemistry parameters in the context of an experimental design is to determine if there are differences associated with an experimental treatment. To be effective, the values obtained must be accurate, reliable, and consistent in order to draw meaningful and valid conclusions.
In research using smaller animals such as mice, core clinical pathology testing is limited by virtue of their relatively small blood volume and a lack of easily accessible vessels. A common approach to overcome this problem is to analyze different analytes using different animals from the same cohort. This practice requires more animals and resources, and limits the ability to interpret results by not being able to provide complete profiles for each individual mouse. This limitation can possibly be resolved by diluting the plasma with a compatible diluent to increase the plasma volume.
From the literature search and to the best of our knowledge, only one publication on plasma chemistry analysis using diluted plasma was found in birds, 1 and there were no publications related to any analyses of diluted plasma samples on a broad panel of clinical chemistry analytes in mice. Due to this lack of published literature with reference to plasma chemistry analyses using diluted plasma in mice, a study to compare the clinical chemistry results obtained using diluted plasma against undiluted plasma samples from mice was conducted.
Materials and methods
All procedures were carried out in the laboratory animal facility of the Suven Life Sciences Limited. The experimental protocol was approved by the competent Institutional Animal Ethical Committee in accordance with the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA), India. The mice used in the study were housed and maintained in accordance with the Guide for the care and use of laboratory animals (National Institutes of Health [NIH], 1985).
Experimental animals
A total of 18 male and 18 female healthy Swiss Albino mice (Mus musculus) of a conventional type (microbiological status not specified) were procured from the National Institute of Nutrition, Hyderabad, India. Mice were housed six per cage in sterilized solid bottom polycarbonate cages (Tecniplast, Buguggiate, Italy) with dimensions of 365 × 207 × 140 mm and a floor area of 530 cm2. Rooms were maintained on a 12:12 h light:dark cycle (from 07:00 to 19:00 h) with temperature of 21 ± 3℃, relative humidity of 50 ± 20% and 13–15 air changes hourly. The mice were fed with an autoclaved extruded rodent feed (Nutrilab; Provimi Animal Nutrition India Pvt Ltd, Bengaluru, India) and had access to reverse osmosis (RO) purified potable water. Cages were lined with corncob bedding (Sparcobb; Sagar Industries, Bengaluru, India), and were suspended on a stainless steel rack.
The mice were allowed to acclimatize for seven days to the experimental conditions. At the time of blood collection, the animals were between 8 and 10 weeks of age and weighed between 23 and 32 g. At the end of the study, all the mice were euthanized by CO2 overdose. Each animal was placed in the euthanasia chamber, and then 100% CO2 gas was introduced at a flow rate of 20–30% of chamber volume per minute. CO2 flow was maintained for at least one more minute after respiratory arrest. CO2 is an inexpensive, non-flammable and non-explosive gas, and poses minimal hazard to personnel when used with properly designed equipment. A flow rate of 20–30% of chamber volume per minute causes animals to lose consciousness before CO2 concentrations can become painful.
Study design
Since assessment of all the analytes from the undiluted sample from a single mouse is not achievable, mice were assigned randomly into three sets (I, II and III) of six males and six females each. Two aliquots were prepared from each set of animals; one aliquot was utilized for dilution and the other one was analyzed undiluted. A limited number of analytes were analyzed from each set of mice: alanine aminotransferase (ALT), alkaline phosphatase (ALP), aspartate aminotransferase (AST), sodium, potassium and chloride were analyzed from Set I; total bilirubin (TBIL), urea, creatinine, total protein (TP), albumin and calculated globulin were analyzed from Set II; and glucose, cholesterol, triglyceride, calcium and phosphorus were analyzed from Set III.
Sample acquisition
Blood samples were collected under isoflurane anesthesia by retroorbital puncture using an untreated capillary tube with blood being dripped into heparinized tubes. Anesthesia was induced using a multi-station anesthesia system (Surgivet; Smiths Medical, Dublin, OH, USA), by adjusting the isoflurane (Aerrane; Baxter Private Limited, Haryana, India) concentration to 5% with a primary O2 flow of 3–4 L/min and exposing the mice in the chamber for 4–5 min. Plasma was separated by centrifugation (1700 × g, 10 min, 4℃) and two aliquots were prepared: one to be used for dilution and the other one was kept undiluted. The first aliquot was diluted 3, 5 and 10-fold with sterile Water for Injection (Nirlife, Ahmedabad, India).
Sample analysis
Different methodologies using the RX Daytona™ automated analyzer for analysis of different analytes.
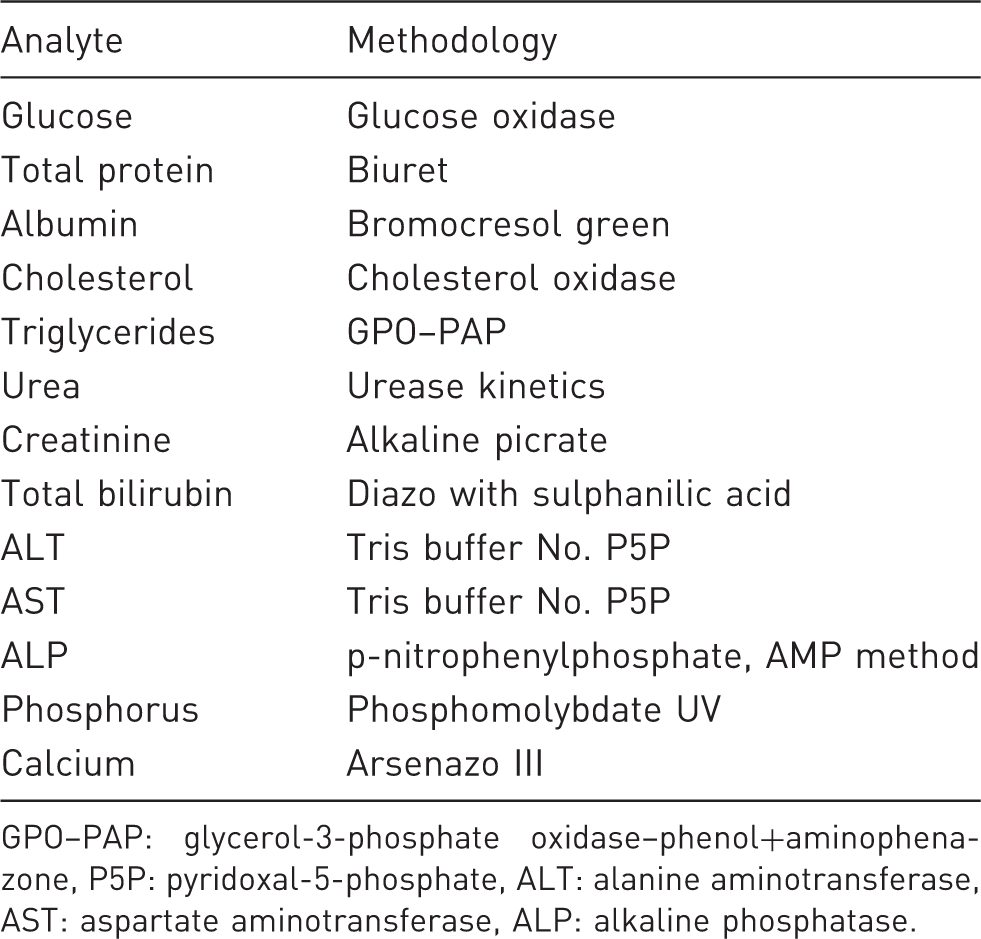
GPO–PAP: glycerol-3-phosphate oxidase–phenol+aminophenazone, P5P: pyridoxal-5-phosphate, ALT: alanine aminotransferase, AST: aspartate aminotransferase, ALP: alkaline phosphatase.
Analysis of results
Results obtained from the diluted samples were multiplied by their respective dilution factors and compared with the results of the undiluted plasma from the same mouse. Mean ± 2 standard deviation (SD) of the undiluted samples and percent differences (inaccuracy) of the diluted samples were considered for comparisons. The linearity of dilution for clinical chemistry analytes was evaluated up to 10-fold dilutions. The correlation coefficient and slope were also calculated.
Statistical analysis
Data of the different dilutions were compared with results of the undiluted samples by Dunnett’s test as follow-up tests to one-way analysis of variance (ANOVA) using GraphPad Prism® version 4 (GraphPad Software, San Diego, CA, USA). Significance level was defined at P < 0.05. The correlation coefficient and slope were calculated using Excel (Microsoft Office 2003, Redmond, WA, USA). There is no absolute number guide for the correlation coefficient to evaluate a low to high degree of correlation. However, values close to −1 or +1 suggest a high degree of correlation; values close to zero suggest no correlation or a low correlation; and values between 0.7 and 0.8 suggest a moderate correlation. 2
Results
Clinical chemistry values of diluted and undiluted samples in male mice.
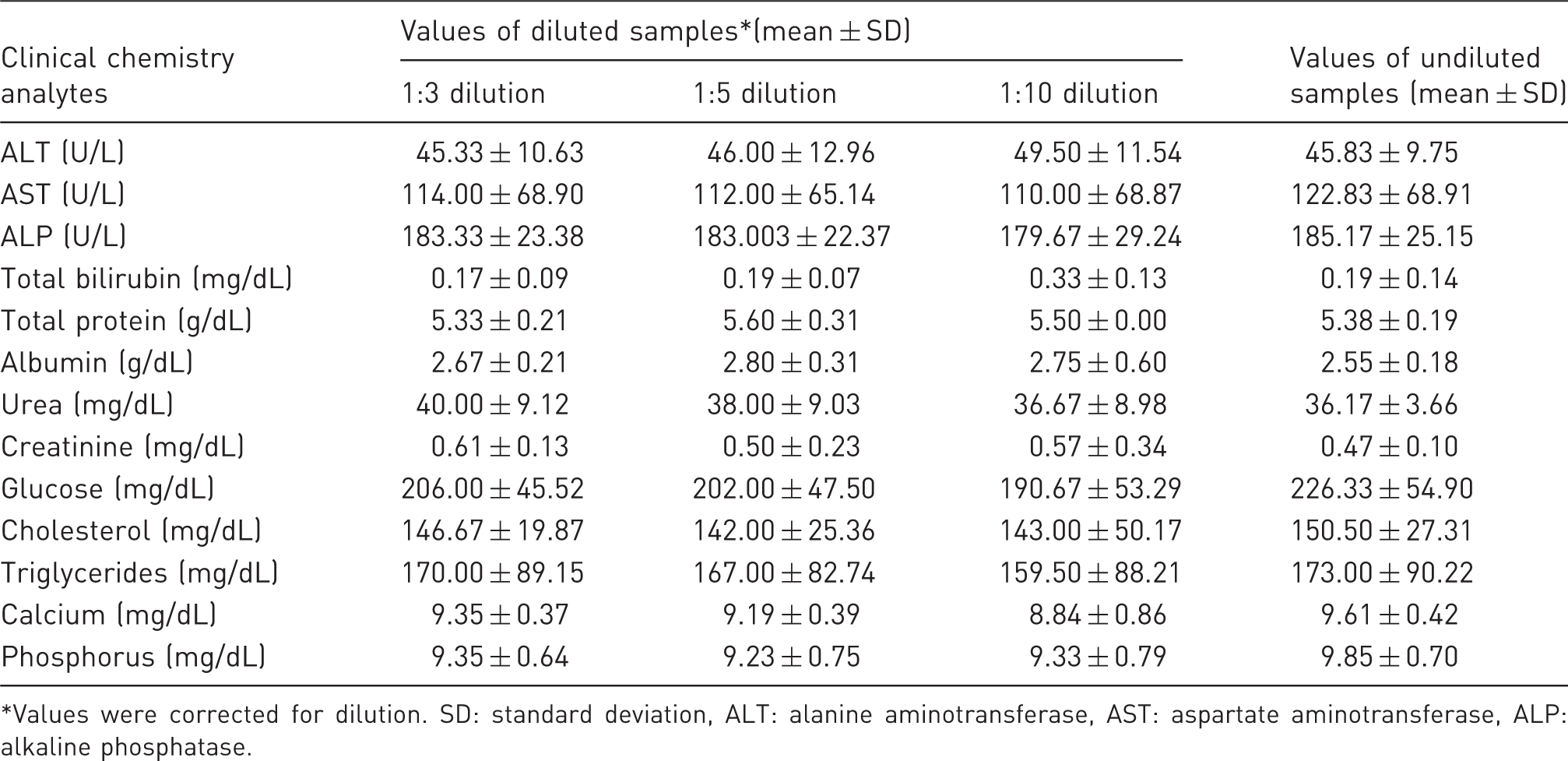
Values were corrected for dilution. SD: standard deviation, ALT: alanine aminotransferase, AST: aspartate aminotransferase, ALP: alkaline phosphatase.
Clinical chemistry values of diluted and undiluted samples in female mice.
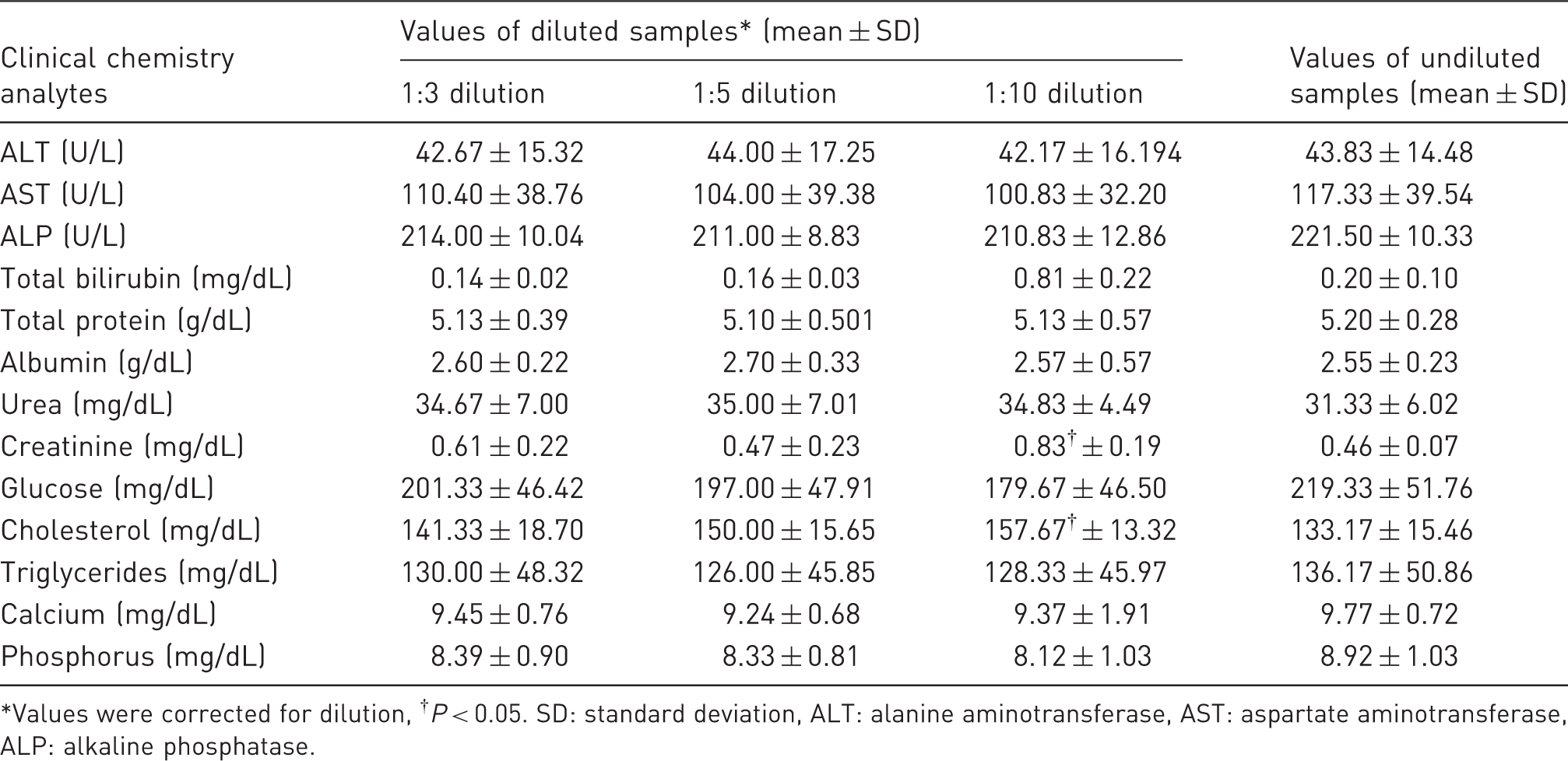
Values were corrected for dilution, †P < 0.05. SD: standard deviation, ALT: alanine aminotransferase, AST: aspartate aminotransferase, ALP: alkaline phosphatase.
Percent differences in clinical chemistry values between diluted* and undiluted samples.
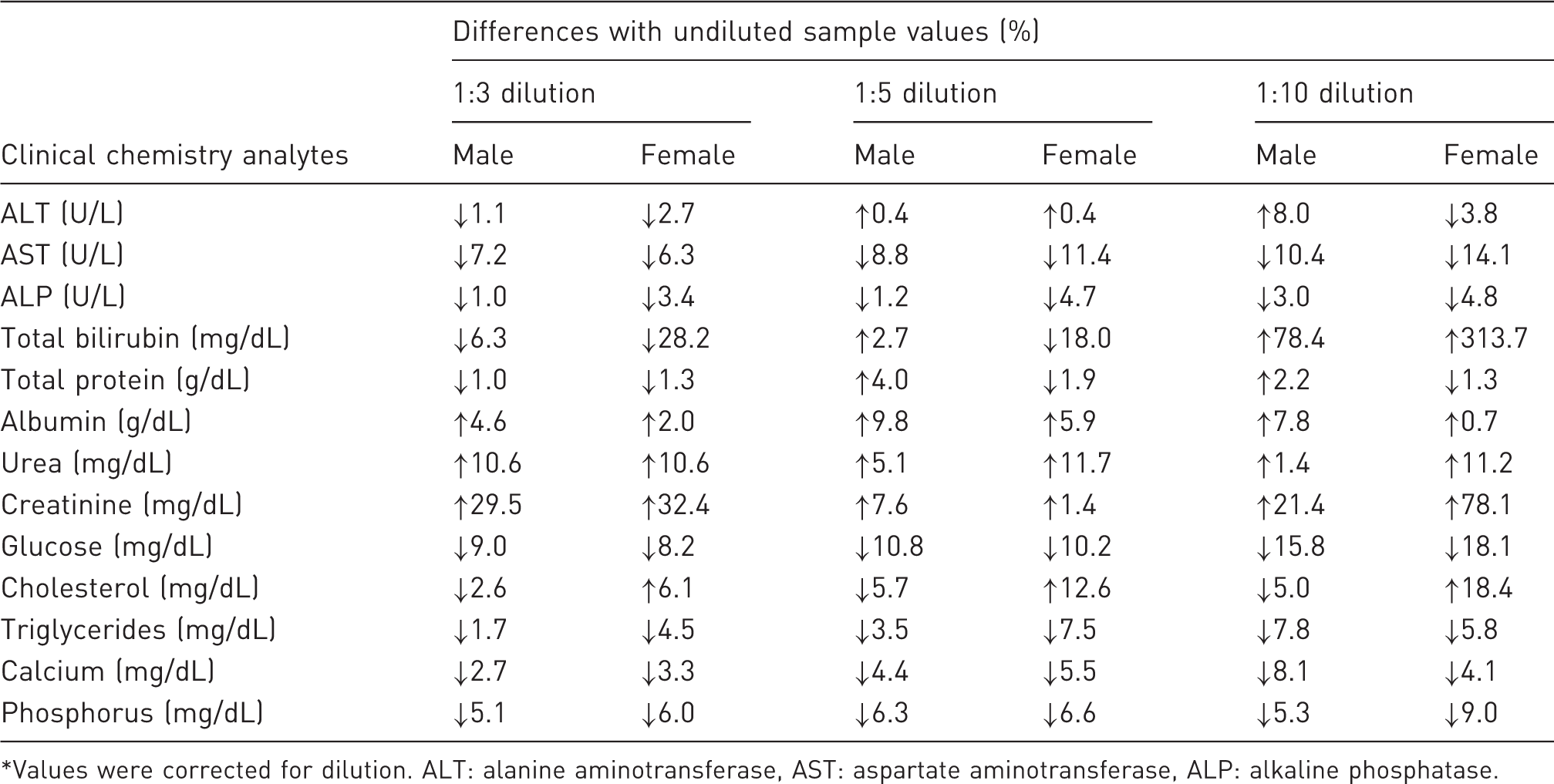
Values were corrected for dilution. ALT: alanine aminotransferase, AST: aspartate aminotransferase, ALP: alkaline phosphatase.
Linearity of dilution of clinical chemistry analytes.
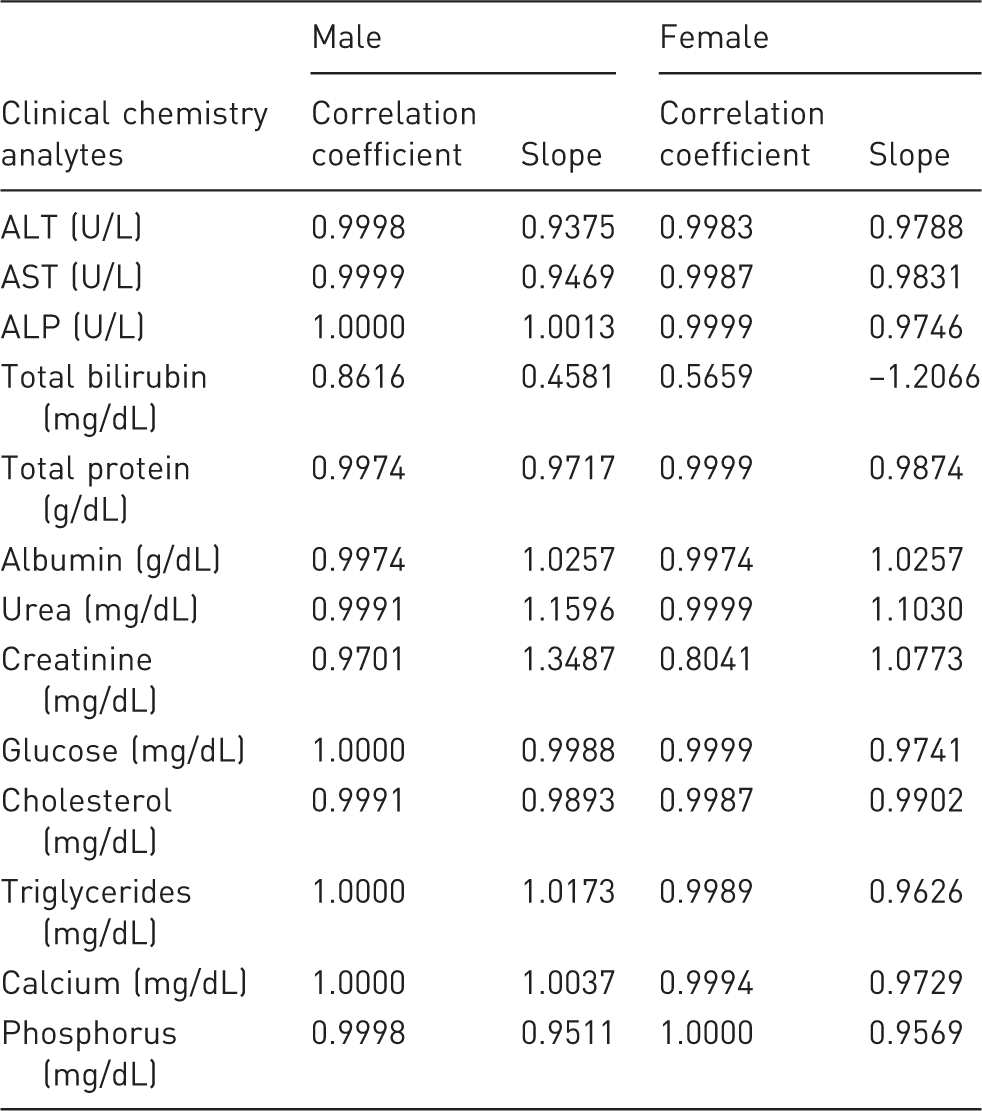
Values for ALT, AST, ALP, cholesterol, triglycerides, urea, TP, albumin, calcium and phosphorus were found to be well within their ranges (within ±2 SD of undiluted plasma and/or with a minor difference) of up to 1:10 dilutions using Water for Injection. Glucose values were lower in all dilutions. Values for creatinine and TBIL in the diluted samples were found to be inconsistent; percent differences (inaccuracy) and coefficient of variation (results not shown) were higher than undiluted ones.
The linearity of the values acquired with the dilutions for all analytes except TBIL and creatinine was high and acceptable since correlation coefficient was varied in the range of 0.99 to 1.00 and the slopes ranged from 0.94 to 1.16. The linearity of the values for TBIL and creatinine obtained after dilution was low or medium.
Discussion
Clinical pathology is an essential analytical tool in many areas of research, drug assessment and development, diagnosis and monitoring of disease. Some government regulatory agencies and professional standards organizations provide guidelines for clinical pathology testing in animal toxicity and safety studies. 3 Clinical pathology tests often provide information not detected by direct examination of organs and tissues, e.g. evaluations of hepatic and renal function, oxygen-carrying capacity of blood, circulating cells of the immune system, hemostatic function, and systemic mineral and electrolyte homeostasis. This information is useful for determining the biological significance of findings in toxicity and safety studies conducted with laboratory animals and the safety testing to be conducted in clinical trials for the development of new drugs and food additives. General clinical pathology data are used to monitor organ function, determine dose-response relationships, and as a measure of overall health.
The core clinical chemistry tests recommended for animal studies are glucose, urea nitrogen, creatinine, TP, albumin, calculated globulin, calcium, sodium, potassium, total cholesterol, and appropriate hepatocellular and hepatobiliary tests.
The use of clinical pathology in smaller animals such as mice has been limited due to their small size and the volume of blood that can be extracted from a single animal. Small volumes (up to 0.5 mL) would suffice for many hematological or biochemical studies, but larger amounts are needed for complete clinical chemistry analyses. Because of this limitation, interim (in-life) blood samples are not collected routinely from mice. Blood samples from mice for clinical pathology testing are collected optimally at terminal euthanasia only.
Some techniques are designed specifically to yield small volumes of blood 4 and so enable a serial study of desired parameters. Other techniques, intended to yield larger volumes of blood, have various limitations, e.g. cardiac puncture gives results which are inconsistent qualitatively 5 and quantitatively, and most are found to be unreliable. 6
Numerous techniques for obtaining a blood sample from a mouse have been described. 7 Most of these techniques are designed to obtain an interim (in-life) sample and, although useful for repeated blood draws, they typically yield an insufficient amount of blood to perform both a complete blood count (CBC) and chemistry profile, as well as other tests. Therefore, if a CBC, complete chemistry profile, and other tests requiring a substantial amount of blood are necessary, interim blood samples from mice are not recommended and are likely not possible. 8 In addition, pooling of samples is also not a recommended practice. 8
Techniques that have been shown to yield greater amounts of blood have been described elsewhere. 9 These techniques require anesthesia and catheterization of a major vessel or exposure of the heart and are terminal. 9 Although the recommendation of obtaining CBC and clinical chemistry profiles from the same animal is not stated explicitly, having this information from a single animal may help in identifying a greater breadth of pathologies and thus provide more useful data for the study. Moreover, the ability to obtain both data-sets from a single animal compared with multiple animals would have considerable ethical and economic impacts, because by doing so it would decrease the number of animals needed for many studies. As stated previously, obtaining an adequate volume of blood to perform both hematology and chemistry testing can be problematic due to the mouse’s small size.
The approximate blood volume in a mouse is 77–80 µL/g. For a 25 g mouse this is equivalent to 1.9–2.0 mL.10,11 Without fluid replacement, the maximum blood volume which can be safely removed for a one-time sample is 10% of the total blood volume or 7.7–8.0 µL/g. For a 25 g mouse, this is equivalent to 193–200 µL. In non-terminal blood collection, the orbital sinus, jugular vein, or saphenous vein typically yield 0.2–0.3 mL of blood for an average-sized adult mouse whereas during terminal blood collection about 0.8–1.0 mL of blood can be collected from the orbital sinus or by cardiac puncture. Other sites such as the caudal vein, tail vein, dorsal pedal vein, posterior vena cava or axillary vessels provide even less blood. Out of one milliliter of blood obtained during terminal sacrifice, some is required for hematology analysis. The remaining blood yields ∼200 µL of serum or plasma, which is insufficient for analysis using a clinical chemistry analyzer, on a broad spectrum of clinical chemistry parameters. 12 For routine biochemistry analysis using modern analyzers, approximately 300 µL (including dead volume) of plasma or serum is needed. Commonly recommended analytes for general toxicity studies includes: ALT, AST, ALP, gamma-glutamyltransferase (GGT), sodium, potassium, chloride, TBIL, urea, creatinine, TP, albumin, calculated globulin, glucose, cholesterol, triglyceride, calcium and phosphorus. Analysis of all these analytes from a single small animal is challenging because of the small blood volume sample. To overcome this problem and to generate sufficient blood for clinical chemistry analysis, more animals are needed in preclinical safety studies which is not in line with the principles of the 3Rs (replacement, reduction and refinement) of animal welfare. In addition to the cost of animals, the overall cost of experiments is also raised significantly due to extra manpower, additional space, increased husbandry expenses, etc. To minimize this constraint, we therefore set out to prove the hypothesis that diluted plasma can be used in toxicity studies using mice to determine various clinical chemistry parameters from the same animal without compromising the accuracy of the data. We thought of increasing the sample volume by diluting the plasma with a compatible diluent. Our literature search could not find sufficient references to guide us in this regard. Due to the lack of published literature related to plasma dilution in mice, we conducted a study to check the feasibility of diluting plasma with any compatible diluents which do not interfere with the assay procedure as well as to give results comparable with those from undiluted samples. The aim of this study was to determine which diluent yields reproducible results relevant to results from undiluted specimens.
In clinical chemistry laboratories, when a sample analyte value exceeds the upper limit of detection or the sample volume is below the minimum requirement, the sample is auto-diluted and corrected for the dilution prior to release by the analyzer. 1 The same principle of dilution can be applied for pre-analysis dilution. The most commonly used diluent in various automated analyzers (e.g. RX Daytona, Vetscan, VetTest) is normal saline, however some researchers do not recommend this as they have experienced difficulties in evaluating sodium and chloride.1,12–14 We therefore considered using diverse diluents other than normal saline, e.g. reagent grade water type I, Cal A (Randox), plasma, and Water for Injection. We limited our quest to sterile Water for Injection. ‘Sterile’ in not just being free from microorganisms, but also to guarantee the minimum probability of finding any minerals or other impurities in the diluent that can interfere with the reaction between reagents and analytes, and thus the outcome. Water for Injection is clean, sterile, non-pyrogenic, distilled water with ≤500 ppb total organic carbon 15 and does not contain any electrolytes. It has a neutral pH which does not interfere with any analytes of plasma, and was therefore chosen as a suitable diluent in this study.
Results of this study demonstrated that the quantity of plasma is not a constraint for estimating clinical chemistry analytes from a single small animal such as a mouse. The values of most of the clinical chemistry analytes, namely ALT, AST, ALP, TP, albumin, urea, cholesterol, triglyceride, calcium and phosphorus obtained from the diluted samples were well within the ranges of the undiluted ones, even up to a 10-fold dilution as shown by the accuracy and correlation coefficient. The values of creatinine and TBIL from some of the diluted samples did not correlate well which substantiate the findings of earlier studies, probably because of the narrow analytical quantification range and the normal values of these analytes in mice. 14 If an analyzer with a higher sensitivity is used, then there is a likely chance of getting the estimations of these two analytes to fall well within the ranges of an undiluted sample even with a higher dilution. For glucose, values from the diluted samples were correlated well with the undiluted ones up to a 5-fold dilution; however, with a 10-fold dilution, either the percentage differences were higher or the values were inconsistent.
Forbes and Brayton found that when plasma was diluted with normal saline, calculated results for the 2× and 3× diluted samples correlated well (>93% similarity), but the 5× diluted sample did not correlate well (<88% similarity) with the results from the undiluted samples. 12 We discovered great accuracy (82–100%) and correlation coefficient (>0.99) for glucose up to a 5× dilution; and for ALT, AST, ALP, TP, albumin, urea, cholesterol, triglyceride, calcium and phosphorus up to a 10× dilution. Prefontaine et al. also diluted (3×) samples with saline and reported that clinical chemistry values obtained with diluted samples were comparable with their historical reference ranges for undiluted samples from CD-1 mice, except for cholesterol and triglyceride. 14 Numerous pre-analytical procedures have been reported to influence the values of analytes, such as blood collection sites, anesthesias used, times of collection, fasting, and handling of animals or samples.
Pre-analytical variations may vary from day to day and from animal to animal. Hence, comparison using historical ranges may not be as reliable as comparing values which are taken from the same animal. Comparisons with historical control references do not mirror comparisons effectively, as sources of variation namely physiological, procedural or artifactual may shift between samples used for dilution experiments and generation of historical ranges. Nonetheless, pre-analytical sources of variation within the limitations of a study can be controlled though not completely stopped.16,17 The key principle is to treat all animals the same with regard to conditions preceding blood collection. As reported in the literature, albumin was not quantifiable in some dilutions, but contrary to this we have found good results of even up to a 10× dilution. 14 There are a few articles expressing that plasma can be diluted with deionized water but only to some extent, i.e. 1× or 2×. Indeed, even with this dilution, the resultant volume may not be sufficient for analyzing all the routine analytes considered for toxicity studies.
Waldoch et al. attempted the dilution of plasma with sterile water but did not specify about the extent of the dilution. What is more, they stored the aliquots of samples to be utilized for dilution at −80℃. They demonstrated that only a few analytes could be measured precisely with the diluted plasma and recommended that sterile water is not a perfect diluent for biochemical analysis. 1 However, results derived from our experiment clearly demonstrated that sterile water can be used for dilution but the quality should be of an injection grade. The underlying cause for the baffling results of Waldoch’s experiment could be due to the quality of water and/or long storage of samples.18,19 Rathkolb et al. utilized deionized water as a serum diluent in their experiment; however the reliability of their data is questionable without earlier validation. 20 Wiedmeyer et al. explored the plausibility of using a single lithium heparin tube for the generation of a CBC, biochemistry profile, and serology profile from mice. They found that CBC, biochemistry, and serology profiles could be obtained more readily when blood samples were placed in a single lithium heparin tube instead of in two separate tubes. 21 However, they were unable to achieve an entire 14-parameter clinical chemistry profile of all the animals as a result of inconsistency in the blood volume drawn. This uncertainty may jeopardize the study objective as it may lead to loss of important parameters if a significant number of mice could not produce enough blood. With this perception in mind, dilution of plasma should be a superior alternative for getting clinical chemistry profiles from all animals.
In view of these observations and comparisons between water and normal saline as diluents, we consider that sterile water is superior to normal saline for diluting plasma. For electrolytes, different diluents such as reagent grade water type I, Cal A (Randox), plasma, normal saline and Water for Injection were attempted, albeit without any success. Electrolyte analysis tends to require a large sample volume and diluting plasma samples for other parameters frees up more undiluted plasma for electrolyte analysis. The freed up plasma can also be used to re-analyze samples in cases where the values of some analytes are below the detection limits following dilution.
Taking everything into account, it is worthwhile diluting plasma, keeping in mind the end goal of obtaining reliable and interpretable data from a single animal as opposed to from a different set of animals which may confound the overall interpretation of a study. This approach has the advantage of allowing the collection of samples during the in-life phase in younger mice without requiring interim necropsies. This results in a reduction of animals needed for a study and provides an opportunity for combining additional blood, plasma or serum endpoints from a given animal (i.e. clinical chemistry, toxicokinetics, immunogenicity and many others).
Footnotes
Acknowledgement
The authors are thankful to Mr Venkat Jasti, the CEO and chairman of Suven Life Sciences Limited for providing necessary support and facilities for this experiment.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
