Abstract
Keywords
Introduction
Posttraumatic stress disorder (PTSD) is a serious mental health condition that can result from surviving a traumatic event and interfere with work performance, interpersonal relationships, and functioning, creating a significant burden on a person’s daily life. PTSD affects 4% to 5% of the population worldwide and between 13.5% and 17% of veterans (Dursa et al., 2014; Hoge et al., 2004; Kessler et al., 2004). People with PTSD are at greater risk of suicide and other comorbidities, including increased chronic pain and cardiovascular disease (Baker et al., 1997; Conner et al., 2014; Edmondson & Cohen, 2013). Available treatments for PTSD include various forms of trauma-focused talk therapies and two selective serotonin uptake inhibitors (paroxetine and sertraline) that are approved by the U.S. Food and Drug Administration (Bradley et al., 2005; Cloitre et al., 2012; Ipser et al., 2006; Lee et al., 2016). However, patients often do not respond to or tolerate available treatments, discontinue treatment, and/or experience relapse, which warrants further investigation into novel treatments (Goetter et al., 2015; Lee et al., 2016).
3,4-Methylenedioxymethamphetamine (MDMA) is a monoamine releasing phenethylamine which possesses unique pharmacotherapeutic properties that may enhance traditional talk therapy by making trauma memory processing more tolerable (Feduccia et al., 2018). MDMA has been proposed to augment therapy through one or more of these processes: reducing fear in the face of emotionally distressing memories, fear-extinction learning, greater compassion for self and others, increased interpersonal affiliation, and an enhanced therapeutic alliance (Carhart-Harris et al., 2014; Gabay et al., 2019; Hysek et al., 2014; Young et al., 2015). MDMA-assisted therapy (MDMA-AT) typically consists of a cotherapist dyad who provides participants with talk therapy in preparatory sessions, followed by active MDMA-AT sessions, and each with several associated integrative sessions. Early Phase 2 trials of MDMA-AT for treatment of PTSD have shown significant and even potentially long-term reductions in PTSD symptom severity (Mithoefer et al., 2011; Mithoefer et al., 2013; Mithoefer et al., 2019; Oehen et al., 2013; Ot’alora et al., 2018). Specifically, pooled analysis of participants across six Phase 2 trials indicated that nearly 67% no longer met PTSD criteria at least 12 months after their last active MDMA-AT treatment (Jerome et al., 2020). Study limitations however including variations in study design and methods require corroboration of these results.
In preparation for the Phase 3 trials, the sponsor conducted two multisite open-label lead-in studies of MDMA-AT to train and select therapists across the United States (MP16 study) and Canada (MP17 study). The studies provided new cotherapist dyads the opportunity to administer MDMA-AT under clinical supervision. To maximize treatment fidelity across multiple study sites, adherence raters systematically evaluated the new cotherapist dyads’ adherence to the therapy manual under clinical supervision by the therapy training team. Cotherapist dyads were cleared to participate in Phase 3 trials following administration and evaluation of MDMA-AT under clinical supervision. The primary aim of the present study was to examine treatment efficacy and safety across multiple clinic sites and therapy dyads. The analysis combined data from the two multisite open-label lead-in studies to assess MDMA-AT treatment adherence, PTSD symptom reduction, and treatment safety.
Method
Participants and Study Design
Across 12 U.S. study sites and 2 Canadian sites, a total of 37 unique cotherapist dyads provided MDMA-AT for treatment under clinical supervision among participants with severe PTSD. Study sites included private practice clinics in Charleston (SC), Boulder (CO), Fort Collins (CO), Los Angeles (CA), New Orleans (LA), San Francisco (CA), New York (NY), Boston (MA), Vancouver (British Columbia, Canada), and Montreal (Quebec, Canada); and the University of California, San Francisco (UCSF, CA), University of Connecticut (UC, CT), University of Wisconsin Madison (WI), and New York University (NY). Sites ranged from one to four cotherapist dyads, and each unique dyad treated one participant. Study participants were recruited from November 2017 to March 2019 via internet advertisements, provider referrals, and by word-of-mouth. Study sites conducted telephone screenings to assess initial eligibility prior to inviting participants on-site for further screening.
Eligibility criteria included confirmation of severe PTSD, which was defined as having a CAPS-5 Total severity score of 35 or greater (Weathers et al., 2017). Participants were asked to agree to the study protocol including lifestyle modifications. Exclusionary criteria included past or present psychotic disorder, Bipolar I disorder, pregnancy or lactation, current diagnosis of a substance use disorder (except for caffeine or nicotine), uncontrolled hypertension, weighing less than 48 kg, and other medical conditions contraindicated for MDMA such as cardiac conditions or cerebrovascular disease. Participants who were at serious risk of suicide or posed a risk to others were also ineligible. Participants with controlled hypertension underwent additional screening to confirm the absence of clinically significant underlying cardiovascular disease. Participants who were enrolled into the study were asked, under the supervision of a physician, to taper off psychiatric medications and any other medications that might have interfered with the effects or metabolism of MDMA.
Treatment
The MDMA-AT therapeutic approach is detailed in the “Manual for MDMA-Assisted Therapy in the Treatment of PTSD,” published by MAPS (MDMA Treatment Manual, available at maps.org/treatment-manual). MDMA-AT was conducted over a duration of 9 to 15 weeks. Treatment periods consisted of three preparatory sessions before the first administration of MDMA and three MDMA experimental sessions, in which each session was followed by three integrative sessions. In preparatory sessions, participants met with their cotherapist dyad to develop therapeutic rapport, discuss their PTSD symptoms, and the upcoming MDMA-AT session. Therapists provided information on what to expect during the MDMA-AT sessions, including drug effects and strategies to manage any challenging experiences that may emerge.
Participants were offered a total of three open-label MDMA-AT sessions that were scheduled 3 to 5 weeks apart. In the first experimental session, participants were administered a divided dose of 80 mg MDMA initial + 40 mg MDMA supplemental (United States) or 100 mg MDMA initial + 50 mg MDMA supplemental (Canada). Supplemental doses were administered 1.5 to 2 hours after the initial dose. The purpose of the supplemental dose was to enable a longer period to process trauma during MDMA-AT sessions without significantly impacting the intensity of pharmacodynamic effects. The second and third experimental sessions utilized slightly higher divided doses of 120 mg MDMA + 60 mg MDMA (United States) and 125 mg MDMA + 62.5 mg MDMA (Canada). The nominal difference in MDMA doses between countries was due to drug availability, where U.S. participants received racemic MDMA synthesized by David Nichols, PhD (Purdue University) and Canadian participants received racemic MDMA from Lipomed AG Switzerland.
Therapy during MDMA-AT sessions consisted of periods of introspection alternating with periods of communication between the participant and the cotherapist dyad. Participants were encouraged to remain with trauma-related memories, feelings, and/or thoughts as the cotherapist dyad provided support. MDMA-AT sessions lasted 6 to 8 hours and ended after drug effects returned to baseline. Participants remained overnight at the site with a night attendant, except for four participants who did not stay overnight as part of a safety substudy. After each MDMA-AT session, participants received several follow-up visits, including three integrative sessions, where therapists facilitated participants’ continued emotional processing, addressed any difficulties following the MDMA-AT session, and helped participants to apply any benefits gained in the MDMA-AT sessions to daily life. Participants worked with the same cotherapist dyad throughout the entire treatment period. The therapeutic approach is detailed in the MDMA Treatment Manual.
Therapy Training and Adherence Rating
As part of the open-label lead-in studies, therapy trainees completed the MDMA Therapy Training Program, which included approximately 120 hours of both online and in-person courses and experiential learning (details about the current MDMA Training Program curriculum available at mapspublicbenefit.com/training). Trainees were recruited from mental health and medical professions and were required to demonstrate experience providing therapy or counseling services to adults affected by trauma. Trainees were paired in cotherapist dyads according to professional training and experience, so that every cotherapist dyad had at least one psychotherapist with extensive clinical experience. Clinical supervisors evaluated each therapy trainee working on the open-label studies to deliver MDMA treatment under clinical supervision. Cotherapist dyads were systematically assessed on their treatment fidelity using an adherence rating checklist that was developed by experienced therapists and trainers.
The aim of the adherence ratings was to measure consistency of MDMA-AT that was delivered across all of the study sites using adherence criteria described in the “Manual for Adherence Ratings of MDMA-Assisted Psychotherapy for Treatment of Posttraumatic Stress Disorder” published by MAPS (available at maps.org/treatment-manual). Raters were graduate-level professionals in psychology, social work, or psychiatry who had a minimum of 1-year experience working with adults affected by trauma and had completed a 50-hour Adherence Training Program and interrater reliability testing. Raters and supervisors reviewed cotherapist dyads’ session videos and evaluated them against the “Manual for Adherence Ratings for Treatment Fidelity.” Dichotomous Adherence Criteria (Yes/No) covered a range of factors, from communicating treatment expectations to nurturing the therapeutic alliance and building trust, across each of the preparatory, experimental, and integrative sessions. For each cotherapist dyad, adherence ratings were performed on at least their first 10 study visits. Each rated session was reviewed in full and rated independently. In the present study, adherence rating percentage scores were calculated as follows: for preparatory sessions, each of the 24 adherence criterion were expected to be met at least once across their three rated preparatory sessions; for experimental sessions, scores were calculated independently, with each of the 20 adherence criterion expected to be met in each rated experimental session; and for integrative sessions, each of the 12 adherence criterion were expected to be met at least once across each set of three integrative sessions that took place after an experimental session. In addition to adherence ratings, raters provided qualitative comments on sessions for clinical supervisors’ review.
PTSD Measure
The primary outcome measure of PTSD symptoms was change in Total Severity scores from the Clinician-Administered PTSD Scale for DSM-5 (CAPS-5). The CAPS-5 is a semistructured 30-item interview to assess past-month presence and severity of PTSD through diagnostic and symptom severity scores, with good internal consistency, concurrent validity, and test/retest reliability (Weathers et al., 2013). CAPS-5 Total Severity scores range from 0 to 80, with higher values indicating greater symptom severity. The CAPS-5 interviews were administered remotely via live video conferencing by a centralized pool of trained independent raters (IRs). IRs were not present during any therapy sessions, remained blinded to the study design, and were scheduled on the basis of their availability. CAPS-5 was assessed at baseline and within three weeks after each MDMA-AT session (visit 8, visit 13, and primary endpoint visit 19). All CAPS-5 assessments for each participant were performed by different IRs.
Safety Measures
Vitals including systolic blood pressure, diastolic blood pressure, heart rate, and body temperature were measured during each experimental session, with measurements occurring once prior to drug administration, at least once 1.5 to 2 hours after initial MDMA dose administration and prior to supplemental dose, and once approximately 6 to 7 hours after initial dose of MDMA.
Adverse events (AEs) were collected throughout the study and gathered during face-to-face visits, telephone calls, and/or correspondence with therapists or site staff. AEs were rated by the site physician as mild (causing no limitations on daily activities), moderate (some limitation in daily activities), or severe (preventing performance of daily activities). The site investigators also assessed for “serious” AEs, which is defined as “fatal, life-threatening, requires or prolongs inpatient hospitalization, or produces persistent and significant disability.” AEs were coded using the Medical Dictionary for Regulatory Activities (MedDRA 20.0) and overseen by a medical monitor. In the present analysis, AEs were counted at the participant-level across an aggregate of the three experimental sessions for day of, 1 day postexperimental, and 2 days postexperimental session.
Suicidal ideation and behavior were measured with the Columbia Suicide Severity Rating Scale (C-SSRS) and were assessed at baseline and during visits and select phone calls throughout the study. The C-SSRS is a clinician-administered structured interview that measures presence and intensity of suicidal ideation and behavior during the assessment period (lifetime or since last assessment; Posner et al., 2007).
Statistical Analysis
All outcome analyses were based on participants who received MDMA in at least one experimental session and had at least one follow-up CAPS-5 assessment. Nominal variables were described in terms of frequencies and percentages. Ordinal and nonnormal continuous variables were described using the sample mean and range and were analyzed by nonparametric statistical tests. Approximately normal variables were described using sample mean and standard deviations, presented as M (SD) throughout, and analyzed by parametric statistical tests. Treatment efficacy was used to estimate the mean change in CAPS-5 Total Severity scores from baseline (Visit 3) to primary endpoint (Visit 19), which was 18 weeks postbaseline. Least-squares means from a restricted maximum likelihood-based model for repeated measures was used to estimate the change in CAPS-5 across timepoints. All analyses were carried out with SAS Version 9.4 (SAS Institute, Cary NC).
Results
Participant Sample
A total of 308 people were screened for study eligibility between November 2017 and March 2019, and 37 participants were enrolled. Across the 14 investigative sites, 37 cotherapist dyads each treated one participant. Only one participant did not complete the primary endpoint assessment; before the start of the second MDMA-AT session, the participant withdrew from the study due to an AE of increased nightmares which was reported as mild. The sample consisted of 15 men (40.5%) and 22 women (59.5%), mean age was 35.6 (10.8) years, and the majority of participants were White (73.0%) and not Hispanic or Latino (94.6%). Five veterans were enrolled, with three (8.1%) having index traumas related to combat. Mean baseline CAPS-5 Total scores were 45.4 (7.2). Many participants reported a history of having had multiple traumas (81.1%) or developmental trauma (78.4%). The mean (SD) duration of PTSD was 10.3 (11.0) years. C-SSRS indicated 89.2% of participants reported a history of lifetime suicidal ideation and 40.5% a lifetime history of suicidal behavior. See Table 1 for demographics and baseline characteristics. The MDMA supplemental dose was withheld by the investigator in two instances for two participants due to occurrence of tachycardia and elevated blood pressure following the initial dose, though neither required any medical intervention or further medical evaluation.
Demographics and Baseline Characteristics.
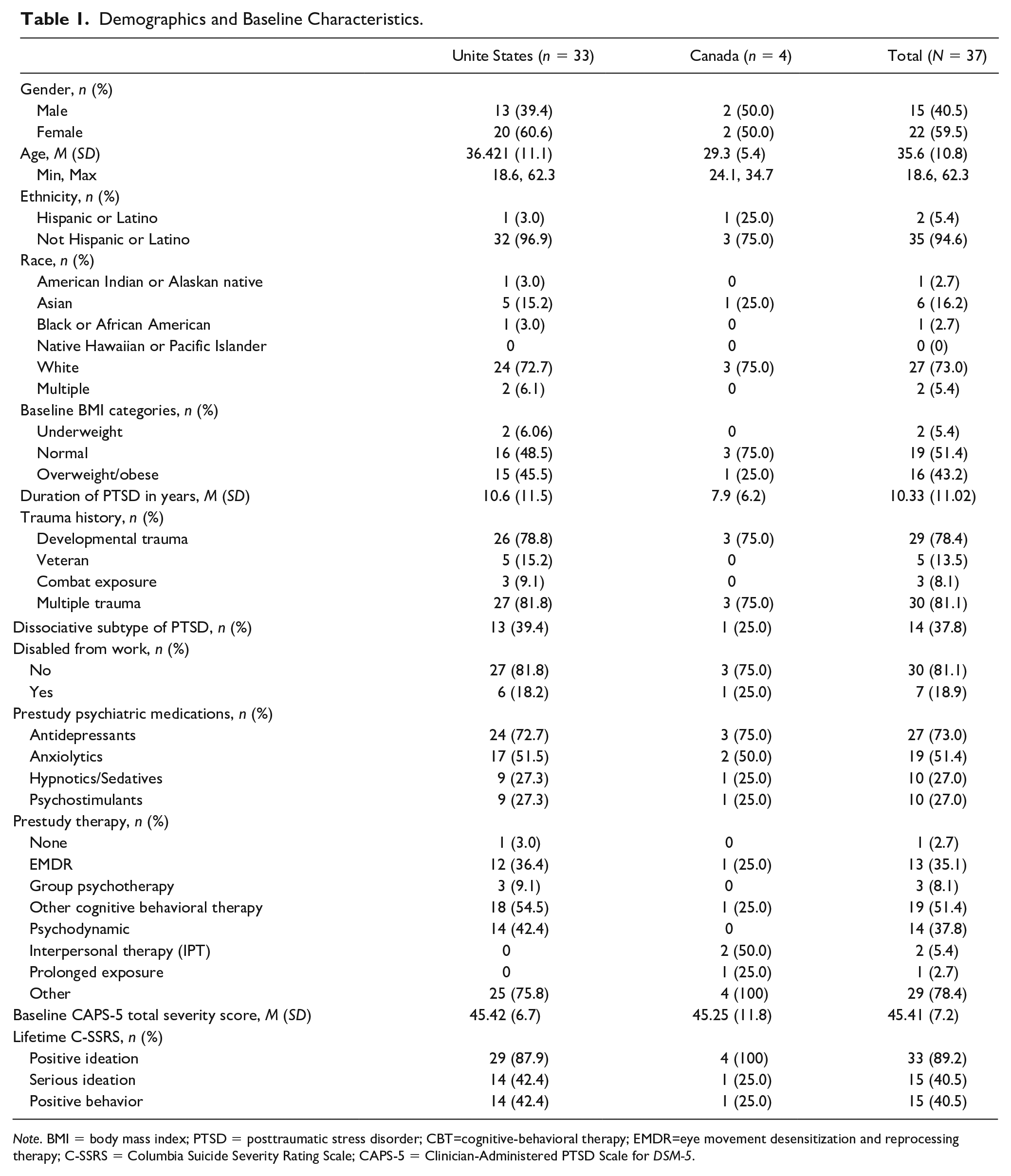
Note. BMI = body mass index; PTSD = posttraumatic stress disorder; CBT=cognitive-behavioral therapy; EMDR=eye movement desensitization and reprocessing therapy; C-SSRS = Columbia Suicide Severity Rating Scale; CAPS-5 = Clinician-Administered PTSD Scale for DSM-5.
Adherence Rating Scores
Across 37 cotherapist dyads, a total of 375 study sessions were reviewed and rated for adherence. An average of 10 sessions were rated per participant (range = 9-14), which consisted of three preparatory, two experimental, and five integrative sessions per cotherapist dyad. Seven sessions that were initially assigned for adherence rating (one preparatory, three experimental, and three integrative sessions) were not included in the analysis due to missing ratings or recording errors. Thirteen additional sessions involving three dyads were rated for adherence in replacement of sessions that were unable to be rated or as requested by a supervisor for further assessment. Overall, adherence ratings were high; cotherapist dyads scored a mean of 95.1% (SD = 3.7%, range = 84.6% to 100%). Across session type, cotherapist dyads scored a mean adherence rating of 92.0% (SD = 7.2%; range = 75.0% to 100%) for preparatory sessions, 98.0% (SD = 3.3%; range = 85.0% to 100%) for experimental sessions, and 93.1% (SD = 9.8%; range = 58.3% to 100%) for integrative sessions. Table 2 presents overall adherence rating scores by site and by country.
CAPS-5 Total Severity Scores by Site.
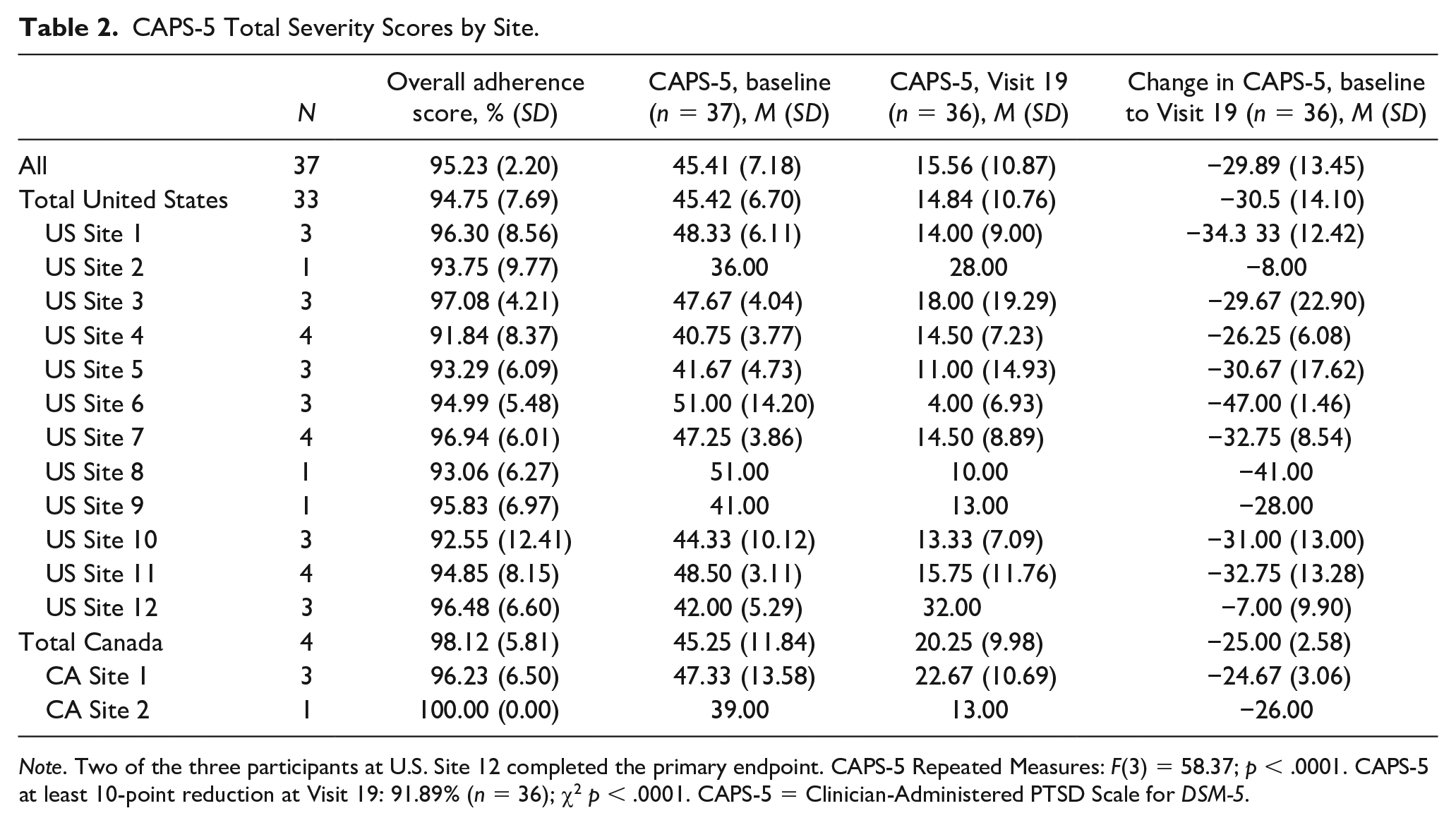
Note. Two of the three participants at U.S. Site 12 completed the primary endpoint. CAPS-5 Repeated Measures: F(3) = 58.37; p < .0001. CAPS-5 at least 10-point reduction at Visit 19: 91.89% (n = 36); χ2 p < .0001. CAPS-5 = Clinician-Administered PTSD Scale for DSM-5.
Efficacy Outcome
The primary efficacy analysis was change in CAPS-5 Total Severity scores from baseline (M = 45.41, SD = 7.18) to the primary endpoint (M = 15.56, SD = 10.87). At the primary endpoint, there was a significant mean change in CAPS-5 scores of −29.89 (SD = 13.45, p < .0001) indicating improvement in PTSD symptoms (Cohen’s d = 2.2; confidence interval [1.97, 2.47]). Figure 1 presents CAPS-5 scores by visit. The primary aim of the open-label studies was to train new cotherapist dyads to lead into the sponsor’s larger Phase 3 trials. In general, efficacy of MDMA-AT was comparable between the United States (mean change = −30.5, SD = 14.1) and Canada (mean change = −25.0 (SD = 2.58). Across all sites, the mean change in CAPS-5 scores ranged from -7.00 (9.90) to −47.00 (1.46). Table 2 presents CAPS-5 scores by country and site. At the primary endpoint (Visit 19), 91.89% (n = 34) of all participants had a clinically meaningful reduction (10 points or greater) in their CAPS-5 scores (p < .0001) and 75.68% (n = 28) no longer met PTSD criteria (p < .001).
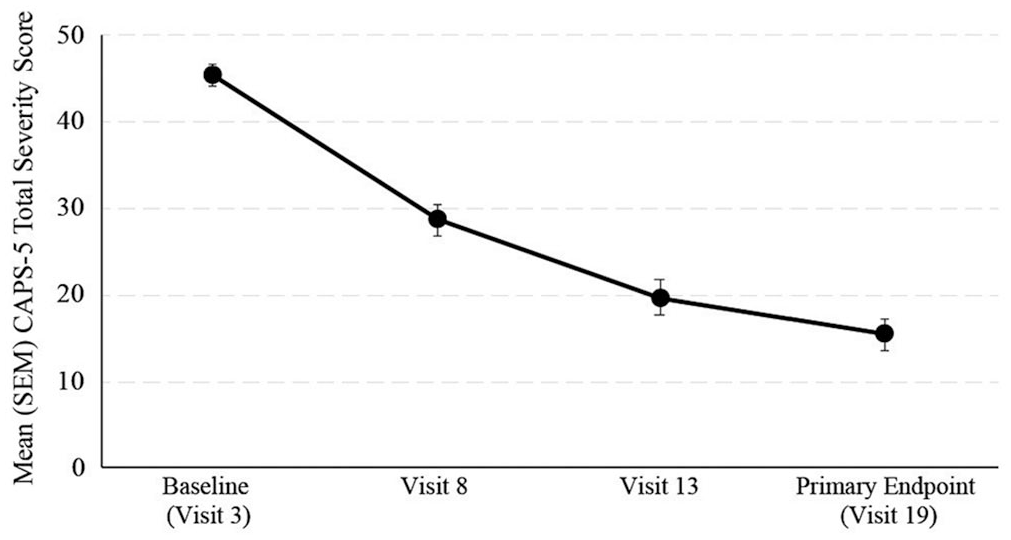
CAPS-5 total severity scores by visit.
Safety Outcomes
During MDMA-AT sessions, there were transient increases in blood pressure and pulse that returned to normal levels by the end of the sessions.
The most common AEs following treatment at the participant-level were headache (68%), muscle tightness (49%), insomnia (35%), anxiety (32%), nausea (30%), fatigue (27%), and suicidal ideation (27%). On the day of the MDMA-AT sessions, the most frequently reported AEs were muscle tightness (73%), headache (62%), insomnia (49%), nystagmus (30%), and nausea (27%). Two days posttreatment, there was one report of headache and three reports of insomnia. Other AEs at two days posttreatment ranged from one to two reports of anxiety, fatigue, pain in jaw, and suicidal ideation. Table 3 presents AEs from days 0, 1, and 2 with an overall prevalence of 10% or greater.
Adverse Events With Prevalence of ≥10% Aggregated Across Three Sessions for Day of experimental, 1 Day Postexperimental, and 2 Days Postexperimental Session.

AE (adverse event) counted once per participant across three separate “Day of experimental” sessions. bAE counted once per participant across three separate “1 Day postexperimental” sessions. cAE counted once per participant across three separate “2 Days postexperimental” sessions.
Most participants reported AEs that were rated as either mild or moderate. Five participants each experienced an AE posttreatment that was rated as severe; however, these AEs did not occur on the day of experimental sessions or 2 days after. All five severe AEs (syncope, headache, anal fissure, exacerbation of suicidal ideation, and suicide attempt) occurred between 5 and 78 days post experimental session. There was one serious AE (SAE) of an aborted suicide attempt in a participant 28 days after their third experimental session. The attempt was aborted by the participant, who was later evaluated at the ER without being admitted and discharged 3 hours after presentation. The study investigator determined that this particular SAE was unrelated to treatment, given that it occurred 28 days after last treatment and MDMA has an elimination half-life of 7 to 9 hours. This participant had a prior history of suicidality due to underlying PTSD, with two previously reported suicide attempts in their lifetime.
At baseline, 51.4% of participants reported the presence of current suicidal ideation (see Table 4). During the study, there were 10 participants who reported suicidal ideation AEs, ranging from one to 20 days postexperimental session. In Table 4, suicidality was counted at the participant-level and, within each experimental session, counts were aggregated across three integration sessions. In general, after the day of experimental sessions, there were transient increases in suicidal ideation and serious ideation during the integrative sessions. At study termination, five participants reported the presence of suicidal ideation, of whom four had a history of suicidal ideation; no participants reported serious suicidal ideation or suicidal behavior. At study termination, participants were provided with an exit plan that included the ability to request referral for further therapy or medical care as needed.
Suicidal Ideation and Behavior as Measured by Columbia Suicide Severity Rating Scale (C-SSRS), N = 37.
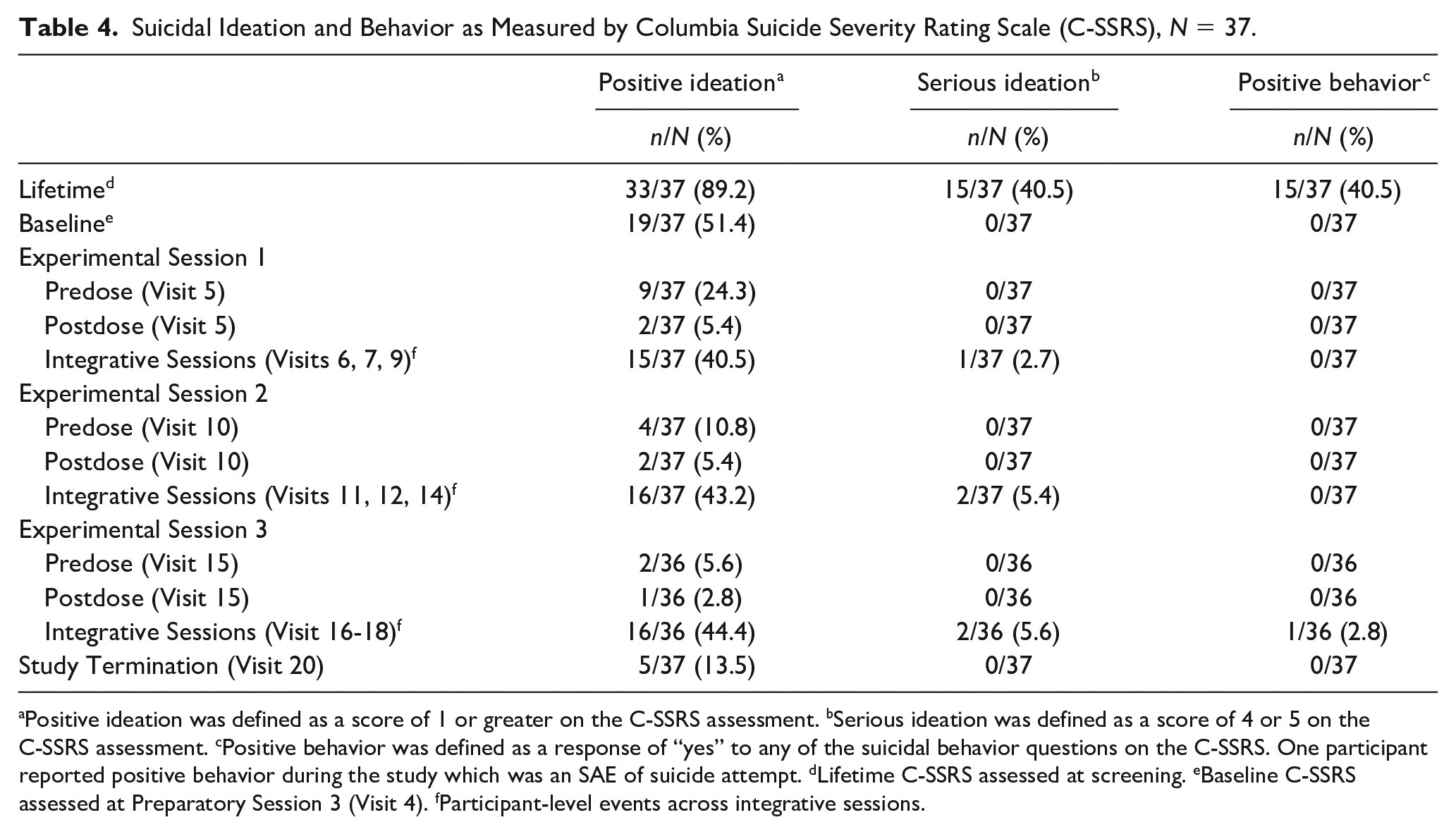
Positive ideation was defined as a score of 1 or greater on the C-SSRS assessment. bSerious ideation was defined as a score of 4 or 5 on the C-SSRS assessment. cPositive behavior was defined as a response of “yes” to any of the suicidal behavior questions on the C-SSRS. One participant reported positive behavior during the study which was an SAE of suicide attempt. dLifetime C-SSRS assessed at screening. eBaseline C-SSRS assessed at Preparatory Session 3 (Visit 4). fParticipant-level events across integrative sessions.
Discussion
These two open-label lead-in studies show that a large treatment response to MDMA-AT can be scaled and replicated by newly trained cotherapist dyads across multiple sites. Further, consistent with earlier Phase 2 studies, MDMA was well tolerated by participants and AEs were generally mild to moderate.
Adherence ratings from these trials demonstrated high levels of fidelity to the treatment manual across new cotherapist dyads, new sites, and session type, and suggest that this treatment modality was reproducible. Importantly, adherence was high in MDMA-AT sessions, which were considered particularly challenging due to their content, length, and novel modality. Ongoing supervision and evaluation were essential components in ensuring that newly trained cotherapist dyads were supported in delivering competent and compassionate therapy in alignment with the treatment model. In addition to supporting ongoing quality assurance, adherence ratings and qualitative comments from adherence raters provided valuable insights into areas where new cotherapist dyads required additional clinical supervision. The benefits of video-recorded sessions in clinical supervision have been well documented (American Psychological Association, 2015; Haggerty & Hilsenroth, 2011; Huhra et al., 2008; Nelson, 2014). Reviewing video-recorded sessions allowed supervisors to more objectively assess therapists’ skills than they might through a supervision discussion or review of session notes; it also provided therapists with opportunities for self-review and reflection, and to more objectively identify their own skills and areas for improvement (Alpert, 1996). Access to session videos for both their own review as well as for formal supervision and adherence ratings and comments from third-party observers allowed supervisors to provide detailed, timely feedback, and guidance to cotherapist dyads as they conducted treatment.
Three sessions of MDMA-AT significantly improved participants’ PTSD symptom severity from baseline to the primary endpoint, which was consistent with previous MDMA-AT studies (Mithoefer et al., 2019). In the present study, participants had a CAPS-5 score reduction of −29.89, which was comparable to a reduction of -26.35 (converted from CAPS-IV score of −44.8) among participants who were in the sponsor’s previous Phase 2 studies across five study sites. Therapists in the prior Phase 2 studies received approximately 50 hours of in-person training, expanded to 120 hours of online and in-person training for the present studies. Across each site, there was a significant reduction in participant’s CAPS-5 scores, ranging from a mean change of −7.00 (SD = 9.90) to −47.00 (SD = 1.46). At the primary endpoint, all but two participants (91.89%) had a clinically meaningful reduction in CAPS-5 scores (10-point reduction or greater), and the majority of participants (75.68%) no longer met criteria for PTSD diagnosis. These results demonstrate that MDMA-AT can be scaled to multiple sites across different countries while maintaining significant efficacy in reducing participant’s PTSD symptom severity.
Overall, MDMA-AT was well-tolerated, with participants experiencing transient increases in blood pressure and pulse that returned to baseline levels posttreatment. Additionally, the type and duration of AEs that were reported in these two open-label lead-in studies were expected and consistent with reports from previous Phase 2 studies (Mithoefer et al., 2018; Ot’alora et al., 2018). Prevalence of AEs occurring during the day of experimental sessions were greatly reduced 2 days posttreatment. Frequently reported AEs on the day of experimental sessions included muscle tightness, headache, insomnia, nystagmus, and nausea, which at 2 days posttreatment were reduced to only one report of headache and three reports of insomnia. Other AEs at 2 days posttreatment ranged from one to two reports of anxiety, fatigue, pain in jaw, and suicidal ideation. These AEs were consistent with AEs reported in a nonsponsor MDMA-AT open-label pilot study, including anxiety, fatigue, insomnia, and headache (Jardim et al., 2020).
A history of suicidality was common in this study sample, where enrollment criteria included a diagnosis of having severe PTSD. Lifetime history and posttreatment prevalence rates for positive ideation, serious ideation, and positive behavior were comparable against an analysis of pooled C-SSRS data from four previous Phase 2 studies that included a total of 68 participants. The previous Phase 2 studies had 15 (24.2%) reports of positive ideation and 1 (1.6%) report of serious ideation at long-term follow-up (Jerome et al., 2019). In the present study, at study termination, there were five (13.5%) reports of positive ideation and no reports of either serious ideation or suicidal behavior.
There was one instance of an SAE, an unsuccessful suicide attempt, self-aborted by the participant, that occurred 28 days following the final experimental session (between Visit 16 and 18), which was resolved by the next day. The participant had a lifetime history of ongoing suicidal ideation and two previous suicide attempts. The study investigator determined that this particular SAE was unrelated to treatment given that it occurred nearly a month after the last MDMA-AT session. Furthermore, clinic notes indicated that the event was likely triggered by a stressful life event. The participant reported PTSD symptoms improvement 47 days after the SAE. Specifically, their baseline CAPS-5 score was 44, which decreased to 23 following the three MDMA-AT session, indicating a clinically meaningful improvement in PTSD symptom severity (Jerome et al., 2019).
MDMA is described as enhancing emotional memory processing of traumatic memories with greater tolerability in therapeutic settings (Carhart-Harris et al., 2014; Mithoefer et al., 2013). After experimental sessions, participants continue to work with their therapists to process these memories during their nondrug integrative sessions. As with other types of therapy that involve extensive review of past trauma, it is possible that recall of such memories can temporarily trigger PTSD symptoms or negative emotions. This phenomenon likely explains increased reports of suicidal ideation several days after the experimental sessions and during the integrative sessions. Future studies should closely monitor suicidality due to the high risk for suicidal ideation and behavior in this population. Overall, prevalence of suicidality was low across all Phase 2 studies, including the present lead-in studies, to suggest that MDMA-AT treatment has a favorable risk/benefit ratio among those with severe PTSD.
Limitations
Although this study reproduced previously reported Phase 2 efficacy findings across multiple sites, there were between one to four participants per site and only four participants in Canada, which limited country- and site-level comparisons. Additionally, the single-arm study design limited the interpretation of the safety and efficacy data, since outcomes may have occurred due to other factors outside of treatment.
The adherence ratings collected in the study provided a valuable standardized method for ensuring consistency of care across teams; however, they could not provide a full picture of how capably a cotherapist dyad provided treatment due to limitations in the binary rating assessment. For example, an adherence criterion such as “therapists described the likely effects of MDMA” could have been rated as unmet because it was not addressed in the session, or because the therapists shared erroneous information—reflecting distinct issues from a quality and supervisory standpoint. Therefore, the quantitative measures summarized here provide only a limited assessment of the therapy adherence that must be paired with an adherence rater’s qualitative comments to fully identify specific areas of deviation from the treatment manual or protocol. Additionally, although the adherence rating checklist can be a useful training tool, it has not been validated.
Another limitation of this analysis is that only a subset of integrative sessions was used for adherence rating (two of the three sessions following the second experimental session). While this was sufficient for clinical supervision, inclusion of these incomplete sets of integrative sessions in the analysis of integrative sessions’ adherence scores would attenuate adherence rating scores. Indeed, when excluding entire sets where an integrative session was unrated due to the initial adherence rating plan or lack of data, overall ratings rose from 93.07% (SD = 9.79%; range = 58.33% to 100%) to 96.49% (SD = 6.32%, range = 75.00% to 100%).
Although adherence raters were trained to review study sessions in an unbiased manner, there was a possibility of leniency bias if raters had a desire for MDMA-AT to be positively evaluated and/ or confirmation bias leading raters to score unmet items as complete. Adherence raters completed interrater reliability testing as part of their training, and across the pool of 13 adherence raters, average adherence scores were consistent to one another (M = 94.47%, SD = 24.33%, range = 90.06% to 100%). However, interrater reliability was not tested within the present studies.
Due to the nature of how adherence criteria were evaluated, this study was not designed to be able to examine if fidelity to specific adherence criteria impacted participants’ PTSD improvement. Future studies would be needed to identify which criteria from the training manual may be most important for therapy efficacy and assess correlations between these criteria and participants’ outcomes.
Conclusion
These multisite studies demonstrate that newly trained therapists can achieve efficacy in reducing PTSD symptom severity and safety. MDMA-AT is a scalable treatment and can be successfully delivered by trained therapists across various locations.
Footnotes
Acknowledgements
The authors would like to thank the MAPS PBC adherence raters (Adrian Scharfetter, Audrey Redfield, Darrick May, Fabrice Nye, Jen Clark, Justin Forman, Linnae Ponte, Lucille Khoury, Marc L’Ecluse, Meghan Kennedy, Richard Knowles, Ryan Stevenson, and Sara Ouimette); therapy teams who worked on the MP16 and MP17 clinical trials for their dedication, expertise, and care; and Terence Ching for his manuscript review.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JW, JL, LB, AC, IJ, AF, CH, EH, SC, RM, AE received salary support for full-time employment with MAPS PBC for this study and other work. AL, BYK, and RD received salary support for full-time employment with MAPS for this study and other work. MM, AM, MOG, and BP received support as a contractor from MAPS PBC for training and supervision of research psychotherapists for this study and other work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This Clinical Trial was sponsored by the Multidisciplinary Association for Psychedelic Studies (MAPS), a 501(c)(3) nonprofit organization. MAPS provided the MDMA and fully funded this study from private donations. MAPS Public Benefit Corporation (MAPS PBC), wholly owned by MAPS, was the trial organizer.
Ethical Approval
The study protocol was reviewed by Western Copernicus Group Independent IRB (Research Triangle, NC), University of California San Francisco Human Resource Protection Program IRB, University of Madison Wisconsin Health Sciences IRB, Western IRB (Puyallup, WA), and University of British Columbia Providence Health Care Research Ethics Board. Studies were designed and conducted in accordance with Good Clinical Practice guidelines. All participants provided written informed consent.
