Abstract
Large mouse litters are often culled based on the premise of better survival and growth for the remaining pups. To test whether the culling of litters does provide the benefits of improved survival and growth, mortality and growth were measured in 468 litters of C57BL/6J × 129S1/SvImJ F1 hybrid mice that were unculled or culled to four or six pups per litter. In addition, a limited number of weanlings were evaluated until three months of age for growth, health and physiological measurements. Most measurements did not differ among the cull groups. These included mortality, organ weights (adrenals, kidneys and testes), bone mineral density, percent fat, 12 of the 17 blood parameters, and three of the seven electrocardiographic (EKG) parameters. Several parameters, including five of the 17 blood parameters and four of the seven EKG parameters, showed statistical differences, but all values were physiologically normal. Unculled weanlings showed a reduced weight of 4%, but this weight difference disappeared by three months. These results suggest that mice in culled litters do not demonstrate improved health compared with those in unculled litters.
The 1996 edition of the Guide for the Care and Use of Laboratory Animals (Guide) 1 recommended that mice weighing less than 10 g, which includes newborns and most pups prior to weaning, be provided with 38.7 cm2 (6 in 2 ) of floor space. Many institutions have culled litters to meet this recommendation. The rationale in the Guide for culling pups is the hypothesis that it improves the survival and growth of the remaining pups due to the increase in access to maternal food and care. The 1996 Guide encouraged studies to test this hypothesis.
Several investigators responded to this suggestion by testing whether the culling of litters was beneficial to the remaining pups. O'Malley and colleagues 2 suggested that a lactating dam and her pups be considered as a single unit and that the minimum floor space recommendations proposed in the 1996 edition of the Guide should not be based solely on the weight of the offspring. This study compared litters that were culled to meet Guide recommendations with litters that were not culled. Reproductive performance and markers of stress did not differ between groups.
Our laboratory also designed a study, reported here, to test whether culling improves the health of the remaining pups. In our study we tested the hypothesis using a hybrid strain, the progeny of C57BL/6J × 129S1/SvImJ (B6129SF1/J). These exceptional breeders regularly produce eight or more pups per litter, exhibit good mothering instincts, and possess a long, productive breeding lifespan. The litters were culled to four mice per litter, to six mice per litter, or were not culled (average litter size of 8.5 pups). The objectives were to compare pup survival and growth among cull groups. We also evaluated the effects of culling on physiological parameters in pups at weaning. Additionally, parents were evaluated for markers of stress. Finally, a subset of pups from each cull group was maintained for 10 additional weeks after weaning to evaluate the long-term effects of culling on performance indices representing growth and health. Before our study was completed, the 2011 edition of the Guide 3 was released, stating that the dam and her pups could be considered as a unit and that culling pups to meet a predetermined space requirement was unnecessary. The completion of our study provided further evidence that culling did not significantly benefit the remaining pups.
Animals, materials and methods
Animals and housing conditions
C57BL/6J × 129S1/SvImJ F1 hybrid mice (B6129SF1/J; JAX® stock number 101043) were obtained from The Jackson Laboratory (Bar Harbor, ME, USA) and housed in trio matings of two females and one male per cage. The animals were housed in pressurized individually ventilated shoebox cages (cage #5, Thoren Caging Systems Inc, Hazelton, PA, USA), providing 504 cm2 ( 78 in 2 ) of floor space. These cages measured 22.2 × 30.7 × 16.3 cm (8.75 × 12.1 × 6.4 in). Mice were given ad libitum access to autoclaved acidified water (pH 2.8 to 3.1) and pelleted, autoclaved, standard laboratory chow containing 6% fat by weight (LabDiet® 5K52, St Louis, MO, USA). All procedures were approved by the Institutional Animal Care and Use Committee at The Jackson Laboratory and were consistent with the United States Public Health Policy on the Humane Care and Use of Laboratory Animals. Mice were euthanized by CO2 asphyxiation at the end of the experiments.
The animal room was supplied with high efficiency particulate air (HEPA)-filtered air at 19 air changes/h and was maintained at 22 ± 2℃, relative humidity of 35% ± 4%, and a 12:12 h light:dark cycle beginning at 06:00 h. Each cage was positively pressurized and set to operate at 60 air changes/h. The cage air input and exhaust system was independent of room air. No cage air was mixed with room air. Bedding, changed weekly, was autoclaved pine shavings (Crobb Box, Ellsworth, ME, USA). The animal room was specific pathogen free, with controlled personnel access. The mouse facility was monitored four times per year and found to be free of 15 viruses (mouse hepatitis virus, reovirus, two mouse parvoviruses, Theiler's mouse encephalomyelitis virus, ectromelia virus, mouse rotavirus, thymic virus, pneumonia virus of mice, Sendai virus, murine cytomegalovirus, lactic dehydrogenase-elevating virus, K virus, mouse adenovirus, and polyomavirus), 17 bacterial species (including Helicobacter spp.), two Mycoplasma spp., external and internal parasites, and Encephalitozoon cuniculi.
A total of 78 trio matings were used, with two females and one male per cage (156 females, 78 males). Each trio was assigned to one of three groups (26 breeding trios per group): no culling (C0); litters culled to six pups (C6); and litters culled to four pups (C4). All trios were maintained until each female had approximately four litters. Birth date, litter size, and mortality during the first two weeks were recorded for all the litters. When pups were exactly 14 days old, the litters were culled using a random number generator. If a litter was smaller than the assigned cull group size (e.g. five pups in C6, three pups in C4, less than six pups in C0), the mice from that litter were not used in the analysis. Culling at 14 days was chosen to align with current practices at The Jackson Laboratory.
When pups were 21 days old, mortality during days 15–21 was recorded and pups were weighed and weaned. Additional analyses were performed on five groups of mice – three groups of pups used for physiology measurements, one group of parents used for body and organ weights, and a group of dams used for fecal corticosterone levels. The three groups of randomly selected pups were: (1) weanlings from each cull group (C0, C6, C4) used to assay organ weights and whole blood at three weeks; (2) a different group of weanlings used to analyze electrocardiogram (EKG) and body composition at three weeks; (3) litters from each cull group housed at weaning in groups of five mice/sex/cage for an additional 10 weeks and maintained on a standard chow diet. This group, which will be referred to as the ‘offspring growth’ group, was weighed weekly and at 11–13 weeks of age; EKG, body composition and organ weights were recorded. The parental group consisted of dams and sires randomly selected at the end of their breeding cycle and used to assay organ weights. Finally, a group of dams from 16 randomly selected matings was used to measure fecal corticosterone.
Measurements to assess health
The following measurements are routinely performed in this laboratory, have been described in detail elsewhere,4–7 and are briefly summarized here.
Body weight
For body weight, the litters were separated by sex at weaning and average group weight per sex was recorded. Mice were weighed using an Ohaus balance (Ohaus Corp, Parsippany, NJ, USA) with InCal calibration to adjust for mouse movement.
Hematology
A 200 µL sample of EDTA-anticoagulated whole blood was obtained from the retro-orbital sinus after administration of a topical anesthetic (tetracaine-HCl). Complete blood counts were determined using an Advia 120 Multispecies Hematology Analyzer (Bayer Diagnostics, Tarrytown, NY, USA) after the administration of a topical anesthetic, tetracaine-HCl. Parameters measured were cell hemoglobin concentration mean, eosinophils, hematocrit, hemoglobin distribution width, lymphocytes, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, mean corpuscular volume, hemoglobin, monocytes, mean platelet volume, neutrophils, platelet count, red blood cells, red cell distribution width, reticulocytes, and white blood cells.
Electrocardiogram
EKGs were obtained from unanesthetized mice using the ECGenie system (Mouse Specifics, Quincy, MA, USA). In this system mice were placed on a platform standing 45.72 cm (18 in) above the laboratory bench and equipped with three pediatric conductance leads. Each mouse was acclimatized on the platform for 5 to 10 min, and readings were initiated and recorded in the next 3 to 5 min whenever three of the mouse's feet simultaneously contacted the leads. Parameters measured were heart rate (HR), heart rate variability (HRV), and intervals for P–Q, P–R, QRS complex, QT-corrected (QTc), and R–R. The EKG trace was dissected into time intervals between defined wave form peaks (P, Q, R, S, T), identified by the Fast-Fourier software analysis package provided with the equipment. Intervals are expressed in milliseconds and therefore represented duration between the P–Q waves, the P–R interval, the Q–R–S complex interval, the Q–T interval, and the R–R interval. The Q–T interval was additionally adjusted to account for HR, providing the QTc measurement.
Body composition
Percent fat and bone mineral density (BMD) were determined in mice anesthetized with 0.02 mL/g body weight of tribromoethanol using dual-energy X-ray absorptiometry (Lunar PIXImus DXA, Luan, Madison, WI, USA).
Fecal corticosterone
Fecal corticosterone was measured in dams from a subset of breeding units. Six trios were used from the C0 group, and five trios each were used from the C6 and C4 groups. Samples were collected from 32 dams. To collect fecal pellets, animals were individually removed from their cages and placed on a paper towel for up to 10 min in a clean cage without bedding. Three to five pellets per animal were collected on the day of culling as pups were culled, to obtain a baseline value, and again 24 h after culling of their two-week-old pups. Dams in the unculled group (C0) had a sham cull; their pups were removed for a period of time equivalent to that required to cull other litters (approximately 10 min). Pellets were stored at −80℃ until analysis. Fecal corticosterone levels were quantified with a double-antibody 125I-corticosterone radioimmunoassay kit (MP Biomedical, Santa Ana, CA, USA) after hormone extraction using an ethanol-based procedure as previously validated. 8 In brief, fecal samples were boiled twice in 90% ethanol with the ethanol from each extraction combined, and dried under air to concentrate the sample; the resulting residue was resuspended in 100% EtOH for assay. Prior to assay, reconstituted extracts were diluted 1:100 in phosphate buffered saline (PBS). Corticosterone concentrations are expressed as nanograms per gram (ng/g) dry weight of feces.
Organ weight
Adrenal glands, kidneys and testes from each male were weighed in pairs and divided by 2 to obtain average organ weight. Like organs (adrenal glands, kidneys) from both females in a trio were weighed together and divided by 4. Body weight was used as a covariate in the analysis.
Statistical analysis
Analyses were performed to determine the effect of cull group on measured parameters using PROC MIXED in SAS (v9.3) or JMP (v10.0.0). The significance threshold was set at P < 0.05. Linear regression models were fit to the data. If assumptions of the analyses (e.g. linear regression) were not met, appropriate transformation of the data was performed (e.g. log, square root, or rank transformation). For each measurement analyzed, the effects of cull group (C0, C6, C4), sex, and their interaction were investigated to determine whether additional analyses, such as a Tukey post hoc test, were required. For organ weights, body weight was fit as a model covariate.
First and subsequent litter sizes were evaluated in the following way: the average size of all first litters, which were not subject to a prior culling, was compared with the average size of the second, third, and fourth litters in each group (C0, C6, C4). The sizes of litters after the first were averaged for each cage; in general this was three litters for each dam for a total of six litters/cage.
Results
Mortality of pups
Mortality recorded among litters for each cull group.
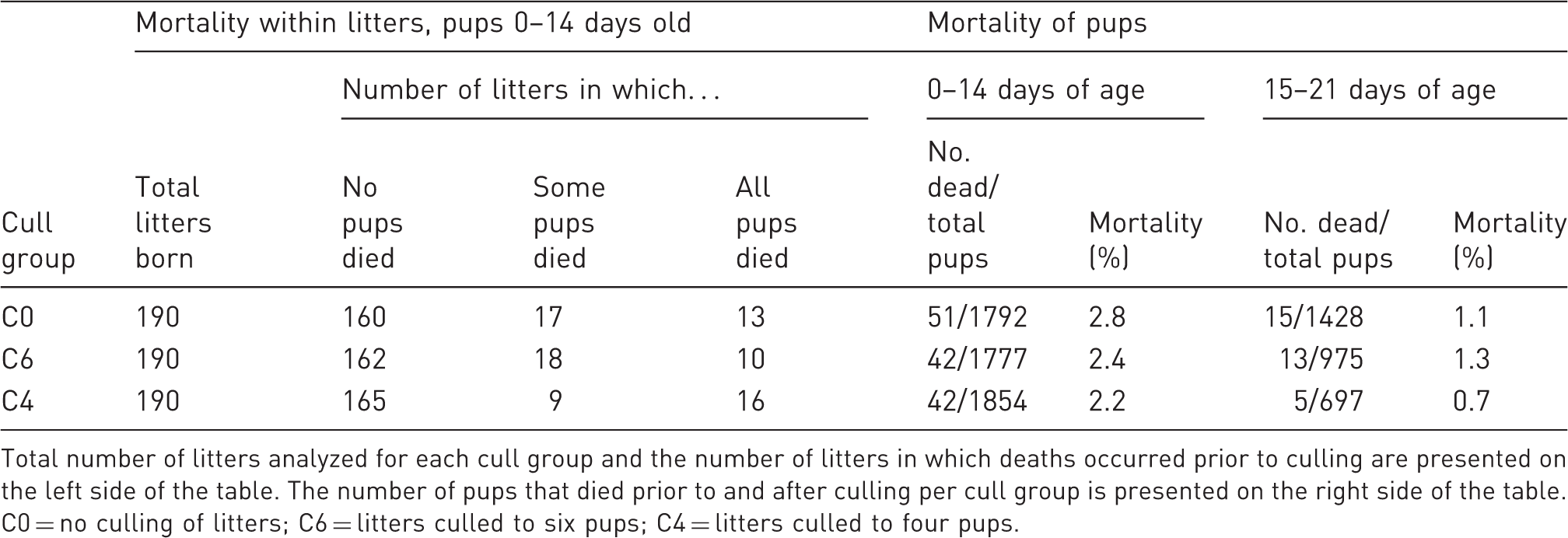
Total number of litters analyzed for each cull group and the number of litters in which deaths occurred prior to culling are presented on the left side of the table. The number of pups that died prior to and after culling per cull group is presented on the right side of the table. C0 = no culling of litters; C6 = litters culled to six pups; C4 = litters culled to four pups.
Weanlings
Body weight
To investigate whether culling improved growth of the remaining pups, we compared the weights of pups in the three cull groups at weaning (Figure 1a). Culling resulted in greater weanling body weight compared with unculled litters (C0 versus C6, P = 0.03; C0 versus C4, P < 0.0001). Pups from the C6 and C4 litters did not differ in weanling weight (P = 0.13). A Tukey multiple test adjustment was made for these data. The group means for females were: C0, 11.4 ± 0.1 g; C6, 11.7 ± 0.1 g; C4, 11.9 ± 0.1 g. The group means for males were: C0, 11.8 ± 0.2 g; C6, 12.0 ± 0.1 g; C4, 12.3 ± 0.1 g. As expected, the males were heavier than the females in all groups, but the interaction between sex and cull group was not significant.
Body weights in weanlings and growth curve in offspring. (a) Weights for weanlings at three weeks of age (n = 910). Numbers of mice per group: C0 = 145 females, 147 males; C6 = 154 females, 155 males; C4 = 151 females, 158 males. *C6 differs significantly from C0 (P = 0.03). **C4 differs significantly from C0 (P = 0.0001). (b) Growth curve of offspring from 3–13 weeks of age. Values are mean weight per sex per cull group at each weekly time point ± SEM. n = 25–29 animals per sex per cull group.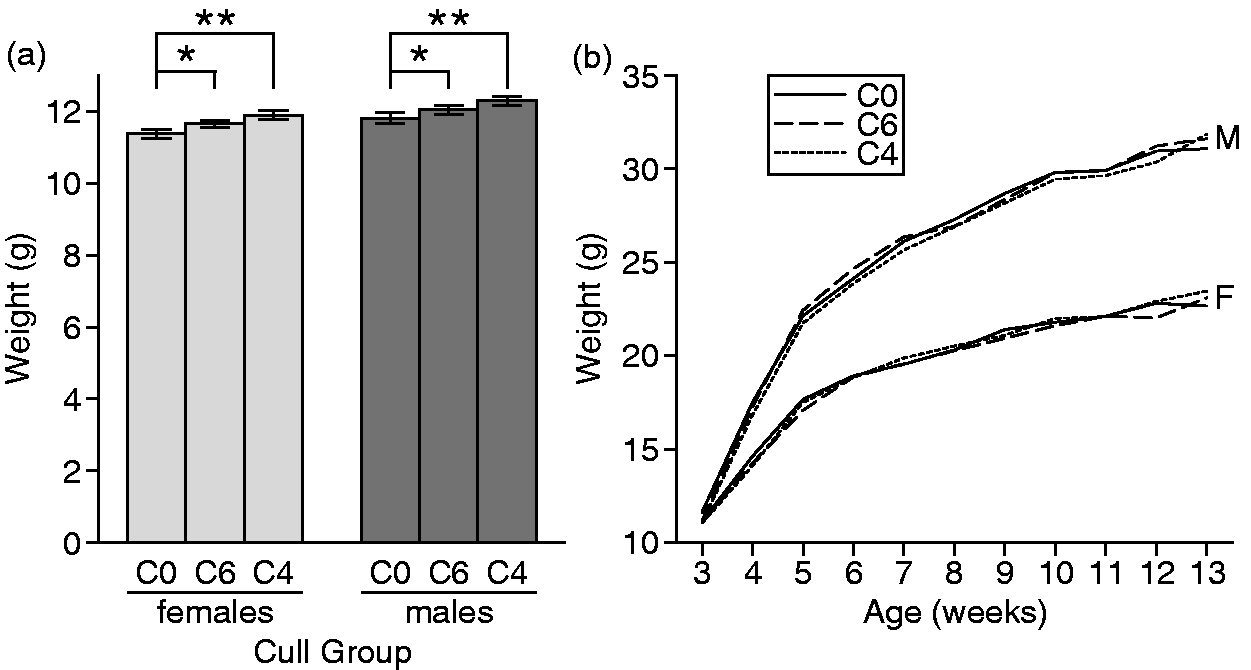
Whole blood analysis
Weanlings: whole blood parameters that differed among cull groups.
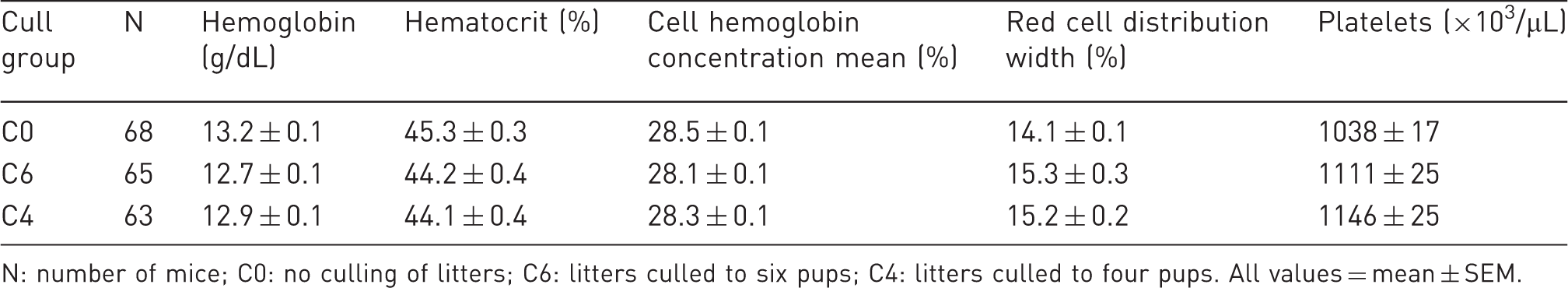
N: number of mice; C0: no culling of litters; C6: litters culled to six pups; C4: litters culled to four pups. All values = mean ± SEM.
Electrocardiogram
Electrocardiogram measurements for weanlings and offspring growth.
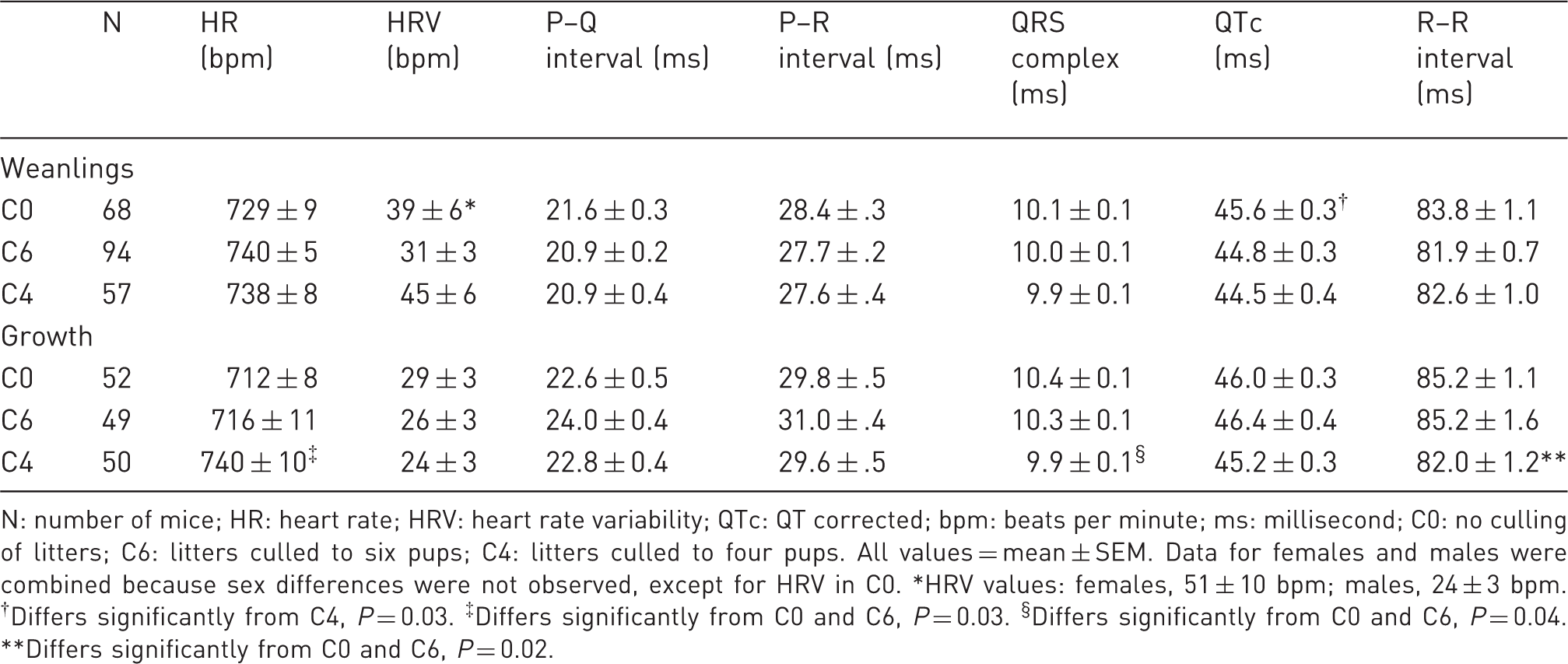
N: number of mice; HR: heart rate; HRV: heart rate variability; QTc: QT corrected; bpm: beats per minute; ms: millisecond; C0: no culling of litters; C6: litters culled to six pups; C4: litters culled to four pups. All values = mean ± SEM. Data for females and males were combined because sex differences were not observed, except for HRV in C0. *HRV values: females, 51 ± 10 bpm; males, 24 ± 3 bpm. †Differs significantly from C4, P = 0.03. ‡Differs significantly from C0 and C6, P = 0.03. §Differs significantly from C0 and C6, P = 0.04. **Differs significantly from C0 and C6, P = 0.02.
Body composition
Body composition was measured when pups were three weeks of age. Neither BMD nor percent fat differed among the cull groups. Average BMD was 0.0329 ± 0.0001 cm2 and average percent body fat was 22.7 ± 0.1%.
Organ weights
Organ weights did not differ among cull groups when body weight was accounted for (data not shown). As expected, sex differences were found, but the interaction between sex and cull group was not significant.
Growth of offspring
To determine whether culling had any effect on post-weaned growth of pups, we measured several factors in animals from 3–13 weeks of age. To assess whether the differences in weaned weight found among cull groups cause a sustained effect on the ability to thrive, body weight was recorded weekly and body composition (percent body fat, BMD) was measured at the end of the study. To further evaluate whether there was any prolonged effect associated with culling, mice were measured for EKG, adrenal weight and kidney weight.
Growth
Growth curves from 3–13 weeks of age did not differ among cull groups (Figure 1b). Unlike the small differences found in body weights at 21 days of age in the weanling group (n = 910; Figure 1a), no differences in body weights at 21 days of age in animals used for the growth study (n = 157) were found among cull groups. As expected, the males in all groups weighed more than the females throughout the 10 weeks of weight measurements (Figure 1b).
Electrocardiogram
EKGs were recorded when mice were 11 weeks of age. Of the seven parameters measured, three differed significantly among cull groups (HR, P = 0.03; QRS complex, P = 0.04; R–R interval, P = 0.02; Table 3). HR was significantly higher in C4 than in C0 and C6; C0 and C6 did not differ from each other. Because HR is inversely correlated to the R–R interval, the significance in R–R followed the same trend as HR among cull groups, as expected. The QRS interval was significantly shorter in C4 than in C0 and C6; C0 and C6 did not differ from each other. The shorter QRS interval in C4 is consistent with the higher HR. Although cull group differences for these three EKG parameters were statistically significant, all values were within normal physiological ranges. 9
Body composition
Body composition was measured when mice were 11 weeks of age. Neither BMD nor percent fat differed among the cull groups. As expected, values differed between sexes. BMD was higher in males than in females (0.0526 ± 0.0003 cm2 and 0.0505 ± 0.0002 cm2, respectively). Percent fat was greater in females than males (18.2% ± 0.25% and 16.3% ± 0.30%, respectively).
Organ weight
The weights of the adrenal glands, kidneys and testes did not differ significantly among offspring from different cull groups when body weights were accounted for.
Parents
To determine whether culling had any effect on the parents, we measured several parameters, including litter size and body and organ weights at necropsy, in a group of parents comprising 21 breeding trios.
Overall, the size of litters from the three cull groups was 4, 6 and 8.5 ± 0.1 SEM for unculled. To determine whether culling had any effect on the size of litters after the initial culling, we recorded the number of pups born in each litter. We compared the size of the first litters from each mother, which would not be affected by prior culling, to the combined average size of subsequent litters (litters 2 + 3 + 4). As expected, the size of the first litters did not differ significantly among cull groups (Figure 2). The size of the first litters ranged from 2–15 pups for all cull groups. We then evaluated whether subsequent litter sizes differed after the first culling took place. All pups were counted at birth and at 14 days, before any culling occurred. The process of culling had no effect on the size of subsequent litters for any cull group. For all cull groups, there was a slight increase in the number of pups in litters 2–4 compared with the first litters, but the differences from the first litter size were not significant (P = 0.09) and were not attributable to the cull group. Sizes of the second through fourth litters ranged from 1–16 for all cull groups.
Effects of culling on litter size. Open bars are the mean litter size for the first litters of all matings in the cull group (n = 26 per group), and gray bars are the mean litter size for litters 2–4 combined for the cull group (26 + 26 + 26 = 78 per group). Values are mean litter size ± SEM.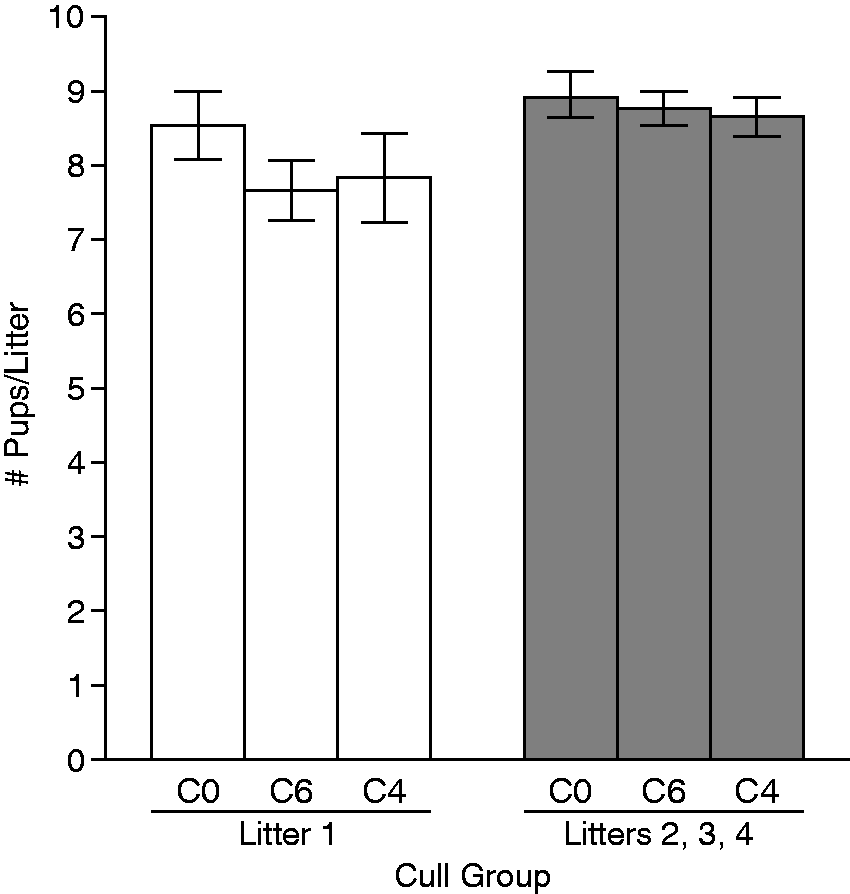
Corticosterone levels of dams.
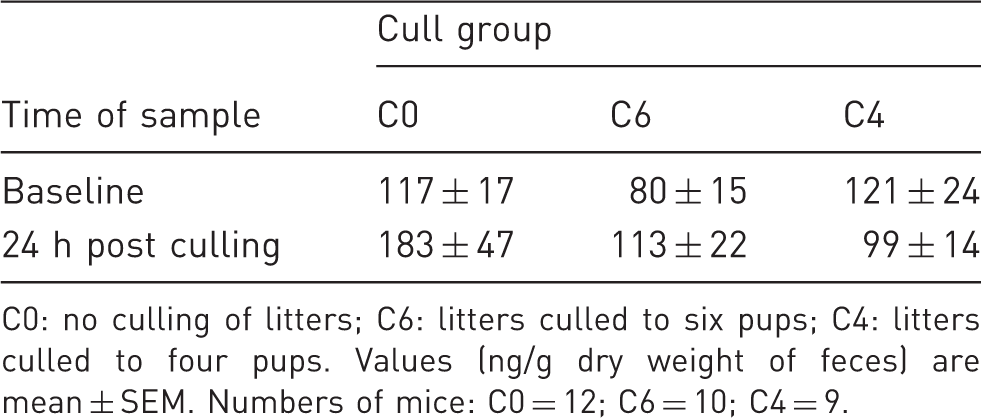
C0: no culling of litters; C6: litters culled to six pups; C4: litters culled to four pups. Values (ng/g dry weight of feces) are mean ± SEM. Numbers of mice: C0 = 12; C6 = 10; C4 = 9.
Parental body and organ weights.
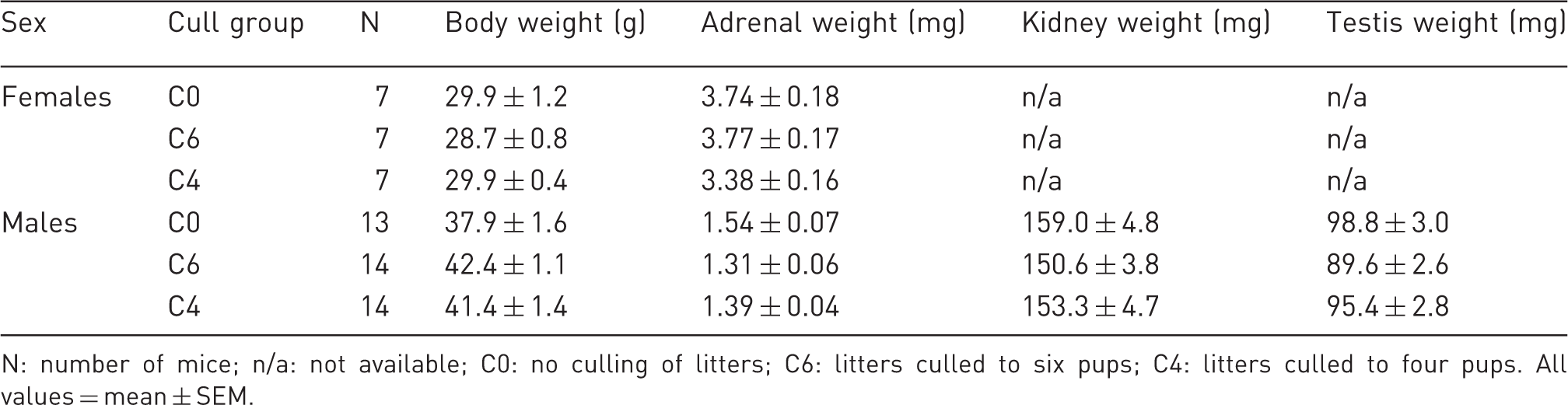
N: number of mice; n/a: not available; C0: no culling of litters; C6: litters culled to six pups; C4: litters culled to four pups. All values = mean ± SEM.
Discussion
Culling of large litters is often practiced in both research and production settings as a precaution to ensure that offspring are not compromised with respect to nutrition and maternal care. Culling is also used to maintain specific cage density goals and to avoid presumed detrimental effects of overcrowding. Our study investigated whether culling pups confers advantages on weanlings and offspring up to three months of age.
Parameters used in this study to evaluate the effect of culling on growth and health were chosen from measurements routinely used in our laboratory to compare physiological differences among genetically diverse mouse strains and are proven indicators of overall health in mice. Stress was directly measured in pups by EKGs, namely in the HR and its variability; and in adrenal weight at necropsy, an indicator of chronic corticosterone production. Stress among parents in culled litters was measured by adrenal weight in both sexes and fecal corticosterone in dams. This study used large numbers of breeding units, litters and pups to provide adequate statistical power to detect culling effects. In addition to unculled litters, two levels of culling were performed to evaluate whether differences in the degree of culling had any effect.
Most measurements of health and physiology did not differ among the cull groups. These included mortality, organ weights (adrenals, kidneys and testes), BMD, percent fat, 12 of the 17 blood parameters, and three of the seven EKG parameters (HRV, P–Q interval and P–R interval).
Small but statistically significant differences were found in a few traits. Weanling weight was 4% less in pups from unculled litters compared with pups from litters culled to six or to four pups (approximately 300 pups per group; Figure 1a). But in the group of mice that was allowed to grow to three months, no differences in weaning weight were observed among cull groups (approximately 50 pups per group; Figure 1b). The larger sample size has more potential to detect small differences, and any failure to demonstrate differences in the smaller group underscores the negligible physiological relevance of the differences found in the larger group.
Some minor differences among weanling cull groups were observed among blood parameters: values for hemoglobin, hematocrit, and cell hemoglobin concentration were significantly greater in unculled litters; values for red blood cells and platelets were significantly lower in unculled litters. None of the red cell-related values indicated a trend toward anemia, nor did the lower platelet values in C0 suggest failure to maintain hemostasis or risk of clotting disorders. Increased red cell parameters in C0 further suggests that nutrition is adequate in unculled litters. Hence, differences in whole blood parameters did not support a lack of good health in any of the cull groups and were all well within normal physiological ranges.
High HR is an indicator of chronic stress. HR was significantly higher in the group with the most pups culled (C4). This result may indicate that culling has a negative effect. Although HRV is also an indicator of stress, it did not differ among cull groups in either the weanling or growth groups in this study.
To evaluate whether parents were affected by the culling of their pups, we measured adrenal weights of both dams and sires and fecal corticosterone levels of dams. Adrenal weight reflects corticosterol production, and increased weight suggests chronic excess corticosterol production. Fecal corticosterone measurements are used to capture prior stressful events. While it is true that fecal corticoid metabolite measures are reflective of circulating levels in the previous 8–10 h, the 24 h sample would integrate over 16 or so hours prior to collection. This includes the dark period, when rodents exhibit elevated corticoid metabolites. However, a stressful stimulus prior to fecal collection will be reflected as an increase in fecal corticoid metabolite levels even as much as 24 h later.8,14 Hence, the effect of a stressor is still measurable if samples include this dark phase effect. No significant differences were found in either adrenal weights or fecal corticosterone levels, suggesting that breeding animals are either unaffected by or rapidly adjust to reductions in litter size.
There is a paucity of literature about the effects of culling in rodent colonies. The design used in the O'Malley study, 2 which found no effect of culling, is the closest to our study. A study using mice in which investigators added pups to enlarge litters by 50% at postnatal day 1 showed increased lifespans in enlarged litters. 15 In rats, culling is controversial and has been discussed in the context of toxicology studies, with opposing conclusions and recommendations. Agnish and Keller 16 argued that early postnatal culling of rodent litters should be a standard practice for controlling pup growth and development and suggested that variability in litter size would affect results of subsequent experiments. Palmer and Ulbrich 17 challenged this position by suggesting that culling may eliminate the opportunity to detect litter size differences in experimental results and that increased growth in culled rat litters could have the long-term consequences of shortening overall and reproductive lifespans.
Our results suggest that culling litters at 14 days of age does not improve the ability of pups to thrive. The small decreases in pup weight in unculled litters are eliminated as animals grow to three months of age. We conclude that pups from unculled litters do not exhibit compromised growth, health or physiology and that culling may be an unnecessary step in animal husbandry practices. Consistent with this conclusion, the most recent Guide 3 now suggests that a dam and her pups are considered as a single biological unit.
Footnotes
Acknowledgements
The authors thank Ann Plaud, Willson Roper III, and Holly Savage for outstanding technical assistance in phenotyping the mice; Vivek Philip for help in data analysis; and Luanne Peters for review of hematological parameters. The authors also thank Jesse Hammer for preparation of figures and Joanne Currer for editorial assistance. This work was supported by grant RR12552 from the US National Institutes of Health (BP).
