Abstract
The mechanisms of production, and gross, microscopic and electrocardiograhic findings of surgically-induced complete heart block (CHB) in the adult rat are presented. This is an effective in vivo model for establishing alternative methods to electronic pacemakers and for providing detailed information aimed at replacement, reduction and refinement of the technique. Sternal thoracotomy was employed to identify the epicardial fat pad by the aortic root, used as a landmark for cauterization of the atrioventricular (AV) node. Stable CHB was produced in 60 rats with a 70% survival rate. The best survival rate was observed in 8-week-old animals weighing 221 ± 27.6 g. Heart rate before cauterization was 387 ± 55 bpm, reduced after cauterization to 126 ± 40 bpm in the survival and to 65 ± 19 bpm in the non-survival groups. At 30 days findings were: elevated left ventricular end-diastolic pressure (21 ± 5.4 mmHg, P < 0.05); maximal rate of rise of left ventricular pressure (LVP) during isovolumetric contraction (2192 ± 235 mmHg/s, P < 0.05); maximal rate of decrease of LVP (−1658 ± 191 mmHg/s, P < 0.05); isovolumetric relaxation constant (5.7 ± 0.8 ms, P < 0.05) with wet-to-dry lung–weight ratio (78.1 ± 0.4, P < 0.05); heart weight/body weight (0.6 ± 0.1, P < 0.05); heart volume (1.8 ± 0.3 mL, P < 0.05); longitudinal diameter (20.2 ± 1.91 mm, P < 0.05); and transversal diameter (17.0 ± 1.4 mm, P < 0.05) with supported dilated cardiomyopathy which culminated in chronic heart failure. CHB hearts had increased preload and replacement of myofibrils by collagen. CHB was achieved reproducibly by cauterization of the rat AV node and/or His bundle. This led to electrophysiological, hemodynamic, and structural remodeling, and could be useful in long-term cardiac remodeling assessments and potential therapy development.
Complete heart block (CHB) is a condition in which the wave of excitation is not conducted from the atria to the ventricles, 1 leading to severe bradychardia and acute heart failure.2,3 It may be inherited, but it is generally acquired as a result of myocardial infarction, cardiac surgery, drug toxicity, or degenerative, infectious and idiopathic diseases. 4
Animal models of CHB are especially important for establishing alternatives to electronic pacemakers, such as gene therapy, cellular therapy and biological pacemakers. 5 Additionally, this model reproduces the clinical circumstances of torsades de pointes, 6 a condition favored by bradycardia as produced by CHB and by drugs that prolong QT duration. 7 Therefore, this animal model is especially appropriate for the understanding of hemodynamics, electrophysiology, metabolic adaptations, volume overload, and atrioventricular (AV) desynchronization secondary to the bradycardia caused by CHB. Efforts to obliterate the AV node emerged in the 20th century, when Frédéricq crushed the AV node with forceps after a right atriotomy in dogs. 2 CHB was produced by cryoablation, 8 section of the AV nodal junction, 9 and electrocauterization 10 during thoracotomy. Fisher et al. 11 introduced a new method in dogs, in which no thoracotomy was needed; instead a catheter was used to inject formalin into the AV node. Thereafter, techniques using electrical ablation, 12 laser, 13 radiofrequency ablation,14,15 and cryoablation 16 were used. In addition to these techniques, CHB was also produced by injecting caustic substances through open and closed chests.17,18 Different methods of AV node obliteration were introduced, such as ethanol infusion in the AV nodal artery, 19 and gene transfer. 20
The goal of this study was to present the description of a reproducible method of producing CHB in rats and its repercussion on the heart, presenting detailed information aimed at replacement, reduction and refinement of the technique.
Animals
Animals were housed in cages, maintained at 23℃, 12 h light/dark cycle, and received food and water ad libitum. All procedures were approved by the Ethics Committee on Animal Experimentation (protocol number 14/2011) which had oversight of the facility in which the studies were conducted. The principles observed in this study are specified in the European Convention for the Protection of Vertebrate Animals and in the National Research Council Guide for the Care and Use of Laboratory Animals.
Sixty-eight male adult Wistar rats (Central Vivarium, Biological Health Science Center of the federal University of Viçosa, Brazil), age from 6 to 9 weeks, and weighing 170 to 272 g were included in the study. The animals were weighed just prior to surgery, and at 15 and 30 days after.
Sixty rats were allocated to two groups (30 rats/group). CHB was induced, and rats were euthanized at 15 and 30 days after surgery (groups 15 and 30 days, respectively). Eight animals were used for cardiopulmonary morphophysiological evaluations, for which four animals underwent CHB (CHB group) and four were submitted to thoracotomy (Sham group).
Materials and methods
Surgical procedures
Antibiotic (enrofloxacin, 10 mg/kg intraperitoneally once a day) was administered preoperatively. The animals were placed individually in an induction chamber, and anesthesia was induced with 3% isoflurane and 100% oxygen at a constant flow rate of 1 L/min. Anesthesia was performed using a BioLITE system (BioTex Inc, Houston, TX, USA) to illuminate the trachea with a fiberoptic stylet inserted within an intravenous catheter (16–20 Ga, 2.54 cm length) adapted as an endotracheal tube. The animals were placed on intermittent positive pressure ventilation (Inspira Advanced Safety Single Animal Pressure/Volume Controlled Ventilators; Harvard Apparatus, Holliston, MA, USA) at a frequency of 50 to 60/min, an inspiratory pressure of 16 to 18 cmH2O, and an inspiratory/expiratory ratio of 1:1. Anesthesia was maintained with 1–2% isoflurane, diluted in 98–99% oxygen.
The animal was positioned in dorsal recumbency on a polyethylene box, such that the hindlimbs were in direct contact with the dispersive pad/electrode of the electrocautery (BP 150; EMAI, SP, Brazil) and the skin was prepared for surgery. During the procedure, the body temperature was maintained at 37.5 ± 1.1ºC by heat transfer from heated water bags placed inside the box used for containment.
Median sternotomy provided access to the thoracic cavity while leaving the manubrium and the xiphoid intact, facilitating closure of the thorax. The thymus was retracted, and a partial pericardiotomy exposed the base of the heart. The right atrium (RA) was gently retracted using a sterilized cotton swab (Johnson & Johnson, SP, Brazil), and the groove between the RA and the aortic root was identified. This maneuver exposed the landmarks for the epicardial approach to the AV node. A fat pad was consistently found between the aortic root and the medial wall of the RA (Figure 1). This fat corresponded, on the outside aspect of the aortic root, to the commissure between the right and non-coronary leaflets of the aortic valve and served as a reference point for the precise location of the AV node.
Schematic drawing showing the landmarks for locating the atrioventricular (AV) node and the acupuncture needle (bent at 90°) used to make contact with the AV node and the active electrode of the electrocautery.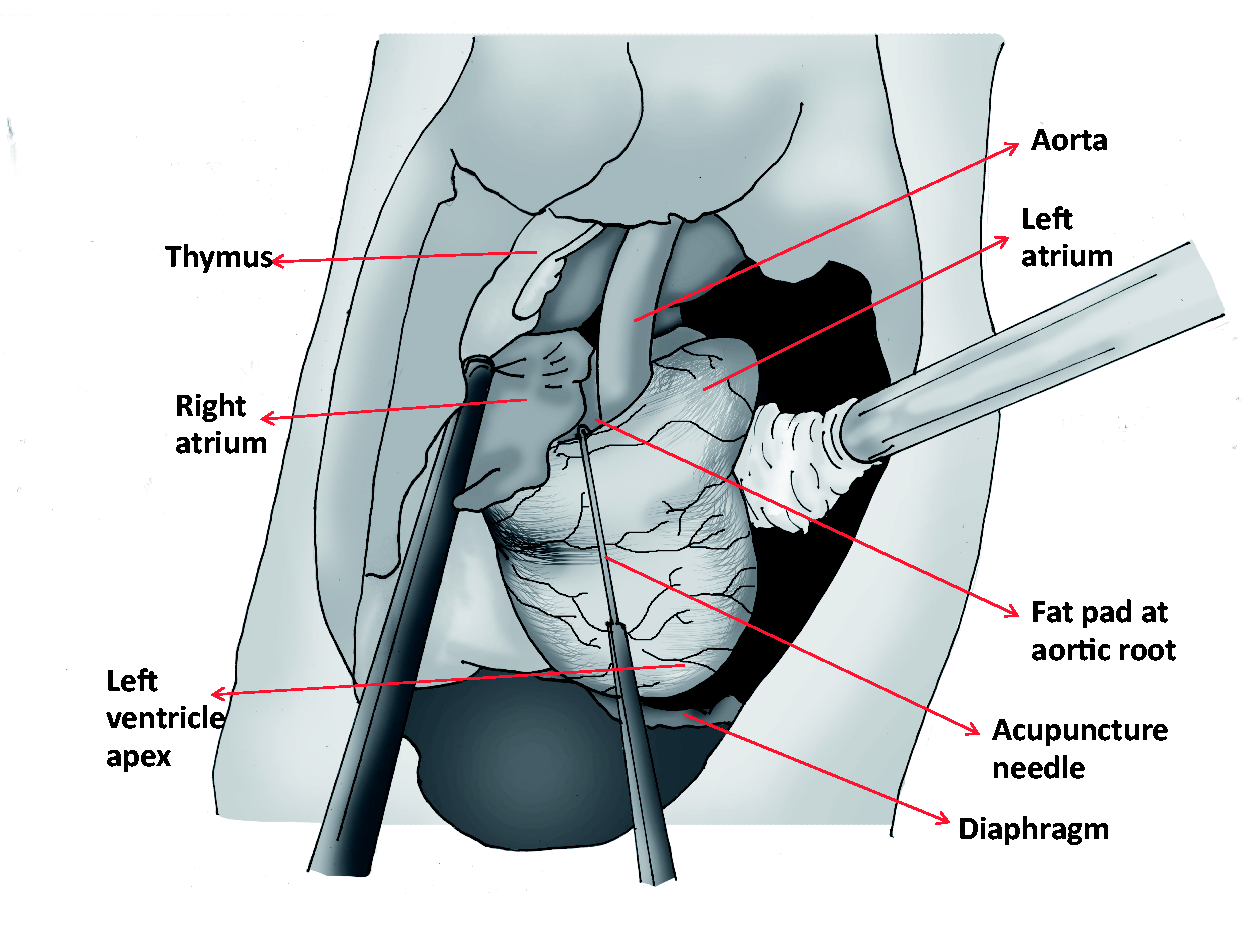
For ablation of the AV node, an acupuncture needle (0.25 mm × 30 mm; Huan Qiu, Shanghai, China) was used. Previously, this needle was bent at a 90° angle 2 mm from the tip (Figure 1) to ensure a sel-limited insertion into the myocardium and to prevent entry into the lumen. The needle was introduced through the fat pad at approximately 1 mm dorsal from the aortic root and parallel to the aorta towards the apex of the heart. The latter approach ensured that the needle stayed in the intramural space. These maneuvers produced momentary interruption of the AV node which confirmed the needle was properly placed for the ablation. The AV block observed on the electrocardiogram (ECG) and the lack of bleeding confirmed that the needle was placed correctly within the myocardium and within the AV node. Immediately after, the active electrode was placed in contact with the needle, activated in coagulation mode, and the power was set at 2, ablating the AV node.
ECGs were acquired before opening the thorax, and before and after ablation/cauterization. Due to interference with the ECG, leads were removed before activation of the electrocautery, but were repositioned immediately to document the production of CHB. If the AV block was first or second degree, or transient, additional cauterizations were applied. The duration of electrocauterization and the number of attempts were registered.
After producing the AV block, the animals were monitored by ECG until closure of the thorax (5 ± 2.5 min). Induction to CHB was considered successful when AV dissociation by block was persistent (Figure 2a and b). Chronic CHB was determined in those cases where blockage persisted up to the 15th and 30th days after induction (Figure 2c and d). P waves, QRS complexes, and any lack of association between them were documented by ECG.
Representative electrocardiograms (ECGs) from rats at (a) baseline, (b) immediately, (c) 15, and (d) 30 days after atrioventricular (AV) node cauterization. Heart rates were (a) 384; (b) 131; (c) 140 and (d) 99. Arrows indicate blocked p waves.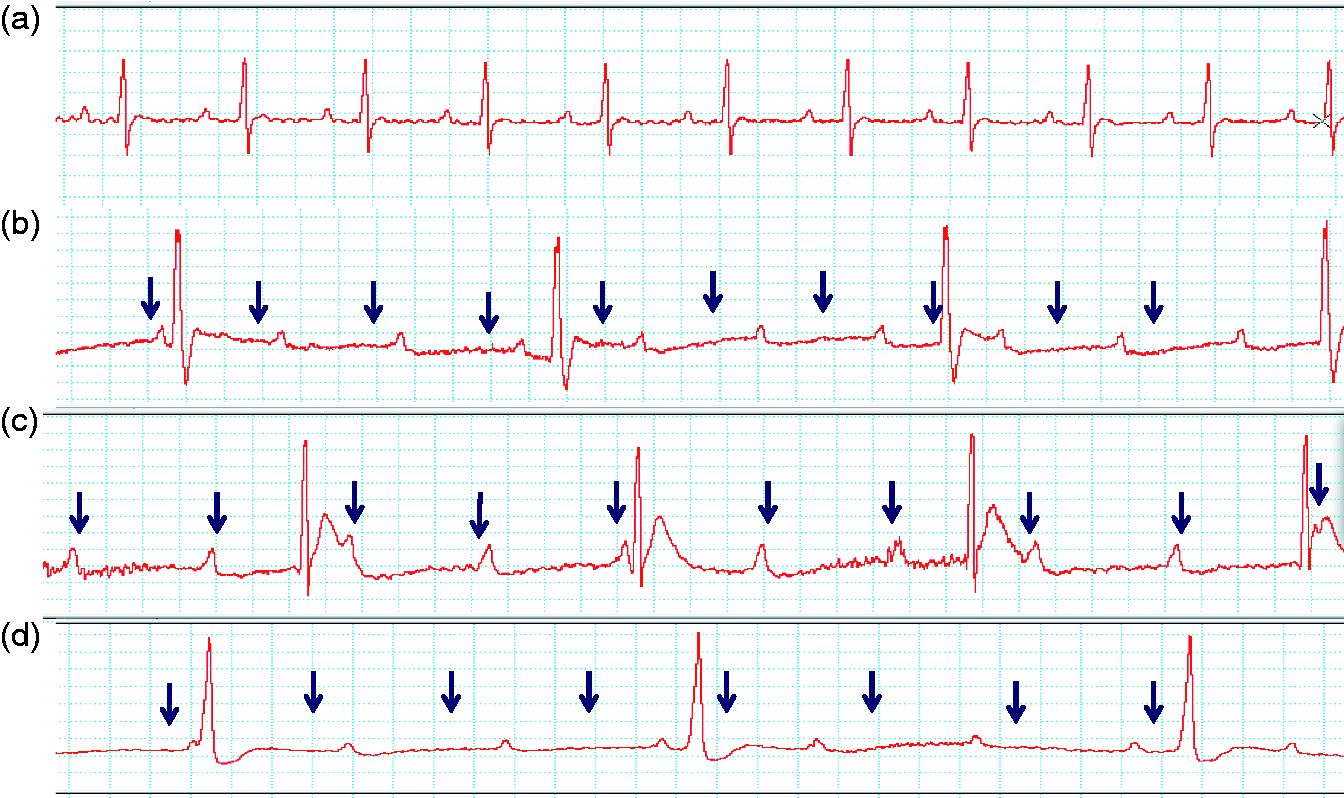
Just prior to complete chest closure using 4-0 mononylon, the lungs were fully expanded for 2 s to minimize pneumothorax and to restore negative intrathoracic pressure. The skin was sutured using 4-0 mononylon (Johnson & Johnson, Norbi, São Paulo, Brazil). The endotracheal tube was constantly aspirated to avoid accumulation of secretions and possible airway obstruction. It was removed after spontaneous breathing was observed prior to regaining consciousness. During recovery from anesthesia the animals were placed in a custom-built wooden box with ambient temperature controlled to remain between 37 and 39℃. All the rats were housed in individual cages and recovery was uneventful.
Tramadol was used for post-surgical analgesia, 4 mg/kg, given subcutaneously every 6 h for 24 h. Atropine was administered subcutaneously at 1 mg/kg to animals with pulmonary secretion during recovery.
Cotton swabs were used to absorb eventual bleeding and for measurement of volume loss through hemorrhage during the surgery. Volume loss was estimated by subtracting the weight of a dry cotton swab from each soaked cotton swab. The soaked swab weighed approximately 0.227 mg. A milliliter of blood equates to 1.06 g (or mL of blood) (http://en.wikipedia.org/wiki/Blood_alcohol_content) and, thus, 0.227 mg is equivalent to 0.240 mL of blood. Volumes were replaced with sodium chloride 0.9% solution intraperitoneally after closure of the thorax.
Electrocardiographic evaluation
On the 15th day (group 15 days) and 30th day (group 30 days), the animals were lightly anesthetized with isoflurane for ECG acquisition. Good quality lead II ECG signals were acquired for at least 2 min. Ventricular (QRS) rate/frequency was averaged from a 10 s period of the ECG trace and used for comparisons between the groups. The animals were then euthanized by anesthetic overdose.
Cardiopulmonary morphophysiological evaluation
Thirty days after surgery, a group of eight animals were lightly anesthetized for ECG acquisition and left ventricle (LV) catheterization. Of these rats, four had CHB and the other four were sham-operated. The anesthetic plane was deepened to a surgical plane for thoracotomy to be performed. The LV was catheterized with a catheter (22 gauge) fluid filled with heparinized saline (50 UI/mL). The catheter was inserted through the epicardium into the LV apex and was connected to a pressure transducer for LV pressure (LVP) recordings using a data acquisition system (Powerlab, model MLT0380; ADInstruments, Colorado Springs, CO, USA). Five minutes were allowed for hemodynamic stabilization before LVP and ECG were registered at a sample rate of 1000 kHz.
Heart rate (HR, bpm), LV systolic pressure (LVSP, mmHg), LV end-diastolic pressure (LVEDP, mmHg), maximal rates of rise (mmHg/ms), and fall (mmHg/ms) of pressure, time constant of fall of isovolumetric pressure (ms) were derived from the LVP curve. For this, five subsequent cardiac cycles free of artifacts were averaged.
After LVP recordings the animals were euthanized by isoflurane overdose. During necropsies the degree of tissue adherence at the surgical site was graded (grade 0: no adherence; grade 1: RA adhered to cauterization area of the AV node; grade 2: thymus or pericardium, or both adhered to the heart; grade 3: lung and thymus adhered to the heart).
The hearts were removed, rinsed in normal saline and gently dried for immediate measurement. Heart weight was acquired using a precision balance, and longitudinal and transverse diameters were measured using a caliper ruler. Heart weight-to-body weight (BW) ratios were calculated. Heart volume (HV) was estimated by the water displacement technique, where the heart was placed within a beaker filled with a known volume of water. The water volume displaced was equivalent to the volume of the organ.
The hearts were dissected immediately below the AV valves, and fixed in 10% formalin solution for 48 h for usual histopathological processing. Four serial cuts containing both the RV and LV were made.
Two sections were stained with Picrosirius Red21 and observed under polarized light microscopy (Olympus BX 53). Four images from each region, RV and LV, were captured with 10× objective, using a digital camera (Olympus DP 73) utilizing the imaging software Cellsens 1.7 (Olympus, Center Valley, PA, USA). The images were analyzed with Image-Pro Plus 4.5 software (Media Cybernetics, Silver Spring, MD, USA). The ‘intensity range selection’ tool was used to determine the colors for the regions of interest. Thick fibers observed as an orange to red hue under polarized light were considered to be collagen type I and mature; and thin fibers, with a green-bluish hue were collagen type II 21 and immature. 22 The area occupied by the different types of fibers was subtracted from the total area of the image and the results were presented as a percentage of the total area. The cell types were evaluated by hematoxylin and eosin (HE) stained slides (n = 2/animal).
To determine whether rats with CHB had developed congestive heart failure, it was hypothesized that pulmonary edema would form in these individuals. Edematous lungs are heavier, 23 so wet-to-dry weights were measured to investigate the hypotheses that chronic heart failure (CHF) was present. Immediately after excision and dissection of adjacent tissues the lungs were weighed wet. The lungs were then dried in an oven (MA035; Marconi, SP, Brazil) at 65℃ for 7 h and weighed again. Wet-to-dry lung–weight ratios and wet-to-dry lung–weight ratios indexed to body weight (W/D−BW) were calculated as indicated: W/D = [(W − D)/W], and W/D − BW = [(W − D)/W]/BW, and posteriorly compared between the groups (CHB versus Sham).
Statistical analysis
Variables that determined the outcome of the atrioventricular node ablation in rats.
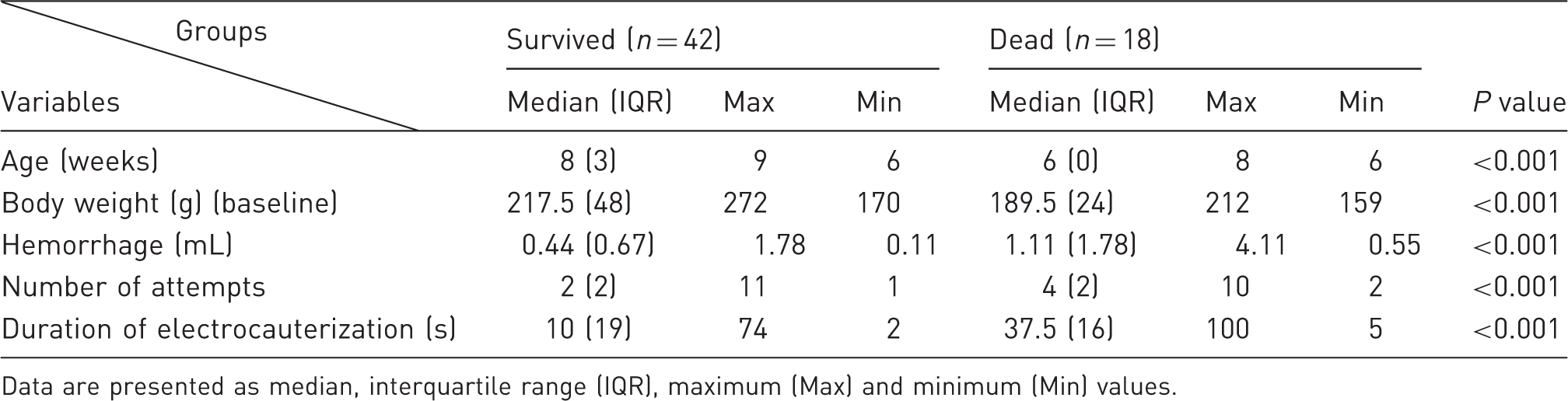
Data are presented as median, interquartile range (IQR), maximum (Max) and minimum (Min) values.
Results
CHB was successfully obtained in all animals. AV block instability, when present, could be observed immediately after cauterization and until thoracic closure, in which instances new energy was applied until establishment of CHB. The number of attempts was 2.5 ± 2.28, comprising a total duration of attempts of 17.5 ± 16.8 s (Table 1). A maximal duration of 10 s was established for each electrocauterization attempt.
In the 30% of animals that had CHB and did not survive the procedure (n = 18), a greater number of attempts (4.5 ± 2) and with prolonged total duration of electrocauterization (38.8 ± 22.4 s) (P < 0.001) occurred (Table 1).
Electrocardiography performed at baseline demonstrated that arrhythmias were not present. On the other hand, ECG findings on day 15 and on day 30 revealed a variety of arrhythmias, as shown in Figure 3.
Electrocardiographic(ECG) abnormalities found 15 and 30 days after atrioventricular (AV) node cauterization in rats. CHB: complete heart block.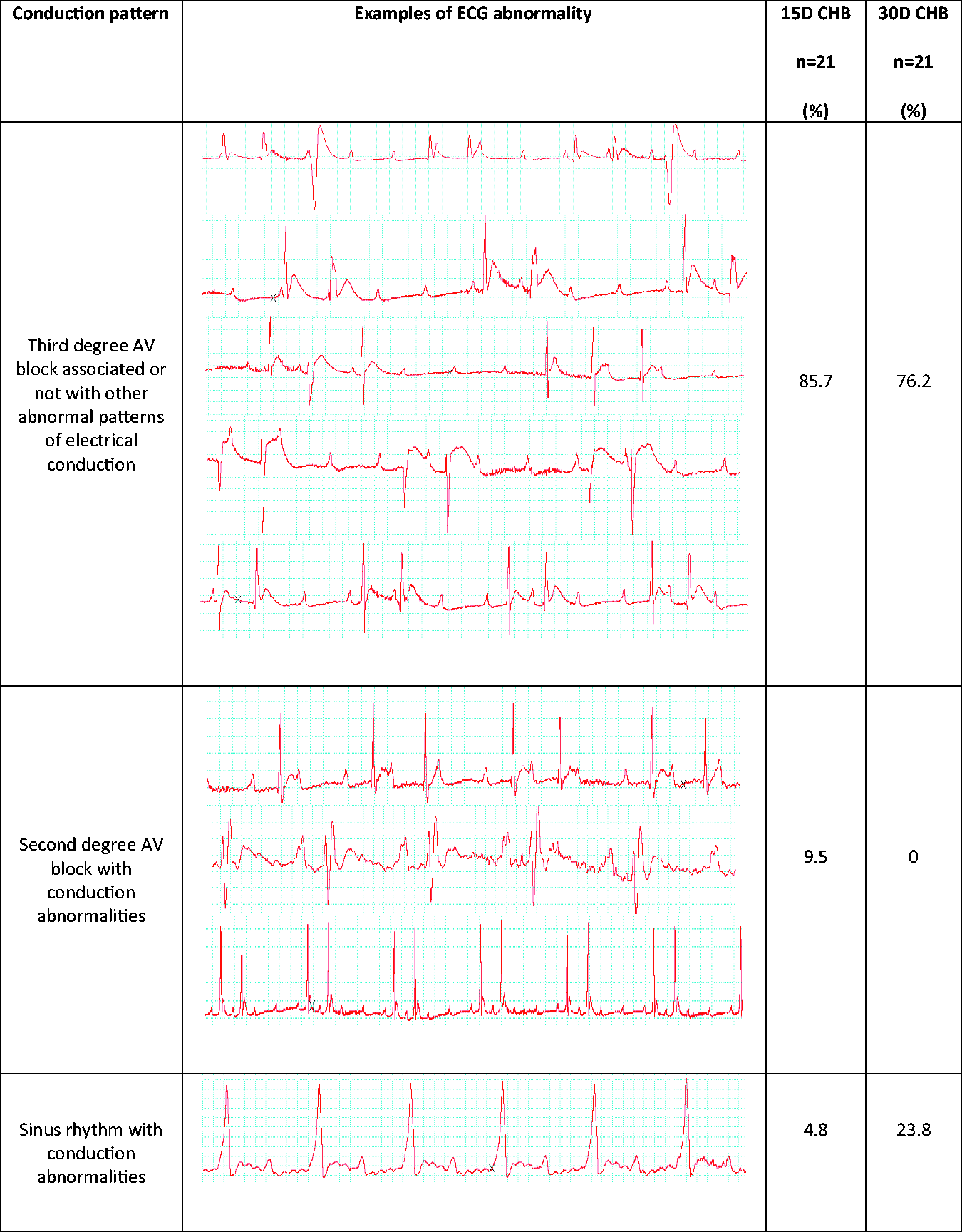
The most frequent rhythm was third degree AV block, present in 18 cases on day 15 and in 16 cases on day 30. Other aberrancies were associated with third degree AV block, with examples presented in Figure 3. These were junctional premature depolarizations with re-entry producing bigeminal rhythm with distorted AV conduction; ventricular premature depolarizations (VPDs); junctional depolarizations in addition to ventricular ectopic depolarizations and junctional beats produced by bigeminal rhythm; and bigeminal rhythm resulting from re-entry from the ventricular or from the AV junction with aberrancy. High grade second degree AV block was observed on day 15, but not on day 30. Variations of the sinus rhythms were present on both days 15 and 30. Sinus rhythms were always associated with some abnormality: bundle branch block; sinus rhythm with first degree AV block with interventricular conduction disturbance (IVCD) combined with accelerated path delta wave. In summary, third degree AV block represented the most common conduction disturbance, followed by sinus rhythm and second degree AV block (Figure 4).
(a) Surface electrocardiograms (ECGs) of rats 15days after atrioventricular (AV) node cauterization, lead II. (I) Third degree AV block with junction re-entry producing a bigeminal rhythm (distorted AV conduction). (II) Third degree AV block, ventricular premature depolarization (VPD) (arrow); last three beats junctional rhythm (dashed circle). (III) Third degree AV block, with re-entry junctional beat (arrow) and single ventricular ectopic depolarization (circle), junctional beats produced by bigeminal rhythm (square). (IV) High grade second degree AV block. (V) Second degree AV block type II with a ratio 2:1 and supraventricular premature depolarization (SVPD) (square) with remarkable first degree AV block (bracket). (b) Surface ECGs of rats 30 days after AV node cauterization, lead I. (VI) Sinus rhythm with bundle branch block (BBB). (VII) Third degree AV block with junction re-entry producing a bigeminal rhythm (distorted AV conduction) (arrows).(VIII) Sinus rhythm with first degree AV block with BBB – accelerated path – delta wave (arrows). (IX) Third degree AV block with bigeminal rhythm (brackets) resulting from re-entry from the ventricular or from the AV junction with aberrancy (arrows).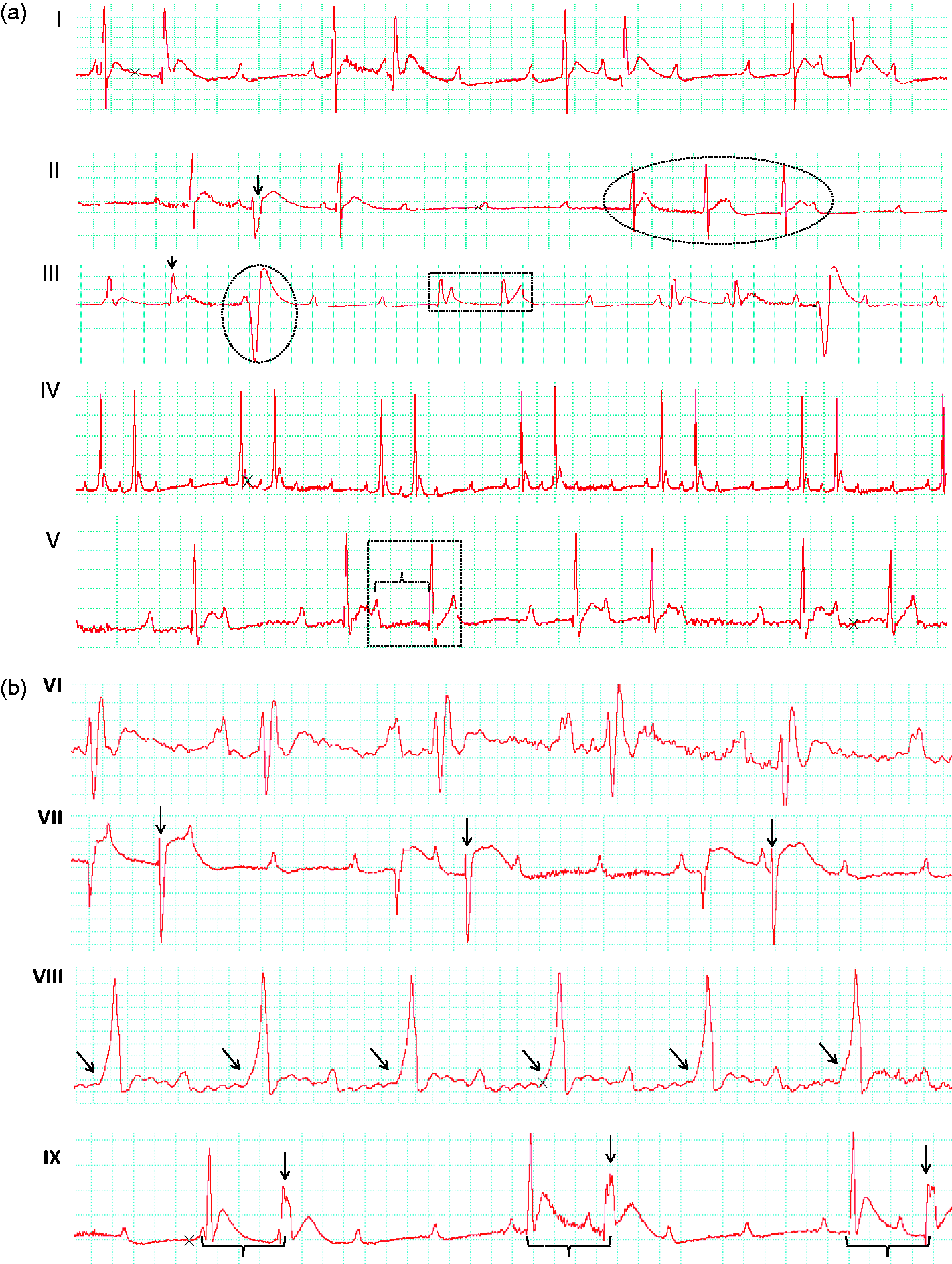
Mean HR before AV block was 387 ± 55 bpm; immediately after CHB it was 126 ± 40 bpm in the group that survived, and 65 ± 19 bpm in the group that did not survive the procedure. Interestingly, HR increased to 158 ± 38 bpm and 163 ± 69 bpm 15 and 30 days later, respectively.
Data regarding age, BW, bleeding, duration of electrocauterization and the number of attempts to acquire CHB are presented in Table 1. The ideal range of BW that produced greater survival after induction of CHB was 221 ± 27.6 g (Table 1). Animals weighing 188 ± 14.9 g did not survive after the procedure (P < 0.001). Similarly, data showed that rats 8-eight weeks of age achieved greatest survival (P < 0.001). Mortality after CHB induction could be associated with greater bleeding volumes, and a greater number of attempts and electrocauterization duration (P < 0.001) (Table 1).
After the first two weeks of surgery, animals demonstrated good tolerance of the AV block, observed by normal behavior of food and water intake. At 15 days animals gained 56 g and at 30 days gained 76 g. Therefore, group 30 days gained only 20 g more than group 15 days and lived 15 days longer (Figure 5).
Box plot graph demonstrating body weights (BWs) of rats at baseline (BL), 15 and 30 days of complete heart block (CHB). BL 15D: baseline BWs of animals from the group of 15 days in CHB; BL 30D: baseline BWs of animals from the group of 30 days in CHB. *Statistical difference between baseline and 15 days of CHB; +Statistical difference between baseline and 30 days of CHB.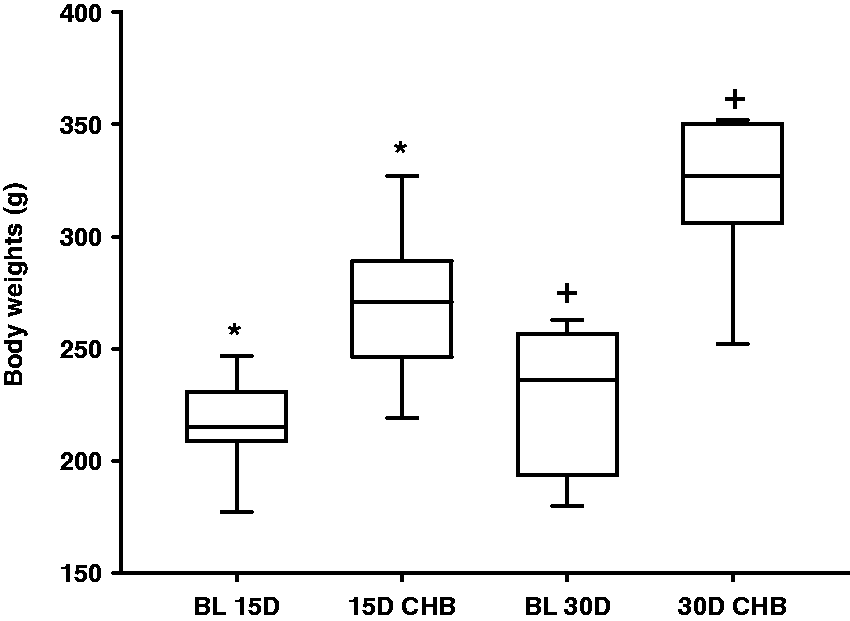
During necropsy, a tendency to worsen tissue scarring involving thoracic organs with time was observed: grade 2 (f = 6) and 3 (f = 5) for the group 15 days, and grade 2 (f = 11) and 3 (f = 8) for the group 30 days.
Hemodynamic parameters, heart dimensions, and wet-to-dry lung–weight ratio in the rat model of complete heart block.
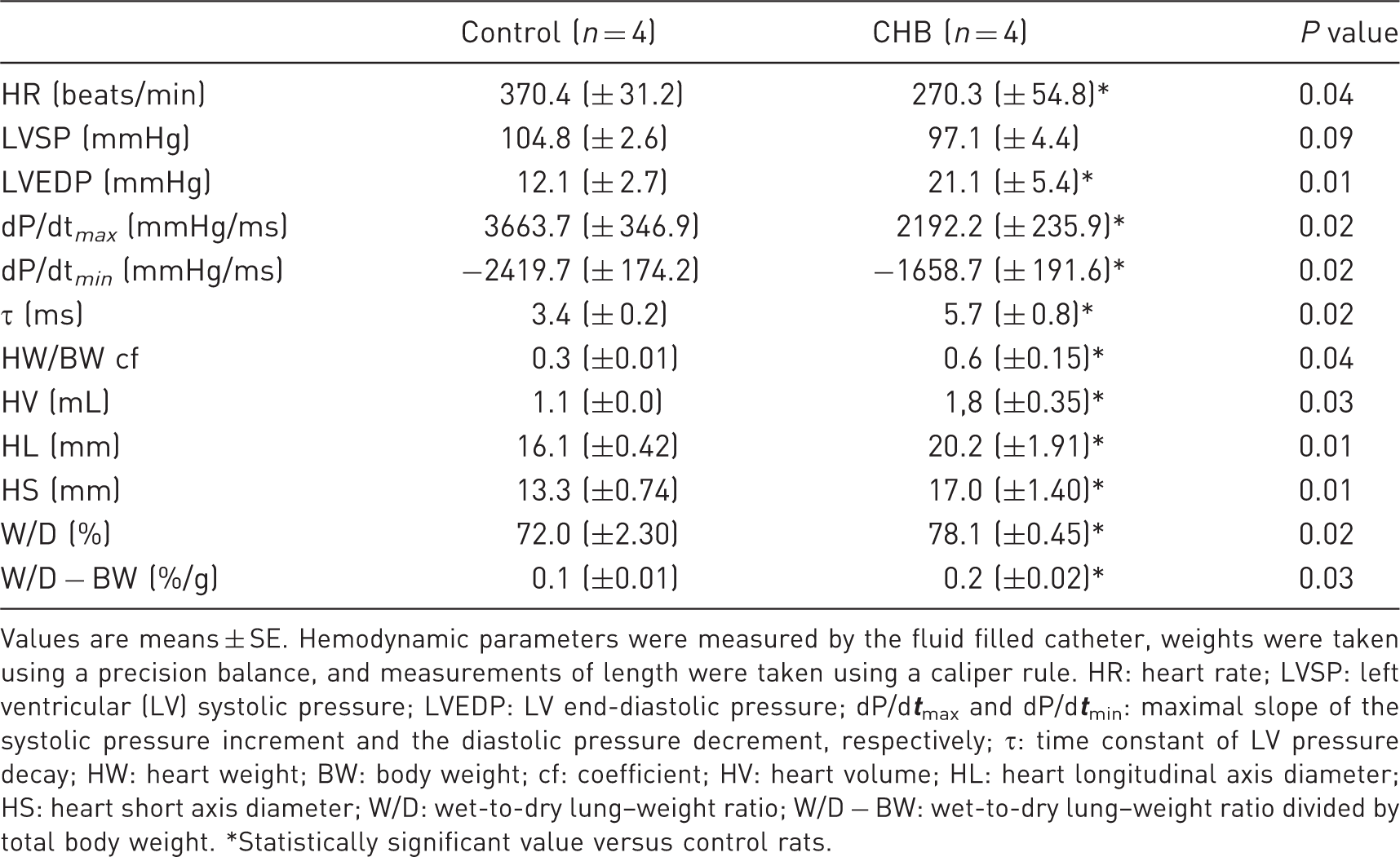
Values are means ± SE. Hemodynamic parameters were measured by the fluid filled catheter, weights were taken using a precision balance, and measurements of length were taken using a caliper rule. HR: heart rate; LVSP: left ventricular (LV) systolic pressure; LVEDP: LV end-diastolic pressure; dP/d
Of the four CHB animals that were part of the cardiac function study, three presented third degree AV block on ECG 30 days later, except for one animal that regressed to first degree AV block. One animal presented signs of CHF with evident edema of pelvic members.
An expected abnormality of left ventricular relaxation was demonstrated by low peak negative dP/dt (−2419 ± 174 versus −1658 ± 191 mmHg) (P < 0.05) and prolonged tau (3.4 ± 0.2 versus 5.7 ± 0.8 ms) (P < 0.05) for animals in the CHB group (Table 2 and Figure 6 ). The elevated LVEDP (21.1 ± 5.4) (P < 0.05) reflected stiffening of the heart as a consequence of the structural alterations in the organ (remodeling).
Electrocardiogram (ECG), left ventricular pressure (LVP) and dP/dt. Original recordings of ECG, LVP and dP/dt signals in one representative rat from the sham-operated and the complete heart block (CHB) groups.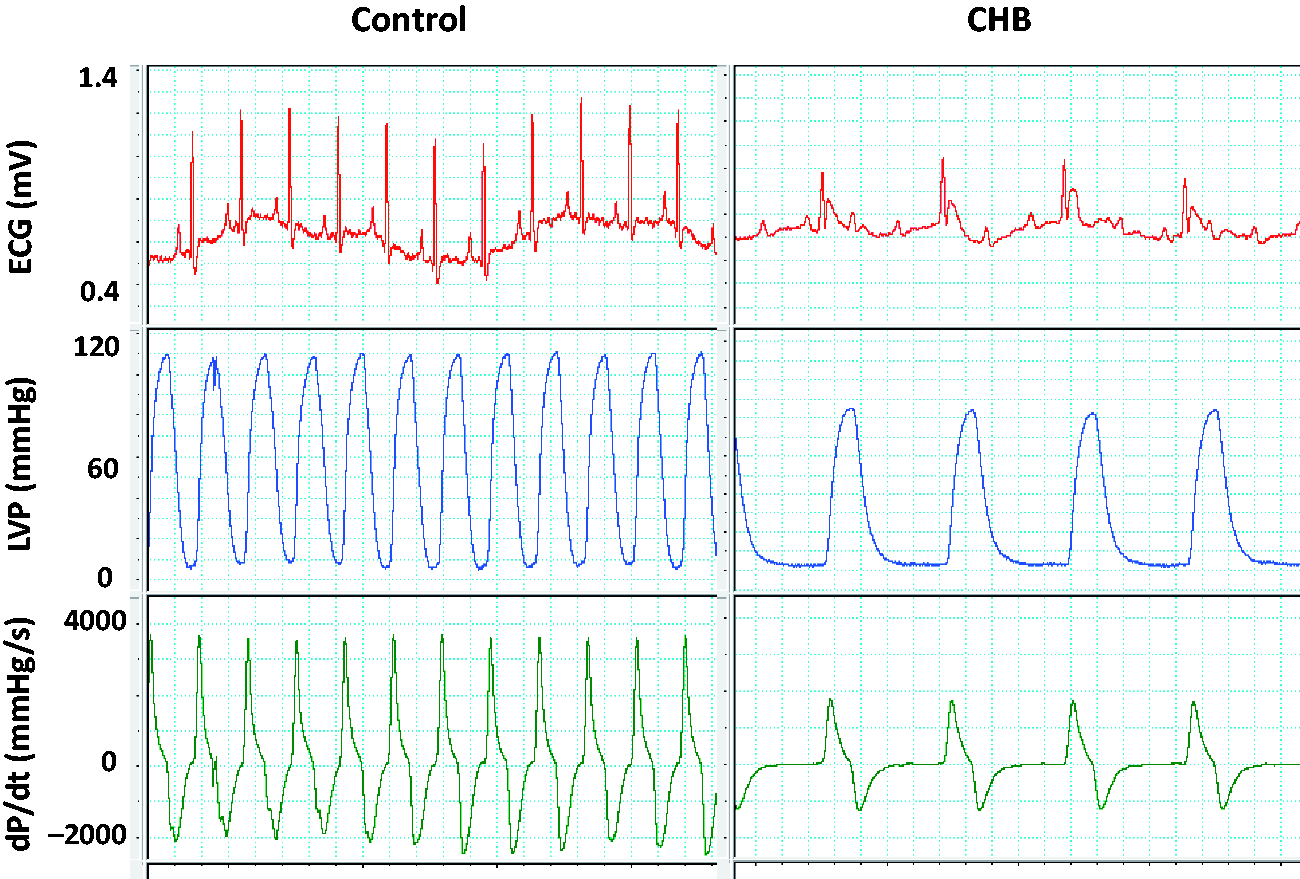
The W/D ratio of the lungs was 78.1%, which was within the normal range. However, when comparing the water content present in the lungs between the two groups (CHB and Sham) there was significant difference for both the W/D ratio and W/D − BW ratio (Table 2).
All the parameters used for measuring heart size were significantly greater for the CHB group (Table 2).
The HE analysis revealed in both groups (CHB and Sham) a thickened and edematous pericardium, and the presence of macrophages, resembling an ongoing inflammatory process probably related to the surgical trauma.
Histopathology of the heart muscle revealed macrophages, polymorphic cells, and loss of transversal striations in cells, along with regions of tissue disorganization. Cytoplasmic fragmentation, nuclei pyknosis, cytoplasmic hydrolysis and acidophilia were encountered. Fibers and muscle bundles were retracted and streaked by conjunctive fibrous tissue. These observations were seen mostly in the LV, but also in the RV (Figure 7).
Series of Picrosirius Red staining sections of the heart 30 days later. (a) Right ventricle of control rats. (b) Same image in (a) viewed with circularly polarized light (note only a few streaks of orange and green collagen). (c) Right ventricle of complete heart block (CHB) rats. (d) Same image in (c) viewed with circularly polarized light (note more prominent streaks of orange and green collagen). (e) Left ventricle of control rats. (f) Same image in (e) viewed with circularly polarized light (note with a fewer streaks of orange and green collagen). (g) Left ventricle of CHB rats. (h) Same image in (g) viewed with circularly polarized light (note more prominent streaks of orange and green collagen).
Collagen evaluations revealed that at 30 days the orange fibers occupied a greater area than did the green fibers, in both the RV and LV (P < 0.05) (Figure 8). Animals with CHB presented a greater amount of both types of collagen (orange and green) fibers than did the Sham animals (P < 0.05) (Figures 7 and 8).
Area of orange (thick) and green (thin) collagen fibers in the myocardium of complete heart block (CHB) and healthy (CO) rats. Values are mean, bars represent SD. The two-tailed t-test for pairwise comparisons were statistically different at P < 0.05. Comparisons between CHB and Sham, between orange and green collagen, and between right (RV) and left (LV) ventricles were statistically different. n = 4.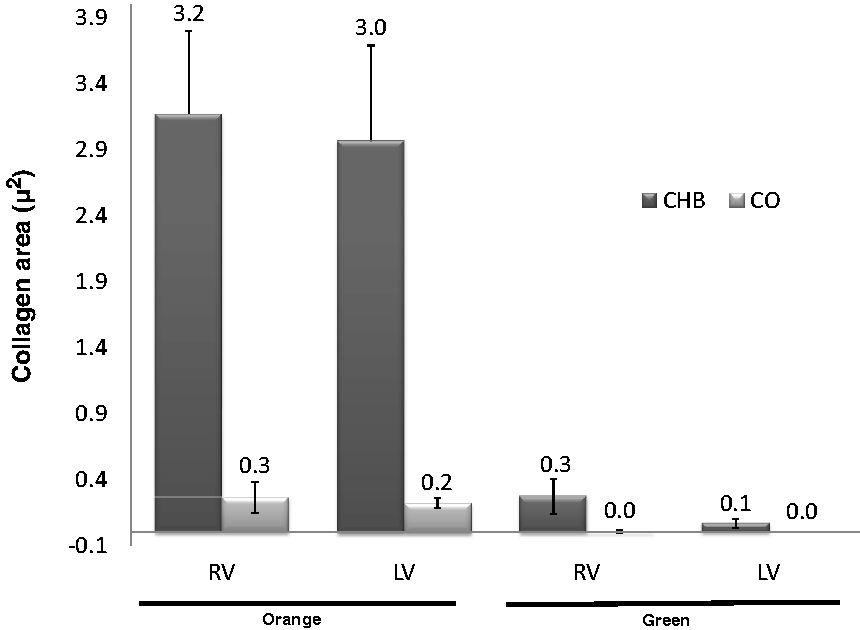
Discussion
Median sternotomy allowed adequate access to the region of interest; however care should be taken not to incise the internal thoracic arteries which run parallel and in great proximity to the midline.
A method of ablation not requiring catheterization was chosen. This approach aimed to reduce the costs and increase efficiency, and to permit direct and accurate access to the AV node. Moreover, the thoracotomy resembled conditions present in the pathological state of AV block after cardiac surgery. This is applicable when testing a potential implant therapy in which the results may be affected by the surrounding scar tissue.
The proposed method for CHB presented a series of advantages over other techniques. For example, it did not require special equipment except for the controlled ventilation system for small rodents; sternal thoracotomy allowed extensive visualization of the thorax and of the heart; it provided easy access and reliable production of CHB, making this model useful for experiments that would require cardiac exposure; and lastly, because animals could be evaluated repeatedly for a long period, this method could be useful in chronic preparations.
A chronic AV block in rats is especially advantageous due to the worldwide availability of many strains that are as near as possible to being genetically identical, which minimizes variability among individuals. Furthermore, this surgical model has proved to be simple, low cost and low maintenance compared with other larger species such as dogs and rabbits, and serves as a ‘relative replacement’ tool in laboratory animal medicine.
Monopolar diathermy was selected since energy is diffusely distributed through the monopolar electrode, constituting a self-limiting process. This happens because when cells reach a completely dehydrated state, the current flow ceases, preserving the neighboring tissues from thermal lesions. In the present study, this was important because electrosurgery was applied in close proximity to hollow structures offering great potential for perforation, mainly in the cases in which cauterization had to be repeated several times.
The thorax was closed just after identification of CHB, but Maclver et al. (2010) waited 30 min to confirm sustainability. Maclver et al. (2010) termed, as acute, CHB that remained up to 30 min from formalin injection, while chronic blocks were those sustained for 12 h, assuming that animals would not recover AV nodal function at this period or later on. In the present study, electrocadiographic evaluations were done at 15 and 30 days after CHB induction. On these occasions, if present, CHB was deemed to be chronic. Thus, our data demonstrate that AV conduction may be recovered some time from 12 h to 15 days after induction, contradicting the findings of Maclver et al. (2010) (Figure 3).
Electrocardiography provided substantial evidence that third degree AV block was maintained up to 30 days in most animals, and that the animals that regressed to lower grades of AV block developed arrhythmias not present at the baseline.
Mean HR (number of QRS complex per minute) increase at 15 (158 ± 38 bpm) and 30 days (163 ± 69 bpm), compared with HR immediately after CHB induction (126 ± 40 bpm), suggests that because of the chronic bradychardia, a specific region or regions in the ventricles manifested increased automaticity that dominated the rhythm. This was an important compensation mechanism in response to the decreased cardiac output. Despite this increase in HR (32 to 37 bpm) at days 15 and 30, HR remained low and characteristic of bradycardia in the rat.
A possible explanation for the 30% mortality rate could be attributed to greater bleeding volumes, a greater number of attempts and the duration of electrocauterization. In addition, respiratory insufficiency due to increased secretions obstructing the orotracheal tube 18 is also likely. This occurred despite frequent aspiration of the orotracheal tube and intraperitoneal administration of atropine. Another potential cause, especially for those occurring within hours of the procedure, could be the individual intolerance to the low cardiac output as a result of CHB.
Despite of significant BW gain at 15 and 30 days compared with baseline, this gain was not as expected. Healthy rats of the same strain and of similar age range26 have a more prominent weight gain. This demonstrates that the AV block was responsible for the reduced weight gain. The possible causes were (1) a cardiac remodeling leading to heart failure and consequently cachexy; 27 (2) an activation of neurohormonal systems 28 accelerating body metabolism and increasing energy expenditure at rest in animals with CHF; 29 and (3) a low cardiac output as a result of CHF. Other reasons for the lower weight gain could be attributed to post-surgical inappetence; however this was not the case since both groups (control and CHB) were subjected to thoracotomy, and only CHB animals had reduced weight gain.
The tissue scarring found at 15 and 30 days demonstrated that the partial pericardiotomy performed aiming at reducing possible adherences was not efficient to prevent them.
The hemodynamic studies revealed that animals with CHB for 30 days develop CHF. In rodents, ventricular dysfunction is characterized by discrete reduction of the systolic pressure and marked increase of the LVEDP. 30 Indeed, in the present study, the LVSP did not differ from that registered for animals in the control group (P < 0.09), while the LVEDP was greater (P < 0.01) for animals in the CHB group. The threshold for formation of pulmonary edema is an LVEDP of about 15 mmHg, reported in a myocardial infarction study in rats with CHF. 31 The LVEDP found in the rats with CHB was 21.1 ± 5.4 mmHg, which was well above the limit previously described. The hemodynamic evaluation (n = 4/group) showed that for almost all parameters the statistical differences were great, even though a low number of animals was included in each group. This demonstrates that a larger number is unnecessary to prove CHF development in this chronic CHB model, and this could facilitate reduction and refinement by other researchers.
Despite the W/D ratio of the lungs of the CHB animals being within normal limits (79 ± 1%) according to previous reports, 23 in the present study the differences in W/D ratio and W/D − BW ratio between the two groups (CHB and Sham) were statistically different. Furthermore, the low variation in water content within the lungs makes this parameter (W/D ratio) a highly sensitive indicator for detecting pulmonary edema, 23 thus demonstrating that this model was indeed capable of increasing the weight of the lungs of CHB animals and suggesting that pulmonary edema may have developed. It is important to consider the variable W/D − BW because animals from the control group were heavier at 30 days.
Along with heart dimensions and HV data, the LVEDP increase and the greater W/D ratio of the lungs in the CHB animals strengthen the belief that the present model was capable of inducing dilated cardiomyopathy with CHF as a potential outcome.
Since pericardiotomy was performed in both CHB and Sham groups, similar findings, such as thickened and edematous pericardium, and the presence of macrophages, resembling an ongoing inflammatory process related to the surgical trauma were observed in the histopathological analysis.
The lesions observed in both ventricles, by histopathology, presented various stages of evolution tending to chronicity providing enough evidence of global dilation of the heart.
Whittaker and Rich 22 reported that the amount of green collagen fibers in the myocardium, considered to be immature, becomes gradually reduced as the lesions progress to chronicity (5 weeks), giving place to orange fibers, consistent with maturation of the collagen fibers. Similarly, in the present study, evaluations at 30 days revealed that the area occupied by the orange fibers was greater than the area occupied by the green fibers, in both right ventricle (RV) and left ventricle (LV). These data confirm that CHB results in excessive increases of preload leading to overstretching and necrosis of the cardiomyocytes, and finally to myofibrils substitution by collagen.
These data provide evidence that this model offers sufficient stimuli to cause biventricular remodeling and possibly induce CHF at later stages. It is assumed that a heart with CHB must adapt itself to chronic bradycardia, volume overload, and increased wall stress. Therefore, the adaptive changes are important for maintaining normal hemodynamics 32 and is a continuous process of alteration in the genomic, molecular, cellular, and interstitial expression which is clinically manifested as modification in size, axis and function of the heart, thus determining progressive myocardial dysfunction.
The most important limitation of this animal model is the requirement of previous training, since complications resulting in death during the surgical procedure are observed at the initial phase. Complications varied from inadvertent perforation of the VE during sternotomy, ventricular arrhythmia, RA rupture with the electrocautery due to excessive energy application, difficulty with orotracheal intubation, accidental tracheal extubation while repositioning the animal, and problems with adjusting the controlled ventilation.
CHB may be achieved reproducibly by cauterization of the rat AV node and/or His bundle. It leads to electrophysiological, hemodynamic, and structural remodeling; and may be useful in long-term cardiac remodeling assessments and potential therapy development.
Footnotes
Acknowledgments
The authors would like to thank Professor Brunna P A Fonseca (Department of Veterinary) for making the heart artwork, and Professor Clóvis Andrade Neves for his assistance with the Picrosirius Red images.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Brazilian National Council of Technological and Scientific Development (grant number 473578/2009-3); and the Minas Gerais State Research Foundation (grant number APQ00933-09).
