Abstract
Ultrastructural localization of a P29 protein of
Dense granule proteins of
Materials and Methods
Parasite Culture
Tachyzoites of
Isolation of T. gondii DNA, RNA, and Protein, and Synthesis of cDNA
A 10-liter secondary suspension culture of HeLa cells infected with the RH strain of
Purified
Cloning and Expression of the Gene Encoding the Novel Antigen
Standard methods were used for isolation and analysis of plasmid DNA, DNA sequence analysis, and Southern analysis (Sambrook et al. 1989). DNA and amino acid sequences were analyzed using the Geneworks software package (Intelligenetics; Mountain View, CA). A
The 1.3-
Cloning and Expression of Other Toxoplasma Antigens
Using the published DNA sequences, PCR primers were generated to clone the genes encoding the following Toxoplasma antigens previously described in the literature into the CKS expression vector (Bolling and Mandecki 1990) by PCR amplification: P22 (SAG2) (Prince et al. 1990); P24 (GRA1) (Cesbron-Delauw et al. 1989); P25 (Johnson and Illana 1991); P28 (GRA2) (Prince et al. 1989); P30 (SAG1) (Burg et al. 1988); P35 (Knapp et al. 1989); P41 (GRA4) (Mevelec et al. 1992); P54 (ROP2) (Saavedra et al. 1991); P66 (ROP1) (Knapp et al. 1989; Ossorio et al. 1992); and P68 (Knapp et al. 1989). The cloned DNA sequences were confirmed by DNA sequence analysis. Expression of these Toxoplasma recombinant proteins in
Generation of the 5-241-178 Mouse Monoclonal Antibody Directed Against the Novel Antigen
Swiss mice (Charles River Laboratories; Wilmington, MA) were infected intraperitonally with 2.5 x 107 tachyzoites of
Gel Electrophoresis and Western Blot Analysis
Approximately 10 mg of Toxoplasma lysate and 10 mg
Immunocytochemistry
After two washes with PBS, pH 7.2, freshly isolated tachyzoites were fixed for 2 hr with 2% paraformaldehyde, 0.5% glutaraldehyde in PBS (pH 7.2) at 4C. Tachyzoites were washed with PBS, then pre-embedded in BSA (diluted 1:4) and glutaraldehyde (25%) in the ratio 4:1, dehydrated with increasing concentrations of ethanol, and embedded in LR White resin. Ultrathin sections (thickness 80 nm) placed on gold grids were immunolabeled as follows. They were incubated first with PBS-1% BSA, pH 7.2, for 30 min at RT and then with the 5-241-178 monoclonal antibody, diluted 1:10,000 in PBS-1% BSA for 1 hr at RT. After three washes with PBS-1% BSA, thin sections were incubated for 1 hr with goat anti-mouse IgG-gold (10 nm) (Biocell; Tebu, France) diluted 1:50 for 1 hr at RT. After two washes in PBS and distilled water, the sections were contrasted with uranyl acetate. Controls without monoclonal antibody were prepared and observed.
Quantitative Immunolabeling Analysis
Conditions for the preparation of samples were designed at the same time to preserve the ultrastructure, to identify labeled organelles, and to retain immunoreactivity. The monoclonal antibody was tested at increasing concentrations up to optimal immunolabeling response, i.e., when the ratio of total signal to nonspecific signal was the highest possible. Under these conditions, we could carry out immunolabeling assays quantitatively, the antibody being bound to almost all accessible epitopes of the antigen. Thus, we could compare the distribution of the antigens quantitatively by evaluating the immunolabeling density (number of gold particles/μm2).
Quantitation was performed with an image analyzer (Bio 500; Biocom, Les Ulis, France) using transmission electron micrographs of ultrathin sections and a semiautomatic quantitative method (Bonnet et al. 1990; Boulanger et al. 1991). After digitizing of images, analysis of labeling densities was performed in three steps. First was identification and selection of the biological compartments: membrane complex, rhoptries, dense granules, reticular network, and membrane of the parasitophorous vacuole. An image segmentation was applied and areas were computed. Tachyzoite membrane complex and parasitophorous vacuole membrane were materialized by two outlines which delimited these two compartments in the section. Therefore, the gold particle distribution was estimated in the area included between these outlines (thickness about 60 nm, which represents the membrane section). Second, extraction of dense markers (gold particles) was performed automatically by mathematical morphology (Serra 1982). Third, quantitation of markers was expressed as number of gold particles/μm2 area. As a reference, we used isolated markers and determined the mean area of one marker. In the case of coalescent particles, the count was estimated from the ratio of their area to the area occupied by the reference marker. Gold particles of uniform size (10 nm) were used and the binding capacity of each particle was assumed to be the same (Bendayan 1984). Because immunolabeling was restricted to the surface of the sections, only those binding sites exposed interact with the specific antibodies. Those sites can be revealed and permit quantitative and comparative evaluations (Bendayan et al. 1987).
The average number of gold particles/μm2 was calculated by subtracting the background determined in the resin (LR White) from the labeling density of the experimental specimens. Quantitation was determined on 13 negatives for extracellular tachyzoites and 20 negatives for intracellular tachyzoites on 50 observed negatives. Only those having a good contrast were taken into account.
State 1 and State 2 of the intracellular state of the tachyzoites are discriminated from the parasitophorous vacuole membrane (PVM) labeling, PVM being the second target after the reticular network of the dense granule exocytosis.
The quantitative evaluation of the immunolabeling is expressed by averages with the standard deviation (SD). To define the significant difference in the immunolabeling among the different states (extracellular, State 1, and State 2), among five different compartments (DG, dense granules; R, rhoptries; MC, membrane complex; RN, reticular network; PVM, parasitophorous vacuole membrane), and among the different compartments during the different states, we used the
Results
Identification of the Novel Antigen as the P29 Antigen of T. gondii
The 5-241-178 monoclonal antibody was used to probe a Western blot containing total Toxoplasma protein and the recombinant CKS-novel antigen fusion protein, which is shown in Figure 1. The 5-241-178 monoclonal antibody reacted specifically with a 29-kD protein present in Toxoplasma lysate (Figure 1, Lane 2), thus identifying the novel antigen as the P29 antigen of

Detection of the novel antigen by Western blot using the 5-241-178 mouse monoclonal antibody. Lane 1, Prestained protein molecular weight markers, kD indicated. Lane 2, Approximately 10 mg of
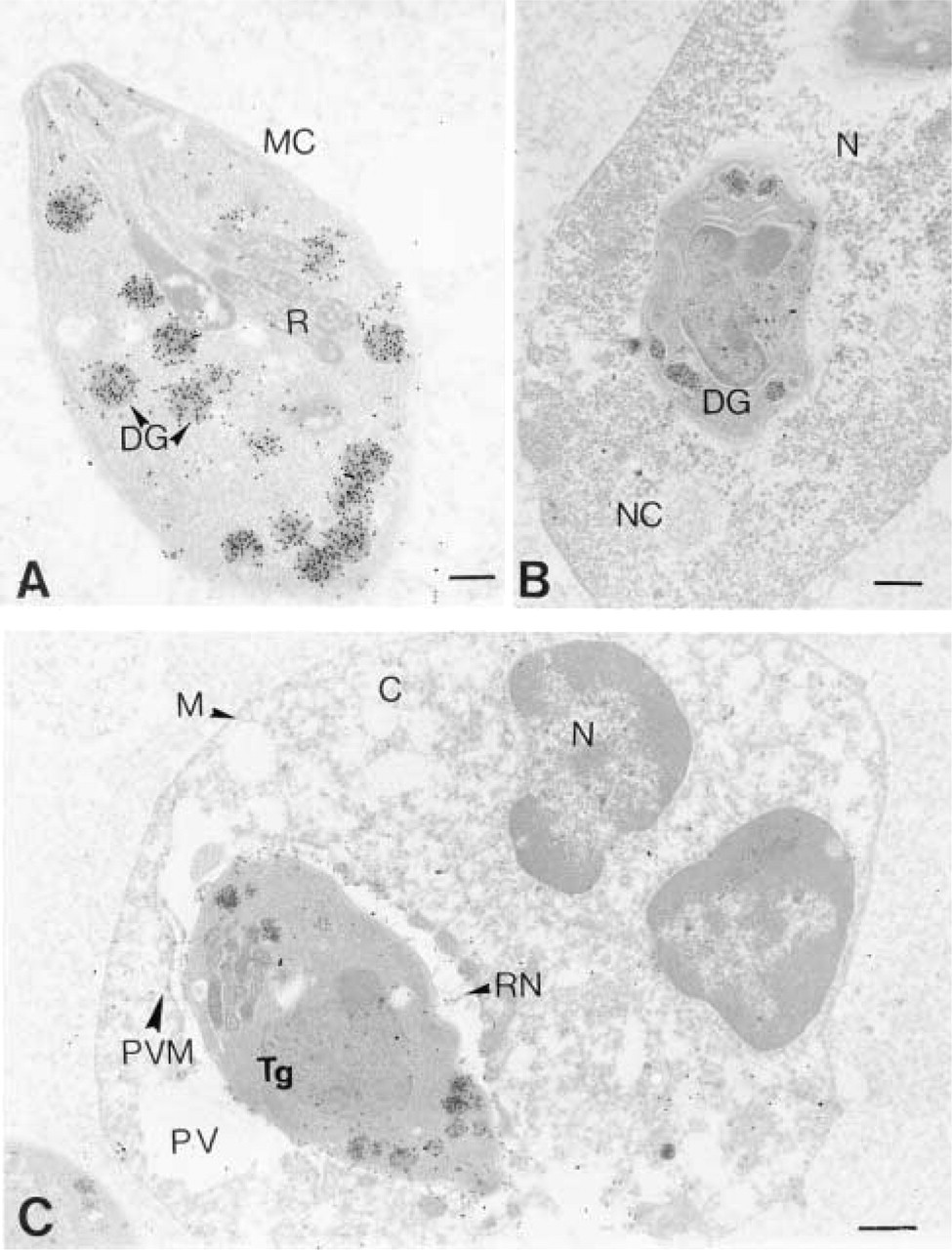
(A) Immuno-EM localization of P29 (GRA7) in extracellular
The distribution of the labeling density of the P29 antigen using the monoclonal antibody 5-241-178 examined in the five compartments-dense granules, membrane complex, rhoptries, reticular network of the parasitophorous vacuole, and the parasitophorous vacuole membrane-is shown in Figures 2-4. The results of the quantitative estimation of the immunolabeling are given in Table 1.
Immunolocalization of the P29 antigen in extracellular tachyzoites demonstrated that the P29 antigen was present inside the dense granules. The P29 antigen was found to be about 25 times more abundant in the dense granules than in either the rhoptries or membrane complex. With the nonparametric
Immunolocalization of the P29 antigen in intracellular tachyzoites as shown in Figures 3 and 4 and in Table 1 demonstrated that there was a differential distribution of the P29 antigen and, from all the negatives observed, we divided the intracellular state of the parasite into two states. In State 1, the P29 antigen present in the tachyzoite dense granules (DG), as in the extracellular state, was also redistributed principally in the membrane complex (MC) of the tachyzoite and in the reticular network (RN) inside the parasitophorous vacuole. In State 2, the P29 antigen distribution was extended and was found in association with the parasitophorous vacuole membrane (PVM). The quantitative P29 immunolabeling repartition is shown on Table 1. The dense granule labeling differences were not significant between the extracellular state and State 1 but were very significant between the extracellular state and State 2 (
Discussion
Dense granule organelles of protozoan parasites Sarcocystis (Entzeroth et al. 1986), Plasmodium (Trager et al. 1992), and Toxoplasma (Leriche and Dubremetz 1990; Cesbron-Delauw 1994) are involved in the installation process within the host cell, presumably by contributing to the structural modifications of the parasitophorous vacuole. As the parasites grow and replicate within the cell, the parasitophorous vacuole is modified (Achbarou et al. 1991). At early times after invasion, the PVM is depleted of intramembranous particles (IPMs) relative to the host cell plasma membrane. IPMs progressively appear in the PVM as the vacuole matures (Porchet-Henneré and Torpier 1983). A tubuloreticular network appears within the vacuolar space (Sibley et al. 1986), progressively increasing as the vacuole enlarges. Although the intravacuolar network is clearly of parasite origin, the source of the IMPs in the PVM is not clear.
Dense granule secretion has all the hallmark features of regulated exocytosis, including storage of secretory proteins in specialized organelles, fusion with the plasma membrane, and release of their contents. This burst of exocytosis occurs soon after internalization. Dense granule antigens, termed GRA proteins, are differentially targeted to either the vacuolar space, the network, or the surrounding membrane of the parasitophorous vacuole.
Sequencing of six genes encoding dense granule proteins has shown typical hydrophobic signal sequences that target them in the secretory pathway. Except for the canonical signal peptide at the N-terminus, their structural features are different and may be related to their differential distribution in the parasit-ophorous vacuole (Cesbron-Delauw 1994). GRA1 (23 kD) is a calcium binding protein uniformly distributed throughout the lumen of the vacuole (Cesbron-Delauw et al. 1989) and is associated with the network through peripheral interactions (Sibley et al. 1995). GRA2 (28 kD), GRA4 (40 kD), and GRA6 (32 kD) are tightly associated with the tubular elements of the membranous network (Charif et al. 1990; Achbarou et al. 1991). Ultrastructural studies suggest that the intravacuolar network and the membrane complex of the parasite are contiguous in localized regions. The molecular structure of GRA4 suggests its association with the intravacuolar network membrane via a transmembrane region (Mevelec et al. 1992). GRA2 may be linked to lipids with two amphipathic helices present in the molecule (Mercier et al. 1993). GRA6 may be a transmembrane fibrous protein implicated in the structures of the membranous network (Lecordier et al. 1995). GRA3 (30 kD) and GRA5 (21 kD) are distributed on the membrane delimiting the PV (Achbarou et al. 1991; Dubremetz et al. 1993; Lecordier et al. 1993), and GRA3 is found both in the network and the PVM locations (Cesbron-Delauw 1994). In a similar manner to GRA5, oligomers of GRA3 associate stably with the vacuole membrane via hydrophobic interactions (Ossorio et al. 1994).
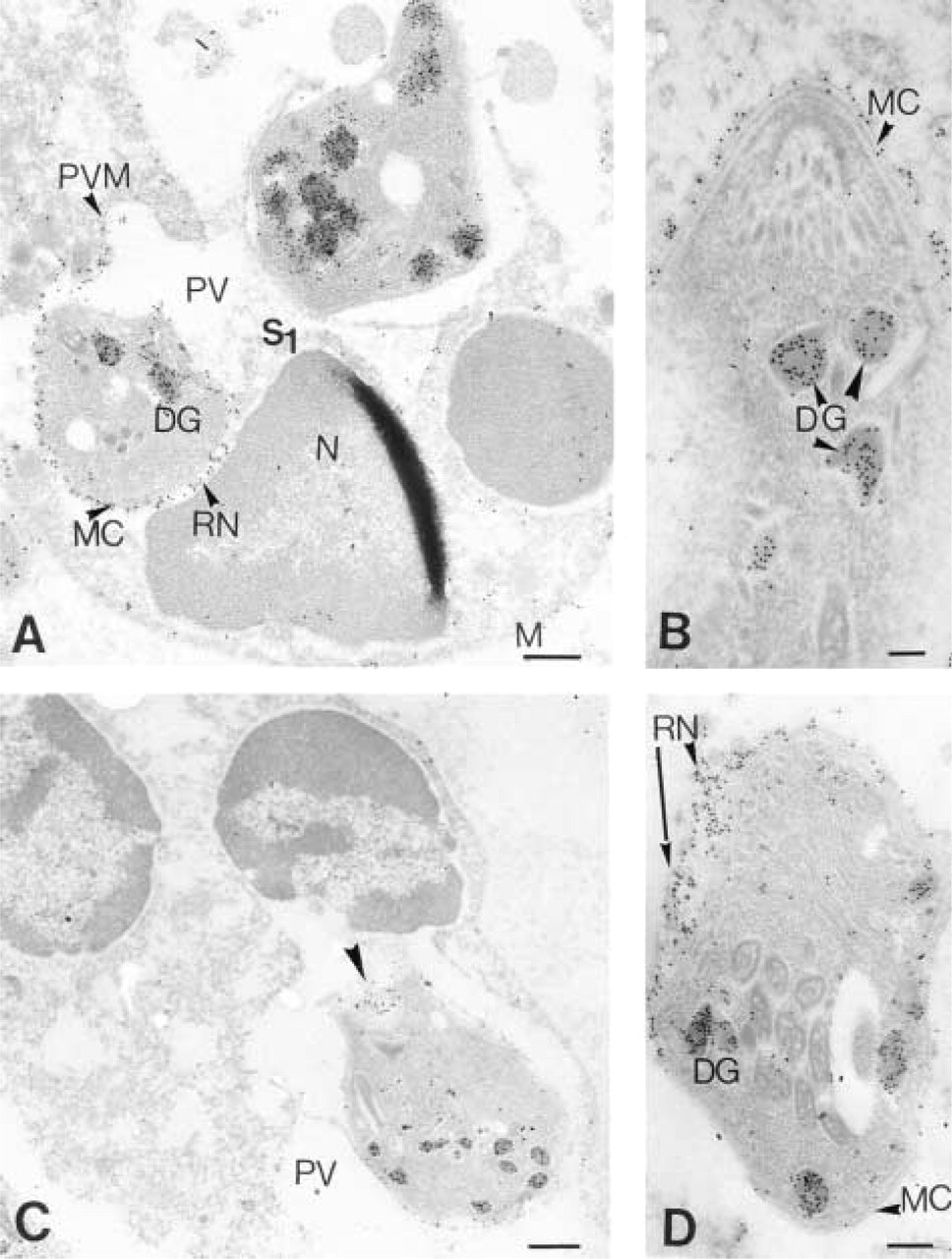
Immuno-EM localization of P29 (GRA7). (
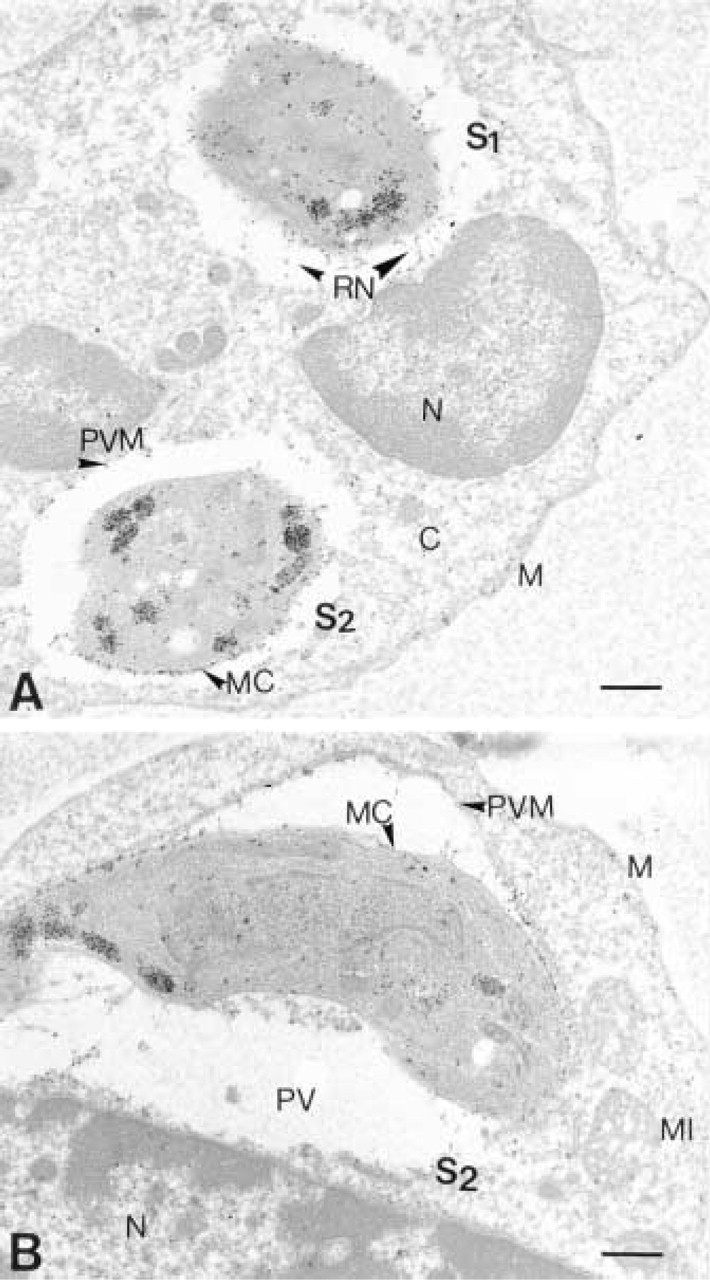
Immuno-EM localization of P29 (GRA7). (
Quantitative P29 immunolabeling analysis in extracellular and intracellular

Immunolabeling of tachyzoites with the new monoclonal antibody 5-241-178, which binds specifically to the P29 antigen of
Analysis of the primary amino acid sequence determined from the DNA sequence of the gene encoding the P29 (GRA7) antigen reveals several interesting structural features consistent with this antigen being a member of the GRA family. First, the P29 antigen contains a canonical signal peptide at the N-terminus to permit the protein to enter the secretory pathway. Second, this antigen contains two hydrophobic domains of 15 amino acids and 29 amino acids. The larger hydrophobic domain has a high degree of α-helical character and is probably the transmembrane region that anchors the protein to the parasitophorous membrane. Third, this antigen contains a potential N-linked glycosylation site just downstream of this transmembrane region, which may also be important in targeting this protein to the membrane. Fourth, no amino acid homology exists between GRA7 and the other GRA proteins. The structural features of the P29 (GRA7) antigen are similar to those of the GRA4, GRA5, and GRA6 proteins in that all of these proteins contain amino acid sequences that fulfill the requirement of transmembrane regions (Mevelec et al. 1992; Lecordier et al. 1993,1995). In addition, both GRA4 and GRA6 contain a potential N-linked glycosylation site upstream of the transmembrane domain, whereas this site is located downstream from this region in the GRA7 protein. The GRA5 protein contains no potential N-linked glycosylation sites. Of the seven GRA proteins identified to date, only three are localized to the parasitophorous vacuolar membrane: GRA3, GRA5, and GRA7. It would be interesting to examine the interplay among GRA3, GRA5, and GRA7 in the biogenesis of the parasitophorous vacuolar membrane on penetration of the tachyzoite into the host cell.
Note Added in Proof
During review of this work, the same gene and protein, and localization of this protein to the dense granules, have been described by Fischer et al. (1998) and Jacobs et al. (1998).
