Abstract
The intraperitoneal application of lipopolysaccharide (LPS) alone or in combination with other hepatotoxins is an experimental model for inducing systemic and hepatic inflammation in rodents applied worldwide. The endotoxin is recognized by the LPS-binding protein. This complex binds together with the lymphocyte antigen 96 (MD2) and the pattern-recognition receptor CD14 to members of the toll-like receptor family. The activated receptor complex in turn transduces signals to well characterized intracellular cascades that result in a multifaceted network of intracellular responses ending in inflammation. The most prominent among these is the activation of the NF-κB pathway and the production of a multitude of inflammatory cytokines. Although the application of LPS is in general easy to perform, unintended variations in preparation of the injection solution or in handling of the animals might affect the reproducibility or the outcome of a specific experiment. Here, we present a well-standardized protocol that allows for an induction of highly reproducible acute hepatic inflammation in mice. Furthermore, examples of appropriate readouts for the resulting inflammatory response are given.
Historic background of the model
Lipopolysaccharides (LPS) are large lipid- and polysaccharide-containing molecules that are found in the outer membrane of Gram-negative bacteria. The toxic activity of LPS, that was originally termed ‘endotoxin’, was first identified in 1904 by the bacteriologist Richard Pfeiffer who worked together with Robert Koch in Berlin. Although Pfeiffer never made use of the term ‘endotoxin’ in his publications, it is historically established that he used this term first in one of his lectures that he gave in Königsberg around 1903/1904. His colleague Alfred Wolff-Eisner then made the term public and assigned it to Dr Pfeiffer in a review article one year later. 1 In brief, Pfeiffer observed that the Gram-negative bacteria Vibrio cholerae released two toxins into the culture medium. One of these, that he called ‘exotoxin’ (also called cholera toxin), is a heat-labile substance that is secreted by living bacteria. The second non-secreted toxin (now referred to as LPS or endotoxin) appeared to be attached to the bacterial cell and is only set free when the bacterium undergoes lysis. This class of macroamphiphiles is the dominating constituent of the outer leaflet of the bacterial membrane and protects the bacterium from the environment, inhibits the entrance of toxic compounds such as antibiotics, and is the key component in the pathophysiological interaction of bacteria with host organisms. 2 The immunogenic potential of such molecules was first shown in pioneering reports from the mid-1950s demonstrating that intravenous or subcutaneous injections of crude extracts of the death mycobacteria (such as Mycobacterium bovis bacille Calmette-Guérin [BCG]) increase the susceptibility of mice to various types of bacteria, and can convert chronic bacterial infections into acute processes that end in death. 3 In later studies, it was proven that small doses of LPS in mice that had been injected with Corynebacterium parvum produced a lethal hepatitis with high serum transaminase concentrations, glycogen depletion, and hypoglycemia. The authors concluded that the observed effects were most likely triggered by the release of hepatotoxic factors from the infiltrating mononuclear cells. 4 One year later, it was reported that the simultaneous treatment of rodents with the hepatotoxic agent D-galactosamine (D-GalN) at 300 mg/kg leads to a several thousand fold increase in their susceptibility to the lethal effects of LPS, most likely by the inhibition of synthesis of different macromolecules resulting in increased cell damage and cell death. 5 It is well known that this amino sugar is metabolized by hepatocytes, induces liver damage, and enhances the production of reactive oxygen species (ROS) in hepatocytes. 6 Therefore, D-GalN alone is also used as a model for acute hepatic failure. 7 An alternative hepatotoxic model is the combination of LPS and BCG which also induces oxidative stress caused by ROS. 8 Further mechanistic studies demonstrated the important role of tumour necrosis factor-α (TNF-α) and its receptor in the hepatocyte apoptosis and lethality associated with LPS and D-GalN administration.9–12 Many laboratories now apply LPS injection or a combination of LPS and D-GalN as an experimental model to induce inflammation and to investigate the underlying cellular and molecular mechanisms within the liver or other organs.
Pathogenic mechanisms of liver damage
Bacterial LPS, also known as endotoxin or lipoglycan, is the major component of the outer surface of Gram-negative bacteria.
13
All LPS molecules are relatively heat-resistant and share a common architecture. The rough-type LPS is composed of an endotoxic fat-like part (i.e. the lipid A) and a core sugar constituent consisting of approximately 10 monosaccharides (Figure 1). An additional O-chain composed of several repetitive subunits of one to eight O-specific monosaccharides is linked to the core sugar constituent in the smooth-type LPS molecules.
2
The lipid A moiety of the LPS molecules was already postulated in 1954 to be responsible for the toxic and immunomodulatory activity of various LPS molecules.
14
It represents a peculiar phosphoglycolipid that is highly conserved in the different LPS molecules.2,15 Pioneering reports have shown that certain cellular constituents of mycobacteria and purified LPS of Gram-negative bacteria enhance acute and chronic infections in mice and induce the appearance of large microbial populations in the murine liver.
3
Interestingly, the sensitivity of different species toward LPS is highly variable and the lethal toxicity of LPS for humans and mice differs by several orders of magnitude.16,17 While a dose of 1µg/kg body weight can induce endotoxic shock in humans, mice are rather resistant to such concentrations, most likely because of the existence of specialized immunoglobulins in mice that defend against severe bacterial infections.
18
During the last few decades the molecular understanding of how LPS harms the host, and which host sensors and intracellular pathways are responsible for the recognition of LPS, has dramatically increased.19–21 Pathogen-associated molecular patterns are recognized by special pattern-recognition receptors.
21
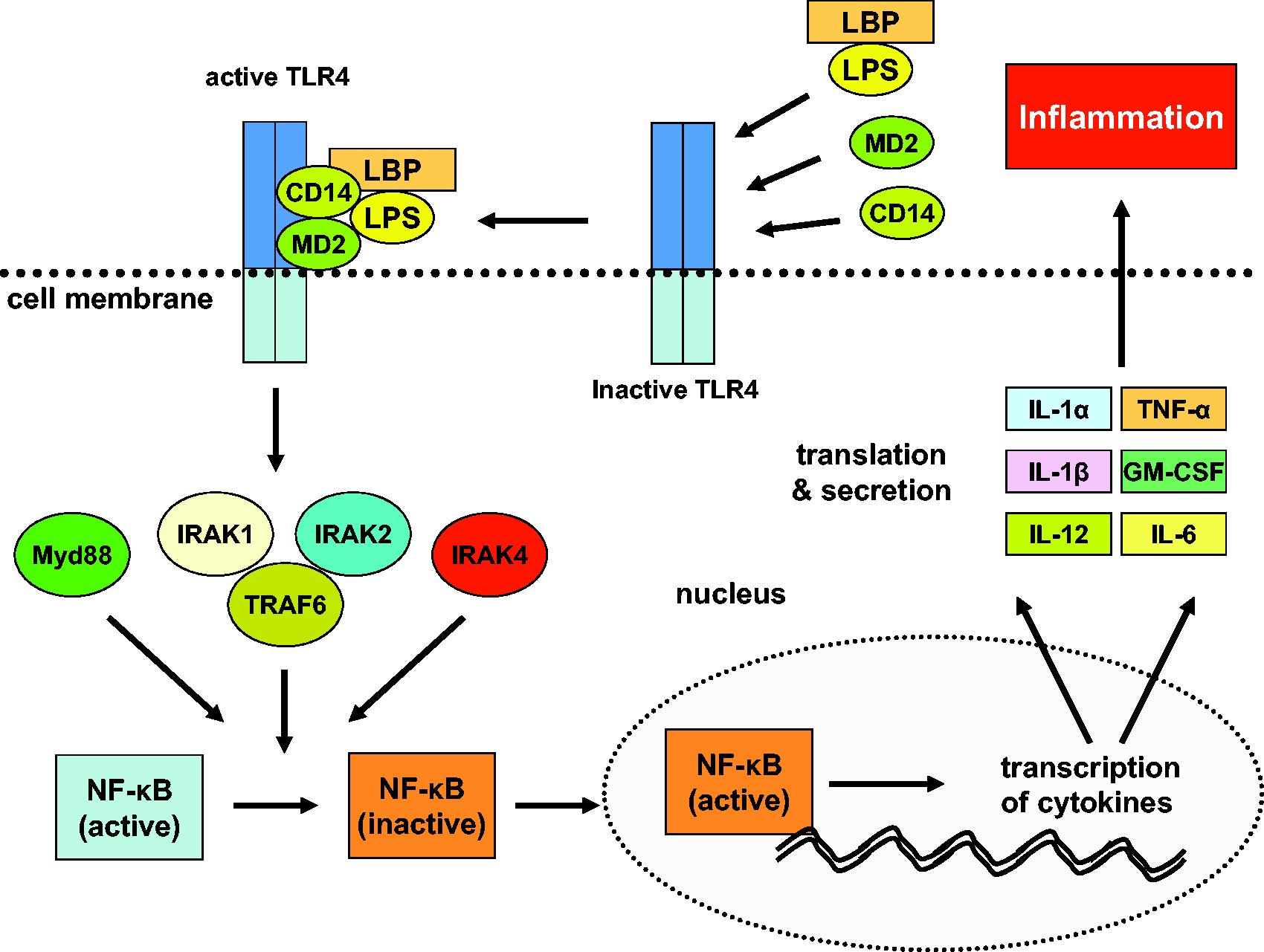
In a first step, LPS binds to the LPS-binding protein. This complex has affinity for the myeloid differentiation factor 2 (MD2) and the cluster of differentiation 14 (CD14) that together connect to the toll-like receptor 4 (TLR4). This activated receptor complex transduces signals to specialized intracellular proteins (MyD88, IRAKs, TRAFs and NIK), resulting in a multifaceted network of cellular responses, activation of the NF-κB pathway, and production of various inflammatory cytokines including TNF-α, interleukin (IL)-1α/β, IL-6, IL-12, IL-18, and granulocyte macrophage colony-stimulating factor (GM-CSF)
22
(Figure 2). As oxidative stress plays a major role in hepatitis, the combination of LPS and D-GalN represents a popular modification of the LPS-induced hepatitis model. D-GalN inhibits protein synthesis by depleting the uridine triphosphate pool which causes the early generation of ROS and finally of apoptosis.23–25
The structure of lipopolysaccharide (LPS). LPS is composed of lipid A containing fatty acids and a disaccharide diphosphate and a sugar core glycolipid of approximately 10 monosaccharides. In the smooth type LPS, an additional O-chain consisting of several repetitive subunits of one to eight O-specific monosaccharides is linked to the core sugar constituent. The figure was drawn using the Ribbons XP Version 2.0 software
47
and is based on the atomic coordinates that are deposited in the Brookhaven Protein Data Bank (PDB) under Access No. 1FCP in which LPS was crystallized together with the outer membrane protein FhuA (TonA) from Escherichia coli.
48
Lipopolysaccharide (LPS)-induced signaling. LPS bound to the LPS-binding protein is recognized by the membrane bound CD14/TLR4/MD2 receptor complex. Once activated, this receptor complex initiates a signal cascade consisting of a large variety of intracellular proteins (MyD88, IRAKs, TRAFs, NIK). As a result the transcription factor NF-κB becomes activated and transduces into the nucleus. It induces transcription of many inflammatory cytokines (e.g. TNF-α, IL-1α/β, IL-6, IL-12, IL-18, and GM-CSF) that are translated into proteins and secreted to drive the inflammatory response. CD14: cluster of differentiation 14; GM-CSF: granulocyte macrophage colony-stimulating factor; IL: interleukin; IRAK1/2/4: IL-1 receptor-associated kinase 1/2/4; LBP: LPS binding protein; MD2: myeloid differentiation factor 2 (lymphocyte antigen 96); MyD88: myeloid differentiation primary response gene 88; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B-cells; TLR4: toll-like receptor 4; TNF-α: tumour necrosis factor-α; TRAF6: TNF receptor associated factor 6.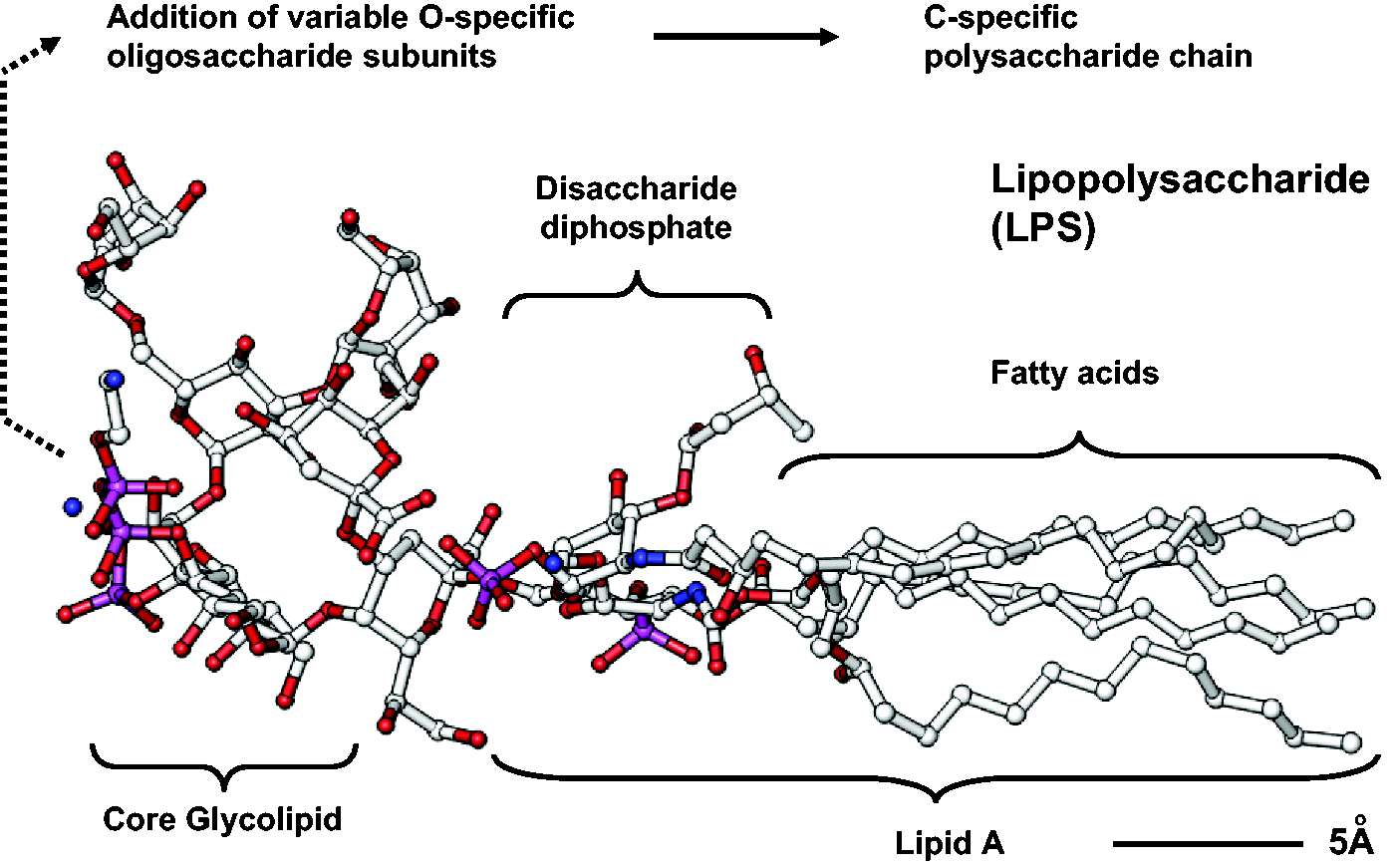
Experimentally and clinically, the role of the liver in the acute and chronic response to exogenous or endogenous LPS has been extensively studied over many years26–28 and the application of LPS in rats and mice as a model system for inducing systemic inflammation and inflammatory liver disease is well established.29–32
Experimental procedure
Overview of the use of lipopolysaccharide (LPS) (and D-galactosamine) in mouse models of inflammatory liver injury.
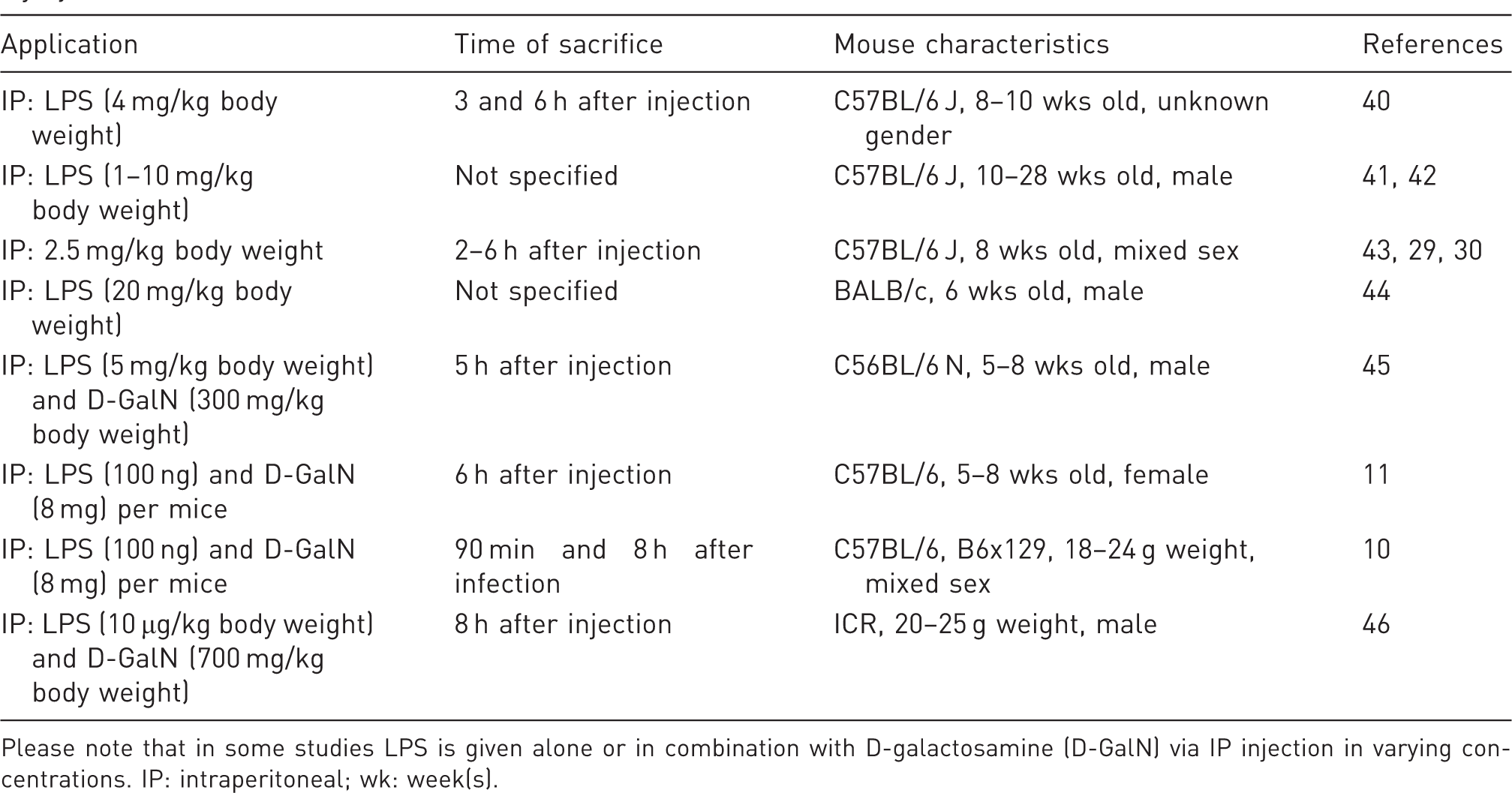
Please note that in some studies LPS is given alone or in combination with D-galactosamine (D-GalN) via IP injection in varying concentrations. IP: intraperitoneal; wk: week(s).
General considerations
Genetic background
As is generally recognized, there are significant differences in inflammation and fibrosis development in mice depending on the susceptibility of the various strains. 35 Thus, as with every liver damage model, the choice of a susceptible strain, the purity of the genetic background, and the use of wild-type littermates as controls are crucial for the interpretation of results. 36 We cannot provide a clear recommendation from the available literature which mouse strain is superior as there has been no systematic analysis of susceptibility towards LPS. If there is doubt, a pilot study that utilizes only small animal cohorts might be helpful before starting a complex set of experiments. As C57BL/6 mice are used in many recent studies investigating LPS-induced liver injury and as there is a considerable availability of genetically-modified mice with a C57BL/6 background, we suggest using this strain whenever possible. This will improve future standardization and further increase the comparability of data obtained from different laboratories.
Age and sex of mice
To reach the highest standardization possible, we recommend using animals of the same age and sex ratio in all experimental sets. In most of the recent studies, the age of the mice was between 6 and 10 weeks. While most of the recent studies used male mice, we suggest using equal numbers of male and female mice. This recommendation is in accordance with the recent guidelines from the National Institutes of Health (NIH, Bethesda, MD, USA). Furthermore, we recommend providing this information along with the average body weight of respective animals in the ‘Methods’ section in every publication. Unfortunately, these important details are often missing.
Application routes
In principle, LPS can be administered in various ways, inter alia by intraperitoneal (IP) injection or via drinking water. 37 The vast majority of investigators use IP administration. Major advantages of this application route are high reproducibility and good standardization. For induction of acute liver damage, LPS is either used alone or in combination with other substances such as D-GalN or BCG as described above. Unfortunately, the applied dosages used in recent studies vary widely and detailed systematic analyses about dose-dependent toxicity are missing. In order to adhere to ethical regulations, specifically the 3R principle, 38 we recommend using the lowest dose possible. Below, we provide recommendations for application modalities that are based on both personal experience and safety considerations.
Duration of treatment
Before LPS injection, mice are kept in a temperature-controlled room with a 12 h light–dark cycle and access to food and water ad libitum. After LPS injection, mice are kept for a few hours before sacrificing them, again with access to food and water ad libitum. In studies published in 2014, mice were mostly analysed between 2 and 8 h after LPS injection. According to the study aim, blood, liver and other organs are collected and preserved for subsequent histological, immunohistological and biochemical analyses.
Biometric calculation of requested cohorts
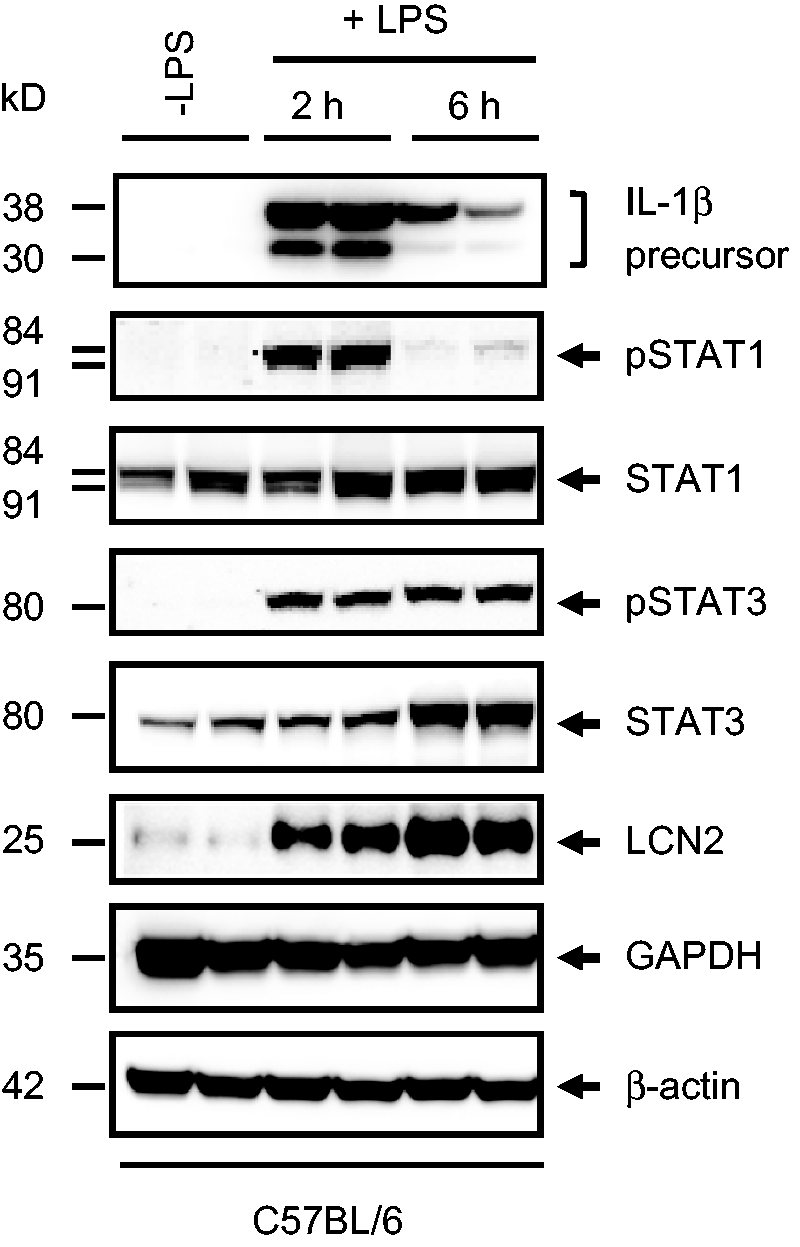
As LPS has various inflammatory effects, we recommend using the expression of inflammatory markers as appropriate readouts to monitor induction of inflammation. In our view, the most suitable markers are TNF-α, IL-1β, IL-6, the monocyte chemoattractant protein-1/C-C chemokine ligand-2 (MCP-1/CCL2) that can be easily measured by quantitative real-time polymerase chain reaction (Figure 3), or quantitative enzyme-linked immunosorbent assay (ELISA). Alternatively, the increased expression of IL-1β, the phosphorylation of the signal transducer and activator of transcription (STAT)1 and STAT3, or the expression of the inflammation-associated marker lipocalin-2 (LCN2) can be analysed by Western blot (Figure 4). The induction of hepatic injury becomes evident due to elevated activities of the serum aspartate transaminase (AST) and the serum alanine transaminase (ALT) that can be measured by standard clinical chemistry. When an inflammation-modifying substance should be tested, it is mandatory to analyse a control group in parallel that had only received LPS and without any further intervention.
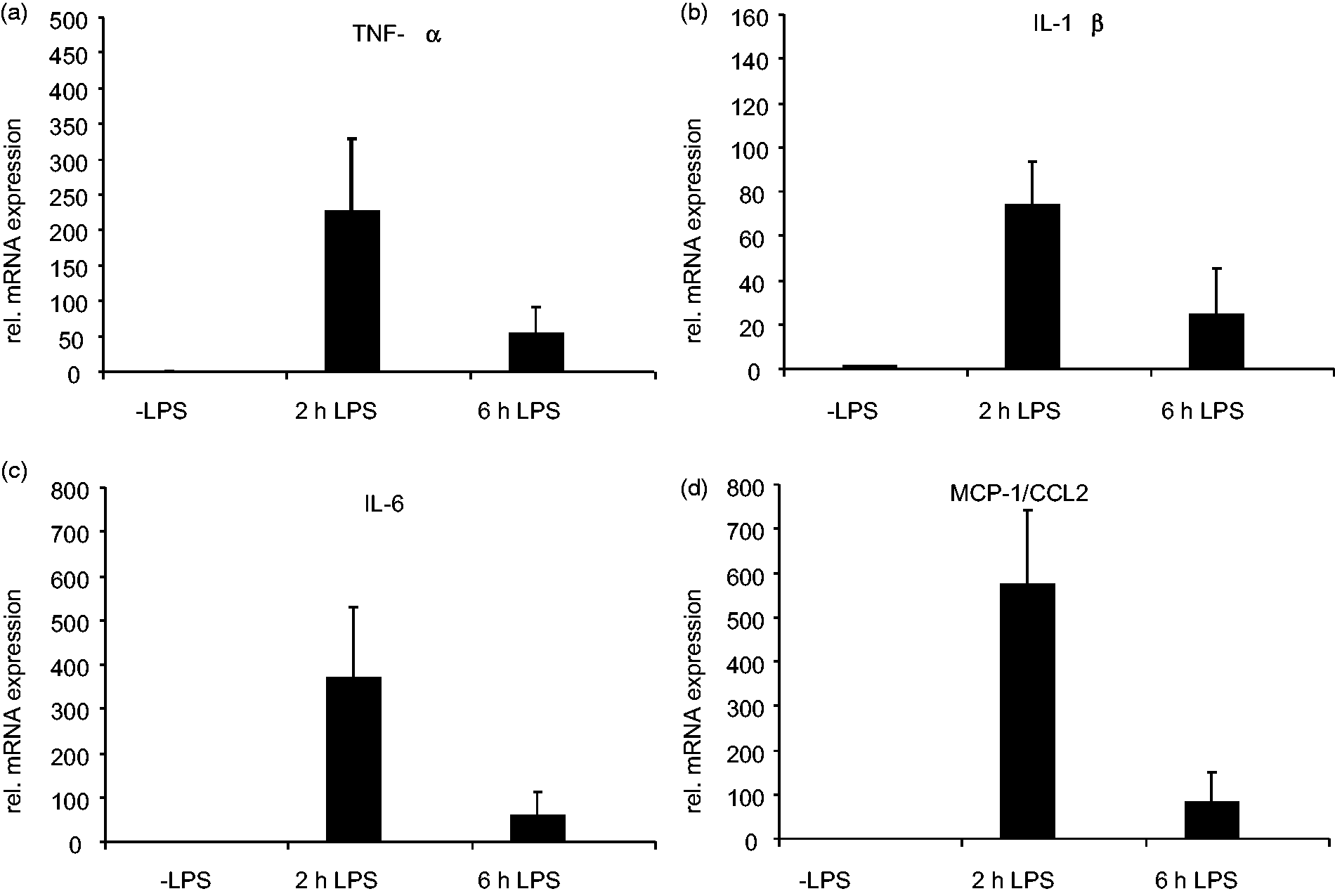
Quantitative real-time polymerase chain reaction (PCR). The expression of TNF-α (a), IL-1β (b), IL-6 (c) and MCP-1/CCL2 (d) was measured by quantitative real-time PCR in mRNA isolated from livers of animals that received 2.5 mg/kg body weight lipopolysaccharide (LPS) for 2 or 6 h. The expression of the individual genes was normalized to the expression that was observed in control animals that did not receive LPS. Details on primers used in this analysis and more experimental details on this study are published elsewhere.
30
Western blot analysis. Liver protein extracts were prepared from two C57BL/6 wild-type mice each that either received lipopolysaccharide (LPS) (2.5 mg/kg body weight) for 2 or 6 h or were left untreated. The proteins were subjected to Western blot analysis and probed with antibodies specific for IL-1β, phosphorylated STAT1 (Tyr701), total STAT1, phosphorylated STAT3 (Ser727), total STAT3, and LCN2. In this analysis the levels of both GAPDH and β-actin served as controls for equal protein loading. More experimental details and exact sources of antibodies used in this study have been published previously.
30
Representative sample size calculation for a typical lipopolysaccharide (LPS) stimulation experiment.
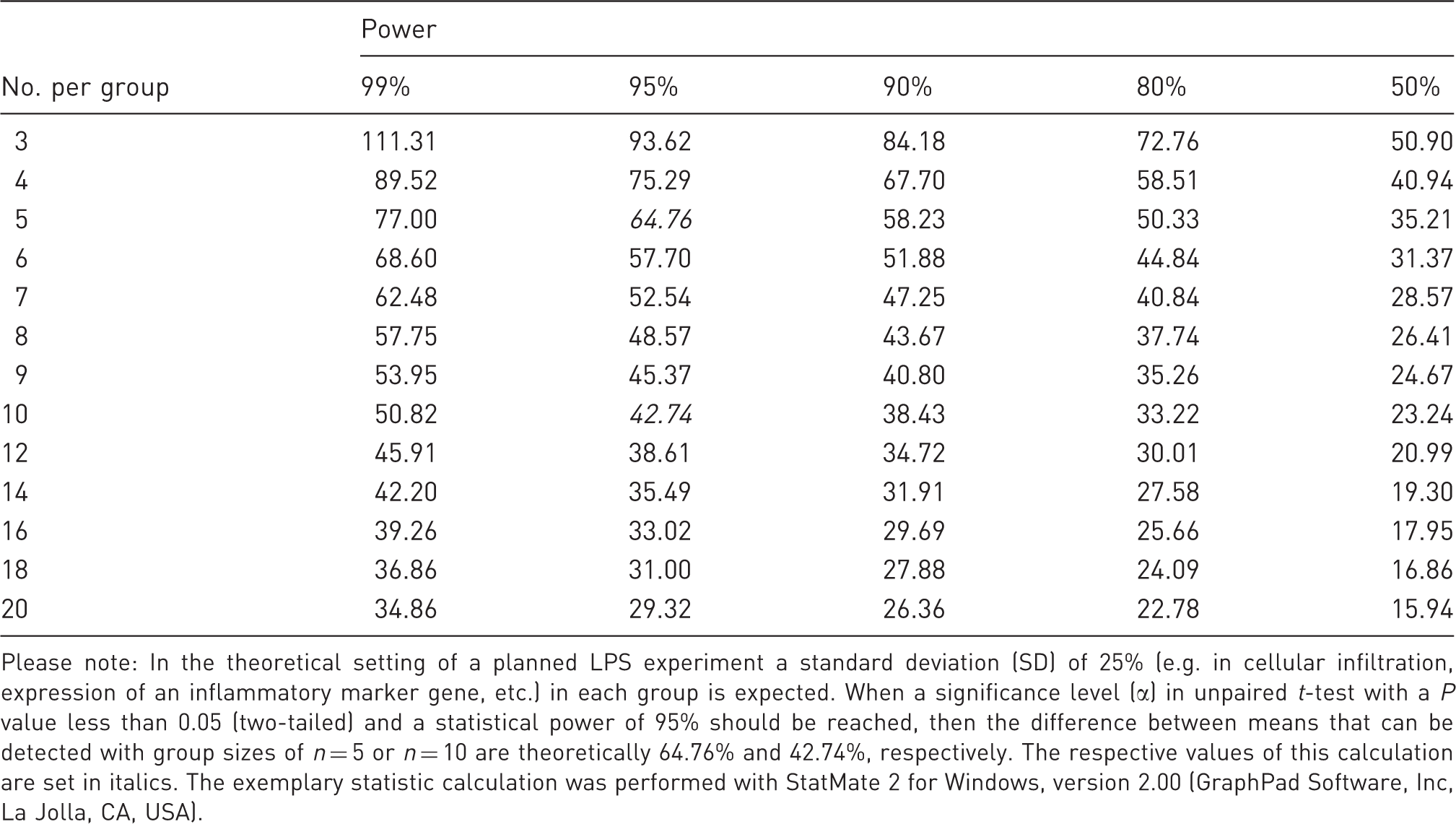
Please note: In the theoretical setting of a planned LPS experiment a standard deviation (SD) of 25% (e.g. in cellular infiltration, expression of an inflammatory marker gene, etc.) in each group is expected. When a significance level (α) in unpaired t-test with a P value less than 0.05 (two-tailed) and a statistical power of 95% should be reached, then the difference between means that can be detected with group sizes of n = 5 or n = 10 are theoretically 64.76% and 42.74%, respectively. The respective values of this calculation are set in italics. The exemplary statistic calculation was performed with StatMate 2 for Windows, version 2.00 (GraphPad Software, Inc, La Jolla, CA, USA).
Practical implementation
Safety and storage
To avoid staff poisoning, the use of personal protective equipment is recommended whenever LPS is handled. To avoid chemical decomposition, LPS should be stored in a cool, dry and safe place. This location should only be accessible to authorized staff.
Preparations
The desired dose of LPS is dissolved either in sterile normal saline (0.9% NaCl) or in sterile phosphate-buffered saline (PBS) to a defined total volume per mouse (e.g. 100 µL). These solutions should be prepared before the experiment and stored in aliquots at –80℃. Control groups receive the same volume of vehicle. The solution should be at room temperature at the time of injection. The routine administration of anaesthesia is generally not recommended for small interventions such as IP injections as it would result in unnecessary stress to the animals and potentially introduce confounding xenobiotic effects. In addition, the novel European animal welfare rules require that only trained laboratory personnel should perform IP injections, ensuring speed and short overall animal handling times.
Please note that the biological activity of different LPS preparations may vary depending on the supplier and the bacterial source from which the LPS batches were prepared. Therefore, we recommend that the biological activity of the individual LPS batches should first be tested in small pilot experiments and the same LPS charge should be used during subsequent experiments.
Procedure
As handling mice (i.e. taking them out of their cage, immobilizing and exposing them to a foreign surrounding) is already stressful, the handler should be well-trained, calm and quick. During treatment, animals should be placed on a heating pad (e.g. HP062; Labotect, Göttingen, Germany) to guarantee an ambient temperature of 37℃ and to prevent hypothermia. Mice should be examined for signs of distress (e.g. changes in respiration or body temperature, rough hair coat, dull eyes, unusual behaviour, hunched posture) to check if the mice can be used for experiments. Mice showing abnormalities should be excluded from the study. Healthy mice should be weighed prior to each injection to accurately calculate the dosage of LPS. The required volume of LPS should be calculated. We recommend using LPS concentrations of between 1 and 20 mg/kg body weight. If a combination with D-GalN is applied, we recommend starting with concentrations of 0.1–5 mg/kg body weight (although even higher concentrations are used in many other protocols, cf. Table 1). For practical reasons, we propose a constant volume (e.g. 100 µL) should always be injected for both treatment and control groups. Therefore, it is useful to have a table listing the doses of LPS that must be diluted from a stock solution and filled up to the required injection volume for a range of different mouse weights. This enables the injection to be undertaken with little or no delay after the animal is weighed, thereby minimizing stress and unnecessary handling. The solution should be injected into the mice using disposable, sterile syringes. To this end, the mouse should be scuffed behind the neck region between the thumb and forefinger. After turning over the hand the animal should rest on the palm against the base of the thumb using a third finger to stabilize the pelvic region. The abdomen should be kept taut, with the head held lower than the rest of the body so that the intra-abdominal organs move towards the diaphragm. LPS should then be injected into the lower side of the abdomen (left or right lower quadrant of the abdomen, preferably the left lower quadrant) using a sterile 27 gauge needle (e.g. 27 G Microlance; Becton Dickinson, Franklin Lakes, NJ, USA). To avoid injury to the bladder, intestine or other intra-abdominal organs, aspiration is recommended before injection. After finishing the injections, all used syringes and needles should be disposed of safely in a sharps bin. Control mice should be injected with an identical volume of vehicle in the same manner as described above. Mice should be re-inspected one hour after injection for abnormalities and every 2 h thereafter. At the end of the experimentation (usually around 2 to 8 h after LPS injection) mice should be sacrificed, e.g. by cervical dislocation, and blood and liver (and other organs) should be collected for subsequent analyses.
Animal burden/side-effects
It is reported that LPS injection induces typical systemic inflammation and its thermoregulatory manifestations (i.e. fever). In addition, the experimental handling and the painful intravenous injection of LPS in the abdomen of the mice are stressful on their own. 39 Furthermore, an uncontrolled temperature during the injection procedure might interfere with the penetrance of LPS effects. 39 We therefore suggest avoiding stress factors such as rough transportation of cages and harsh removal of the animals from their cages. As already outlined, the animals should be placed on a heating pad during the procedures. In addition, each mouse should be well restrained so that it cannot move during the procedure and the needle should be inserted into the abdomen at about a 30 degree angle while the animal’s head is facing downward and its abdomen is exposed. This prevents traumatization of organs. Side-effects, especially unexpected ones, as well as the mortality rate should be reported in the ‘Methods’ section of any publication. In addition, the biological source and the provider of the LPS used for injections should be documented.
Classification of severity of procedure
According to Article 15 of the EU Directive 2010/63 (http://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:2010:276:0033:0079:en:PDF) the estimated degree of pain, suffering, distress or lasting harm of the animals subjected to LPS application should be classified following a scoring system that contain procedures to be classified as ‘non-recovery’, ‘mild’, ‘moderate’ or ‘severe’.
Details about the classification criteria that underlie this assessment have been established by the Expert Working Group on severity classification of scientific procedures performed on animals. These can be found at: http://ec.europa.eu/environment/chemicals/lab_animals/pdf/report_ewg.pdf.
The LPS application to induce liver failure with no major impairment of liver function is classified as a moderate procedure according to Article 15 of the above-mentioned EU Directive 2010/63. The induction of liver failure and death due to the typical complications of acute liver failure, such as cerebral edema and encephalopathy, coagulopathy, renal, hemodynamic and cardiorespiratory failure, is classified as a severe procedure. In line with the 3R principle of Russell and Burch, the procedure should undergo a refinement and humane endpoints must be implemented with frequent observation points to restrict pain, suffering, distress or lasting harm to the animals. Then the procedure can be reclassified as moderate.
Concluding remarks
Since the identification of the ‘endotoxin concept’ in 1904, the biological activities of bacterial LPS have been researched extensively. The receptors and the molecular pathways of LPS that promote the synthesis and secretion of proinflammatory cytokines, chemokines, ROS and many other types of mediators were identified. As LPS is largely responsible for most of the clinical manifestations of infections with Gram-negative bacteria, the application of LPS or mixtures of LPS with other hepatotoxins into mice or other rodents has become an attractive, highly reproducible experimental model for studying various aspects of endotoxemia, inflammation, septic shock and bacteremia-induced organ failure. With regard to experimental liver research, the application of LPS is well established, and many scientific issues could be resolved with this model. Many LPS-sensitive target genes have been identified over the last few decades that correlate well with the severity of the inflammatory response. Therefore, we recommend the use of this model when new anti-inflammatory drugs should be tested or involved pathways analyzed.
Footnotes
Ethical statement
All experiments were approved by the official State Animal Care and Use Committee (LANUV, Recklinghausen, Germany).
Funding
This work was supported by grants of the German Research Foundation (SFB/TRR57, projects P13 and P28).
