Abstract
Early adverse experiences are known to influence the risk of developing psychiatric disorders later. To shed further light on the development of laboratory mice, we systematically examined the influence of a prenatal or postnatal olfactory stressor, namely unfamiliar male mouse faeces, presented to pregnant or nursing mouse dams. Maternal and offspring behaviours were then examined. Maternal behaviours relative to controls revealed changes in nest building by the pregnant dams exposed to the unfamiliar faeces. There were no differences among groups on pup retrieval or exploration by the dams. Behavioural phenotyping of male and female offspring as adults included measures of exploration, anxiety, social and depressive-like behaviours. Additionally, serum corticosterone was assessed as a marker of physiological stress response. Group differences were dependent on the sex of the adult offspring. Males raised by dams that were stressed during pregnancy presented elevated emotionality as indicated by increased numbers of faecal boluses in the open field paradigm. Consistent with the effects of prenatal stress on the males only the prenatally stressed females had higher body weights than their respective controls. Indeed, males in both experimental groups had higher circulating corticosterone levels. By contrast, female offspring of dams exposed to the olfactory stressor after parturition were more anxious in the O-maze as indicated by increased latencies in entering the exposed areas of the maze. These findings emphasize the necessity for researchers to consider the pre- and postnatal environments, even of mice with almost identical genetic backgrounds, in designing experiments and interpreting their data.
With the introduction of rodent strains that are selectively bred for specific research purposes, mice, especially the C57/BL6 strain, have become the most popular animal model. Moreover, the ever increasing numbers of genetically-manipulated lines have amplified the need to know more about these mice. This was recently highlighted in an article by Couzin-Frankel that suggested a revision of current research standards in animal science. 1 This suggestion has led us to consider the overall breeding environment of laboratory mice.
Many diseases were originally believed to be related to a limited number of aetiological factors, with an emphasis on genetics. It is now recognized that diseases often have a multifactorial background, and important contributions come from the environment. Thus gene × environment interactions are especially significant, particularly regarding psychiatric disorders. Pre- and postnatal conditions, such as variations in maternal care, are known to influence vulnerability to certain stress-associated disorders.2–4 Some prominent examples include anxiety, depression and schizophrenia, in which a strong perinatal component has been identified.5–10
The implication is that conditions in breeding colonies could have profound effects on maternal and offspring behaviours.11,12 Various experiments investigating the perinatal environment have offered confirmation of the impact of this critical ontogenetic phase on emotional phenotypes later in life.10,13–17 Notably, sex-dependent differences are common. 18
To evaluate the effects of an exemplary early-life disturbance, we presented a well-described stressor, odours from unfamiliar adult males, to dams during pregnancy or after parturition. That even subtle odours can have an impact on dams and offspring was revealed in a recent study. Sorge et al. reported that odours from the T-shirts of animal caretakers or researchers appeared to influence the behaviours of the animals. 19 Here, we wanted to expand these and other findings20–22 by analysing emotional, depressive-like and stress physiological features in male and female offspring from similarly stressed mothers. Moreover, we designed the study to find whether mice were more sensitive to the odours presented to mothers pre- or postpartally. The working hypothesis was that challenging early-life conditions produce long-lasting effects on the emotional phenotype of offspring as adults. We furthermore hypothesized that environmental stress during pregnancy and lactation has a significant effect on the stress physiology of the progeny.
Materials and methods
Animals
Multiparous female C57BL/6N mice aged 3–4 months were obtained from Charles River (Sulzfeld, Germany). After arrival the animals were individually housed in Makrolon type III cages (Tecniplast, Buguggiate, Italy), with wooden chip bedding (ABEDD LTE-001; Lab & Vet Service, Vienna, Austria) and nesting materials composed of cellulose. Food and water were available ad libitum (Rod16A; Lasvendi, Soest, Germany). Animal housing was standardized with a 12:12 h dark–light cycle (dark phase: 09:00–21:00 h) at a room temperature of 22 ± 2℃. Humidity was set at 50%. The hygienic status was specific pathogen-free (SPF) according to recent Federation of European Laboratory Animal Science Associations (FELASA) recommendations. 23
Stress exposure
Animals were allowed to acclimatize over 14 days. Mating was performed with one male and two female mice aged 14–16 weeks. The females were separated from the male when pregnancy was ascertained by positive vaginal plug check and indicative weight changes. Females were then housed individually under the conditions outlined above.
The stressor was four faecal boluses from an individually-housed and mating-experienced unfamiliar male mouse placed into the bedding of the female. Each female in the two experimental groups was exposed twice to the stressor, namely the odour from the faeces.
For the stress protocol the pregnant females were separated into three groups (Figure 1):
Control (n = 5): These pregnant females were not exposed to the faecal stressor. Prepartum stress (n = 6): The pregnant females were exposed to the faecal materials on day 8 (E8) of pregnancy and again on E15. E8 was chosen for the first exposure, because stress-induced disruption of pregnancy could then be excluded as far as possible
24
and E15. Postpartum stress (n = 7): The initial stress exposure was on postnatal day (PND) 7 with a second exposure at PND14. Course of experiments. Overview of the three different groups and time points of stress exposure. The experiment comprises maternal as well as offspring trials. Analysis of offspring started at postnatal day (PND) 28 with weekly body weight assessments. The behavioural analyses of offspring were between 8 and 10 weeks of age. NC: novel cage; NT: nest test, OF: open field, NO: novel object, OM: O-maze, DLB: dark–light box, SR: social recognition, FST: forced swim test, HP: hotplate, FC: fear conditioning.
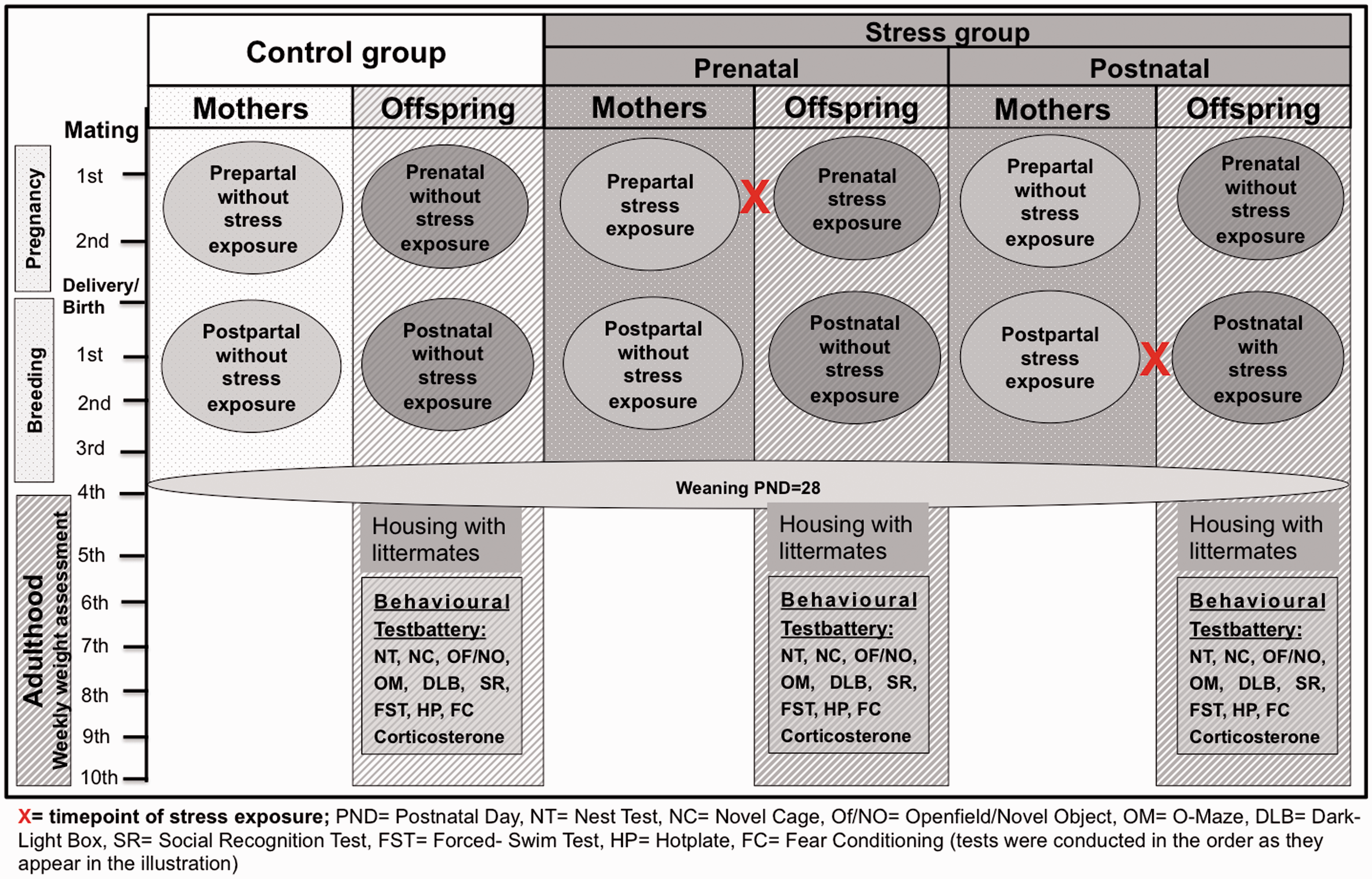
Weaning took place on PND28 after offspring were earmarked for future identification. Sex-separated siblings were group-housed in type III cages as outlined above.
Maternal behaviour
Maternal behaviour was observed during weekly cage changes in the light phase between 08:00 and 09:00 h.
Exploration and nest building
Dams were placed in a new Makrolon type III cage, containing a layer of new bedding material. Latency to the first rearing and total numbers of rearings were measured for 5 min. Subsequently a nestlet (PLEXX, Arnheim, The Netherlands) was introduced, and nest building was analysed after 5 and after 24 h using a scoring system suggested by Deacon. 25 A score of 1 was assigned if the nestlet was completely untouched. A score of 2 was when the nestlet was partly pulled to pieces, a score of 3 when the nestlet was completely pulled to pieces, a score of 4 for a recognizable nest, and a score of 5 for a completely covered nest.
Pup retrieval testing
On PND7 pup retrieval behaviour was tested by placing two pups in the far corners of the home cage. The latency to retrieve the pups to the nest was recorded, 26 with a maximum of 300 s.
Offspring
The behavioural procedures were performed in adult offspring at 8–10 weeks of age, and always during the dark (active) phase. In order to avoid ‘litter effects’ only one pup/sex/litter was tested, resulting in small but representative groups. The representative animal/litter was chosen according to the median of litter weights.
Our study complies with regulations on animal experiments in Germany. The protocol was approved by the local authorities (§15 Kommission, Regierungspräsidium Karlsruhe, permit number: G-279/12) and the animal welfare committee of the University of Heidelberg. To minimize distress, all animal care and testing were carried out by the same (female) person.
Weight monitoring
From weaning until the 10th week of age, each offspring was weighed weekly during cage changes.
Behavioural test battery
Before each test the animals were acclimatized to the behavioural test room for 30 min. To avoid artefacts due to test–test interactions, the sequence of tests in the battery started with the least stressful and ended with the most invasive paradigm.27,28 Between test sessions the apparatuses were cleaned with 70% ethanol.
Nest test
The nest test is a procedure for assessing this aspect of maternal care performance, but it also detects inadequate behaviour in non-breeding mice, 25 since nest building is part of the natural behavioural repertoire. 29
As assessed for their mothers nest building performance of the offspring was separately rated 5 and 24 h later with scores by Deacon. 25
Novel cage test
The novel cage test assesses the level of vertical exploration in a new environment. 30 To reduce handling, this test was performed after the weekly cage change. The test mouse was placed in a new type III cage with clean bedding. Numbers of rearings were measured for 5 min as well as the latency until the first rearing. The mouse remained in the new cage and the untested littermates were transferred to the fresh cage.
Open field/novel object test
In the open field test changes of activity, anxiety and exploration can be analysed. 31 In our experiments the animal was placed in a 50 × 50 × 50 cm black open field arena placed on an infrared light surface. Four mice were tracked simultaneously in separate boxes for 10 min at 25 lux. A camera positioned above the arena (Ikegami Digital, Ethovison 4.0; Noldus Information Technology, Wageningen, The Netherlands) evaluated ‘distance moved’, ‘velocity’ and ‘centre time’. After 10 min a water-filled falcon tube (50 mL) was placed in the centre of the apparatus to serve as a novel object. Exploration and neophobic behaviours were measured for 10 min by assessing the latency and total number of approaches to the novel object. Subsequently, faecal boluses were counted as indicator of emotionality.32–34
O-maze test
The O-maze is an elevated circular arena with two open and two closed arms, representing a test for anxiety. The apparatus in our set-up is made of rough high-grade steel (outer diameter: 45 cm, arm width: 6 cm, elevated 50 cm above ground). Anxiety was measured by recording time in the open arms, numbers of exits from open to closed arms, and full crosses (circumnavigation of the O-maze). The test was performed for 5 min with lighting at 25 lux. 35
Dark–light box test
The dark–light box (54 × 22.5 × 32 cm L/W/H) consists of a dark (22.5 cm) and a light compartment (31.5 cm) connected by a 5 × 5 cm aperture and is used to assess conflict–avoidance behaviour. 36 The light compartment was illuminated by 600 lux. The tests began by placing the mouse in the dark compartment. The time in the dark box and the numbers and latencies of exits into the light compartment were measured for 5 min. 30
Social recognition test
The social recognition test is to assess socially-induced motivation. The apparatus (50 × 50 × 50 cm) consists of three compartments, which are connected by transit openings. The test consisted of four phases. Phase 1 consisted of 5 min in a central compartment with closed doors. Phase 2 was 5 min of free access to all compartments. In Phase 3, an unfamiliar mouse of the same sex and age as the subject animal was placed in a wire frame cage in the corner of one outer compartment. The opposite external compartment contained an empty wire frame cage. The analysis comprised the time spent within the centre and the external compartments. Additionally, the time spent in close vicinity to the unfamiliar animal was measured. Tracking lasted 10 min. In Phase 4, the first unfamiliar mouse was removed from the cage and a second unfamiliar animal was introduced into the opposite cage. Phase 4 consisted of 10 min trials under a light intensity of 25 lux.
Forced swim test
A mouse was placed in a 2000 mL beaker filled with 1400 mL 21℃ water to assess depressive-like behaviour. Floating (i.e. complete lack of movement), and the latency to its first display (i.e. a form of ‘helplessness’) was measured for 6 min under red-light conditions using EthoVision software (Noldus Information Technology). The mouse was then allowed to dry in front of a red light in its home cage. After 24 h the mouse was retested under the same conditions. 30
Hotplate test
A hotplate (Ugo Basile Srl, Monvalle, Italy) was used to assess pain thresholds. The apparatus consists of a plate and a glass cylinder to prevent the mouse from escaping. The temperature of the plate was 52℃. The test took place at 100–130 lux, lasted 45 s, and latency until licking of hind paws was measured. The hotplate test was performed to identify possible artefacts due to different pain thresholds induced by different stress conditions. 37
Emotional learning test
A fear-conditioning system (TSE Systems, Bad Homburg, Germany) was employed for the analysis of hippocampus- and amygdala-associated emotional learning. The paradigm comprised three days of exposure.
Day 1 (shock): For initial conditioning, the mouse was placed in a cylinder onto a grid. After 120 s of habituation, followed by a 30 s tone (conditioned stimulus, 2800 Hz, intensity 67), a brief footshock was applied (unconditioned stimulus, 2 s, 0.8 mA). Inter-trial cleaning was performed with 70% ethanol in this and all phases. Day 2 (context conditioning): 24 h after shock exposition the mouse was placed in the identical experimental chamber for 5 min without any stimulus. Freezing, a complete absence of movement apart from respiration, was measured to analyse context conditioning. Day 3 (cued conditioning): Freezing behaviour was analysed in a different context on day 3. Here, the mouse was placed in the same chamber but the context was changed using a different rectangular cylinder with a flat surface cleaned with chamomile tea to induce a different olfactory environment. After 180 s of habituation, the conditioned stimulus was presented for 180 s. Freezing was evaluated as percentage freezing/time.
Corticosterone analysis
Trunk blood was obtained by decapitation and blood plasma was collected and determined by radioimmunoassay (ICN Biomedicals, Eschwege, Germany); assay sensitivity was 12.5 ng/mL.
Statistical analysis
Data were analysed with InVivoStat (invivostat.co.uk) statistical software. Maternal and behaviour were evaluated by one-way analysis of variance (ANOVA). Offspring behaviours were analysed by two-way ANOVA with ‘odour stress’ and ‘sex’ as factors. The assessment of changes in body weight during development used a repeated measurement analysis in which the factor of ‘week’ was added. Tukey’s post hoc tests were used to determine statistically reliable group differences. Statistically significant effects were set at P ≤ 0.05.
Results
Because perinatal stress has been implicated in a variety of emotional behaviours, we first examined maternal behaviours before or after exposure to a stressful odour. Next, we assessed the emotional phenotype of the offspring.
Maternal behaviour (nest building) was changed with prepartum stress
Nest building was measured 5 h after introduction of a new nestlet in the third week of nursing. The factor ‘odour stress’ achieved statistical significance (F(10,18) = 5.70; P = 0.014). Tukey’s post hoc testing revealed higher nest scores in prepartum stressed dams compared with postnatal stressed dams and controls (P = 0.012), which did not differ (Table 1).
Prenatal stress in dams led to increased body weights in female offspring
Evaluation of pre- and postpartally applied ‘odor stress’ in dams.
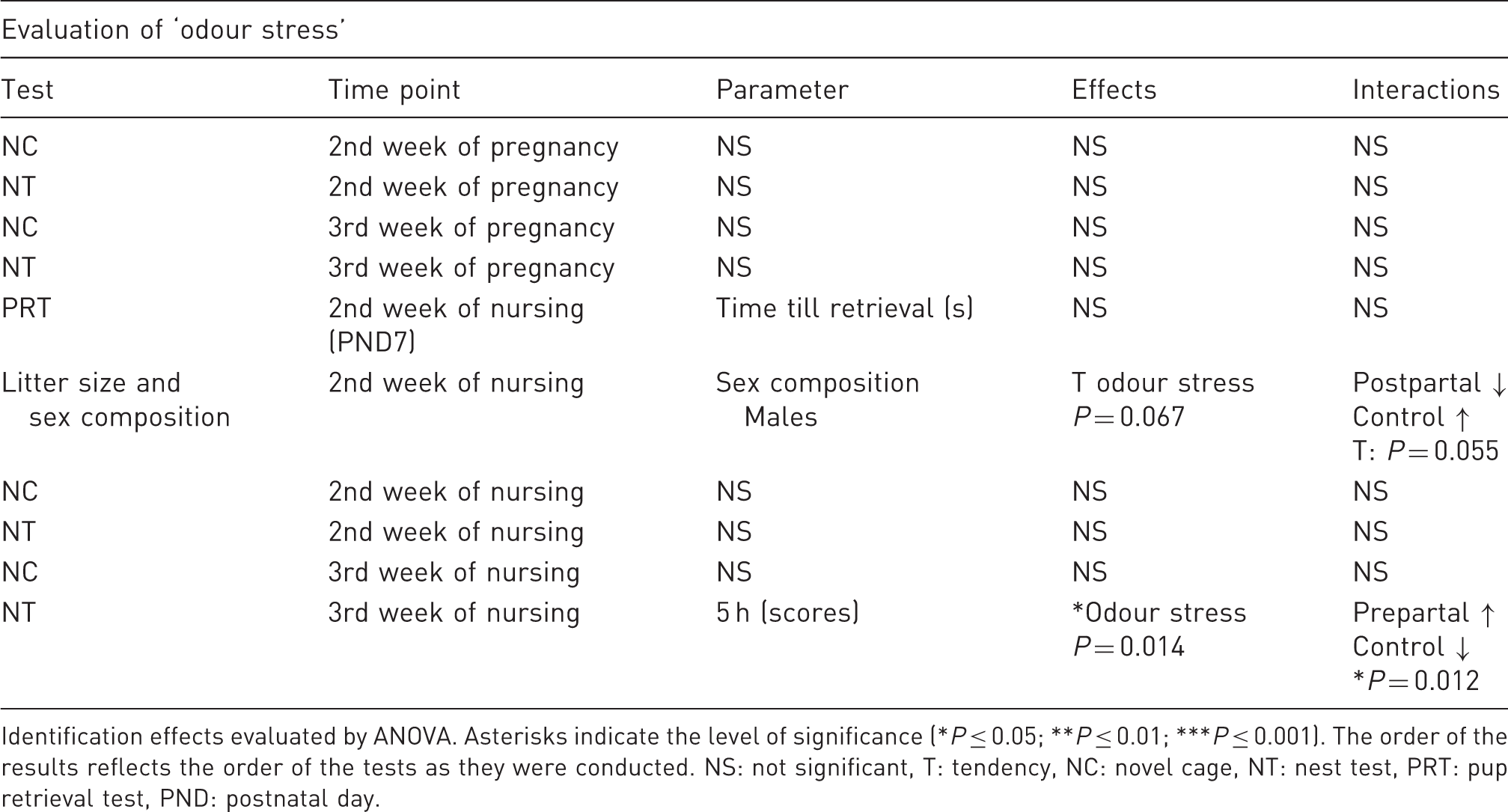
Identification effects evaluated by ANOVA. Asterisks indicate the level of significance (*P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001). The order of the results reflects the order of the tests as they were conducted. NS: not significant, T: tendency, NC: novel cage, NT: nest test, PRT: pup retrieval test, PND: postnatal day.
Evaluation of perinatal ‘odour stress’ and ‘sex’ effect on adult offspring.

Illustration of effect sizes evaluated by analysis of variance (ANOVA). Asterisks indicate the level of significance (*P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001). The order of the results reflects the order of the tests as they were conducted. NS: not significant, NC: novel cage; NT: nest test, OF: open field, NO: novel object, OM: O-maze, DLB: dark–light box, SR: social recognition, FST: forced swim test, HP: hotplate, FC: fear conditioning, f: female, m: male.
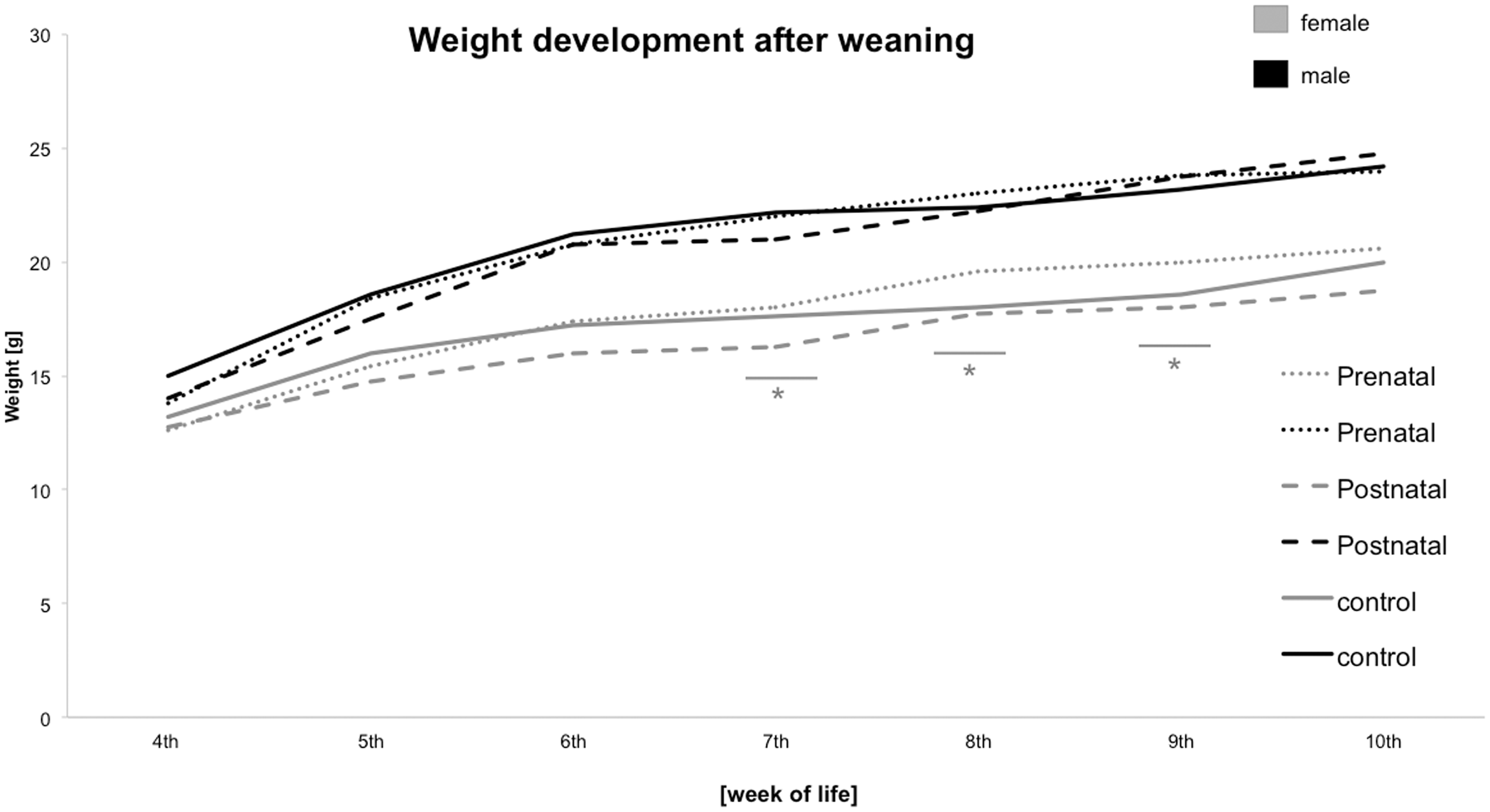
Weight development after weaning. Changes in body weight after weaning with males displaying greater body weights than females. Adult females of prenatally stressed dams had higher weights in comparison to female offspring of dams exposed to postnatal stress and control females at 7, 8 and 9 weeks of age. *P ≤ 0.05; ** P ≤ 0.01; ***P ≤ 0.001.
Male offspring of postpartum stressed dams had higher levels of emotionality in the open field
Faecal boluses are a measure of emotionality in an open field. The sex × odour stress interaction revealed a statistically reliable value (F(1,28) = 7.68; P = 0.003) (Table 2, Figure 3).
Open field: faecal boli. Males from prenatal stress conditions showed higher numbers of faecal boluses in the open field than females from the comparable group and from controls. The differences from adult male and female offspring were also statistically reliable from their comparable prenatal groups. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001.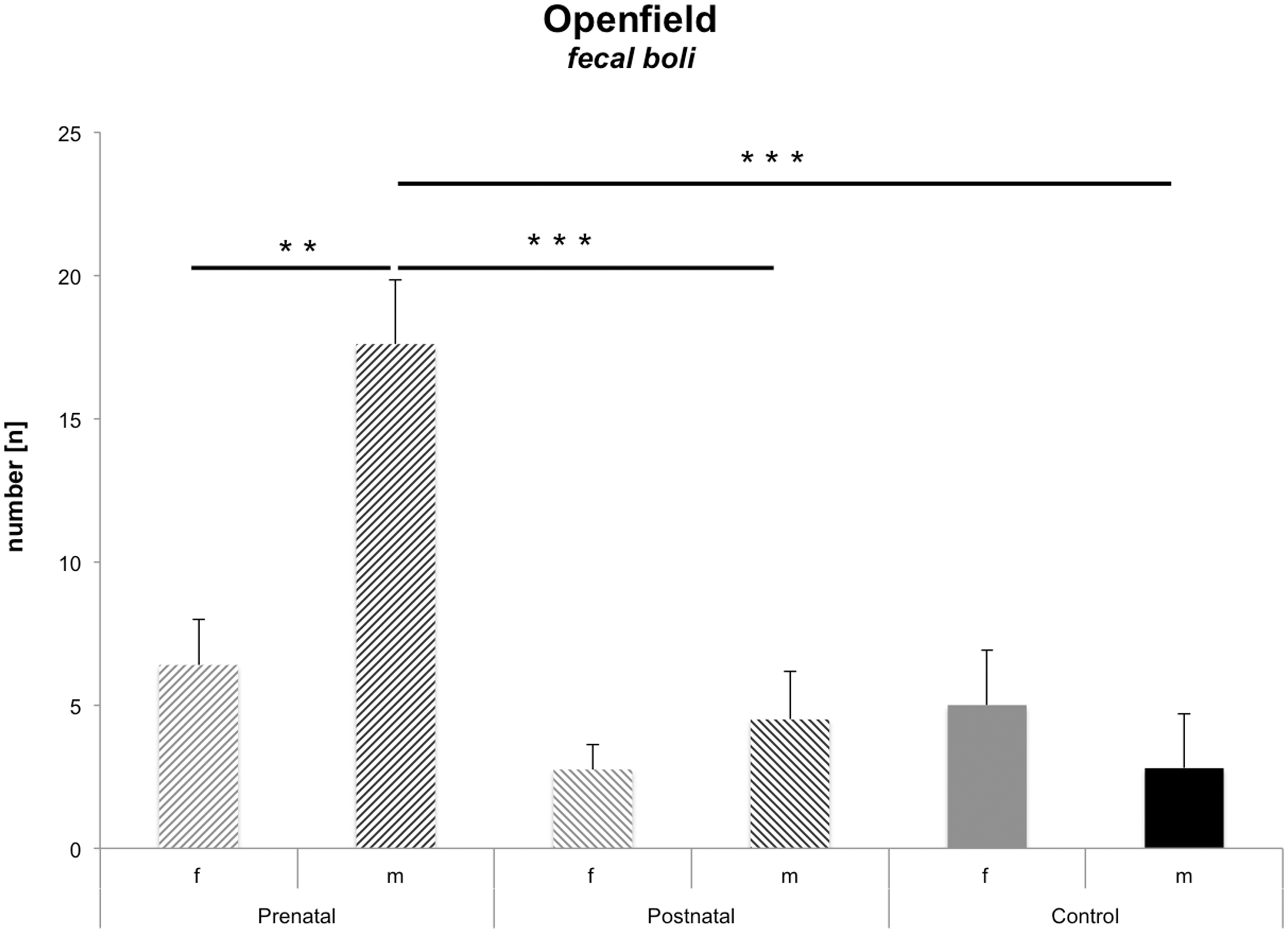
Post hoc testing revealed that males from the prenatal dam group had elevated numbers of faecal boluses compared with females of its comparable group (P = 0.002), as well as compared with males of the postnatal groups and of the control groups (both P = 0.001). No other between-group comparisons reached statistical significance.
Female offspring of postpartum stressed dams showed increased anxiety
Analyses of latency to enter the open compartments of the O-maze revealed a statistically reliable interaction of ‘sex:odour stress’ (F(1,28) = 10.22; P = 0.001). Subsequent Tukey’s post hoc tests indicated that females of the postnatally stressed group had increased latencies, a measure of anxiety, compared with control females (P = 0.001), and compared with females of the prenatal group (P = 0.001) (Table 2, Figure 4).
O-maze: latency 1st exit on to open arms. Females from postpartally stressed dams present a higher level of anxiety indicated by an increased latency to enter open arms. This becomes highly significant when compared with females from the prenatal and control groups. Females from the postpartum group demonstrated increased latencies compared with males. *P ≤ 0.05; **P ≤ 0.01; ***P ≤ 0.001.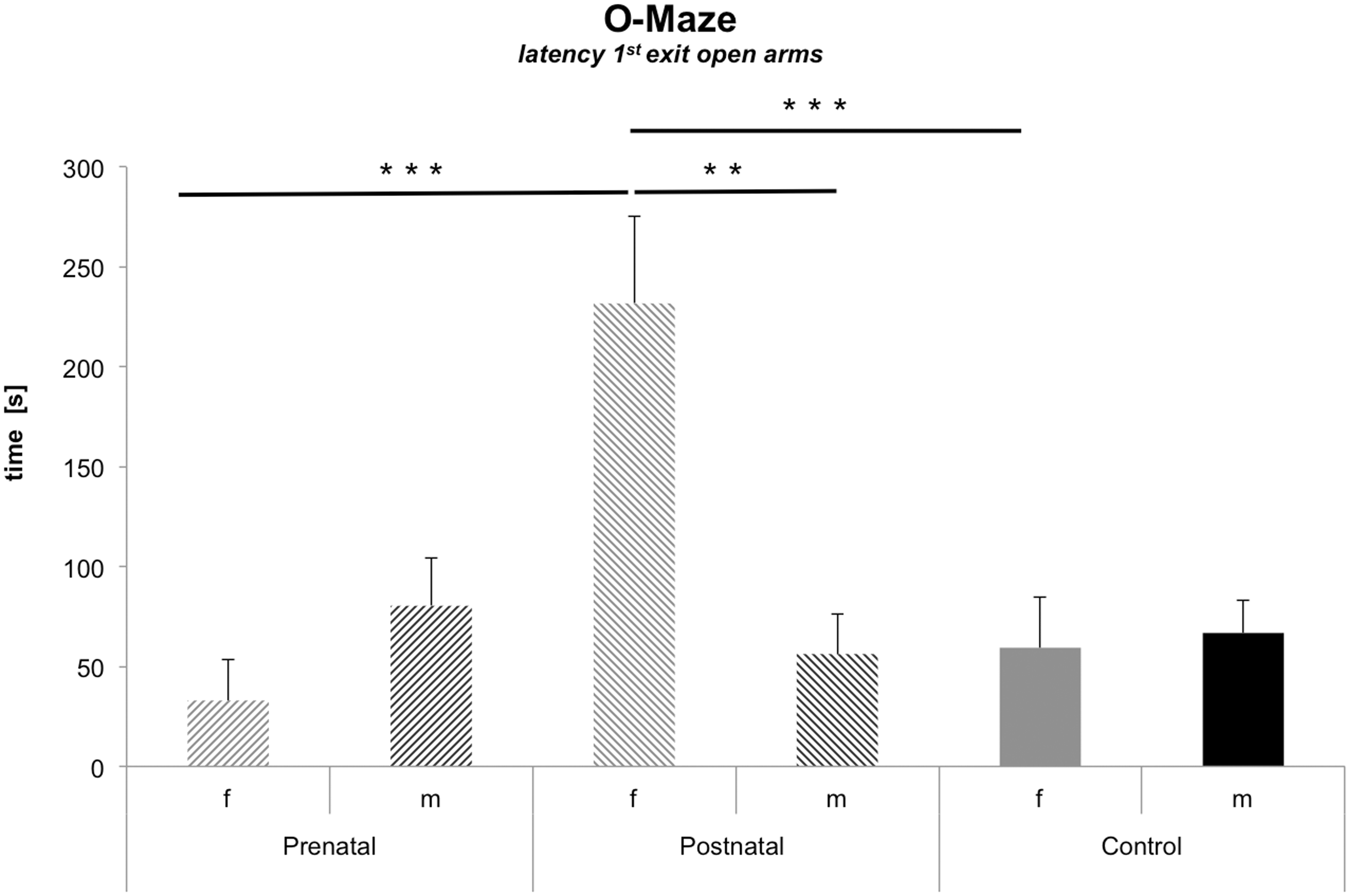
No general effects of perinatal stress conditions on social recognition emotional learning and depressive-like behaviour
Examination of data from the social recognition paradigm yielded a significant value for the sex × odour stress interaction (F(1,28) = 3.74; P = 0.04, Table 2, Figure 5). However, subsequent group comparisons failed to find any differences between specific groups, suggesting that no group showed impaired social behaviour. There were no significant effects on measures of fear conditioning in context or in cued conditioning. Similarly, there were no detectable statistical differences in the forced swim test.
Social recognition: entries Stranger 1 Phase 3. The figure shows the interaction of ‘odour stress:sex’. When regarding this interaction the previous findings match with the postulated anxious postnatal stressed females. Because postnatal stressed females show the lowest social interests. *P ≤ 0.05.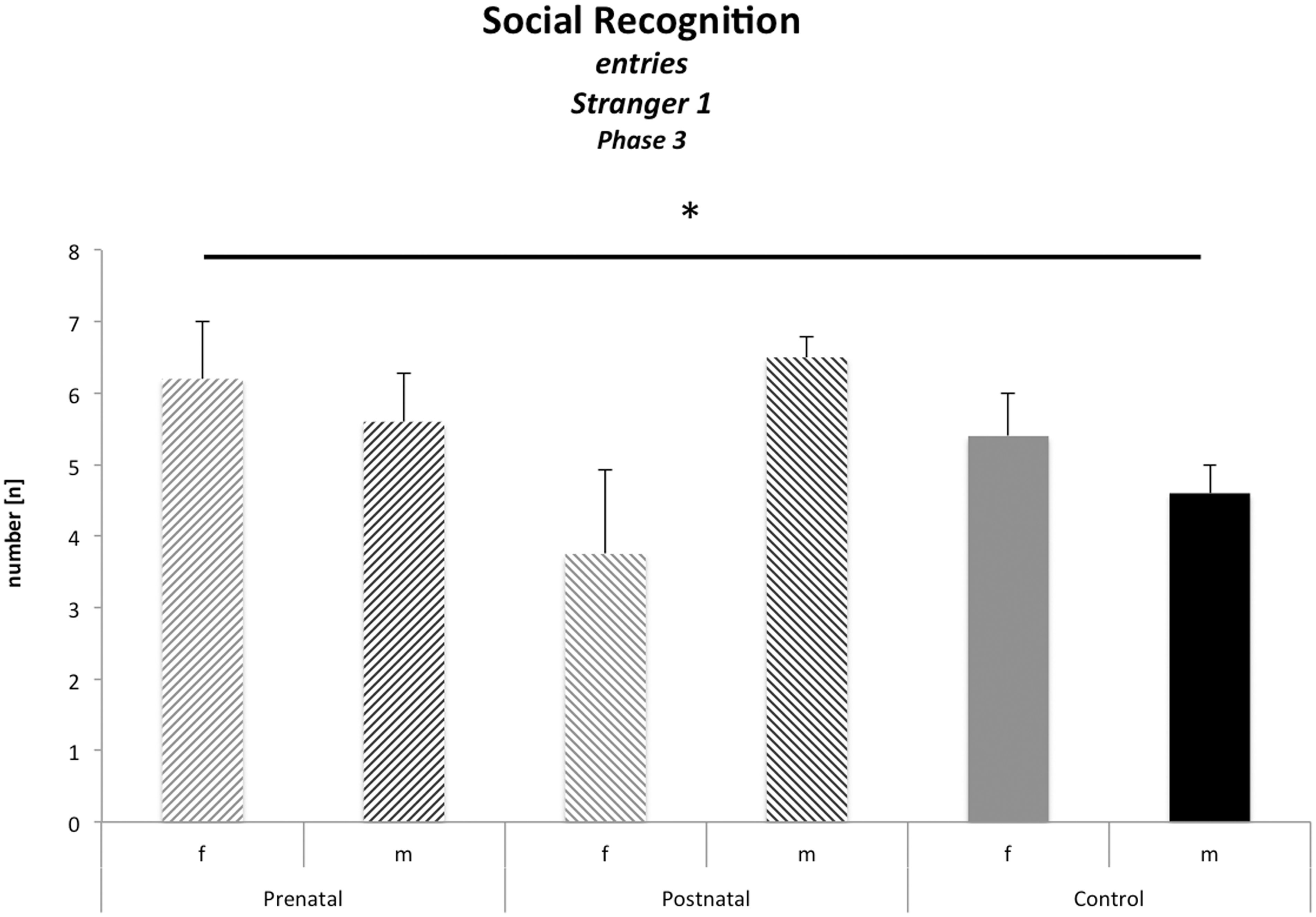
Corticosterone levels
Analyses of circulating corticosterone yielded non-significant differences for the odour stress main effect or for the interaction of sex and odour stress. However, the main effect for sex was statistically different (F(1,24) = 5.22; P = 0.035).
Discussion
Our study demonstrates that olfactory stress administered to pregnant females or to dams after parturition affects emotionality in their offspring in a sex-specific manner. These data are in line with several studies focusing on potential predator stress during the critical pre- and postnatal phases.38,39
Animal models need to fulfil ‘face’, ‘construct’ and ‘predictive’ validity,40–44 which closely mimic the focused syndrome, its origin/pathophysiology and its reversal by ‘gold standard’ treatments. Given these principles, it seems doubtful that animal models – especially regarding psychiatric illnesses – may provide ‘construct’ validity without consideration of decisive ‘pathophysiology-shaping’ aspects during the perinatal phase.
The primary aim of the experiment was to address the effects of pre- and postpartum odour stress on mothers and the emotional behaviour of adult offspring. Indeed, alterations of maternal nest building behaviour could be detected in the third week of nursing when prepartum stressed dams showed higher nest building scores. Other measures of maternal behaviour showed no differences with the non-stress control groups.
Despite these findings in mouse mothers, pre- and postpartum olfactory stress evoked altered emotionality in adult offspring. Higher numbers of faecal boluses in the open field suggested that higher emotionality in the males and observed anxiety in the O-maze were, therefore, not mediated by any obvious features of the maternal care that we measured.
Several studies on predatory olfactory stress have shown that rat odour significantly affects the hedonic and anxiety behaviour of male mice.45,46 Further investigations have demonstrated odour-transmitted alterations in maternal behaviour, which in turn affects the offspring’s stress response and fear system in a sex-specific manner. 22
Similarly, predator odour has been shown to affect many parameters such as the release of acetylcholine and corticosterone, hippocampal cell proliferation, and avoidance responses.47–52 Since most of these studies were not conducted during a particular sensitive phase, it seemed likely to us that exposure to a potentially dangerous situation during pregnancy or lactation would evoke significant effects by epigenetic programming. This is supported by studies describing anxiety-related gene × environment effects when mouse mothers were exposed to soiled bedding 21 or to an unfamiliar male during late pregnancy. 53 Therefore one may assume that odour represents one of the key cues for rodents – crucial for the survival of many species.
Facilities that breed mice for biomedical studies typically have standard operating procedures (SOPs) for mating, raising and weaning but details of these protocols are seldom described in publications. Our data and findings from other laboratories point to the experiences of mothers being a risk factor to the development of psychiatric disorders. 14
An example from our study was the increased emotionality in the open field in males raised by prepartum stressed mothers. Also, females from postpartum stressed dams exhibited increased anxiety. O-maze testing revealed an increased latency to enter the exposed compartments by adult female offspring experiencing postnatal stress. A sex difference in anxiety levels is well documented. The suggestion here is that females may be more sensitive to postnatal stress.54,55
Fear and anxiety represent behavioural adaptations for an individual raised in a challenging environment.56,57 It is, moreover, likely that different parameters are of distinct relevance for each sex, and the sexes are affected differently.
Conclusion
Our results and those of others consistently demonstrate that the influence of the perinatal environment may have a great impact on studies of emotional behaviours. It is therefore noteworthy to consider describing potential influences of the breeding environment (e.g. ARRIVE guidelines58,59). It is our recommendation that breeding facilities and researchers preempt conditions that could confound experimental results. Consequently, our study emphasizes the importance of the animals’ early histories and supports current guidelines requesting precise description of all animal procedures.
Footnotes
Acknowledgements
We thank Charles River (Sulzfeld, Germany) for donating the mice for this study as well as the caretakers of the IBF. We also appreciate the great help of Professor George Taylor in proofreading this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was in part supported by a grant from the Deutsche Forschungsgemeinschaft to PG. (SFB 636-B3).
