Abstract
Repeated blood sampling from laboratory animals is desirable in certain experimental designs and also for reducing the number of animals used in research. Biochemical methods for analysing blood samples require only small blood volumes to be collected (typically 20–40 µL). In juvenile mice, the small blood volume of the animals also requires only small samples to be taken. Furthermore, for behavioural studies it is desirable to have a method that does not require anaesthesia or the use of invasive indwelling cannulae. We report the validation of a refined method for repeated blood sampling (up to 3 times at 24 h intervals) in juvenile and adult mice using the tail incision method to sample from the lateral tail vein. This method is not stressful, as assessed by low basal levels of the stress hormone corticosterone. Since repeated blood samples can be collected from the same animal at multiple time points, it is not necessary to increase group size for terminal sample collection. Thus, in addition to being a refined method requiring no warming of the tail, no anaesthesia and only gentle restraint, this method also reduces the numbers of mice used for experiments.
Numerous techniques exist for the collection of blood from mice. 1 While taking blood samples from mice is a common practice in the laboratory setting, the procedure can be stressful due to the handling, restraint or anaesthesia involved in the procedure.1,2 Taking repeated blood samples from the same mouse allows a significant reduction in the number of mice used over the course of an experiment. Hence a refined minimally invasive method of blood sampling, which allows multiple samples to be taken from the same animal, is desirable. 3 However, in the UK, Home Office guidelines limit repeated sampling and indicate that no more than 10% of the total blood volume from a mouse may be taken on each occasion, with no more than 25% being taken over a 28-day period.2,4
In our research we are interested in studying the behavioural effects of stress, over time, in juvenile mice and assessing the impact of stress via measurements of neuroendocrine function, e.g. corticosterone analysis. This presents several challenges when considering repeated blood sampling methodology. Firstly, the stressful nature of the blood sampling technique itself may confound the results of corticosterone analyses. Secondly, when working with juvenile mice (3–6 weeks old), which weigh as little as 12 g, this limits the volume that can be taken on each occasion to less than 60 µL. The lateral tail vein is an appropriate route for repeated sampling of small blood volumes from mice without anaesthesia, although vasodilation may be required.2,4 We compared three different blood collection techniques in adult mice before adapting and validating the method developed by Fluttert et al. 5 as a refined method for repeated blood sampling in juvenile mice.
Male BALB/cAnNCrl mice (Charles River, Margate, UK), aged 3–4 weeks old (juvenile) or 7–8 weeks old (adult), were housed individually in 35 × 20 × 15 cm polysulfone cages (Plexx, Elst, The Netherlands) with woodchip bedding (Datesand, Manchester UK) and paper nesting material (Lillico/LBS Biotechnology, Horley, UK). The mice were maintained in a temperature (21 ± 1℃) and humidity (50–60%) controlled environment, under a 12 h light/dark cycle (lights on 07:00 h), with food and water available ad libitum. All the mice were allowed to acclimatize to the animal facility for at least one week prior to blood sampling. During this time the animals were handled by gentle cupping 6 for 2–3 min on 2–3 days prior to blood sampling. All procedures were carried out under a Home Office project licence held in accordance with the Animals (Scientific Procedures) Act 1986.
Adult mice had a blood sample taken from the lateral tail vein by one of three methods (n = 5 per treatment group). All the samples were collected in heparinized capillary tubes (Fisher Scientific, Loughborough, UK) between 11:00 h and 13:00 h. The first method involved warming the mice in a thermostatically controlled warming chamber, at 38℃ for 15 min in line with guidelines on the NC3Rs microsampling website. 2 The mice were then briefly restrained in a restraint device, a 25 G needle was inserted into the lateral tail vein, and resulting blood droplets were collected. The second method involved holding the mice by hand, and immersing the tail in warm water at 42℃ for 20 s. A 25 G needle was again inserted into the lateral tail vein, and resulting blood droplets were collected. The third method was the tail incision method 5 which involved one person gently cupping the mice by hand, 6 while the operator held the tail gently on the bench and made a small nick (approximately 2 mm wide × 0.5 mm deep) in the tail with a razor blade, perpendicular to the tail vein, approximately 2 cm from the tip of the tail. Blood droplets were directly collected into capillary tubes. In the tail incision method blood flow was encouraged by gently stroking the tail and in the majority of cases, blood flow stopped spontaneously when stroking was stopped. On occasion it was necessary to apply a small amount of pressure to the tail to stop bleeding. In all cases, the blood samples were immediately transferred to microcentrifuge tubes containing EDTA (final concentration in sample 3 µg/µL), and stored on ice. Twenty-four hours after the blood sample was taken, all the mice were culled by cervical dislocation, rapidly decapitated and a second, terminal blood sample was collected directly into microcentrifuge tubes containing EDTA (final concentration in sample 3 µg/µL). All the tubes were centrifuged at 2000 relative centrigual force (rcf) for 20 min at 4℃. Plasma was removed and stored at −20℃ until analysis. The concentration of corticosterone in each plasma sample was determined using a corticosterone enzyme-linked immunosorbent assay (ELISA) (IBL International, Hamburg, Germany). Plasma was diluted 1:10, and the ELISA was carried out according to the manufacturer's instructions.
In adult mice (Figure 1), two-way repeated measures mixed model analysis
7
revealed a significant main effect of sampling method (F(2,12) = 7.08, P < 0.01), baseline or terminal sample (F(1,12) = 21.16, P < 0.001) and a significant method*sample interaction (F(2,12) = 10.78, P < 0.005). Post hoc analysis revealed that corticosterone was significantly higher in samples taken by the warming cabinet method compared with both the tail warming method (330 ± 58 ng/mL versus 110 ± 13 ng/mL, P < 0.005) and the tail incision method (330 ± 58 ng/mL versus 74 ± 13 ng/mL, P < 0.001), clearly indicating that the warming cabinet was stressful (Figure 1). There was no significant difference between the tail warming and tail incision methods (110 ± 13 ng/mL versus 74 ± 13 ng/mL, P = 0.18), or between the tail warming/incision methods and their respective terminal samples (Figure 1).
Effect of three different blood sampling methods on plasma corticosterone levels in adult (7–8 weeks old) BALB/cAnNCrl male mice. ‘Baseline’ samples were obtained from the lateral tail vein following either warming in a cabinet, or tail warming by immersion in water or tail incision method (no warming). Animals were killed 24 h later by cervical dislocation and ‘terminal’ samples were obtained. Results are expressed as mean ± SEM, n = 5/group. **P < 0.005, ***P < 0.001 (two-way repeated measures mixed model analysis).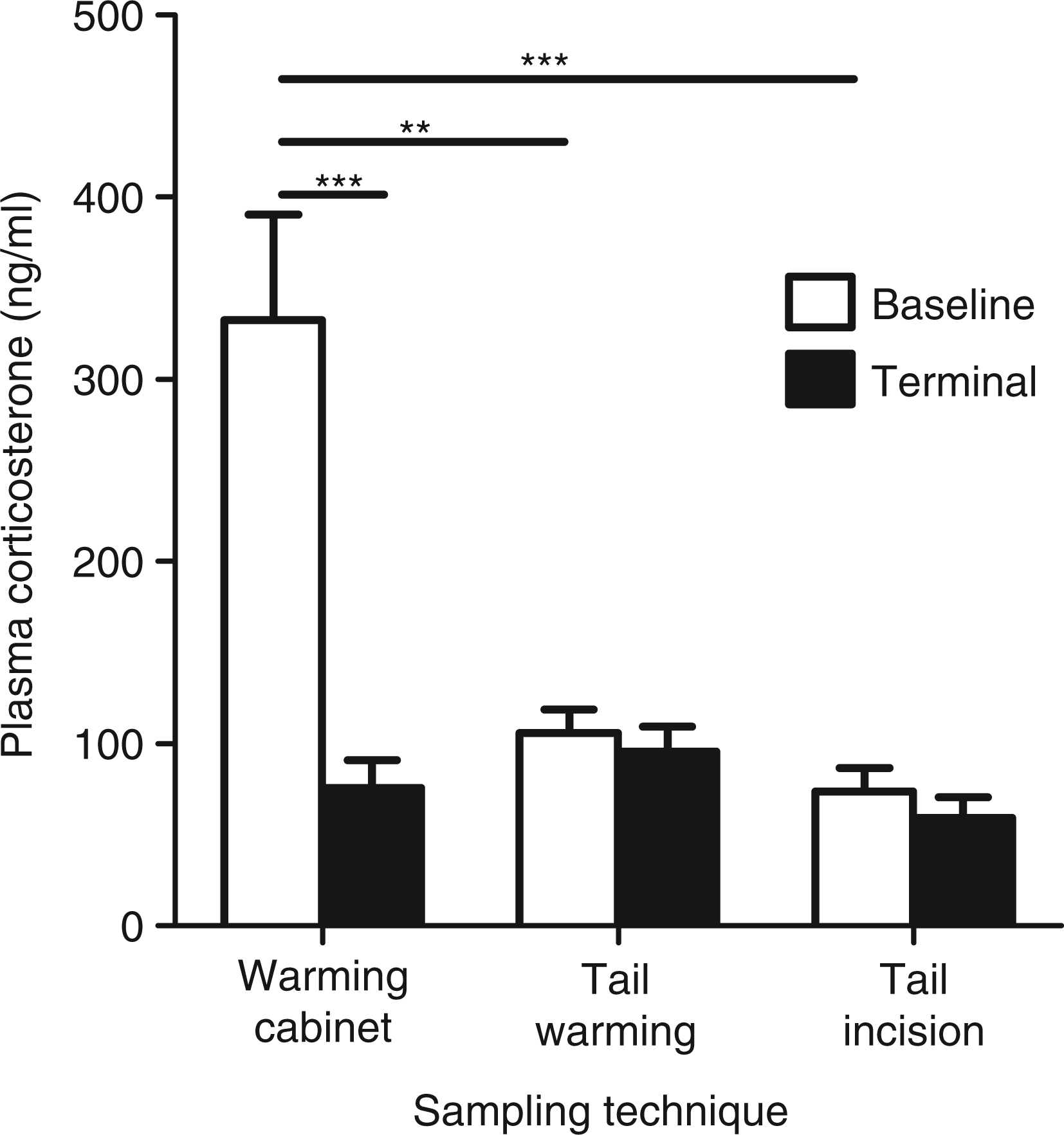
We then went on to develop the tail incision method for repeated sampling in juvenile mice (n = 4/5 per treatment group). Blood samples were taken from juvenile mice (1) at baseline, (2) 24 h later following a 2 h restraint stress and (3) 24 h post-stress. A tail incision was made starting approximately 2 cm from the tip of the tail, and on repeated sampling a different location was used, working towards the base of the tail in 0.5 cm increments. Corticosterone was significantly increased by 2 h of restraint stress compared with baseline (720 ± 59 ng/mL versus 21 ± 9.8 ng/mL), and this increase had returned to baseline within 24 h following stress (24 ± 3.4 ng/mL versus 720 ± 59 ng/mL, Figure 2a). We then confirmed that heating and restraining mice to take a blood sample was stressful in juvenile mice in the same way as in adults. Mice had a blood sample taken at baseline by the warming cabinet method. Twenty-four hours later they were restrained for 2 h and a second blood sample was taken in the same way. The results confirmed our previous findings that this method of taking blood is stressful (Figure 2b).
Effect of stress on plasma corticosterone in juvenile (4–5 weeks old) male BALB/cAnNCrl mice following repeated blood collection. (a) Blood samples were taken on three successive days using the tail incision method at baseline (day 1), immediately following stress (day 2) and 24 h post-stress (day 3). (b) Blood samples were taken by the warming cabinet method at baseline (day 1) and immediately following stress (day 2). Results are expressed as mean ± SEM, n = 4–5/group. *P < 0.05, ***P < 0.001 (two-way repeated measures mixed model analysis (a), paired t-test (b)).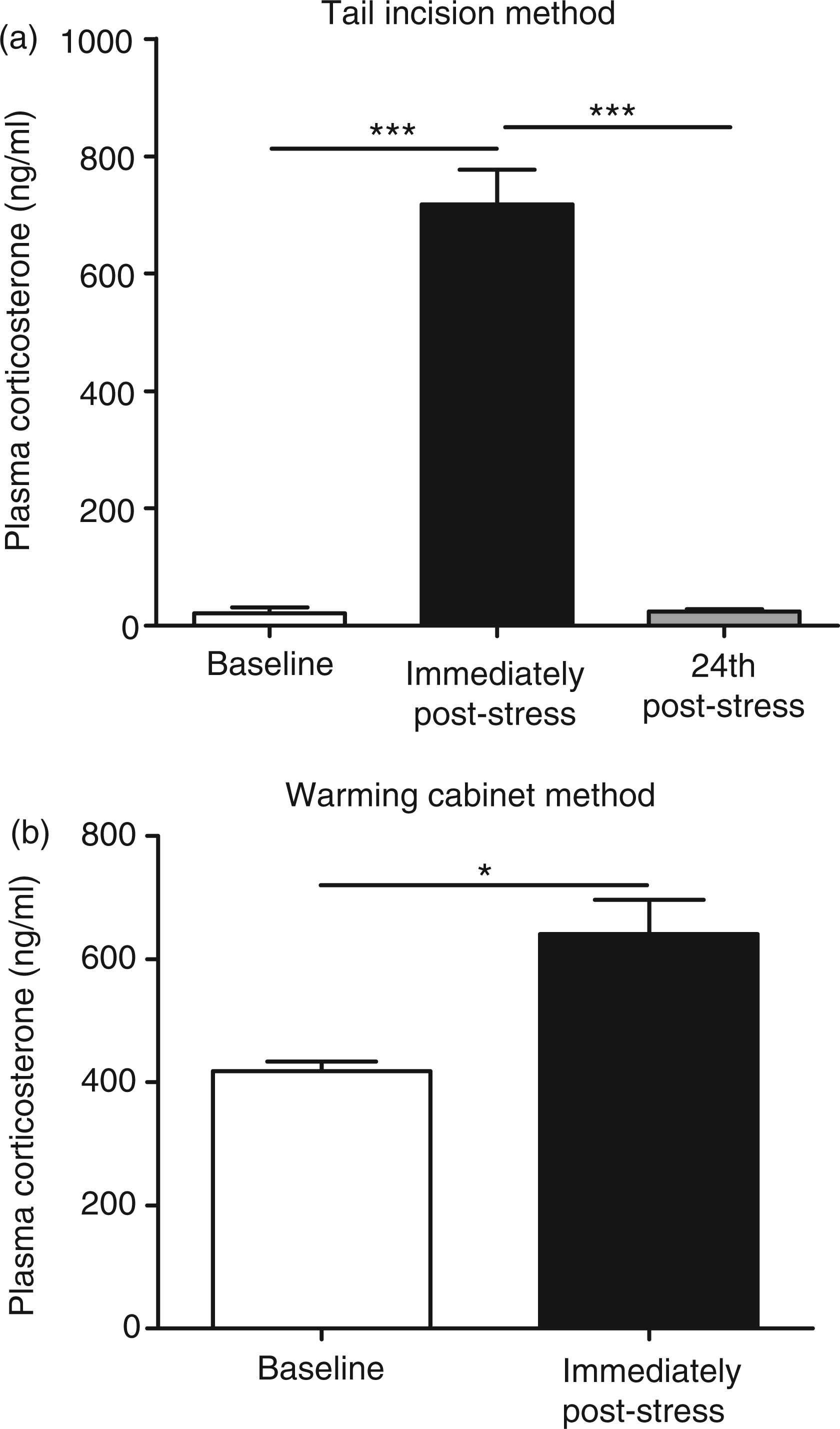
Here, we have demonstrated that the tail incision method is a refined method of taking repeated blood samples from juvenile mice. The low basal levels of the stress hormone corticosterone obtained using the tail incision method compared with the warming cabinet method indicate that this is not a stressful procedure. Furthermore, the repeated nature of sampling is not stressful as the third sample taken using the tail incision method did not increase plasma corticosterone levels above the baseline sample on the first day of sampling (Figure 2a). This method was originally developed for use in adult rats, 5 particularly for use in behavioural studies where implantation of indwelling cannulae is not desirable or methods like tail-cuts are unsuitable for repeated sampling. Our data demonstrate that this methodology is applicable to juvenile mice. Furthermore, we have shown that in both adult and juvenile mice, the use of a warming chamber is stressful, producing a significant ∼3-fold increase in plasma corticosterone levels. The stressful nature of these techniques therefore precludes their use in studies examining effects of stress. An additional benefit of the tail incision method is that it allows blood flow to be started and stopped easily, facilitating the collection of very small volumes of blood (typically 20–40 µL) from juvenile mice. Pilot studies in our laboratory involving the use of the saphenous vein were not successful in this respect. Finally, the ability to take repeated samples from the same mouse at multiple time points reduces the number of animals required for an experiment, compared with the taking of terminal blood samples from large numbers of mice, and is thus in keeping with the principles of both reduction and refinement of animal use in laboratory research. 3
Footnotes
Acknowledgements
Lesley Moore, Jean Tye, Jane Graham and Alan James in the Biosciences Unit at the University of Bath assisted in these studies. AMS was funded by an MRC Doctoral Training Grant Studentship (MR/J500318/1) and an MRC In Vivo Strategic Skills Award Supplement (SJB: G1000380).
