Abstract
Conventional genotoxicity tests are technically difficult to apply to oocytes, and results obtained on somatic cells cannot be extrapolated to gametes. We have previously described a comet assay (original–CA) on denuded mouse oocytes, but, in vivo, oocytes are not isolated from their surrounding follicular cells. Our objective was to develop a comet assay on cumulus–oocyte complexes (COC–CA) for a more physiological approach to study the genotoxicity of environmental factors on oocytes. For COC–CA, whole COC were exposed directly to exogenous agents after ovulation and removal from oviducts. Three conditions were studied: a negative control group, and two positive control groups, one of which was exposed to hydrogen peroxide (H2O2) and the other group was incubated with cerium dioxide nanoparticles (CeO2 NPs). With both tests, DNA damage was significant in the presence of both H2O2 and CeO2 NPs compared with the negative control. COC–CA offers an interesting tool for assaying the genotoxicity of environmental agents towards germinal cells. Furthermore, COC–CA is less time-consuming and simplifies the protocol of the original–CA, because COC-CA is easier to perform without the washing-out procedure.
Conventional genotoxicity tests such as micronucleus test, comet assay (CA) or Ames test1–3 are technically difficult to apply to oocytes, and results obtained with somatic cells cannot be extrapolated to gametes. 4 In ovaries, follicle and oocyte development is dependent on communication between oocyte and somatic cells of cumulus–oocyte complexes (COC), thanks to paracrine pathways. 5 CA is a well-validated technique for assessing low-level DNA damage in individual somatic cells. 6 Regarding oocytes, only a few studies have used CA to assess DNA damage in mammalian oocytes.7–15 In a previous work, we adapted CA to denuded mouse oocytes with zona pellucida (ZP). 12 However, in vivo, oocytes in ovaries are dependent on COC and are not isolated from follicular cells. Our objective was to adapt the CA for a more physiological approach for studying the genotoxicity of environmental factors on oocytes.
Materials and methods
Reagents used were from Sigma Aldrich (Saint Quentin, Fallavier, France), and prepubescent female CD1 mice were from Charles River Laboratory (L’Arbresle, France). IRB approval (No. 12-18042012) was obtained from the National Ethics Committee on Animal Experimentation. Metaphase II oocytes were collected after superovulation and mouse euthanasia, as described previously.12
Exposure conditions
Two CAs were compared: (i) the original CA (original–CA), as previously described, with denuded oocytes,
12
and (ii) the CA on COC (COC–CA), as described below. Two agents inducing DNA damage toward mouse oocytes (already described with the original–CA) were used to validate the COC–CA: hydrogen peroxide (H2O2) and cerium dioxide nanoparticles (CeO2 NPs).
16
Three conditions were studied:
Negative control group: denuded oocytes (original–CA)/COC (COC–CA) incubated 2 h in M16 medium (37℃, 5% CO2). H2O2 positive control group: after incubation over 2 h in M16 medium (37℃, 5% CO2), oocytes were placed in 250 mmol H2O2 solution for 5 min at 4℃. For COC–CA, hyaluronidase was added after incubation with H2O2. CeO2 NP group: incubation with CeO2 NPs at 100 mg/L for 2 h in M16 medium (37℃, 5% CO2).
At least 40 denuded oocytes for the original–CA and 40 COC for the COC–CA were used for each condition; all conditions were carried out in triplicate.
Alkaline CA on COC
The original–CA was performed as previously described by Berthelot-Ricou et al. 12 For the COC–CA, modifications of the original–CA were performed at the stage where hyaluronidase was used to separate the oocyte from their surrounding follicular cells. For the original–CA, hyaluronidase (10 mg/L over 10 min) was added before exposure. Then, oocytes and follicular cells were washed out to remove hyaluronidase and were incubated separately with the tested agents before the CA. For COC–CA, COC were removed from the oviducts, then incubated immediately with the tested agents. After incubation, hyaluronidase was added to separate the oocyte from the follicular cells, and the CA was performed immediately without the washing-out step.
Main outcome measures and statistical analysis
Examination was performed after propidium iodide staining using a fluorescence microscope (X200, Olympus BX-60; Olympus, Rungis, France). Pictures were obtained with Visilog software version 6.7 (Noesis SA, Saint Aubin, France). DNA damage was assessed with olive tail moment (OTM) analysed by Komet software (version 5.5; Andor Bioimaging, Nottingham, UK). OTM was defined as the product of the tail length by DNA% contained in the tail of the comet. 1 An analysis of variance (ANOVA) analysis was performed followed by Fisher LSD post hoc test using Statview® 5.1 for Windows (Abacus Concepts, Berkeley, CA, USA). For each condition, results were expressed as mean OTM values ± SEM. Results were considered significant at P < 0.05.
Results
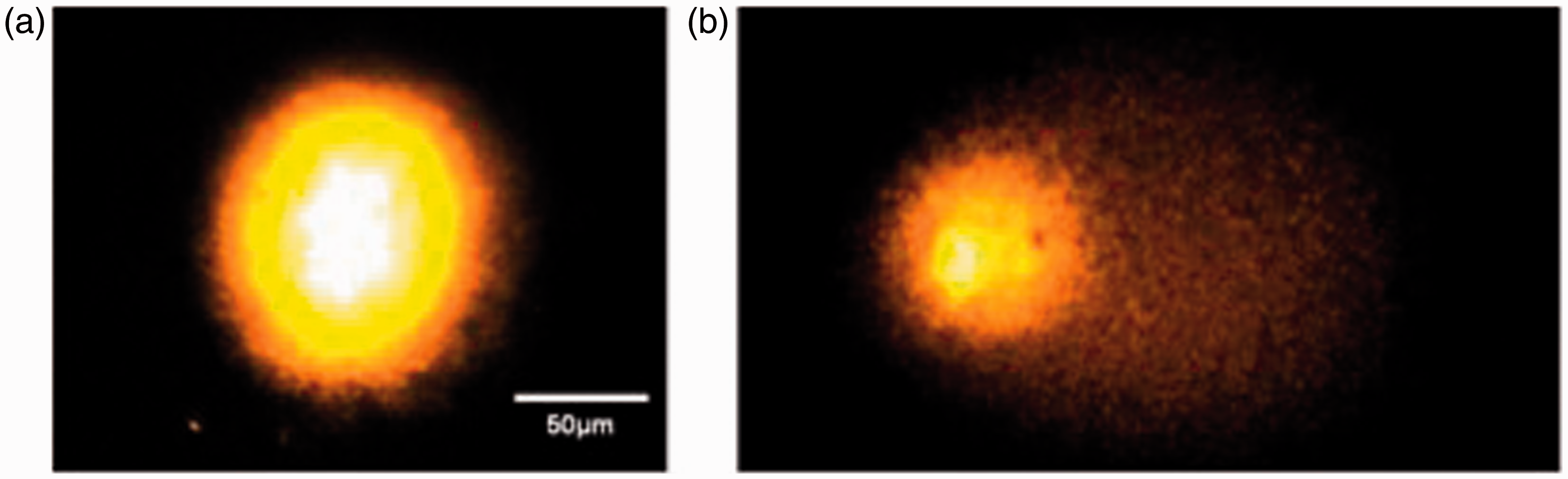
An average of 100 oocytes were analysed by condition. With the original–CA (Figure 1a), significant DNA damage was observed following H2O2 (OTM = 38 ± 1.3) and CeO2 NP (OTM = 11.6 ± 0.8) exposures compared with the negative control (OTM = 2.9 ± 0.2). With COC–CA (Figure 1b), significant DNA damage was observed following H2O2 (OTM = 36 ± 1.3) and CeO2 NP (OTM = 20 ± 1.6) exposures compared with the negative control (OTM = 5.4 ± 0.6). Figure 2 shows that comet pictures obtained with COC–CA looked the same as those obtained with the original–CA.
Genotoxicity assessment with the original comet assay (a) and the comet assay performed on cumulus–oocyte complexes (b). *P ≤ 0.05. OTM: olive tail moment; CeO2 NP: cerium dioxide nanoparticles. Images obtained with comet assay on cumulus–oocyte complexes for negative control group (a) and positive control group (b).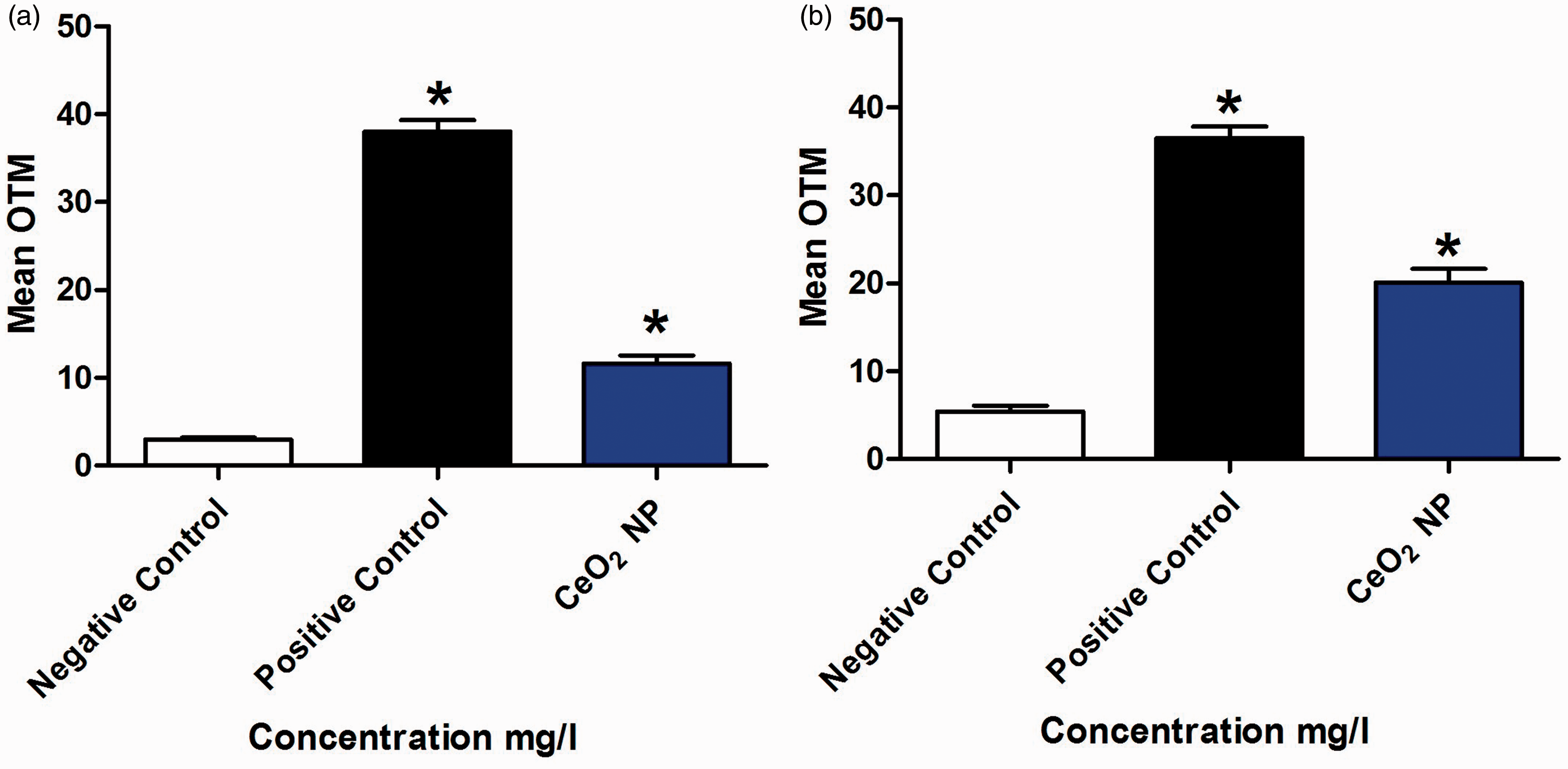
Discussion
Summary of different studies using comet assay on mammalian oocytes.
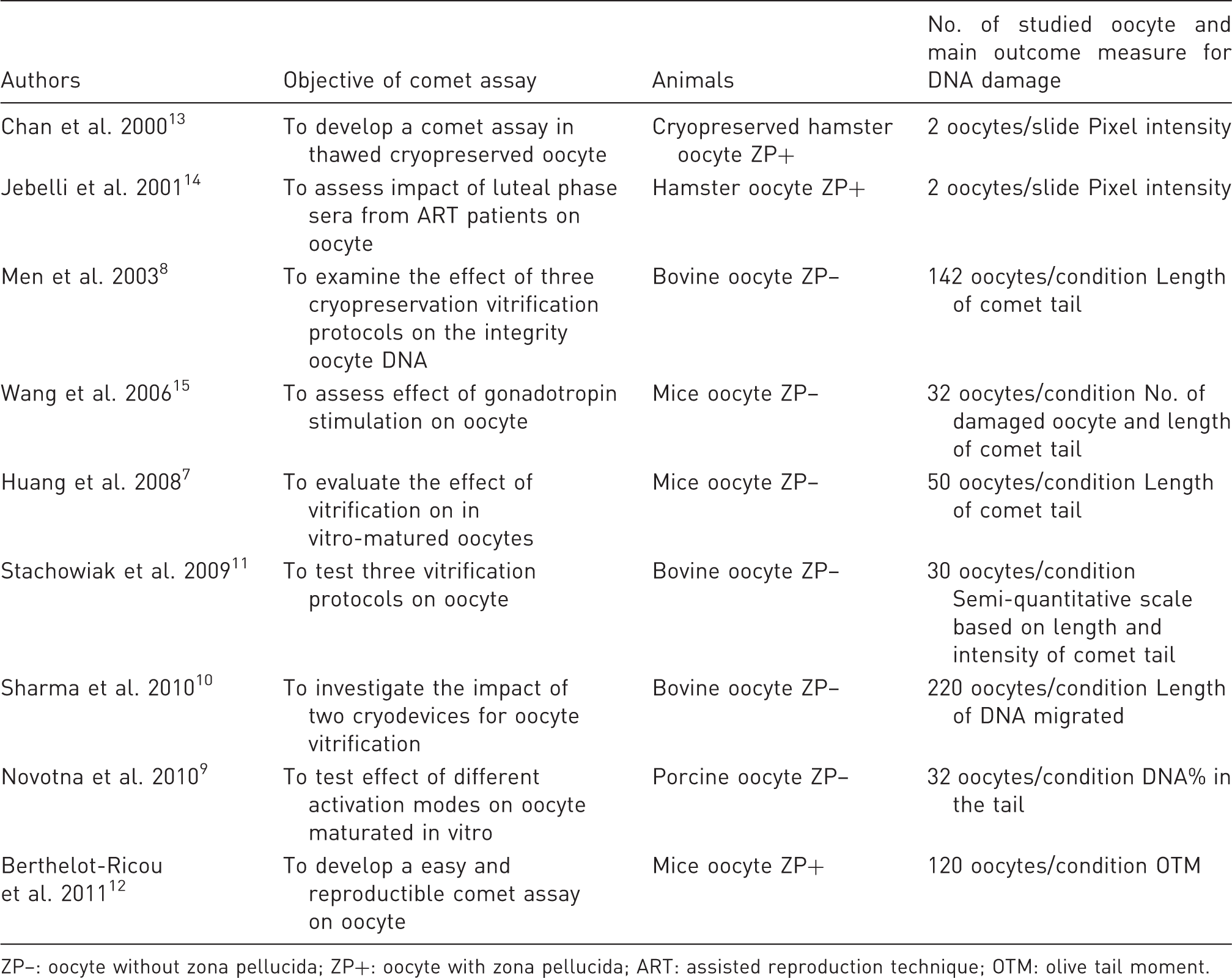
ZP–: oocyte without zona pellucida; ZP+: oocyte with zona pellucida; ART: assisted reproduction technique; OTM: olive tail moment.
Footnotes
Declaration of competing interests
FG was awarded a scholarship from the ‘Foundation, health, sport and development’, Aix Marseille University for a master's degree. JP, MA, TO, and BC have nothing to disclose.
Funding
This work is a contribution to the LABEX SERENADE (No. ANR-11-LABX-0064) funded by the French Government program of the French National Research Agency (ANR) through the A*Midex project (No. ANR-11-IDEX-0001-02).
