Abstract
The collection of cerebrospinal fluid is necessary in order to determine its composition. It can then be used to diagnose various diseases. The aim of the study was to develop and optimize a technique for performing safe centesis for the collection of cerebrospinal fluid in piglets and its injection through the cisterna magna. The study was divided into three phases: (1) anatomical study of cadavers, (2) in vivo application of the technique and (3) observation of recovery time. The proposed technique resulted in a safe puncture of the cisterna magna. The authors identified and confirmed the correspondence of the crista occipitalis and the wings of the atlas with the external landmarks on the cadaver by means of direct radiological visualization. The punctures were performed successfully at the first attempt in 11 out of 12 anaesthetized piglets. The technique herein described provides a reproducible safe and easy route for approaching the cisterna magna for cerebrospinal fluid collection, drug administration and gene delivery.
The cerebrospinal fluid (CSF) puncture technique has been described in several animal models: rats, 1 rabbits, 2 guinea pigs, 3 primates 4 and adult pigs. 5 Moreover, the intrathecal delivery of drugs, or other compounds, into the CSF is a convenient method for targeting the central nervous system (CNS). By so doing, the obstacle of crossing the blood brain barrier (BBB) following systemic administration and unwanted exposure of the visceral organs and involvement of the renal and hepatic metabolisms was avoided.4,6,7 In pigs, the injection of drugs or substances has been used to experimentally reproduce human diseases8–10 and to study delivery methods for widespread gene transfer to the CNS.11,12
In order to develop a specific and reliable centesis/injection technique in newborn piglets (which have size- and age-dependent anatomical specificities), a fast reproducible sampling and compound administration technique with minimal side-effects is necessary.
The aim of the study was to develop and optimize a technique for performing safe centesis in piglets for the collection of CSF and its injection through the cisterna magna (CM).
The experiment (in vivo study) was conducted in accordance with the provisions of European Economic Community (EEC) Council Directive 86/609 adopted by the Italian Government (DL 27/01/1992 No. 116). The total number of animals used was 18: 12 female and 6 male large white (LW) crossbreed piglets (Az. Agricola Pasotti SS, Imola, Italy); (age range: 2–30 days; weight range: 0.8–10 kg).
The study was divided into three phases:
anatomical study of cadavers in vivo application of the technique observation of recovery time
Six LW female piglet cadavers (age range: 2–30 days; weight range: 1–10 kg) were used for this phase of the experiment.
After hand palpation and identification of the crista occipitalis (occipital protuberance), a surgical pen (Viscot Medical LLC, East Hanover, New Jersey, US) was used to draw a line caudally along the spine extending from the occipital protuberance. This was done in order to identify the median spinal line. A second line was traced between the cranial margins of the right and left wings of the atlas. The spinal needle (22 gauge × 75 mm, Pic indolor, Artsana, Grandate, Italy) was introduced along the median line to a depth of 4 mm, 5 mm posteriorly to the intersection of the two lines; the tip of the needle was directed cranioventrally using the cranial margin of the wings as an external landmark. The cadaver was placed in right lateral recumbency without any flexion of the head throughout the entire procedure. The centesis of the first female cadaver (8 kg) was performed using C-arm X-ray (Technix SpA, Grassobbio, Italy) to evaluate the correct correspondence between the anatomical and the suggested external landmarks; then the centesis was performed without any radiological guidance for the remaining five female cadavers which were also always placed in right lateral recumbency without any flexion of the head. Small adjustments regarding the insertion site (range 4–7 mm caudally to the intersection of the two lines) and the depth of the needle placement (range: 4.5–7 mm) were required according to the varying size of the cadavers; 2.5 mL of radiopaque contrast medium was injected for radiological evaluation to verify the correct distribution of the radiopaque contrast medium.
Six male piglets (age range: 2–30 days; weight range: 1.2–8 kg) were used in the in vivo study.
General anaesthesia was induced and maintained with sevoflurane (SevoFlo; Abbott Laboratories, Chicago, IL, USA). Non-invasive blood pressure, heart rate, electrocardiogram measurements, hemoglobin saturation, respiratory rate, end tidal CO2 and rectal temperature were monitored.
The piglets were positioned as reported in Phase I. The collection site (5 × 5 cm) was clipped and surgically prepared. The lines described in Phase I were drawn using a sterile surgical pen (Figure 1), and puncture of the CM using a 75 mm 22 gauge spinal needle was carried out. The CSF collected (range: 1–2.5 mL) was replaced with an equal volume of radiopaque contrast medium, and a radiographic examination was performed. At the end of the procedure, the piglets were sacrificed.
The external landmarks (CO: crista occipitalis; AW: atlas wings) and the lines drawn on the piglet skin in order to identify the correct site for carrying out the centesis with the spinal needle. (a) lateral view; (b) dorsal view. The photo was obtained with the piglet in sternal recumbency (in the study, the centesis was performed with the animals in lateral recumbency) in order to achieve better visualization of the landmarks.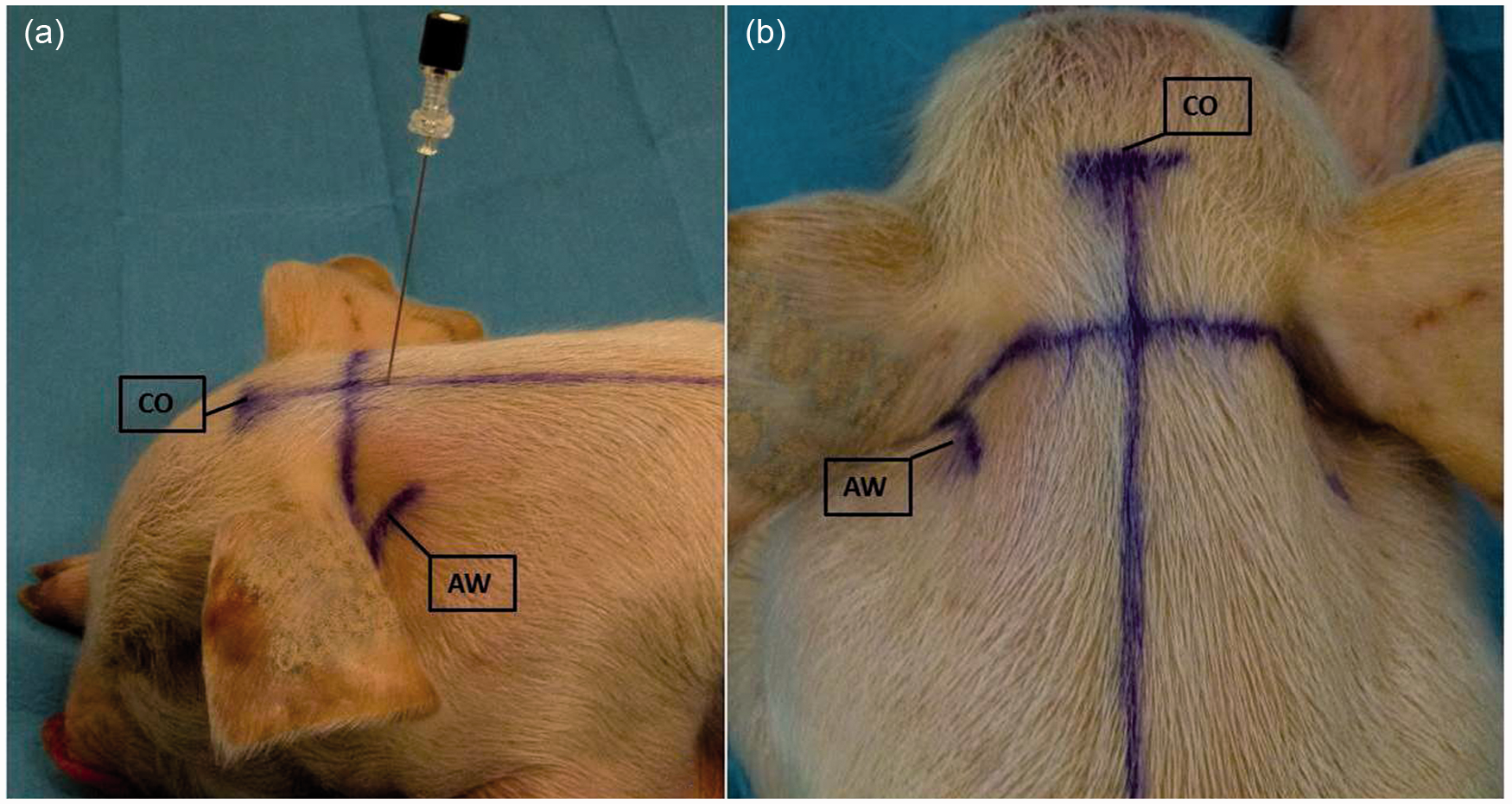
Moreover, six female piglets (age range: 2–5 days; weight range: 0.8–1.8 kg) were used to observe the recovery periods. All the animals were anaesthetized as in the preceding phase. Only two piglets (control animals) were anaesthetized; in the other four animals, the centesis of the CM was carried out using the above-mentioned technique; a one-millilitre liquor sample was collected, and one millilitre of saline solution was injected. The recovery time and neurological alterations were recorded.
The technique described allowed the puncture of the CM to be carried out. In Phase I, the authors identified and confirmed the correspondence of the crista occipitalis and the wings of the atlas with the external landmarks on the first cadaver by direct radiological visualization; the correct orientation of the needle was also tested and verified. After drawing the lines, the spinal needle was successfully placed into the CM of four out of five animals at the first attempt. In the last case, the spinal needle was oriented too caudally, and so encountered the bone surface. The operator had to reinsert the needle with a more cranial orientation in order to carry out the CM puncture.
In all cases, the radiopaque medium confirmed the correct placement of the spinal needle. The contrast medium was identified by a sharply marginated thin column in the subarachnoid space up to the seventh cervical space; it was clearly present in the cranial cavity. First step of Phase II was the induction of general anaesthesia which was rapidly accomplished in about 2 min. Intubation was challenging in the newborns due to the particular anatomy of the laryngeal inlet. In Phase II of the study, the punctures were carried out successfully in five out of six piglets at the first attempt. In one subject, the operator accidentally drew a paramedian rather than a median line, thus making it impossible to carry out the puncture; it was necessary to draw a second line to obtain the correct landmark and the correct puncture of the CM. When the collection of the CSF was begun, the fluid showed mild blood contamination in five out of six animals due to accidental puncture of the dorsal venous plexus. In each case, the radiopaque medium confirmed the correct placement of the spinal needle (Figure 2). The cranial diffusion of the contrast medium was observed in six out of six anaesthetized animals.
Contrast radiography of the head and cervical spine, lateral view (anaesthetized piglet): the contrast medium diffusing along the cervical tract of the spinal cord is clearly present in the cranial cavity.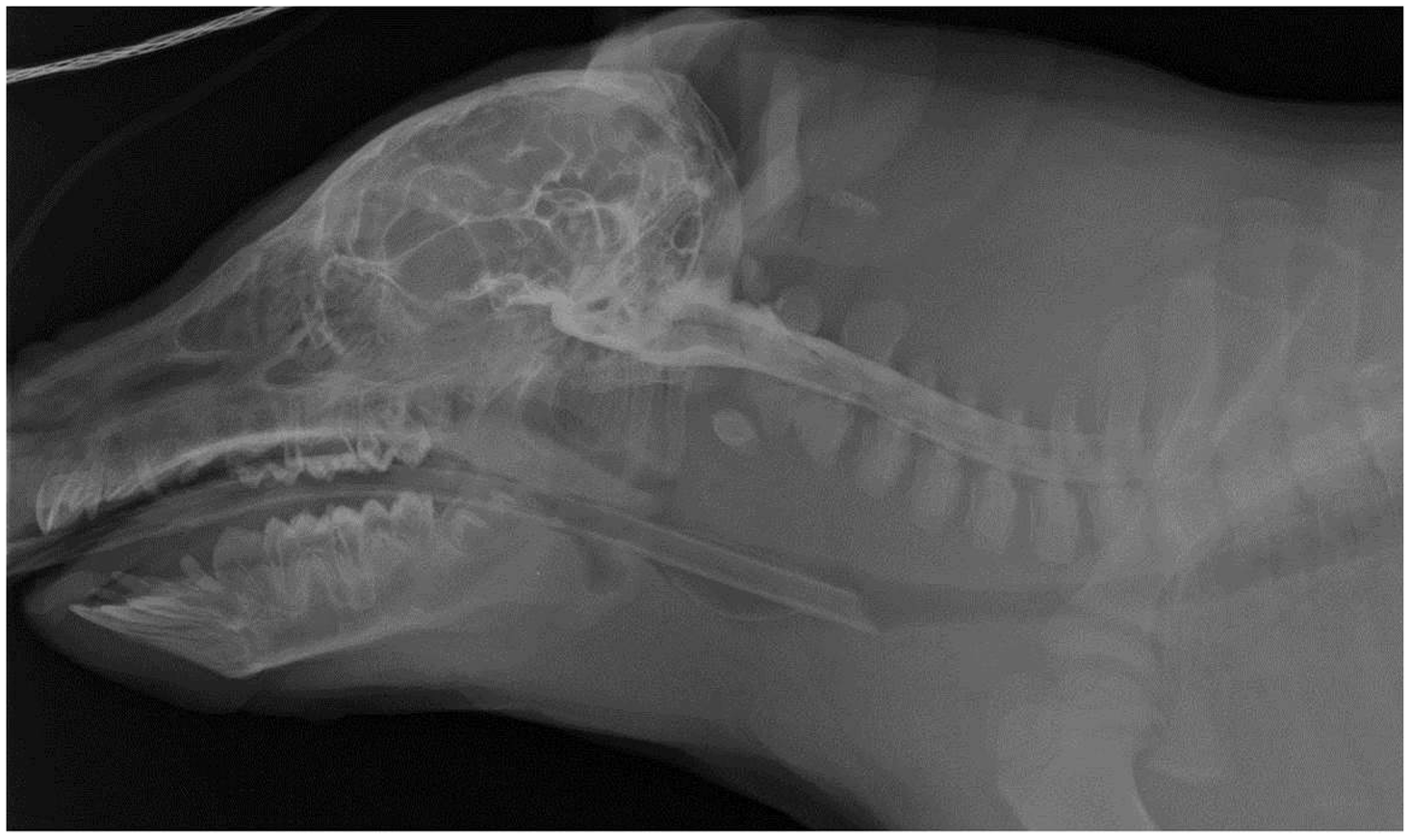
Moreover, in Phase III, the punctures were successfully carried out in six out of six piglets at the first attempt. The recovery of all piglets (six animals) was very rapid when the sevoflurane was discontinued (3 ± 0.8 min). Mild ataxia was observed during the first hour after complete recovery in all piglets.
The technique herein described provides a reproducible and easy route for CSF collection, drug administration and gene delivery through the CM. The anatomical landmarks validated during Phase I allowed the puncture of the CM and the collection of CSF in the six anaesthetized piglets.
All the punctures were carried out by the same experienced operator to minimize variables and to evaluate the ‘learning curve’, which was rapid and relatively problem free. The correct drawing of the reference lines (the median spinal line and the line traced between the cranial margins of the wings of the atlas) was of primary importance in the procedure since any mistake, such as a misdrawn line, would have precluded achieving the correct injection into the CM. The lines were drawn with the patient in lateral recumbency; however, the results suggested that the same procedure performed in sternal recumbency would have helped in the correct positioning of the reference lines.
The anatomy of newborn piglets is substantially different from adults since the occipital protuberance has a rounder and less inclined caudal profile. 13 This is the reason why the operator decided in this study to insert the needle more caudally along the median line and with a more cranioventral orientation, as compared to the injection site described in the technique used for adults.5,14 It was necessary to adjust the distance between the intersection of the lines and the insertion point of the needle in order to adapt to the different sizes of the animals. In fact, in the smaller ones, it was necessary to puncture more caudally by 1 or 2 mm. The wings of the atlas are not easily palpable, and the cartilaginous consistency of the bones makes it difficult to determine the correct positioning of the needle during its insertion, since the operator strongly relies on the different consistency of tissues.
As shown by the anatomical study in neonatal patients, 15 the dura contains an inner vascular plexus which is larger in the infant. The vascular plexus in the piglet is extremely wide, making it difficult to avoid puncturing the vessel, thus obtaining blood-contaminated CSF. After puncturing the vascular plexus, no clinical signs of cerebral hypertension were reported in this study; however, blood contamination of the CFS could represent a limitation when performing a cell count or biochemical analysis.
Performing the procedure without flexing the head allowed the piglets to breathe normally. The technique herein described demonstrates an easily accessible route to the CSF and widespread diffusion of the radiopaque contrast medium into distinct CNS areas, allowing for both sampling and administration of substances in different medical and experimental procedures. Recovery was rapid in both the control animals and in the injected piglets. The authors did not observe any neurological alterations related to the injection of the fluid or the puncture of the neurological structures, such as plegia, lasting ataxia, deficit of cranial nerves and states of impaired sensorium.
In conclusion, the proposed technique can be carried out safely in piglets.
Footnotes
Acknowledgements
This research was supported by a Ricerca Fondamentale Orientata (RFO) grant from the University of Bologna.
