Abstract
Oral administration of non-steroidal anti-inflammatory drugs (NSAIDs) can lead to adverse effects such as gastrointestinal distress. The complexation of different groups of active substances with β-cyclodextrin (β-CD) has drawn considerable interest over recent years. The purpose of this study was to analyze the ketoprofen/β-cyclodextrin (K/β-CD) conjugate complex as well as to assess its anti-inflammatory effect after oral administration (doses of 30 mg/m2 and 15 mg/m2 of body surface), compared with ketoprofen. The studies were done on two models of experimentally-induced acute inflammation in rats (n = 48, 6/group), by means of intraplantar administration of a 10% aqueous kaolin suspension and intraperitoneal administration of a 1% sodium thioglycolate solution. The dynamics of the acute inflammatory process and the anti-inflammatory effects were monitored using plethysmometric determinations after 3, 6, 9, 12, 24 and 48 h (plantar inflammation), and the absorbance of the exudates (spectrophotometrically read) and nucleated cell counts after 24 h (peritoneal inflammation). The coupling of ketoprofen with β-CD resulted in increased solubility (100% in 60 min) of the newly-formed product, which further resulted in a higher bioavailability compared with ketoprofen (<40% in 120 min). In both models of experimentally-induced inflammation, the K/β-CD complex had a higher anti-inflammatory activity than ketoprofen.
Keywords
Ketoprofen is a propionic acid derivative, [(RS) 2-(benzoylphenyl)-propionic acid], non-steroidal anti-inflammatory drug (NSAID), with analgesic, anti-inflammatory and antipyretic properties.1–4 It acts by inhibiting cyclooxygenase, phospholipase A and prostaglandin synthase, and can also produce central analgesia by acting at a supraspinal level. 5
Ketoprofen is still an important part of current veterinary therapy, being frequently used to treat pain and acute or chronic inflammation.
When administered orally, ketoprofen achieves the highest plasmatic concentration in 1 to 2 h. After parenteral administration (intramuscular and subcutaneous) the highest plasmatic concentration occurs in less than one hour.6,7 The apparent distribution volume is 0.28 L/kg, and the time of plasmatic halving is 6–12 h.8–10
Most NSAIDs are insoluble in aqueous media, which poses a major problem for their transportation across biological membranes, including their absorption into the body.4,11–13 In addition, NSAIDs produce a range of well known side-effects in the gastrointestinal tract, kidneys, liver and heart.2,14–18 A number of different methods can be used to increase the bioavailability of active pharmaceutical ingredients (APIs) and their therapeutic effects (analgesic and anti-inflammatory activities) while decreasing potential side-effects.19–21 Cyclodextrins are frequently used in the pharmaceutical industry for this type of application.
Cyclodextrins are crystalline colorless solid natural polymers obtained by using a relatively simple technology – the fermentative prehydrolysis of starch.22,23 They are a family of non-reductive cyclic linear oligosaccharides that consist of six, seven or eight D(+)-glucopyranoside units, linked by α-glycosidic bonds, called α, β and γ cyclodextrins, respectively, of which β-cyclodextrin (β-CD) is the most widely used. 22
β-CD has been used with various APIs to form inclusion complexes which increase their rate of dissolution, improve stability, mask unpleasant odor and taste, and increase their solubility 10 to 1000 fold.24,25
In vivo tests involving animal models, especially rats, are used in biomedical research to assess the effects of different active substances or to establish more effective clinical interventions.26,27 Experimental models of acute inflammation in rats are used to screen for analgesic and anti-inflammatory effects of NSAIDs.28,29 Experimental inflammation models are based on the use of irritants that cause edema/swelling which lasts from several hours to several days and involves the activation of the arachidonic acid cascade giving rise to the formation of the main mediators of inflammation (prostaglandins, thromboxanes). Carrageenan, 30 Freund adjuvant, 31 kaolin, 32 turpentine oil 33 and sodium thioglycolate 34 are the most frequently used inductive agents of inflammation.
Materials and methods
Substances
The substances used in the study were ketoprofen, 99% pure, and β-CD, 98.6% pure (Sigma-Aldrich, Hamburg, Germany). Other reagents used were kaolin and sodium thioglycolate of analytic quality (Shandong Xinda Fine Chemical Co Ltd, Zibo, People’s Republic of China).
Animals
The studies were conducted on 48 young white specific-pathogen free male Wistar rats, weighing 200 ± 10 g, (‘Cantacuzino’ National Research and Development Institute for Microbiology and Immunology, Baneasa, Bucharest, Romania). The rats were acclimatized to the environment, in a room with controlled temperature and humidity (23℃ and 55% relative humidity), for five days before starting the study. They were fed with standardized feed and water which were available ad libitum. The protocol was approved by the Ethics Commission of the Faculty of Veterinary Medicine, ‘Ion Ionescu de la Brad’ University of Agricultural Sciences and Veterinary Medicine of Iasi, Romania by notification No. 43 of 02/03/2012, and complied with the international ethic regulations regarding the handling and use of laboratory animals, using the method recommended by the OECD guidelines for the Testing of Chemicals 425, 17 December 2005.
Preparation of the ketoprofen/β-cyclodextrin inclusion complex
The ketoprofen/β-cyclodextrin (K/β-CD) complex was synthesized by freeze drying (Alpha 1-2 LD plus freeze dryer; Martin Christ GmbH, Osterode, Germany) at the ‘Ilie Murgulescu’ Physical Chemistry Institute of Bucharest, Romania. The substances were used in an equimolar ratio, taking into account the molecular mass of each product (0.245 g ketoprofen and 1.435 g β-CD). Ketoprofen powder was dissolved in ethyl alcohol to produce a 0.5% solution, and β-CD was dissolved in water to produce a 0.5% aqueous solution. The complexes were formed in an aqueous medium. To improve the solubility of the active substance, PEG 400 and a phosphate buffer were used. The solutions were mixed for 30 min in a blender equipped with a temperature sensor (Heidolph sensor; Heidolph Instruments GmbH & Co KG, Schwabach,Germany), until the complete homogenization of the dispersion, then the obtained complex underwent solvent evaporation for 2 h, at 45℃, at a constant temperature with steering. After solvent evaporation, the complex was freeze-dried for 12 h, then analyzed using a scanning electron microscope (JEOL JSM 5510-LV microscope; Joel Ltd., Tokyo, Japan). The size and morphological structure of the crystals were assessed.
The solubility of the K/β-CD complex and of the simple ketoprofen were determined spectrophotometrically (MR-96 A Mindray spectrophotometer; Mindray, Shenzhen, People’s Republic of China), at a wavelength of 253 nm. The apparent 1:1 bond constant of the formed complex was calculated from the slope and intercepts of the straight lines in the phase-solubility diagram.
Experimental design
The study was done on eight groups of six rats each (C1, C2, C3, C4; T1, T2, T3 and T4). In the C1, C2, C3 and C4 groups, plantar inflammation was experimentally induced by injecting 0.15 mL of 10% aqueous kaolin suspension in the right posterior limb. The animals were first anesthetized using isoflurane (Abbott Laboratories Ltd, Maidenhead, UK). The limb of each rat was marked up to a point of immersion of the acropodium into the measuring cell, then its diameter was measured (moment 0–m0) and calculated in cm3 by using a digital plethysmometer (Hugo Basile Plethysmometer 7140; Ugo Basile, Varese, Italy). At 1, 3, 6, 9, 12, 24 and 48 h (moment – m1, m3, m6, m9, m12, m24 and m48) after the induction of inflammation, the dynamics of the inflammatory process and the anti-inflammatory and analgesic effects of the studied substances were assessed.
Peritoneal inflammation was induced experimentally in groups T1, T2, T3 and T4 by intraperitoneal administration of 5 mL of 1% sodium thioglycolate solution. After 24 h, the peritoneal exudate was collected into Eppendorf microtubes (Sigma-Aldrich) from the rats in all four groups, after anesthetizing the animals with isoflurane. To harvest peritoneal cells, the abdominal cavity of the rats in groups T3 and T4 was washed with 3 mL of phosphate buffer solution. The peritoneal fluid was centrifuged at 2500 rpm (EBA 21 Heltrich centrifuge; DJB Lab Care Ltd, Newport Pagnell, UK), then spectrophotometry (MR-96 A Mindray spectrophotometer; Mindray) was performed and the nucleated cells present in the peritoneal exudate were counted by direct microscopy (Micros MC300 microscope; Micros, Wien, Austria), with the result expressed in terms of 103 cells/mm3.
Test substances were administered orally, by means of a gastric probe, one hour before the induction of inflammation, as follows: C1 and T1 represented the untreated control groups; groups C2 and T2 received 30 mg ketoprofen/m2 body surface (approximately 5 mg/kg); groups C3 and T3 received 15 mg K/β-CD/m2 body surface (approximately 2.5 mg/kg); and groups C4 and T4 received 30 mg K/β-CD/m2 body surface (approximately 5 mg/kg). The drug dosage for each rat was calculated by determination of the precise total body surface area (TBSA) using the metabolic weight by applying Meeh’s formula: TBSA = kW0.67, where W = rat weight (in grams) and k is a pre-established constant having the value of 9.83 according to Gouma et al. 35 This mode of calculating the doses using the metabolic weight (per m2 body surface) is more accurate than the usual one (per kg body weight) as it minimizes the differences of size and amounts of active tissue between animals.
Statistical analysis
The results were summarized using descriptive statistics. To compare the overall magnitude of changes during treatment the Friedman ANOVA non-parametric test for repeated measurements was used. A value of P < 0.05 was considered significant.
Results
Preparation of the inclusion complex
Freeze-drying produced a fine, white, water soluble precipitate. Scanning electron microscopy revealed the morphological structure and the agglomeration degree of particles after the elimination of water from the K/β-CD inclusion complex.
Dissolution profiles of ketoprofen and K/β-CD demonstrated that, after complexation, dissolution improved significantly and water solubility was greatly increased. The dissolution rate of K/β-CD was approximately 100% at 60 min, while the maximum dissolution rate of ketoprofen in water, at 35℃, was less than 40% at 120 min (Figure 1).
Dissolution profile of ketoprofen (K) and ketoprofen/β-cyclodextrin (K/β-CD).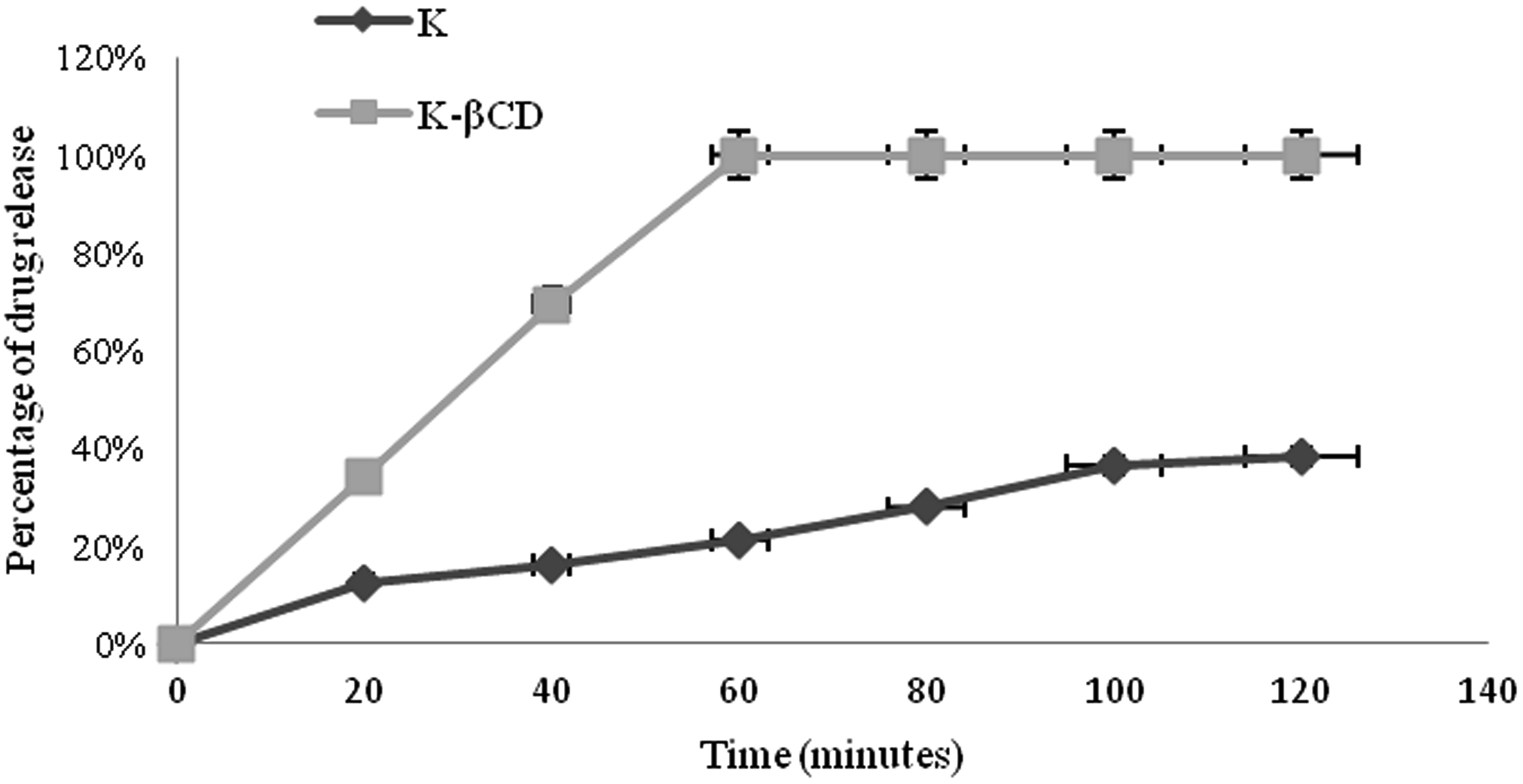
Induction of the experimental inflammation at plantar level
Average values obtained in the plethysmometric assessment of the plantar edema at different times.
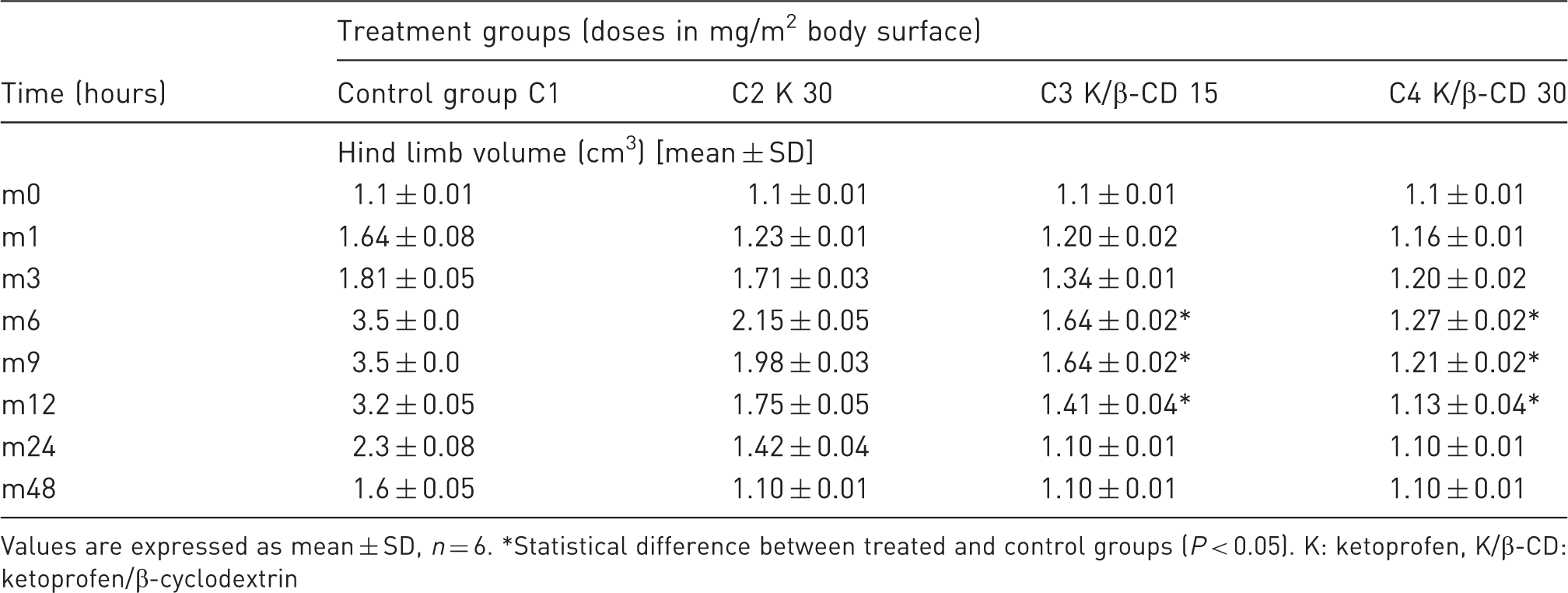
Values are expressed as mean ± SD, n = 6. *Statistical difference between treated and control groups (P < 0.05). K: ketoprofen, K/β-CD: ketoprofen/β-cyclodextrin
In the control group all the definitive clinical signs of a local inflammatory process were obvious. They occurred shortly after applying the stimulus as reddening, edema, temperature increase and pain. The clinical signs intensified gradually from m1 up to m9 when a maximum inflammation of over 100% was reached; these values persisted up to m12, then the inflammation slowly regressed, but remained visible after 48 h. In the treatment groups, the dorso-plantar diameters were markedly reduced compared with the control group. At m6, plethysmometry showed a marked inhibition of the limb edema in groups C4 and C3: 63.71% less, under the dose of 30 mg K/β-CD/m2 body surface, 53.14% less, under 15 mg K/β-CD/m2 body surface, and 38.57% less, in the group treated with ketoprofen. The regression of limb edema in the rats of these groups was visible at each stage of the plethysmometric assessments. The visible onset of the therapeutic (anti-inflammatory) effects was much faster and more intense in the groups treated with K/β-CD compared with the group treated with ketoprofen, even at a dose rate of 15 mg/m2 body surface, and there were no clinical signs of pain (Figure 2).
The dynamic plantar effect in the studied groups of rats.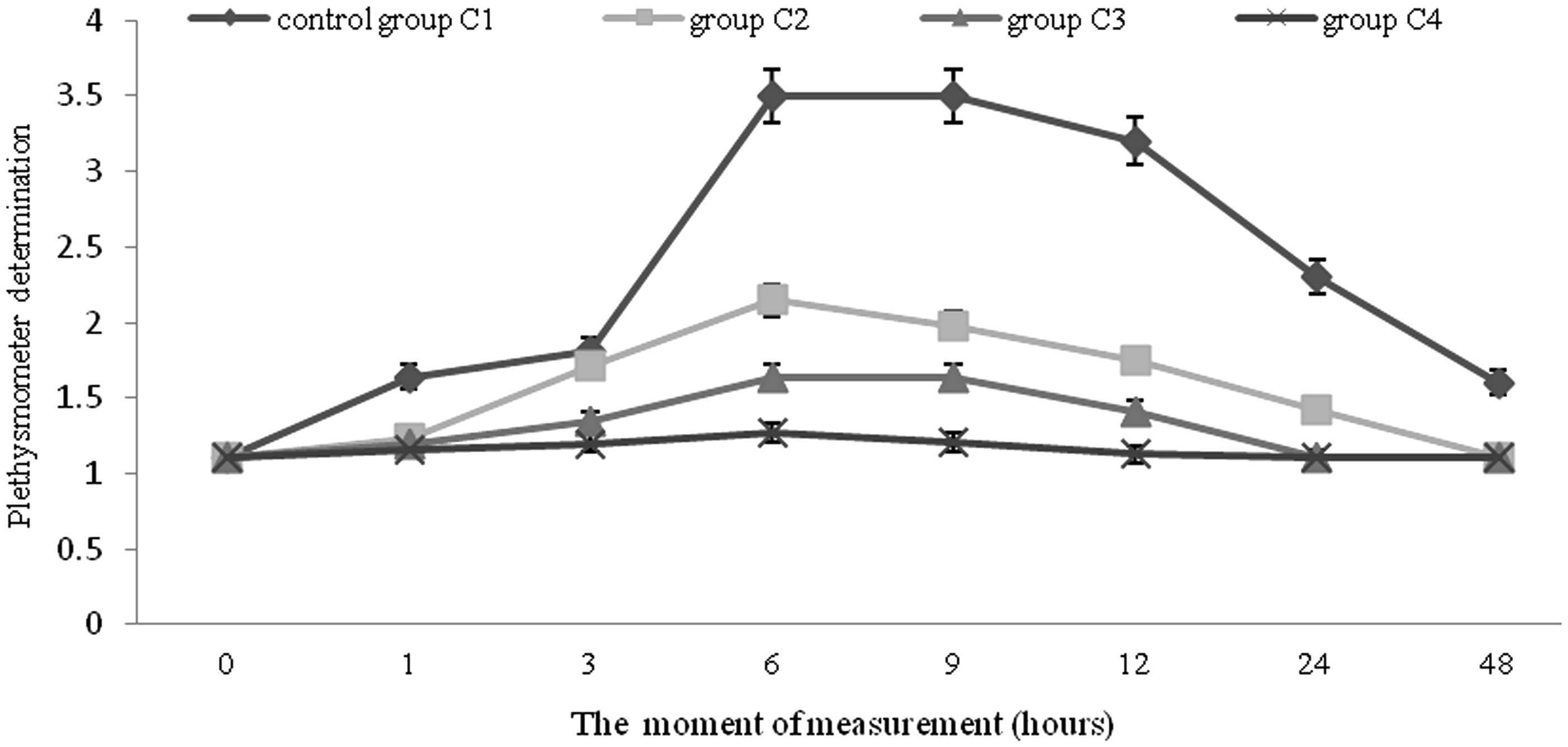
Induction of the experimental peritoneal inflammation
In the groups with induced peritoneal inflammation, the rats showed no changes in physical status. They were euthanized with isoflurane 24 h after NSAID administration and their abdominal cavities were opened to collect peritoneal fluid. Considerable quantities of exudate were collected (2 mL/rat) from the control group and fibrin deposits were observed at the perihepatic level, especially on the visceral side of the hepatic lobes as well as on the mesentery, reflecting the intensity of the inflammatory process. In the ketoprofen-treated group (T2) no fibrin deposits were observed and the quantity of peritoneal fluid collected was moderate (0.75–1 mL/rat). In groups T3 and T4, where animals were treated with K/β-CD, no fibrin deposits were observed and there were no peritoneal fluids.
Spectrophotometric values were high in the control group and lowest in group T4, showing that the intensity of the inflammation was directly correlated with the absorbance readings, at a wavelength of 450 nm. The higher the absorbance, as observed in the control group, the more intense was the inflammatory process. The degree of peritoneal inflammation decrease was inversely proportional to the absorbance value: the lower the absorbance value, the higher the anti-inflammatory effect of the NSAID administered (Figure 3).
Spectrophotometric absorbance values in the studied groups of rats.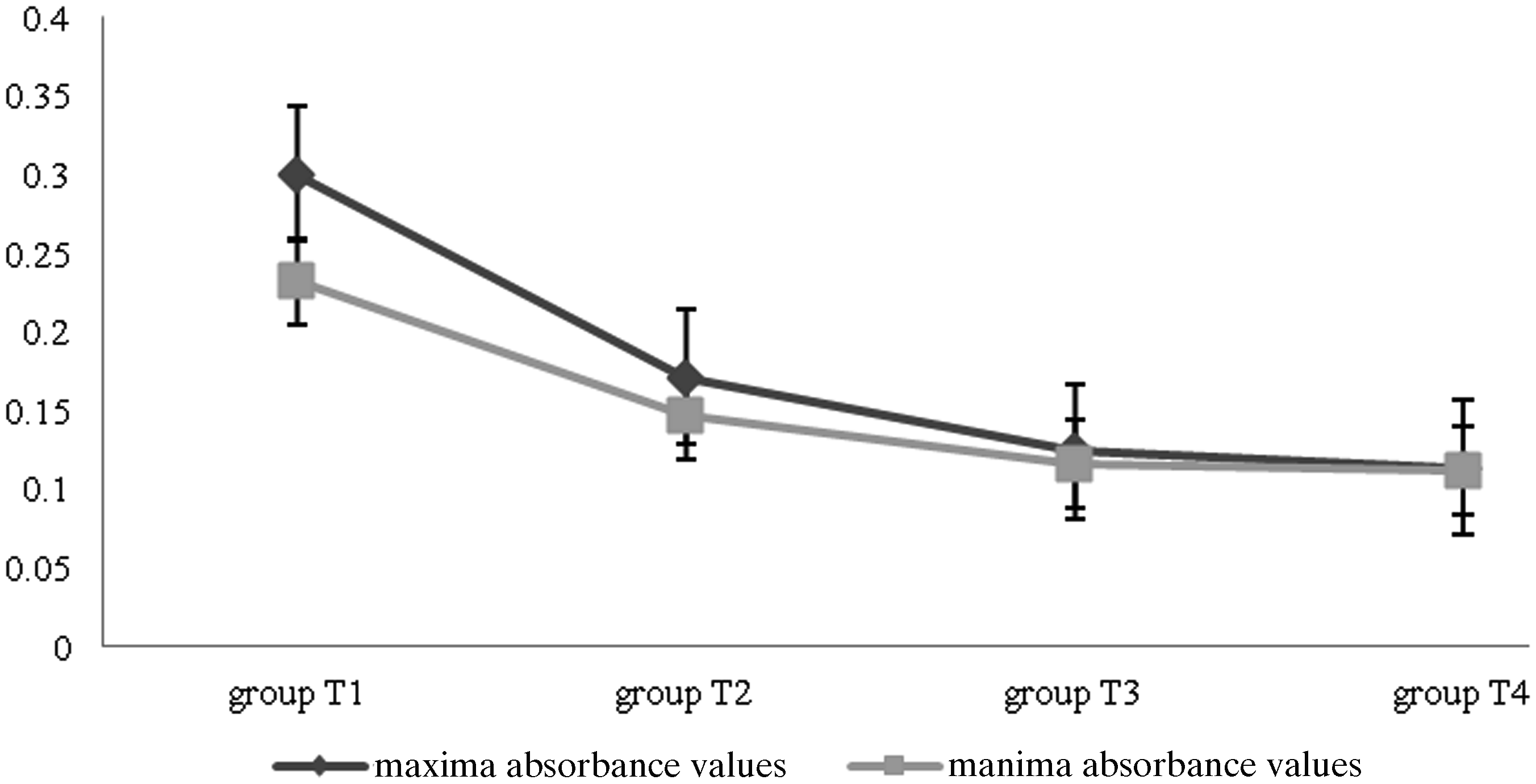
On direct examination of the peritoneal cells from the exudate samples, a high concentration was observed in group T1 and a moderate one in group T2. They were almost absent in groups T3 and T4. In the peritoneal fluid, macrophages and granulocytes were prevalent.
Discussion
The plethysmometer used in our experimental studies allowed reasonably precise and sensitive measurements of plantar edema.
The development of drugs with improved features is a current concern for both fundamental research and the pharmaceutical industry. Encapsulating APIs in cyclodextrins, natural polysaccharides synthesized through a process of enzymatic degradation of starch by cyclodextrin glucosyl-transferase synthesized by Bacillus macerans,20,22,24 has yielded some promising results. Cyclodextrins form inclusion complexes in aqueous media and can improve in vivo formulation dissolution, even if complexation occurs after ingestion. 22 Much research36–38 has focused on describing the improved transport capacity and permeation of drugs from complexes formed with cyclodextrins. The increase of the apparent solubility induces a rise in the thermodynamic activity of the formulation in the transport mechanisms of the cell and, therefore, leads to a rapid release at this level. The significant increases of solubility and dissolution rates of K/β-CD compared with ketoprofen alone were demonstrated in our studies, which found a linear relationship between the quantity of dissolved API and the concentration of cyclodextrin in the solution.
Complexation decreases the speed of decomposition, polymerization and autocatalytic reactions, and reduces sensitivity to light while increasing thermal stability.22,39 This is highly important in the pharmaceutical industry as it allows the preparation of stable aqueous solutions of water insoluble APIs without the need for organic solvents. Loftsson and Duchêne (2007) showed that cyclodextrins can also stimulate the release of API. Enhancement of API release increases absorption by improving bioavailability.40,41 This was confirmed in the present studies, which showed that K/β-CD had a higher efficiency than the non-complexed API. The anti-inflammatory and analgesic effects of the complex were significantly higher and occurred faster, even at low dose rates (15 mg/m2 body surface), in both types of experimental inflammation.
Similar studies of NSAIDs complexed with β-CD23,42–45 have demonstrated that there is an increase in the therapeutic effects (analgesic and anti-inflammatory activities) and stability of the API, and a reduction in gastrointestinal irritation.
Both experimental models of acute inflammation used in this study are reproducible and are very sensitive with respect to the pathophysiology of the process, clinical signs and obtained results. The experimental model of inflammation, with the ability to measure inflammatory edema and other changes at the level of the plantar acropodium in rats, has proved to be very important in the testing and assessment of the analgesic and anti-inflammatory effects of the ketoprofen alone or complexed with β-CD. For peritoneal inflammation, evidence of acute signs of the inflammatory process could be seen using spectrophotometric analysis of peritoneal fluid samples and nucleated cell counts. The absorption spectrum was sensitive enough to reveal structural information with amplitude depending on the API concentration. The intense migration of macrophages and granulocytes into the peritoneal fluid, as well as the high spectrophotometric absorbance values reflected pronounced inflammation in the control group of rats, less inflammation in the ketoprofen-treated group and little inflammation in the K/β-CD-treated groups. In summary, it is possible to confirm that forming an inclusion complex compound containing an API (K/β-CD) contributes to the increase of the dissolution and solubility of the API, which leads to increased bioavailability resulting in a longer and more intense therapeutic activity compared with ketoprofen alone.
In conclusion, there was a significant difference in the anti-inflammatory and analgesic activity in K/β-CD-treated rats, even when the dose rate was halved, compared with the therapeutic effect of the ketoprofen-positive control in both models of inflammation. This type of β-CD complex may be a suitable formulation for NSAIDs used in the treatment of pain and inflammation in both veterinary and human medicine.
Footnotes
Acknowledgements
The authors wish to thank Nina Davidescu for technical support, and Bogdan Minea for assistance with English language.
