Abstract
Due to its similarity to humans, the pig has proven to be a suitable biomodel for both research purposes and for training medical professionals, particularly in surgical specializations. For example, new implant materials have been tested on pig jaws and pigs have also been used in the development of new surgical techniques. For optimizing the effectiveness of such research or training, detailed data on the anatomy of their particular features are needed. At present, however, only limited information related to surgical and imaging anatomy of the facial and orbital areas of the pig and its comparison to human structures from the experimental surgery point of view is available in the literature. The aim of this study was to obtain such data and to compare the morphological structures of the porcine and human orbital regions and to lay down the foundation for practical use in experimental surgery. Ten pig heads were examined using computed tomography (CT) and magnetic resonance imaging (MRI) and, subsequently, a dissection of the orbit was carried out. Attention was focused on the structure of the orbit (floor, rim and nerves) frequently affected by pathological processes in humans (such as trauma, infection or tumours) and which consequently are frequently the subject of maxillofacial surgery. The porcine orbit is suitable for use in experimental medicine. However, if used in experiments, its anatomical peculiarities must be taken into consideration. Our study presents a foundation of basic knowledge for researchers who plan to use the pig as a biomedical model to investigate alternative treatments in the head region.
The pig has become popular as an experimental model for human systems in many biomedical fields due to its anatomical and physiological similarity to humans as well as for economic reasons. Because of the physiological similarities, the extrapolation of results acquired in pigs to human conditions gives more relevant results than those acquired from other experimental animals such as the mouse, rat or rabbit. 1 Experimental studies in terms of different surgical approaches to the facial skeleton have been performed in various model animals including rabbits, dogs, sheep or goats.2–8 A variety of animal models for implant biomaterial research have been described in a recent extensive literature review. 9 In training and practising oral surgical procedures, pig heads are a well-established model.10–12
However, only limited information has been obtained using modern imaging methods, or is available comparing the anatomy of the pig orbit to human structures from the perspective of experimental surgery. Compared with humans, the pig orbit is relatively small (Figure 1).
Morphometric assessment of the porcine orbit. Computed tomography 3D reconstruction of the porcine orbit. OW: orbital width; OH: orbital height; a: line connecting processus zygomaticus ossis frontalis and processus frontalis ossis zygomatici.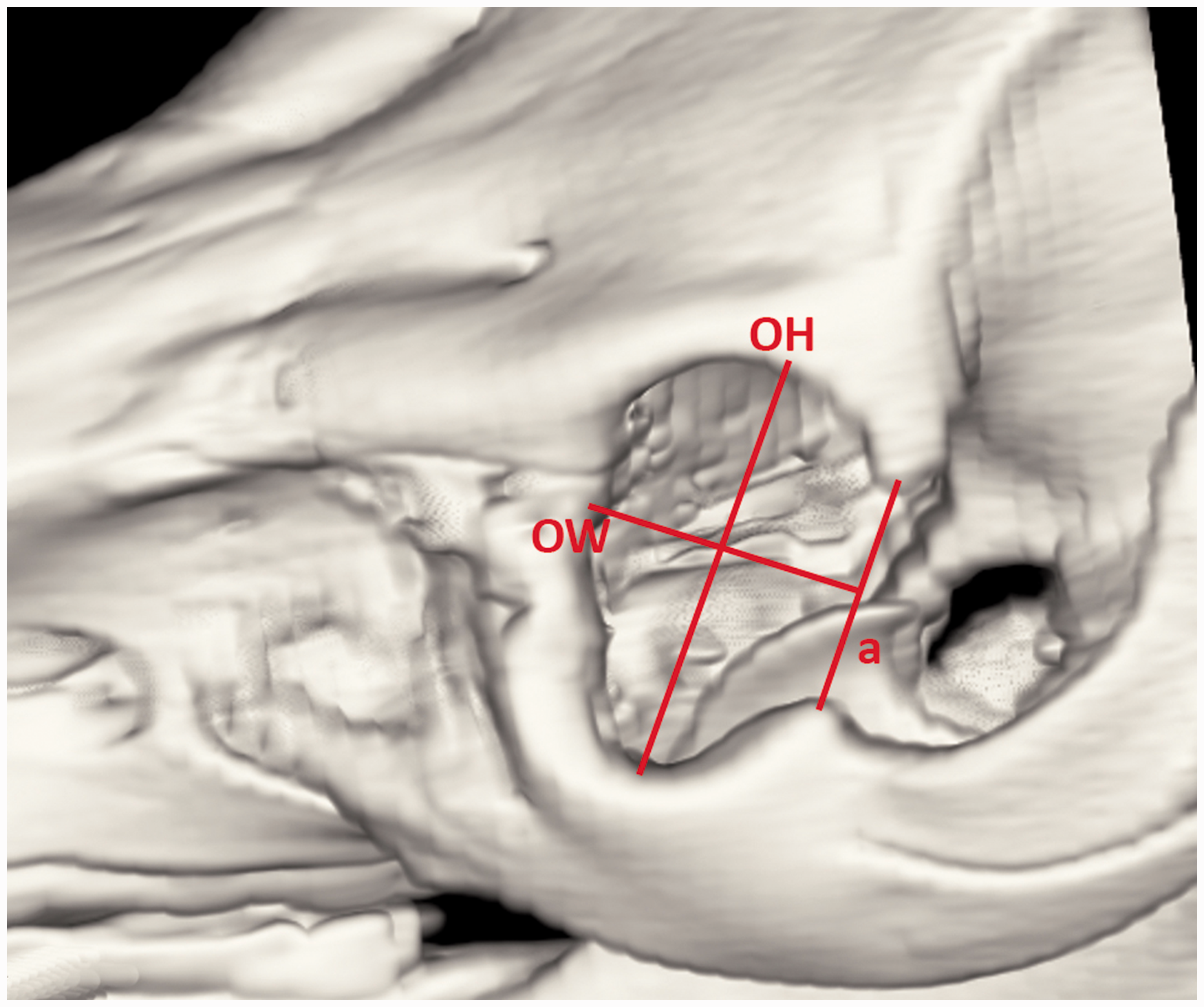
The eyelids, lacrimal system, orbit and conjunctiva together play an important role in the protection and function of the eye. Many diseases or trauma altering the structure or function of the eyelids, lacrimal system and/or the orbit can be detrimental to vision. Periorbital and facial injuries are mainly caused by assaults and falls and may at times involve the forehead. 13 Such injuries necessitate cranial and orbital reconstructions to correct both aesthetic and functional defects. 14 In order to achieve this, the surgeon needs to have a proper understanding of the human orbital structure, its relationship to both intra- and extracranial structures, and associated key surgical/anatomical landmarks. In general, the pig is a widely used animal model in medical research disciplines as it is very similar to humans in both physiological and individual anatomical aspects.1,15 However, little information is available about surgical procedures affecting the facial area and the orbit in animal models. 16
The aim of this study was to describe the surgical anatomy, imaging and morphometric findings of the orbit required when planning surgery of maxillofacial aspects of the porcine orbital region. This information could be helpful both in the training of maxillofacial surgeons to enable them to perform trial surgeries on a cheap model similar to humans, and in the research of new surgical methods that could be later extrapolated to humans.
Materials and methods
Ten heads of 24-month-old neutered male pigs were obtained from a slaughterhouse (Steinhauser, Tišnov, Czech Republic). All procedures were conducted following a protocol approved by the ethical committee of the university (VFU Brno, Czech Republic, No. 67985904). All the heads were fully developed and free of any obvious pathology in the facial area. The animals had no local or systemic illness likely to cause any pathological tissue alteration. The health status of the animals was inspected and confirmed by a certified veterinarian prior to and following the slaughter. Heads were examined by computed tomography (CT) and magnetic resonance imaging (MRI) and subsequently a dissection of the orbit was performed.
Diagnostic imaging
CT was performed using a single slice CT scanner (Synergy; GE Healthcare, Milwaukee, WI, USA). The following protocol was used: 120 kV, 100 mA, 3 mm slice thickness/3 mm spacing, DFOV 25 cm, standard reconstruction algorithm. MRI analysis (Magnetom Trio 3 T; Siemens AG, Erlangen, Germany) of the pig heads was undertaken by the Institute for Clinical and Experimental Medicine, IKEM (Prague, Czech Republic). Forty-eight transverse and sagittal T1-weighted images were acquired using the above parameters.
Morphometric assessment of the orbit was carried out on the 3D reconstructed CT images and on the CT sagittal sections. Three measurements of the orbital rim and the orbit were collected. Aditus orbitae was assessed using orbital width (OW) and orbital height (OH). OW was measured from the most medial aspect of the orbital rim to the level of the imaginary line connecting laterally zygomatic process of the frontal bone with the frontal process of the zygomatic bone. OH was a line connecting the most proximal aspect of the supraorbital margin with the most distal aspect of the infraorbital margin (Figure 1, Table 1).
Orbital dissection
Dissection of the orbit was carried out using a stratigraphic approach based on surgical approaches used in human orbital surgery with adaptations to porcine anatomy. Each layer dissected was photographed for future description and comparison using a Nikon D 5100 camera (Nikon Europe BV, Amsterdam, The Netherlands). Detailed anatomic description of the images and dissected surgical approaches were carried out and served as a background for a comparison to human orbital anatomy and surgical approaches described in the literature.17,18
Results
CT findings
The orbit of the pig is oval shaped and its longer axis vertically positioned, such that its average height is 53.0 mm and average width 34.1 mm.
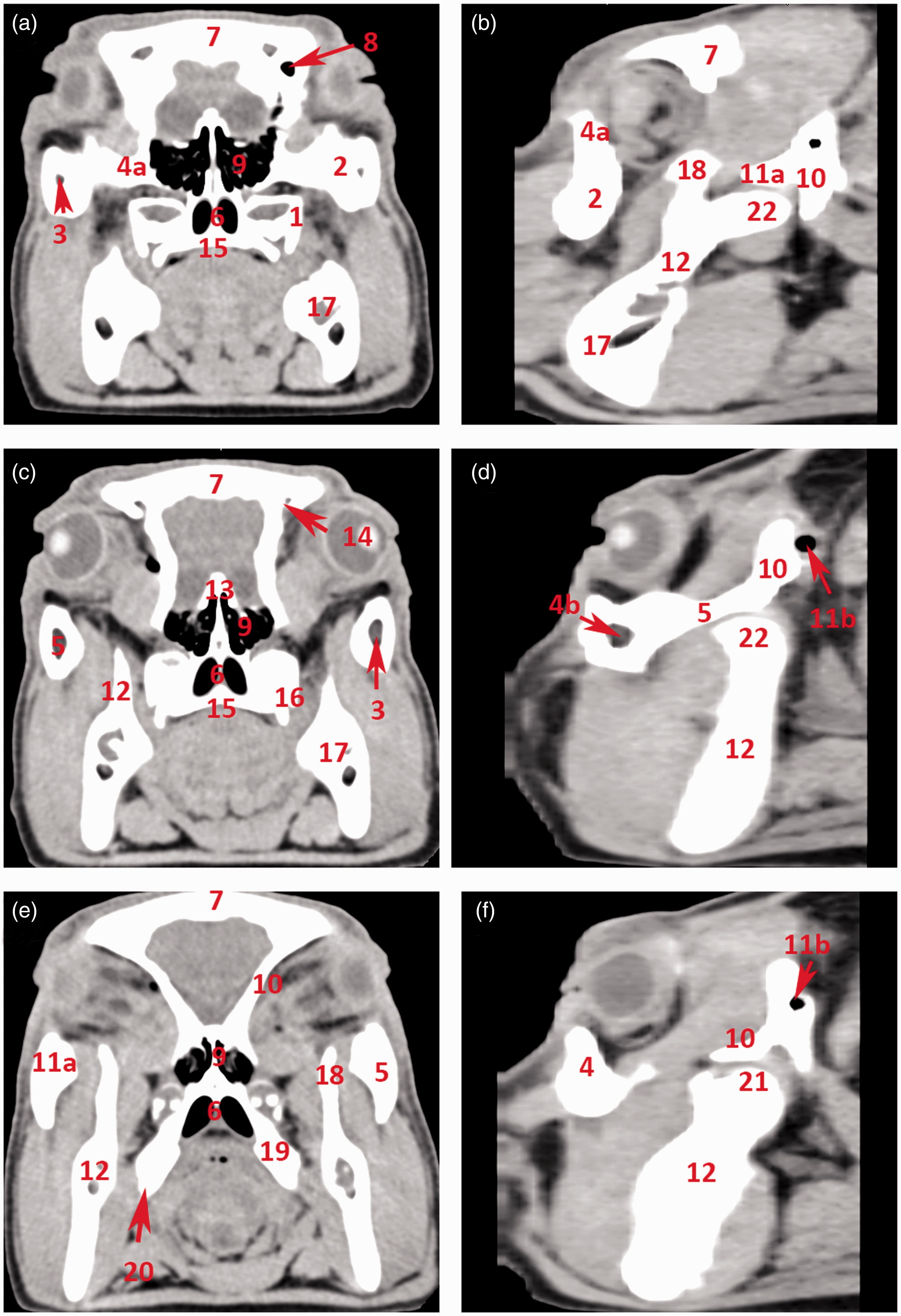
Its infraorbital margin is formed by the lacrimal and zygomatic bones and the supraorbital margin of the frontal bone. Its bony margin is incomplete caudolaterally and is completed by the orbital ligament, which connects the processus zygomaticus ossis frontalis with the processus frontalis ossis zygomatici (Figure 1). The cavity is limited ventrally by a ridge on the frontal and lacrimal bones and is separated by a crest from the temporal fossa. The medial wall is perforated dorsally by the orbital opening of the canalis supraorbitalis and ventrally by the optic and ethmoidal foramina. Two lacrimal foramina can be found on or close to the rostral margin, and these open into the large open fossa sacci lacrimalis which is located in the medial orbital canthus on the orbital face of the lacrimal bone. The zygomatic bone continues caudally with the processus temporalis, which together with the processus zygomaticus of the temporal bone forms a thick, dorsally convex zygomatic arch (Figures 2 and 3). The zygomatic, lacrimal and frontal bones that form the aditus orbitae are all pneumatized bones. The lacrimal sinus (sinus lacrimalis) is an excavation into the lacrimal bone. It borders ventromedially on the lacrimal canal and the maxillary sinus, medially and dorsally on the rostral sinuses, and caudally on the rostromedial wall of the orbit. The zygomatic bone is pneumatized by the maxillary sinus extending all the way to the processus temporalis ossis zygomatici. The sphenoid sinus (sinus sphenoidalis) is a paired sinus (right and left sides) divided by a septum (septum sinuum sphenoidalium). It excavates into the presphenoid, basis phenoid and temporal bones, creating a sinus in the medial wall of the orbit, in close proximity to the chiasma opticum. It extends laterally and dorsally, excavating into the squamous part of the temporal bones, and reaching into the zygomatic process (Figures 2 and 3).
Computed tomography 3D reconstruction of the porcine orbit. (a) Overview of the skull: lateral aspect. (b) Overview of the skull: rostral aspect. (c) Orbit: dorsal view. (d) Orbit: dorsolateral view. (e) Orbit: ventrolateral view. (f) Orbit: lateral view. 1: os frontale; 1 a: processus zygomaticus ossis frontalis; 1 b: os frontale – facies orbitalis; 1 c: margo supraorbitalis; 2: foramen supraorbitale – frontal opening; 2 a: foramen supraorbitale – orbital opening; 2 b: sulcus supraorbitalis; 3: os lacrimale; 3 a: os lacrimale – orbital surface; 3 b: foramen lacrimale; 4: maxilla; 5: foramen infraorbitale; 6: os zygomaticum; 6 a: processus frontalis ossis zygomatici; 6 b: processus temporalis ossis zygomatici; 6 c: margo infraorbitalis; 7 a: processus zygomaticus ossis temporalis; 7 b: squama ossis temporalis; 8: foramen ethmoidale; 9: arcus zygomaticus; 10: processus coronoideus mandibulae; 11: incisura mandibulae. Computed tomography transverse sections through the orbit. (a) Transverse section through the cranial aspect of the orbit. (b) Sagittal section through the lateral aspect of the arcus zygomaticus. (c) Transverse section through the mid-part of the orbit. (d) Sagittal section through the middle of the arcus zygomaticus. (e) Transverse section through the caudal part of the orbit. (f) Sagittal section medially from the arcus zygomaticus. 1: maxilla; 2: os zygomaticum; 3: sinus maxillaris; 4: margo infraorbitalis; 4 a: os lacrimale; 4 b: sinus lacrimalis; 5: arcus zygomaticus; 6: choana; 7: os frontale; 8: sinus frontalis rostralis; 9: labyrinthus ethmoidalis; 10: os temporale – squama ossis temporalis; 11 a: os temporale – processus zygomaticus; 11 b: os temporale – meatus acusticus externus; 12: ramus mandibulae; 13: os ethmoidale; 14: foramen supraorbitale – orbital opening; 15: palatum durum; 16: processus pterygoideus ossis palatini; 17: corpus mandibulae; 18: processus coronoideus mandibulae; 19: os pterygoideum; 20: hamulus pterygoideus; 21: processus condylaris mandibulae.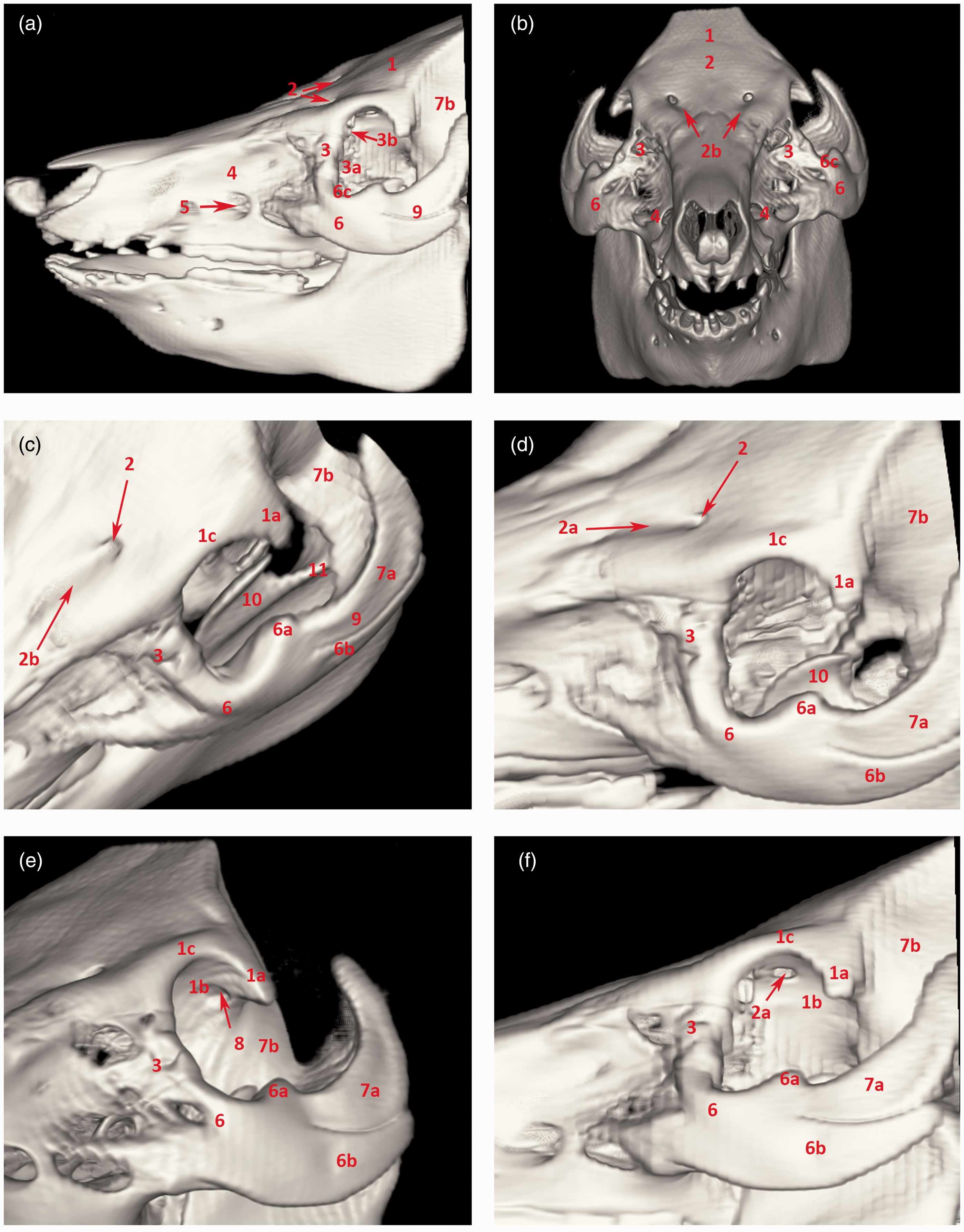
MRI findings
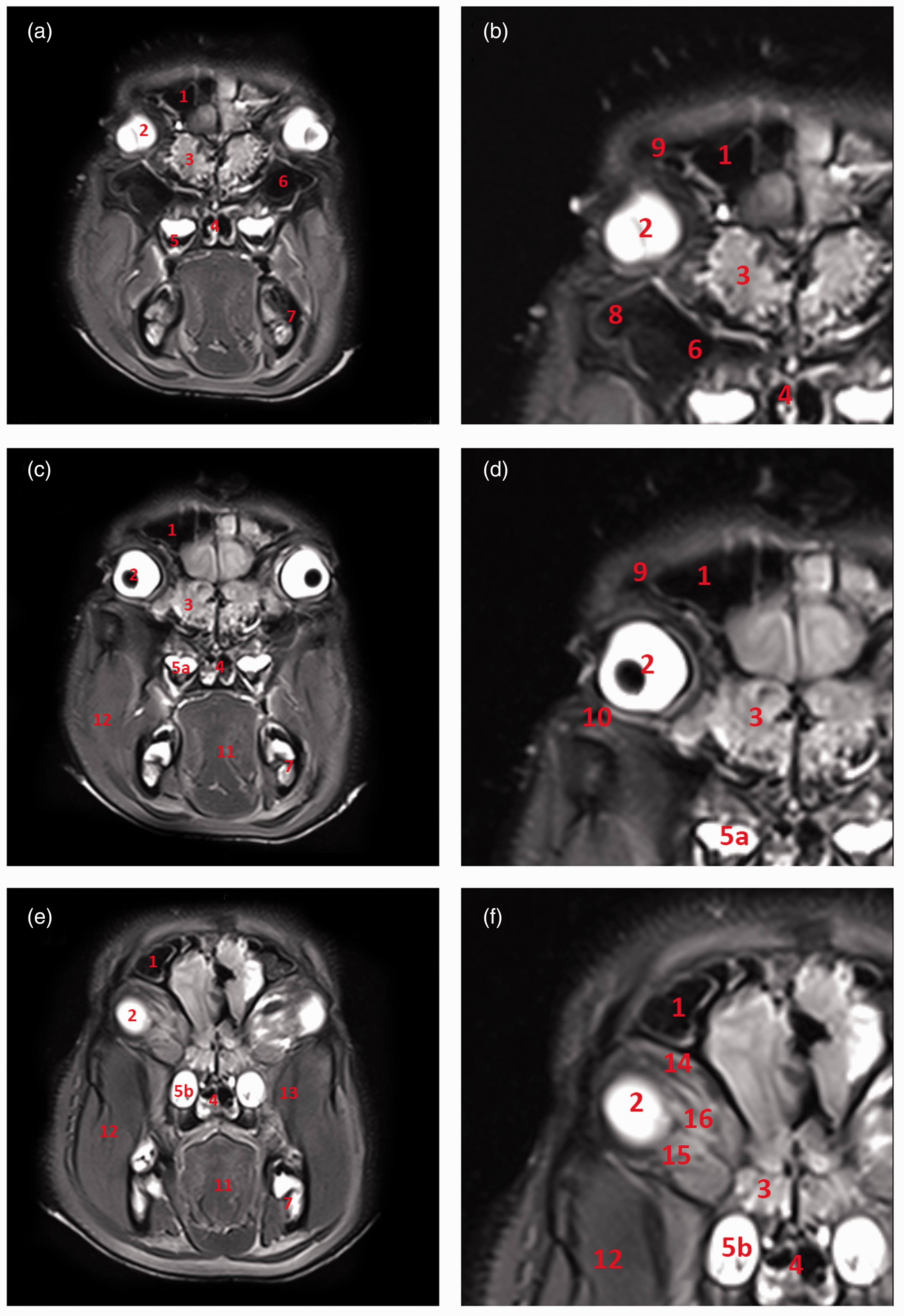
Soft tissue structures such as the mucosal covering of the conchae and paranasal sinuses, the detailed structure of the ectoturbinalia and endoturbinalia of the labyrinthus ethmoidalis, and muscular tissue are all shown in clear detail in the MR images (Figures 4 and 5).
Magnetic resonance imaging: T1-weighted sagittal sections of the orbit. (a) Sagittal section of the porcine head at the level of the medial orbit. (b) Detail of the orbit. (c) Sagittal section of the porcine head through the centre of the orbit. (d) Detail of the orbit. (e) Sagittal section of the porcine head at the level of the lateral orbit. (f) Detail of the orbit. 1: sinus frontalis; 2: sinus lacrimalis; 3: musculus temporalis; 4: musculus pterygoideus lateralis; 5: fourth mandibular molar tooth; 6: musculus levator nasolabialis; 7: bulbus oculi; 8: squama ossis temporalis; 9: musculus rectus bulbi dorsalis; 10: musculus rectus bulbi ventralis; 11: musculus retractor bulbi; 11 a: meatus acusticus externus; 12: os frontale; 13: auris externa; 14: condylus mandibulae; 15: discus articularis. Magnetic resonance imaging: T1-weighted transverse sections of the orbit. (a) Transverse section of the porcine head at the level of the second maxillary molar tooth. (b) Detail of the orbit. (c) Transverse section of the porcine head at the level of the third maxillary molar tooth. (d) Detail of the orbit. (e) Transverse section of the porcine head at the level of the fourth maxillary molar tooth. (f) Detail of the orbit. 1: sinus frontalis; 2: bulbus oculi; 3: labyrinthus ethmoidalis; 4: choana; 5: second maxillary molar tooth; 5 a: third maxillary molar tooth; 5 b: fourth maxillary molar tooth; 6: sinus maxillaris; 7: corpus mandibulae; 8: sinus zygomaticus; 9: os frontale; 10: os zygomaticum; 11: lingua; 12: musculus masseter; 13: musculus pterygoideus lateralis; 14: musculus rectus bulbi dorsalis; 15: musculus rectus bulbi ventralis; 16: musculus retractor bulbi.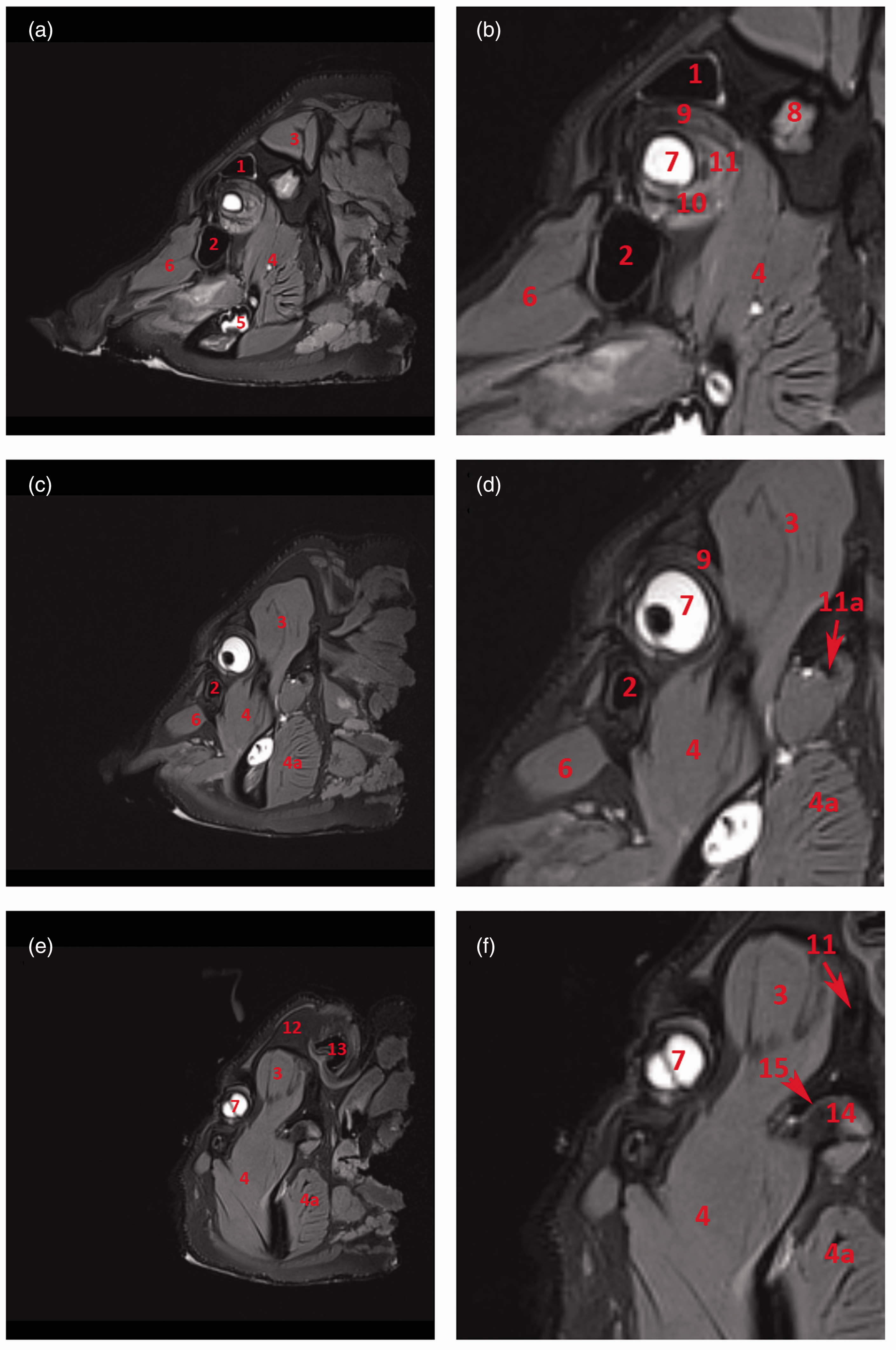
The globe and the other orbital tissue structures are surrounded by orbital fat. This orbital fat starts as a dense connective tissue at the aditus orbitae and extends dorsally towards the roof of the orbit and ventrally towards the pterygopalatine fossa as an intermuscular fat pad. The orbit is lined by a continuous fibrous membrane, the periorbita, which forms a dense condensation in close proximity to the orbital margin.
MRI clearly facilitates the differentiation of extraocular muscle groups. The dorsal and ventral oblique muscles of the eye bulb (m. obliquus dorsalis and ventralis oculi) are easily discernible from straight muscles of the bulbus (m. rectus bulbi dorsalis, ventralis, medialis and lateralis) which however cannot be distinguished from the retractor muscle of the bulbus (m. retractor bulbi) except at their origins on the basis of sphenoid and presphenoid bones. The temporal muscle (musculus temporalis) originates in the temporal fossa or planum parietale and inserts into the internal side of the coronoid process of the mandible (Figures 4 and 5). This insertion occupies the entire rostral region down to the level of the third molar tooth.
Surgical anatomy of the orbit
The pig orbit is a cavity delineated by four walls, all but one of which is formed by bones. The medial wall is almost rectangular. It comprises the lacrimal bone, the zygomatic bone rostrally and medially and maxilla ventromedially. The thick bone of the medial wall covers the underlying sphenoid paranasal sinus caudally and the frontomaxillary sinus rostrally. It also adjoins the optic canal and the supraorbital opening for the same artery and nerve. The lacrimal bone carries two lacrimal foramina (Figure 2). Ventrally it continues as the lamina pterygopalatina bordering the ventrally located fossa pterygopalatina filled with pterygoid musculature.
The roof consists of the frontal bone and a wing of the sphenoid bone (Figures 2 and 3). It is composed of very thick bone pneumatized by the frontal lateral sinus (Figure 4). The ventral aspect of the orbital roof lodges a large and shallow fossa for trochlear attachment of the dorsalis obliquus muscle. The lacrimal gland is housed in a shallow fossa in the rostrolateral aspect of the roof where the zygomatic process of the frontal bone connects with the ligamentum orbitale (Figure 6).
Surgical anatomy of the orbit. 1: palpebra superior; 2: palpebra inferior; 3: canthus medialis; 4: canthus lateralis; 5 a: musculus orbicularis oculi pars inferior; 5 b: musculus orbicularis oculi pars superior; 6: musculus malaris; 7: musculus zygomaticus – tendo originis; 8: nervus zygomatico facialis accessorius; 9: nervus zygomaticofacialis; 10: ramus communicans cum nervus buccalis; 11: arteria malaris; 12: margo supraorbitalis; 13: nervus trochlearis; 14: ligamentum orbitale; 15: processus zygomaticus ossis frontalis; 16: processus frontalis ossis zygomatici; 17: corpus adiposum periorbitae; 18: glandula lacrimalis; 19: musculus temporalis.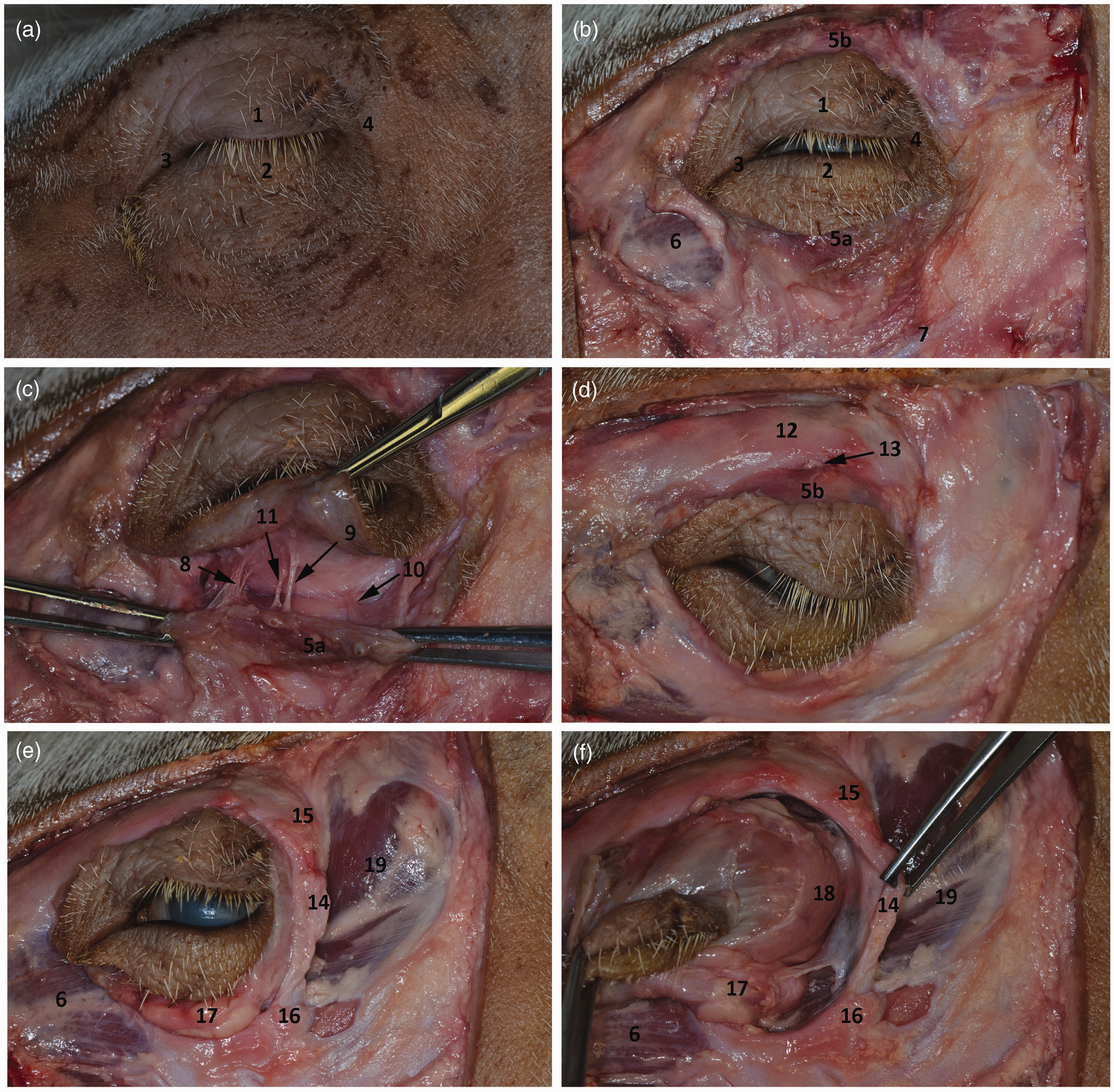
The floor of the orbit is very short, extending only one centimetre behind the infraorbital edge and descending sharply into the fossa pterygopalatina (Figure 6). This is occupied mainly by an infraorbital fat pad (Figure 6) which continues ventrally into the fossa pterygopalatina between the lateral and medial pterygoid muscles, protecting the large neurovascular bundle comprising the maxillary artery, and its branches and branches of the trigeminal nerve.
The caudolateral wall is composed of soft tissues represented by a ligament (ligamentum orbitale) that extends between the zygomatic process of the frontal bone and the frontal process of the zygomatic bone (Figures 2, 3 and 6). Caudally and ventrally it opens widely into the infratemporal fossa, which contains the terminal fibres of the temporalis muscle, the maxillary artery, and the maxillary vein.
The orbicularis muscle underlying the eyelids is composed of two layers of muscular tissue and is anchored with short ligaments to the medial and lateral aspects of the orbital opening. Its innervation arises from the trigeminal origin of the zygomatic nerve branches located on the ventral margin of the orbit and dorsally from the trochlear nerve. The blood supply of the ventral part of the orbit is provided by the arteria malaris, and of the dorsal part by the arteria supraorbitalis (Figure 6).
Discussion
Orbital rim measurements.
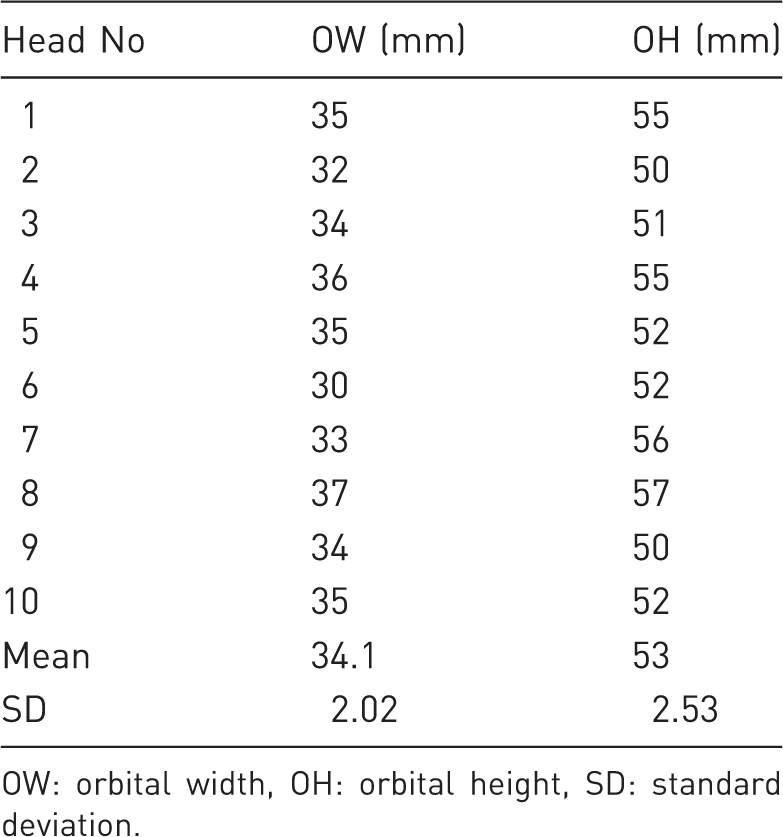
OW: orbital width, OH: orbital height, SD: standard deviation.
As well as differing from the the porcine orbit in size and shape, the human orbit is considered to be enclosed while the porcine orbit is open or incomplete. 21 The enclosed orbit is formed by bones along its whole circumference, whereas the open orbit, such as in the pig, lacks bones at its caudolateral aspect. This gap is bridged by a strong band of collagen fibres, the orbital ligament, which completes the orbit. It is believed that the enclosed orbit is essential for protection and skull reinforcement during dominance rituals (such as the one in a large animal) and chewing; whereas the open orbit allows a better ability to open jaws widely, with reduced orbital closure. 21
All the bones contributing to the formation of the orbit in pigs are pneumatized, forming paranasal sinuses. Some reports regarding the extent of the paranasal sinuses in pigs are contradictory.17,20 The extent of paranasal sinuses in our study was found to be the same in all the skulls investigated. The disagreements between different anatomical studies and textbooks could be explained by different breeds of pigs, with differences in development of the skulls varying from almost brachycephalic to dolichocephalic breeds. Paranasal sinuses in pigs seem to be more extensive than those of the human skull, which could be explained by the size of the skull. Pigs’ skulls are larger and heavier, and the paranasal sinuses create empty cavities within the bone, adding to the bone mass while maintaining a low weight. 17
Orbital fasciae in pigs are separated by the adipose tissue that fill the dead space in the orbit and between the extraocular muscles, which acts as a protective cushion for the eye bulb and muscles and therefore makes the periorbital and orbital anatomy in this aspect identical to that of humans. 18 Porcine eyelids are relatively thick and their movement is limited due to their considerably thicker dermis and also to the amount of fat under the skin and the conjunctiva. The upper eyelids contain heavy cilia but the lower eyelids lack them, unlike in humans. The porcine conjunctiva, unlike that of humans, has developed a third eyelid which is located in the medial canthus of the eye. It contains a cartilaginous plate which serves as an attachment of smooth muscle and third eyelid gland. It is believed to be involved in immunological defence of the conjunctiva, photoprotection and pheromone production. 22
Despite certain anatomical differences between human and porcine orbits, the pig is an important anatomical and surgical model as described in other studies23–25 and has to be considered in many areas of maxillofacial trauma and aesthetic surgery training. The porcine model can be very useful in simulating reconstruction of an injured eye bulbus, eye muscles, oculomotor or optic nerves of the lateral, upper and/or lower orbital wall, or reconstructions after oncological resections.1,18
However some conditions and surgeries cannot be simulated in the pig model due to differences in the structure and thickness of the orbit-forming bones between pigs and humans. Most orbital injuries in humans occur due to impact forces acting on the very thin (sometimes described as ‘paper-thin’) bone forming the floor and the medial wall. Upon impact on the bulbus, hydrodynamic forces lead to breakage of these thin bones with subsequent prolapse of the orbital tissues and their incarceration into the fracture lines, 26 known as a ‘trap door’ fracture. This type of fracture however cannot be modelled in the porcine orbit due to the different anatomical arrangement of the bones, namely formation of the caudolateral wall of the orbit by fibrous tissue in the form of a ligament, and the significantly thicker orbital bones.
There are many other pathologies affecting the human orbit. In addition to orbital trauma, pathological processes such as damage to the brainstem or cortical centre scan can lead to individual orbital nerve defects. 26 Defects of mobility or binocular vision which are caused by mechanical disruption or by changes of orbital volume are also common in humans. The causes might be infraorbital and/or extraorbital and may be due to trauma, tumours or other orbit-located expansive processes (such as orbital thyreopathy).26,27 Other causes include pathologies leading to expansion of the paranasal cavity or myogenic pathologies caused by illness or by incarceration of eye muscles. The location and composition of the porcine orbit do not allow modelling situations requiring coordination of both eyes.26,28 It is not possible to identify a disruption of binocular sight after carrying out the simulated surgical procedure. It is also impossible to simulate surgeries aimed at modifying optical properties of the eye. 29
Surgical approaches into the orbit
Surgical approaches to the orbit used in human medicine are well described, and require particular attention to ensure a good aesthetic result and to prevent damage to the optic nerve, oculomotor muscles and nerves, and damage to the eyelids (entropion and ectropion). These surgical approaches are based on detailed knowledge of human orbital anatomy. The surgeon must be aware of the limited volume of the orbit, which must not be altered during the surgical procedure. Any change in the volume or violation of the closed space can lead to a shift of the bulbus and thus to disruption of binocular vision.27,30–33 The surgical approaches involving the skin of the lower eyelid – subciliary, subtarsal, or transconjunctival – are employed for surgeries concerning the edge of the orbit or of the orbit itself. The decision about a particular incision the surgeon is going to perform is in large part dependent on the experience and surgical skills of the surgeon.34–36
A supraorbital approach offers direct access to the upper lateral edge and fronto-zygomatic suture. Disadvantages include a skin scar and possible disruption of branches of the trigeminal and facial nerves or the lacrimal gland.26,27,37–39
These routes of access can also be practised on the pig; it is however necessary to take into consideration the differences in the supraorbital nerve outlet and in the course of the facial nerve. In addition, the lacrimal gland is located dorsomedially from the orbital ligament.
The transconjunctival incision is another of the possible approaches into the lower part of the orbit. With this approach the skin remains intact, the scar is hidden in the conjunctiva (although this approach could be extended through the lateral cantus as a skin incision), and if the surgery is performed correctly there is a minimal risk of complications. The incision can be made in both the preseptal or retroseptal area, however this method requires a certain degree of surgical experience and practice.40–42
Since the porcine orbit is a suitable model for this type of approach, appropriate experience can be gained using the pig.
A transantral endoscopy-assisted approach provides direct access to the bottom of the orbit through the maxillary sinus. Due to possible complications associated with incisions to the lower eyelids, this approach is currently used to reposition tissues of the orbit and to insert osteosynthesis mesh. Its advantage is the avoidance of damage to soft tissues around the eye, resulting in no scarring and no risk of extropy. The disadvantages however lie in the long surgical time required in comparison with other surgical approaches, in the requirement for specific instrumentation, and the need for surgeons experienced in endoscopic surgery.43–45 The skin remains intact but this approach is technically difficult due to the limited space within the maxillary sinus.
The porcine model is however not suitable for practising this approach due to anatomical differences between pigs and humans, particularly in the layout of the orbit and the maxillary sinus.
The aim of surgeries of the orbit is to restore function and to reconstruct the orbit in order to achieve an outcome as close as possible to its normal anatomy. There is no ‘one size fits all’ surgical approach, and a wide range of techniques, materials and approaches are available for specific case scenarios. Therefore, careful pre-surgical planning is necessary and the porcine orbit can serve as a training model and can help to establish the advantages and disadvantages of each technique in different situations.
In conclusion, the porcine orbit is a suitable model for experimental medicine. However, if used in research, its anatomical differences and the breed of pigs must be taken into consideration. Consequently, the porcine orbit could serve as a suitable model for the training of surgeons, allowing them to gain necessary experience. All the most common surgical approaches used in human orbital surgery can be performed on a porcine head; therefore, the pig is a suitable model for practical training.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Ministry of Health of the Czech Republic (project 3 RVO-FNOs/2014).
