Abstract
Bone marrow transplantation in mice is performed by intravenous administration of haematopoietic repopulating cells, usually via the lateral tail vein. This technique can be technically challenging to carry out and may cause distress to the mice. The retro-orbital sinus is a large area where there is a confluence of several vessels that provides an alternative route for intravenous access. Retro-orbital injection, although aesthetically unpleasant, can be performed rapidly without requiring mechanical restriction or heat-induced vasodilation. In addition, this technique can be easily learned by novice manipulators. This route of administration has been reported for use in bone marrow transplantation but there is no comparison of retro-orbital and tail vein injections reported for this specific purpose, although both routes have been compared for many other applications. Here, we provide for the first time a comprehensive comparison between tail vein and retro-orbital injections for two different bone marrow transplant scenarios in P3B and B6D2F1 mice. In both cases, no significant differences regarding donor engraftment were observed between mice transplanted using each of the techniques. Haematological counts and leukocyte subpopulation distribution were practically identical between both animal groups. Moreover, donor engraftment levels were less homogenous when cells were transplanted by tail vein injection, probably due to a higher risk of failure associated with this technique. All these data suggest that retro-orbital injection is a compelling alternative to conventional tail vein injection for bone marrow transplant in mice, providing similar and more homogenous haematopoietic reconstitution.
The mouse, Mus musculus, is the most commonly used laboratory animal. One of the most common and efficient routes of administration or injection in mice is the intravenous (IV) route. 1 It can be used to administer cells, chemical agents, infectious disease agents, drugs and any other reagent that needs a systemic delivery.
IV vascular access is technically challenging in the adult mouse. The most common technique for IV delivery is tail vein (TV) injection, which requires practice and accuracy to be correctly performed. Mice have two superficial lateral tail veins, and a single artery along the ventral side of the tail. Intra-arterial administration is usually avoided because of potentially severe complications including blindness, cerebrovascular stroke, permanent motor deficits, and limb gangrene. 1 The lateral tail veins are therefore the route of choice in most situations. These veins are thin and prior warming up of the mouse is required to promote vasodilation. A very useful but not very popular alternative to this procedure is injection in the retro-orbital (RO) venous sinus of the mouse. The RO sinus is a large area with a substantial amount of blood flow in which several vessels meet, including the supraorbital vein, inferior palpebral vein, dorsal nasal vein and the superficial temporal veins. 2 An accurate description of the RO sinus injection technique in mice was lacking until a very detailed work was published in 2011. 2 In addition, some authors have compared these two routes in some specific situations. It has been demonstrated that the RO route can be used equivalently to the TV route for IV administration of monoclonal antibodies, cells and chemical agents.3,4 The authors did not find any differences, either in organ distribution or blood decay profiles, between the two groups. Moreover, in some cases the RO route has appeared to be superior to the TV route. Socher et al. found that the RO administration of contrast media for cardiac perfusion studies in mice using computed tomography provided better cardiac imaging results than TV injections. 5
Some researchers have also claimed that RO administration causes less stress to the animals, although it was not until 2008 that Steel et al. published some data to support this. The authors performed a direct comparison of TV and RO injections as IV drug delivery routes in mice. Although no differences were observed in the effects caused by the administered drug between the two routes, they found that, at least in BALB/c mouse strain, injection via the RO route was much less stressful than via the TV. In addition, they measured stress-related parameters, finding much lower stress-related scores in RO-injected mice than in TV-injected mice. 6
However, apart from the works mentioned above, there is still very little information about particular procedures using this route in the literature. One of these particular procedures is bone marrow transplantation (BMT).
The haematopoietic system follows a hierarchical organization in which haematopoietic stem cells (HSCs) are at the top of the pyramid. They are minimally present in the bone marrow (BM) and are characterized by their ability to generate the various cell types in the blood and by their self-renewal capability. Mouse haematopoietic progenitors are defined as a heterogeneous population with short- and long-term repopulation ability that do not express the typical lineage markers expressed by mature blood cells, so they can be defined as lineage negative (Lin–) cells. This Lin– cell fraction contains a population of cells expressing the Sca1 and c-kit markers known as Lin–, Sca1+ and c-Kit+ (LSK) cells, which correspond to the most primitive mouse HSCs. 7
BMT is a routine technique applied when studying the haematopoietic system. It involves the IV administration of haematopoietic repopulating cells into specific recipients. Interest in BMT goes beyond the basic knowledge of the haematopoietic system as it can be a therapeutic option for many haematopoietic and non-haematopoietic diseases. BMT in mice is usually performed by TV injection, and no comparisons of TV and RO routes have been published for this particular application. The only published data, to our knowledge, was produced in the context of xenotransplantation and conditioning studies for HSC transplantation. The authors found that limiting numbers of human HSCs could be transplanted by the RO route, reaching higher human chimerism levels than when an equal number of cells were transplanted by the TV route. 8 There are also some data comparing intra-femoral and RO administration of HSCs in the context of syngeneic BMT in mice, demonstrating that these two routes are also equally efficient. 9
Many authors would find it interesting to have another choice other than IV injection, but the lack of expertise and information in the literature about the actual rate of success and engraftment using the RO route for BMT might deter them. Evidence is still needed to determine whether the route of administration might have an effect on HSC engraftment or in different multilineage reconstitution. Thus, we present here an accurate comparison between TV and RO routes for BMT in mice. We demonstrate that RO injections can be superior in some circumstances and that, on balance, both routes can be considered as equivalent for BMT.
Materials and methods
Animals
B6.SJL-Ptprca/bPep3b/BoyJ (P3B 10 ); B6D2F1 and (B6.SJL-Ptprca/bPep3b/BoyJx-DBA/2) F1 (P3D2F1) colony founders were obtained from the Jackson Laboratory (Bar Harbor, ME, USA) and bred in-house for a maximum of five generations at the CIEMAT Animal Facility, Madrid, Spain (registration number ES280790000183).
The vavHS21/45ΔhCD4 (vavΔhCD4) transgenic mice were previously generated in the P3B strain (CD45.1+) in our laboratory. 11 These mice express a human CD4 truncated protein (ΔhCD4) which is incapable of intracellular signalling or of association with the major histocompatibility complex (MHC) class II in all cells of the haematopoietic system.
The mice were maintained under high standard conditions and were routinely screened for pathogens in line with Federation of European Laboratory Animal Science Associations (FELASA) recommendations for the health monitoring of mouse colonies in breeding and experimental units.12,13 The animals were maintained with HEPA-filtered air with an exchange rate of 16–20 changes per hour, and with regulated temperature of 20 ± 2℃ and humidity of 55 ± 10%. An artificial light/dark cycle of 13/11 h was followed with lights on at 07:00 h. They were fed a 20 Gy-irradiated standard diet (Teklad Global Diets 2914; Harlan Laboratories Inc, Indianapolis, IN, USA) and water ad libitum. The water was obtained from the human supply network, filtered through 5 microns, ultraviolet-irradiated, acidified at pH 2.5 and autoclaved. Dust-free and heat-treated soft wood pellets from the white poplar (Populus alba) was used as bedding (Souralit, Girona, Spain). The animals were housed five to six mice per group in polyphenylsulfone cages with a 435 cm2 area (Type 1145T; Tecniplast, Buguggiate, Italy) enriched with cellulose non-woven blocks (Nestlets; Bioscape, Castrop-Rauxel, Germany) and paper wool (Souralit, Girona, Spain).
All experimental procedures were carried out according to Spanish and European regulations (Spanish Royal Decree 53/2013 and Law 6/2013, which translate and comply with the European Directive 2010/63/UE about the use and protection of vertebrate mammals used for experimentation and other scientific purposes). At the end of the experimentation all animals were euthanized using carbon dioxide.
BMT studies
Two different BMT scenarios were used for the comparison of TV and RO routes. In the first study, 10 P3B female recipients (8 week old) with a mean body mass of 22 ± 3 g were lethally irradiated with two doses of 4.75 Gy spaced 24 h apart and were then transplanted with 3 × 106 total bone marrow (TBM) cells obtained from three 6-week-old vavΔhCD4 male mice with a mean body mass of 21 ± 3 g. Five mice were transplanted by RO injection (experimental group) and the other five by TV injection (control group). Peripheral blood (PB) was collected monthly and analysed for donor engraftment levels (determined by flow activated cell sorting [FACS] as the percentage of hCD4+ cells). Multilineage distribution was also determined by FACS as the percentage of B220+, CD3+ and Gr-1+/CD11b+ cells at three months post transplantation (mpt). Haematological counts were determined at 1 and 3 mpt. At 3 mpt, mice were sacrificed and BM cells were collected and analysed for donor engraftment in the total BM population and inside the LSK subpopulation.
In the second study, ten female B6D2F1 mice (8 week old) with a mean body mass of 24 ± 3 g were sublethally irradiated with one dose of 5 Gy and 24 h later they were transplanted with 1500 FACS-sorted LSK cells. These cells were obtained from three 6-week-old female P3D2F1 mice with a mean body mass of 25 ± 3 g. Five mice were transplanted by RO injection (experimental group) and the other five by TV injection (control group). The same analyses and the same timeline were applied here as the ones described for the first study; with the exception of donor chimerism level determination, which was performed on the basis of the percentage of CD45.1+ cells.
The irradiation conditioning was carried out using an X-ray equipment MG324 (300 kV, 12.8 mA; Philips, Hamburg, Germany) placed in the Animal Facility.
TV injections
TBM cells or FACS-purified LSK cells were IV transplanted through lateral TV injection. The mice were placed under a heat lamp to promote peripheral vasodilation and were then mechanically restrained using a home-made Plexiglas chamber. The tail was then kept horizontally straight to allow the manipulator to perform the injection. Syringes with 25-gauge needles were used for the TV administration of the cells. A maximum volume of 200 µL was injected. After manipulation the animals were returned to their cages for recovery.
Retro-orbital venous sinus injections
TBM cells or purified LSK cells were IV transplanted through RO sinus injection. The mice were anaesthetized using continuous inhalation of isoflurane (IsoFlo, Abbott Laboratories Ltd, Queenborough, Kent, UK; 4–5% as the induction dose and 1.5–3% as the maintenance dose in one LPM medicinal oxygen as the carrier gas) and a Plexiglas induction chamber. When the required depth of anaesthesia was reached, as indicated by the absence of the corneal reflex and toe pinch, the mouse was taken out of the chamber and placed in a left lateral recumbency position with its head facing to the right. This position was optimal for right-handed manipulators. Then, gentle pressure was applied to the right eye socket to partially protrude the mouse’s eyeball. A 31-gauge needle was carefully introduced, bevel down, at an angle of approximately 30°. A maximum volume of 200 µL was injected. When the needle was withdrawn, usually no bleeding was observed. When the injection was complete the mice were placed on a heating pad to ensure body temperatures of about 34℃, and were then placed back into their cages for recovery.
PB collection
PB collection was carried out monthly after transplantation as well as at the end of the experiments when the animals were culled. PB was mixed with 20 µL of ethylenediaminetetraacetic acid (EDTA) to prevent blood coagulation. For the PB periodical collection, mice were placed under a heat lamp to promote peripheral vasodilation and were then mechanically restrained using a home-made Plexiglas chamber. Then, after local administration of anaesthetic (EMLA [lidocaine/prilocaine]; AstraZeneca, Madrid, Spain), a small transverse cut was performed in the lateral TV with a sterile blade. Blood drops were collected with a micropipette and mixed with EDTA. A maximum volume of 100 µL of blood was obtained. After blood collection a slight pressure was applied to ensure that the bleeding would stop and the mice were put back into their cages for recovery.
When the animals were culled at the end of the experiment, PB was collected by cardiac puncture. Immediately after euthanization with carbon dioxide, the animals were placed in a right lateral recumbency position. The abdomen skin was moistened with 70% alcohol and then cut to visualize the abdomen wall. A one-millilitre syringe with a 25-gauge needle was slowly and perpendicularly inserted between ribs 5 and 6 and close to the sternum. After blood collection the needle was carefully withdrawn and cervical dislocation was performed to ensure that the animal was deceased. A maximum volume of 800 µL of blood was obtained.
BM cell collection
For the collection of the BM cells, mice were sacrificed and both tibiae and femora were surgically extracted. BM was harvested by flushing the tibiae and femora under sterile conditions with Dulbecco's modified Eagle medium + GlutaMAX (DMEM; Gibco/Life Technologies/Thermo Fisher Scientific, Waltham, MA, USA) and washed twice with phosphate buffered saline (PBS; Sigma-Aldrich, St Louis, MO, USA).
Haematological counts
To determine haematological counts, 50 µL of EDTA-anticoagulated PB were analysed using an Abacus Junior Vet Haematological Analyser (CVM, Navarra, Spain).
Flow cytometry analyses
Forty microlitres of PB samples or 5 × 105 BM cells were stained with different moAbs, listed in Supplementary Table 1 (see Appendix), over 30 min at 4℃. After that, erythrocytes were lysed in ammonium chloride lysis solution (0.155 mmol/L NH4Cl + 0.01 mmol/L KHCO3 + 10−4 mmol/L EDTA). The stained cells were resuspended in flow cytometry buffer (PBS containing 0.5% BSA and 0.05% Azide) containing 1 µg/mL 4',6-diamidino-2-phenylindole (DAPI) as a viability marker and analysed in a BD LSRFortessa™ Cell Analyser (BD Biosciences, San Jose, CA, USA). A minimum of 105 viable cells were acquired. Off-line analysis was performed using the FlowJo software package (TreeStar Inc, Ashland, OR, USA).
LSK sorting
HSCs with an LSK phenotype were purified from BM samples. The BM cells were lysed and stained for LSK phenotype using the moAbs listed in Supplementary Table 1 (see Appendix) and FACS-sorted using a BD Influx (BD Biosciences).
Statistical analyses
The statistical analysis was performed using GraphPad Prism Software version 5.00 (GraphPad Software, San Diego, CA, USA) for Windows. For all the analyses a non-parametric Mann–Whitney test was performed. Significances are expressed as P < 0.05(*).
Power analyses was performed using DSS Research online tool (see https://www.dssresearch.com/KnowledgeCenter).
Results
To perform a direct comparison between the two selected routes for IV injection in the context of HSC transplantation we first performed a classical BMT in congenic mice. A scheme of the procedure can be observed in Figure 1a. vavΔhCD4-P3B mice (CD45.1+ΔhCD4+)
11
were used as donors while P3B mice (CD45.1+ΔhCD4–) were used as recipients. A total of 3 × 106 TBM cells were administered by either RO or TV injection into lethally-irradiated P3B recipients. Similar volumes were used for both injections in order that exactly the same conditions were achieved.
Engraftment analyses in TBM transplants. A total of 3 × 106 vavΔhCD4-P3B TBM cells (CD45.1+ΔhCD4+) were transplanted into lethally-irradiated P3B mice (CD45.1+ΔhCD4–). Donor engraftment was determined at 1 and 3 mpt in PB, and at 3 mpt in BM when the animals were culled by the FACS analyses of the ΔhCD4+ population. (a) Scheme of the transplantation experiment. (b) PB donor engraftment. (c) Donor engraftment obtained in recipients’ BM at 3 mpt. (d) Donor LSK cell engraftment obtained in recipients’ BM at 3 mpt. (e) Lineage distribution of donor engraftment in recipients’ PB. (f) PB haematological counts at 3 mpt. Significance of differences between groups are expressed as P < 0.05(*). BM: bone marrow; FACS: flow activated cell sorting; LSK: Lin–, Sca1 +, c-Kit+; mpt: months post transplant; n.s.: not (statistically) significant; PB: peripheral blood; PLT: platelets; RBC: red blood cells; RO: retro-orbital; TBM: total bone marrow; TV: tail vein; WBC: white blood cells.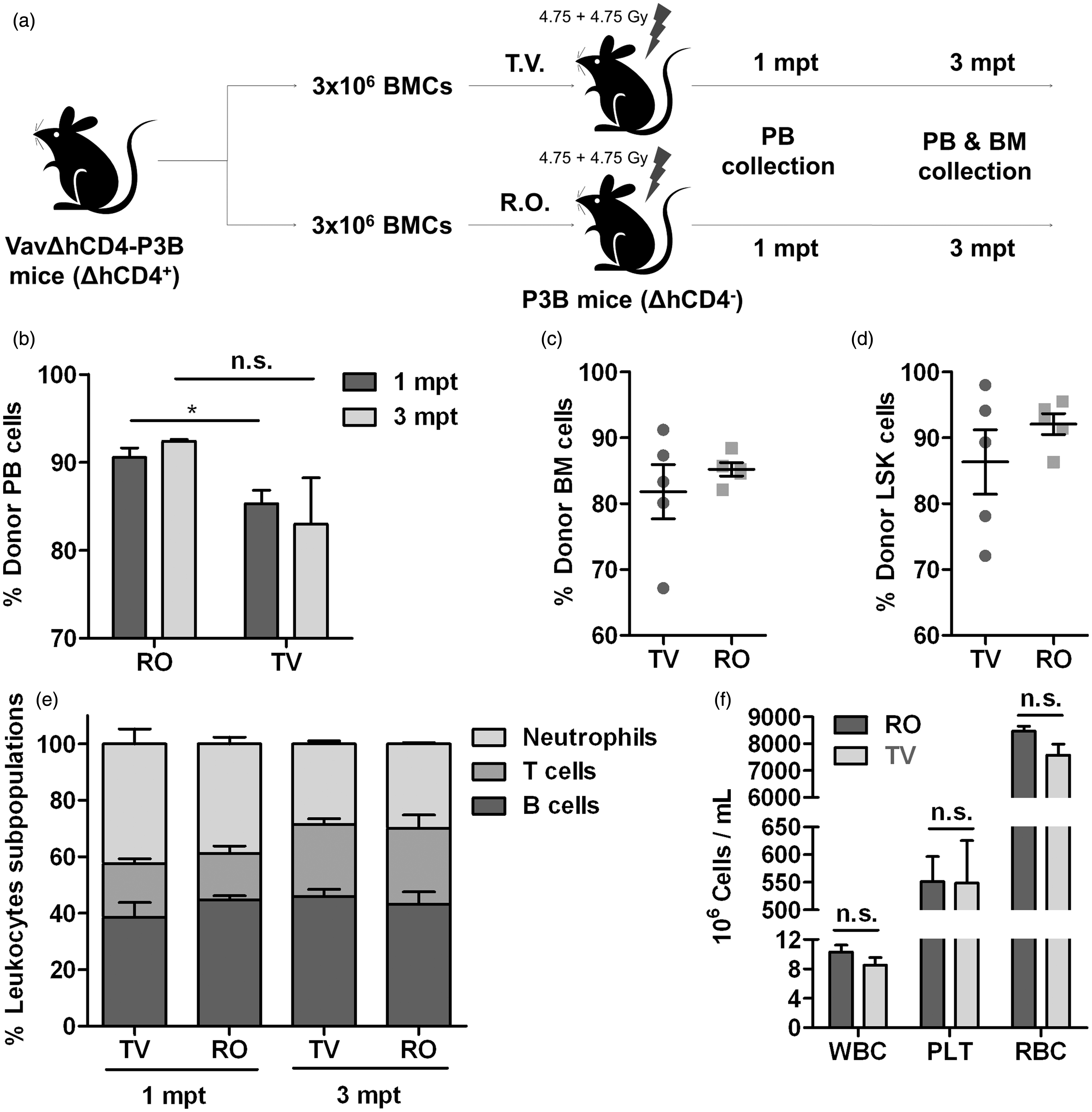
After transplantation, the recipient mice were analysed to assess donor PB engraftment. The analyses revealed that, although almost full donor chimerism was achieved in all cases, RO-administered TBM cells provided significantly higher levels of engraftment than TV-administered cells both at 1 mpt (90.56 ± 2.42% vs. 85.32 ± 3.38%) and 3 mpt (92.4 ± 0.46% vs. 82.98 ± 11.78%) (Figure 1b). When BM was analysed at the endpoint of the experiment (3 mpt), these differences were not significant, as donor chimerism in both TBM cells and LSK subpopulations was similar in both groups (Figures 1c and 1d). However, a more homogeneous distribution of engraftment levels in the RO-injected group than in the TV-injected group was found, as shown by the coefficient of variation (0.50 vs. 14.20) (Figure 3a). Therefore, in a BMT study, similar PB and BM engraftment levels can be obtained with both RO and TV routes. These results were confirmed when haematopoietic reconstitution was studied. No significant differences were observed in the distribution of B cells, T cells or neutrophils between both groups (Figure 1e), or in the absolute numbers of leukocytes, erythrocytes and platelets (Figure 1f).
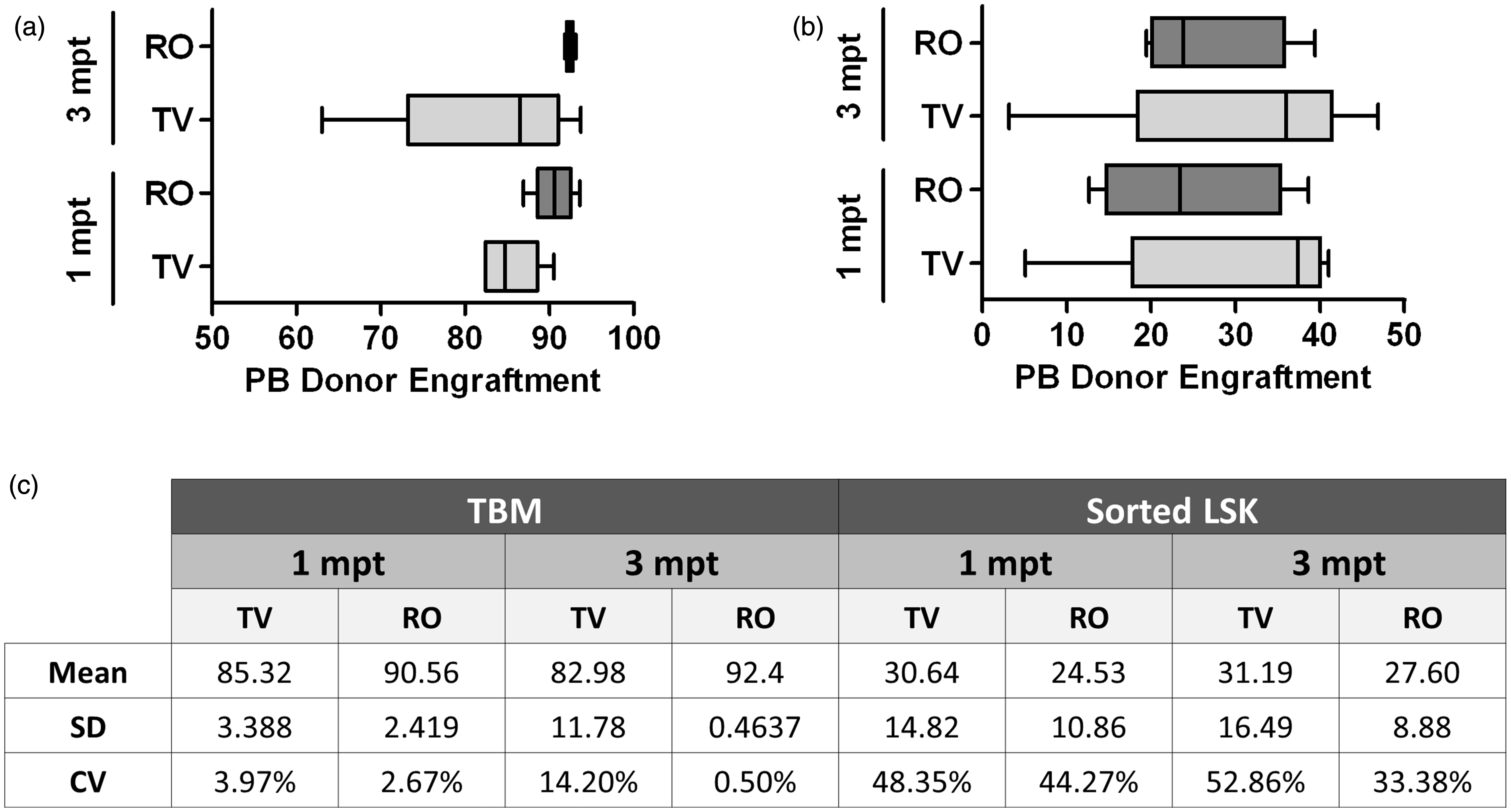
Engraftment analyses in LSK cells transplants. A total of 1500 P3D2F1 LSK cells (CD45.1+CD45.2+) were transplanted into submyeloablated B6D2F1 (CD45.1–CD45.2+) recipients. Donor engraftment was determined at 1 and 3 mpt in PB and at 3 mpt in BM when the animals were culled by the FACS analyses of the CD45.1+ CD45.2+ population. (a) Scheme of the transplantation experiment. (b) PB donor engraftment. (c) Donor engraftment obtained in recipients’ BM at 3 mpt. (d) Donor LSK cell engraftment obtained in recipients’ BM at 3 mpt. (e) Lineage distribution of donor engraftment in recipients’ PB. (f) PB haematological counts at 3 mpt. BM: bone marrow; FACS: flow activated cell sorting; LSK: Lin–, Sca1+, c-Kit+; mpt: months post transplant; n.s.: not (statistically) significant; PB: peripheral blood; PLT: platelets; RBC: red blood cells; RO: retro-orbital; TV: tail vein; WBC: white blood cells. Data homogeneity. (a) Box and whisker plot distribution of donor engraftment values (ΔhCD4+ cells) in PB deduced from the analysis of P3B recipient mice at 1 and 3 mpt. SD, median and range values (central line in boxes) are shown. (b) Box and whisker plot distribution of donor engraftment values (CD45.1+ CD45.2+ cells) in PB deduced from the analysis of B6D2F1 recipient mice at 1 and 3 mpt. SD, median and range values (central line in boxes) are shown. (c) Mean, SD and CV values for donor engraftment data obtained in PB at 1 and 3 mpt for both transplantation experiments. LSK: Lin–, Sca1+, c-Kit+; mpt: months post transplant; TBM: total bone marrow; PB: peripheral blood; RO: retro-orbital; TV: tail vein; SD: standard deviation; CV: coefficient of variation.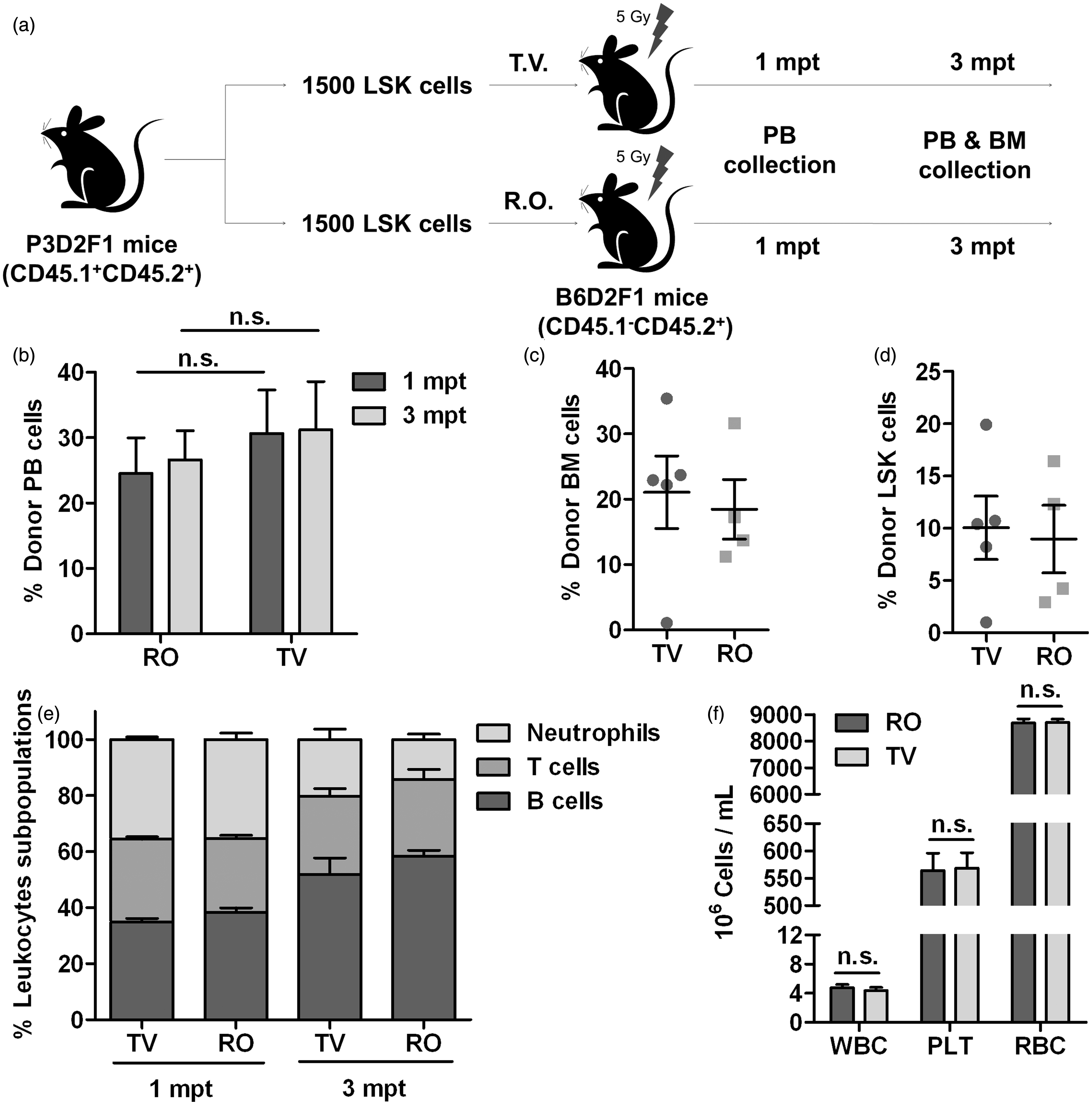
A power calculation analysis based on data from the analyses of the first month showed adequate power (0.94) with an alpha error level of 5% by using five mice in each group.
In BMT studies it is not always possible to treat recipients with a high conditioning regimen or to have a high number of cells available when using defined progenitors rather than TBM cells. To establish a different approach we decided to perform a BMT experiment in which conditions were not ideal so we could infer whether or not both administration routes were good under more restraining conditions. LSK cells from P3D2F1 mice (CD45.1+CD45.2+) were FACS-purified and transplanted in a limiting number (1500 cells per mouse) into sublethally-irradiated B6D2F1 mice by either RO or TV injection (Figure 2a). Similar volumes (200 µL) were used for both injections. After transplantation the recipient mice were analysed to assess PB engraftment using the CD45.1 and CD45.2 pan-leukocytic markers up to 3 mpt.
Donor chimerism level analyses revealed that LSK cells administered by RO and TV routes provided similar levels of engraftment at 1 and 3 mpt (RO engraftment levels: 24.53 ± 10.86% and 32.15 ± 10.82%, TV engraftment levels: 30.64 ± 14.82% and 37.44 ± 18.7%; Figure 2b). When BM was analysed at the end of the experiment, donor chimerism in both TBM cells and LSK subpopulations was also similar for both groups (Figures 2c and 2d). Although, again, a more homogeneous distribution of data was observed in the RO group (Figure 3b). These results were confirmed when haematopoietic reconstitution was studied in the recipients (Figure 2e). No significant differences were observed in either the distribution of B cells, T cells or neutrophils between both groups or in the absolute numbers of leukocytes, erythrocytes and platelets (Figure 2f).
Taking into account all these results, we can conclude that there are no significant differences between the engraftment levels obtained both in PB and BM with both IV injection routes. However, it is worth mentioning the higher homogeneity obtained in the results for the RO-injected mice in comparison with the TV-injected group, as shown by the values of the standard deviation and the coefficient of variation (Figure 3). In addition, no adverse effects were detected in any experiment.
Discussion
BMT is a procedure intended to replace damaged or ablated BM with healthy cells. This procedure may be used with different purposes in experimental mice: the study of different functions of haematopoietic and non-haematopoietic cells, studies of haematopoietic system regulation or ex vivo gene therapy protocols,14–16 among others.
A BM graft is delivered through the IV route. In mice the TV has usually been the most common choice.11,14–17. Other sites for delivery include the RO venous sinus,18,19 the BM cavity itself,20–22 and the spleen.23–25 The splenic route is not commonly used for delivering HSCs as it offers a very low rate of success in repopulating recipient BM in comparison with the other routes. The intra-bone delivery of HSCs is meant to increase the possibility of cells homing in the BM of the host, as they are delivered directly into the target tissue, thus avoiding homing through circulation. However, this is a very difficult procedure that requires expert manipulators, deep anaesthesia of the mice and a much reduced injection volume (up to 30 µL). Theoretically, one true HSC is sufficient for long-term BM engraftment. However, additional BM cells are required to ensure the survival of the mice in the early stages after transplantation as HSCs require time to engraft and differentiate into the different lineages. These support cells have to be co-administered, thus increasing the total volume necessary for inoculation. RO injection is easier to perform, but it requires the mouse to be anaesthetized and, as very little data are available regarding reconstitution after BMT, the route of choice is usually the TV.
In preparation for TV venepuncture, the mouse must be mechanically restrained and placed under a heat lamp to promote peripheral vasodilation. This approach can cause distress to the animals, especially if the initial venipuncture is unsuccessful and repeated attempts are made. If the needle is not correctly inserted inside the vein, cells will be delivered into the surrounding subcutaneous tissue, resulting in skin swelling. Also, in white mice the veins are generally easy to detect as dark lines along either side of the tail, but in brown or black mice it is much more difficult to detect these lines. Of course, all of these drawbacks are minimized in the appropriate expert hands but they highlight the need for extensive training and expertise to carry out this procedure with accuracy and reproducibility.
RO injection may seem aesthetically distasteful, but it is an easily-mastered technique and is ultimately more humane than the alternative IV injection technique in mice. It is easier and faster to perform as a mouse can be injected in as short as 15 s. Of course, it also has some drawbacks: the maximum volume injectable through this route can be a bit lower than through the TV, injection close to the eye-ball can be disagreeable for some people, and there is the possibility that some cells or reagents might not be delivered in this situation.
In all these methods, manipulator skills are very important and can have great impact on the final results, even determining the difference between the success and failure of the graft. Thus choosing an easy procedure can lead to more reproducible and homogenous results.
Taking into account all of these considerations we have tried to perform a deep characterization of the RO injection-mediated BMT in mice. Two different approaches of syngeneic BMT were chosen: transplantation of high numbers of TBM cells and transplantion of limiting numbers of purified HSCs (defined as LSK cells). In both approaches we analysed engraftment levels, lineage distribution and haematological counts, trying to give an overall vision of the procedure.
We found that both RO and TV routes provided an equal haematopoietic reconstitution, as similar engraftment levels, lineage distribution and haematological counts were achieved. If anything, the RO route could provide higher levels of engraftment, as shown in the PB analyses of TBM-transplanted mice (Figure 1b). Remarkably, the level of variability among the different engraftment methods was lower in the RO-injected group (Figures 1c, 1d, 2c, and 2 d). Both by the representation of PB engraftment in a box and whisker plot (Figures 3a and 3b) and by the calculation of the standard deviation value and the coefficient of variation (Figure 3c), we can clearly observe that the distribution of the results is much more homogeneous among the RO-injected mice than among the TV-injected ones, independent of the experiment type.
In conclusion, we have demonstrated that RO injection is a good alternative as an IV route for BMT in comparison with the more commonly used TV injection. It can reduce the time required for manipulation of the mice, thus reducing mouse and manipulator distress. The shortening of the procedure time can also be beneficial as cells will be in tubes for a shorter period prior to injections. As mentioned above, other studies have been published comparing the TV and RO administration routes that show similar results when delivering drugs, antibodies or cells in similar situations.2–6,9 Also, in our hands, RO administration has shown greater consistency regarding donor chimerism levels due to the ease of the technique and the lower rate of failure.
Altogether, a higher homogeneity of data, equal or even higher engraftment values, and lower rates of failure offer significant improvement that could allow for a reduction in the number of animals used in a BMT experiment. Moreover, although both techniques cause mild suffering, RO injection is much easier to learn which in turn implies a reduction in the distress and suffering of the mice as it is performed under anaesthesia. All of these considerations will eventually satisfy the principle of the 3Rs (replacement, reduction and refinement).
Footnotes
Acknowledgements
The authors would like to thank Aurora de la Cal, Sergio Losada and María Carmen Sánchez for their collaboration in the administrative work, and Miguel Angel Martín and Edilia de Almeida for their careful maintenance of the animals.
Declaration of conflicting interests
The authors declare no conflict of interest.
Funding
This work was supported by the following grants: Dirección General de Investigación de la Comunidad de Madrid (CellCAM; S2010/BMD-2420), Fondo de Investigaciones Sanitarias, Instituto de Salud Carlos III (RETICS-RD12/0019/0023) and Ministerio de Economía y Competitividad (SAF2012-39834). DLR was supported by a Ministerio de Educación FPU grant (AP-2009-4129).
