Abstract
1,25-Dihydroxy-22-oxavitamin D3 (OCT) is a new synthetic analogue of 1,25(OH)2D3 with a low calcemic effect. This study utilized quantitative receptor autoradiography to determine the dose-related receptor binding and saturation among the vitamin D target cells: parathyroid chief cells, kidney distal and proximal tubule epithelium, duodenal absorptive epithelium, and epidermal keratinocytes. Rats were injected with 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, or 16.0 μg/kg bw of [26-3H]-OCT and sacrificed 1 hr afterwards. Then autoradiographs were prepared under identical conditions. In these target cells, nuclear uptake of radioactivity increased with dose and then achieved a plateau. However, their saturation doses showed differences: parathyroid chief cells 1-2 μg; duodenal absorptive epithelium, distal tubule epithelium, and epidermal keratinocytes 4-6 μg; proximal tubule epithelium 8 μg (per kg bw). In contrast, in nontarget cells, such as liver and duodenal smooth muscle, radioactivity did not concentrate in the nuclei but increased in the cytoplasm with dose, without plateauing. These results provide the first information on the relative saturabilities of various target cell populations with a vitamin D ligand. Parathyroid chief cells required the relatively lowest receptor saturation dose. This suggests a high sensitivity and response to OCT treatment with related therapeutic potential for the regulation of parathyroid function.
1,25-D
1,25-Dihydroxy-22-oxavitamin D3 (OCT) is a synthetic analogue of 1,25(OH)2D3 that retains many of the therapeutically useful properties of 1,25(OH)2D3 but has less calcemic activity compared to 1,25(OH)2D3 (Nishii et al. 1991). OCT has also been shown to induce differentiation of WEHI-3 myelomonocytic cells (Abe et al. 1987), to inhibit proliferation of psoriatic fibroblasts (Morimoto et al. 1989), and to suppress PTH secretion and gene expression (Brown et al. 1989b; Naveh-Many and Silver 1993).
OCT has some different biological properties compared to 1,25(OH)2D3. The binding potencies of OCT to chick intestinal 1,25(OH)2D3 receptor (VDR) and serum vitamin D binding protein (DBP) were approximately 1/8 and 1/600 of the respective values of 1,25(OH)2D3 (Kobayashi et al. 1994). OCT circulates as an intact form mainly bound to low-density lipoproteins (LDLs) (Teramoto et al. 1995). In addition, OCT reduces the plasma concentration of 1,25(OH)2D3 by suppression of the renal 25-hydroxy vitamin D3 1α-hydroxylase (Grieff et al. 1992). However, OCT was demonstrated to bind to the same receptor as 1,25(OH)2D3 in developing bone (Stumpf et al. 1994), skin (Stumpf et al. 1995b), and stomach (Stumpf et al. 1995a).
OCT has been suggested to have various therapeutic potentials, e.g., for secondary hyperparathyroidism, psoriasis, and breast cancer (Abe et al. 1991), mimicking specific actions of 1,25(OH)2D3 in each of the target tissues without causing hypercalcemia. Therefore, a careful exploration of the in vivo target tissues for OCT is necessary to obtain further indications about the therapeutic potential for this low-calcemic vitamin D analogue.
To understand the mechanisms of vitamin D and its analogues, it is essential to know their target sites in vivo. Receptor autoradiography has high resolution and high sensitivity for the demonstration of ligand- receptor binding at the cellular-subcellular light microscopic level (Stumpf 1976). Such information is difficult or impossible to obtain by radioassay using tissue homogenates, whole body autoradiography, imaging plate analysis, and in vitro studies with cultured cells. Thaw-mount receptor autoradiography permits the study of noncovalently bound hormones and drugs by avoiding translocation and leaching during histological tissue preparation, and has been used to identify target cells for 1,25(OH)2D3 in more than 50 different tissues (Stumpf 1995).
The present study was designed to obtain information about relationships between the administered dose and the level of nuclear uptake and binding in select target cells after i.v. injection of [26-3H]-OCT. Quantitative autoradiography combined with computerized image analysis was utilized.
Materials and Methods
The animals used in this experiment were treated in accordance with Chugai Pharmaceutical's ethical guidelines, which adhere to generally accepted international criteria for humane treatment. Eight-week-old male Sprague-Dawley rats (JCL:SD; Japan Clea Laboratory, Tokyo, Japan) were fed normal rat chow (CE-2) and water ad libitum and kept under a 14:10 light-dark cycle. Two animals each were injected i.v. with 0.25, 0.5, 1.0, 2.0, 4.0, 8.0, or 16.0 μg/kg bw of [26-3H]-1,25-dihydroxy-22-oxavitamin D3 ([26-3H]-OCT), with a specific activity of 76 Ci/mmol, dissolved in ethanol-isotonic saline (1% ethanol, 0.01% Tween 20). [26-3H]-OCT was synthesized by the Chugai Pharmaceutical Co. with a purity better than 95%. One hour after the injection, duodenum, kidney, thyroid-parathyroid, skin, and liver were dissected, individually placed on tissue holders, and frozen in dimethylbutane cooled with liquid nitrogen, then stored in liquid nitrogen until sectioning in a microtome cryostat (Microm HM 500M; Heidelberg, Germany). The frozen sections at a thickness of 4 μm were thaw-mounted on nuclear emulsion (Konica NR-M2)-coated slides and exposed in light-proof desiccator boxes at 4C for 1 month, 4 months, or 15 months. After the exposure, slides were developed in Konicadol-X for 8 min at 20C, rinsed, fixed in Konicafix for 15 min at 20C, rinsed, and stained with methylene blue-basic fuchsin, then air-dried and coverslipped. This thaw-mount autoradiographic technique has been described in detail (Stumpf 1976).
Competition studies were performed by co-injection of [26-3H]-OCT at 2 μg/kg bw with a 1000–fold excess of unlabeled 1,25(OH)2D3 (Wako Pure Chemical Industries; Osaka, Japan) or unlabeled OCT (Chugai Pharmaceutical Co.).
As additional controls against artifacts, autoradiographs from experimental tissues with 0-day exposure time were compared with autoradiographs with 1 month, 4 months, or 15 months of exposure time. There was no evidence for positive chemography.
Quantitative analysis was performed with a computerassisted image analysis system (Olympus SP-500) by counting the number of silver grains per nuclear area of all cells of a defined region (between 60 to 100 cells each in 10 fields of observation) with automatic background (extranuclear tissue radioactivity) subtraction. Background was calculated by multiplication of the number of silver grains per 100 μm2 (density) by nuclear area (size). Autoradiographs were evaluated at 1-month exposure time, because the clusters of silver grains increased at excess exposure and they caused loss of information for grain counting.
Results
Cell Localization
After the injection of [26-3H]-OCT, specific cells (target cells) displayed a higher number of silver grains over the nuclear region compared to the cytoplasmic region.
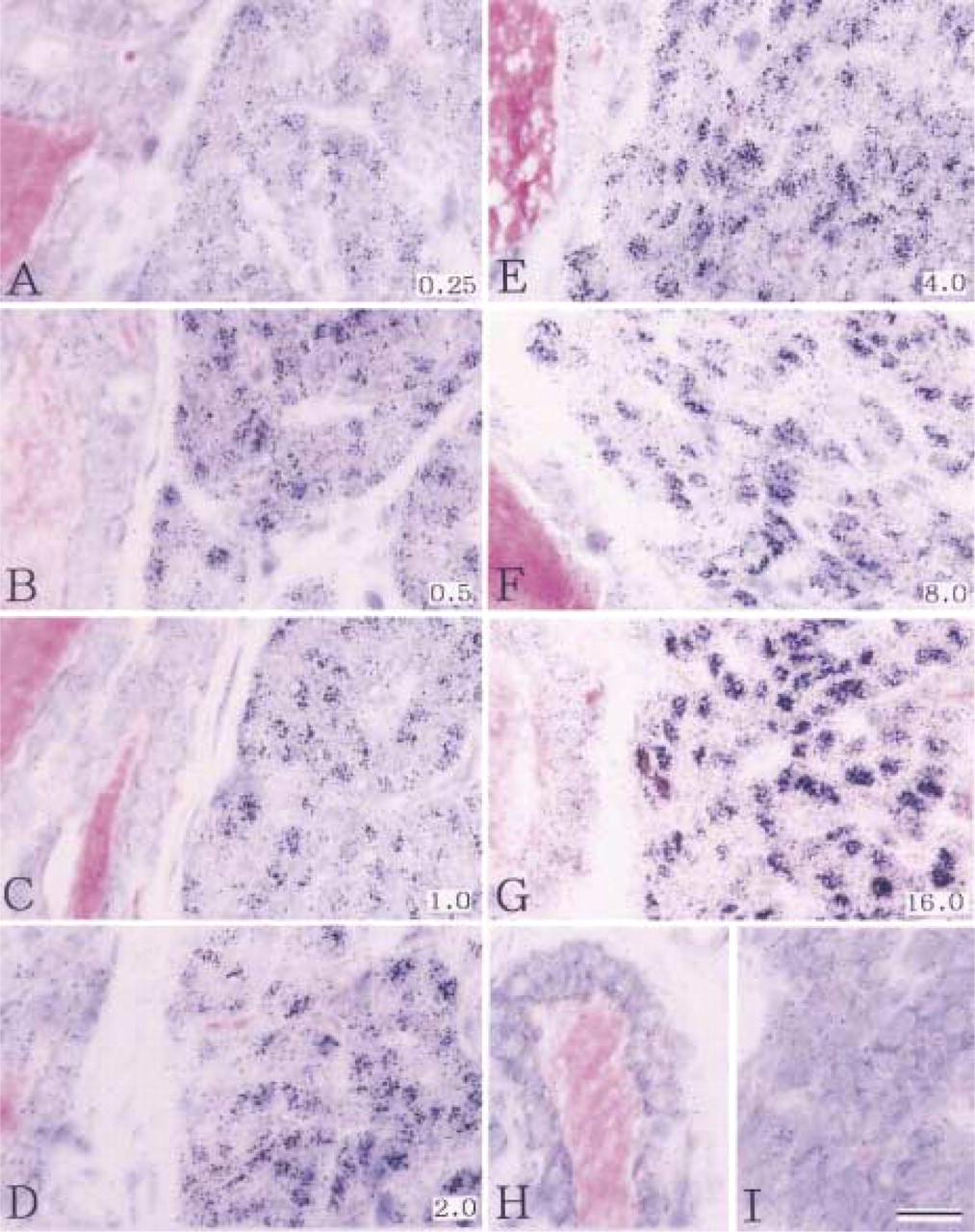
Parathyroid-Thyroid. Most of the parathyroid chief cells showed a high nuclear concentration of radioactivity, i.e., silver grain levels were well above that of adjacent cytoplasm and extracellular space (Figure 1, 1-month exposure; Figure 2, 15-month exposure). Connective tissue cells of the parathyroid, in general, did not show nuclear accumulation of silver grains. In the adjacent thyroid tissue, radioactivity levels were low compared to the parathyroid chief cells (Figure 2). Thyroid follicle epithelial cells showed a slight elevation of cytoplasmic radioactivity compared to extracellular radioactivity levels. Occasionally, in follicle epithelial cells, a low to intermediate accumulation of nuclear radioactivity was noted. In the lumen (colloid) of small follicles, no or only very little radioactivity was present, whereas in the lumen of larger follicles radioactivity was accumulated, especially at the border of the follicle epithelium. This predominantly marginal radioactive labeling of the colloid increased in correlation with the increasing size of the follicles.
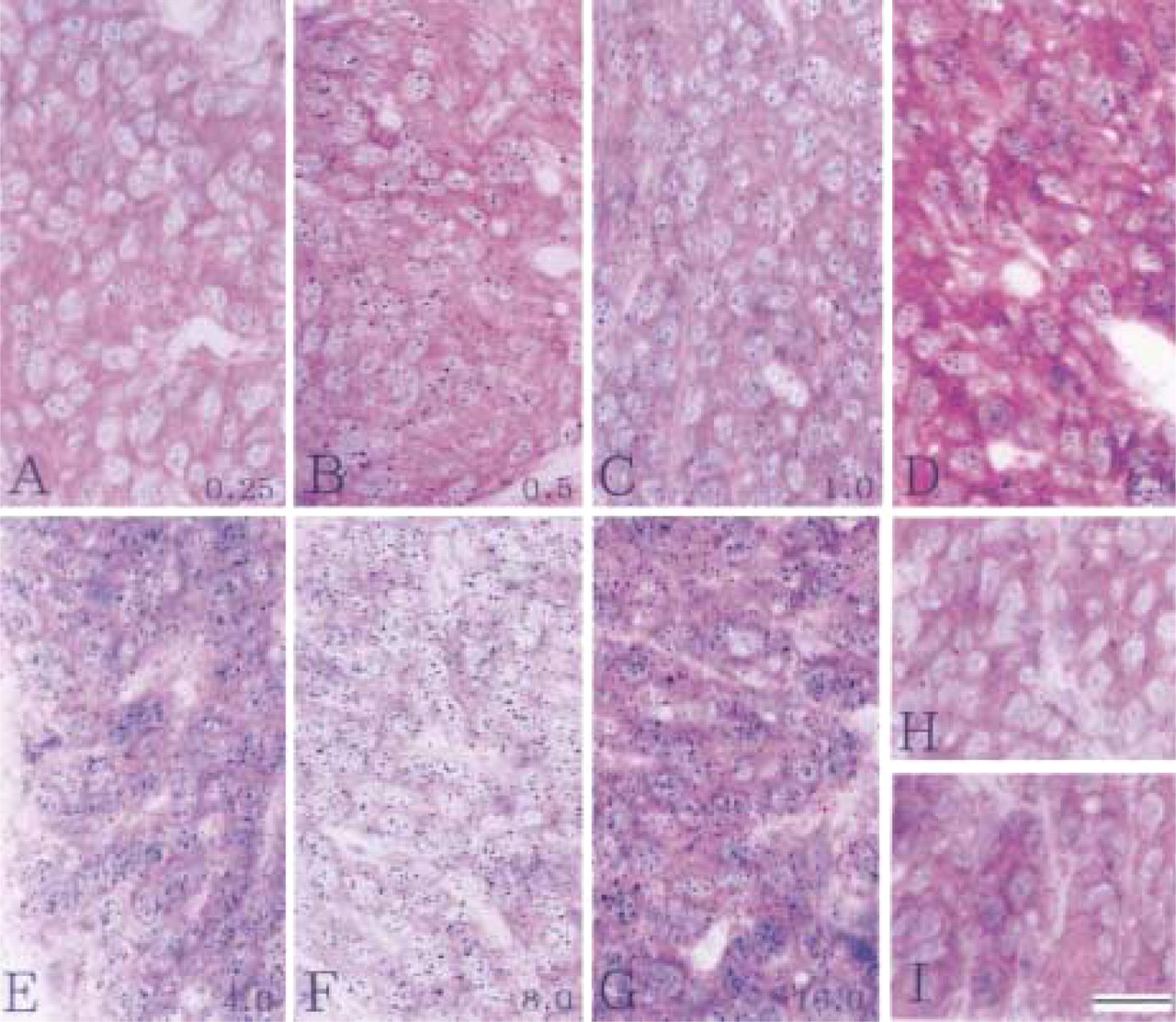
Autoradiographs of parathyroid (
Most of the extrafollicular cells did not show a nuclear accumulation of radioactivity, except for an occasional cell or group of a few cells with distinct nuclear labeling.
Duodenum. Midportions of villi were selected. Absorptive epithelial cells exhibited nuclear accumulation of silver grains, in contrast to goblet cells and most of the connective tissue cells, which did not display nuclear labeling but exhibited only low levels of tissue “background” radioactivity (not shown).
Kidney. Among the various cell types in the kidney, the distal tubule epithelium showed the strongest nuclear labeling. In the proximal tubule epithelium, nuclear accumulation of radioactivity was also visible but was less intense than that in distal tubules. Glomerular podocytes and epithelial cells of the thick and thin limbs of the loop of Henle showed weak to intermediate nuclear labeling. In the cytoplasm of proximal tubule epithelium, silver grain density was high compared to the cytoplasm of adjacent distal tubule epithelium. Intertubular connective tissue cells were generally unlabeled (not shown).
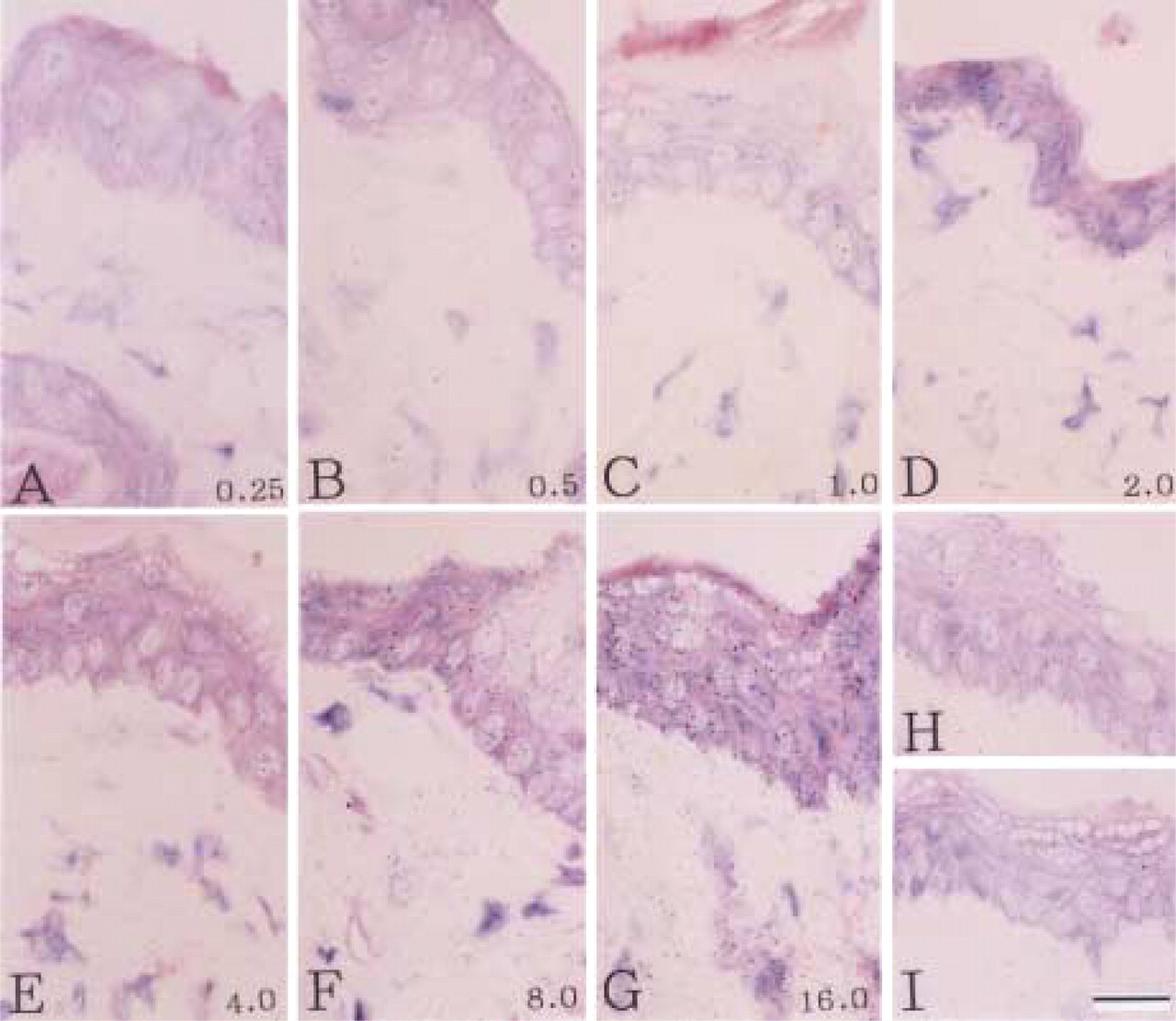
Skin. In epidermis, the strongest nuclear concentration of radioactivity was present in keratinocytes of the stratum spinosum (Figure 3, 1-month exposure). Cells of the basal layer of the epidermis showed relatively weak nuclear labeling under the conditions of the experiment. Labeled keratinocytes of hair follicles were not evaluated. Most of the fibroblasts of the dermis were devoid of nuclear labeling.
Liver and Duodenal Muscularis Externa. Hepatocytes and smooth muscle cells did not show nuclear accumulation of radioactivity (Figure 4A, 1-month exposure; Figure 4B, 4-month exposure).
Quantitative Evaluation of Autoradiographs
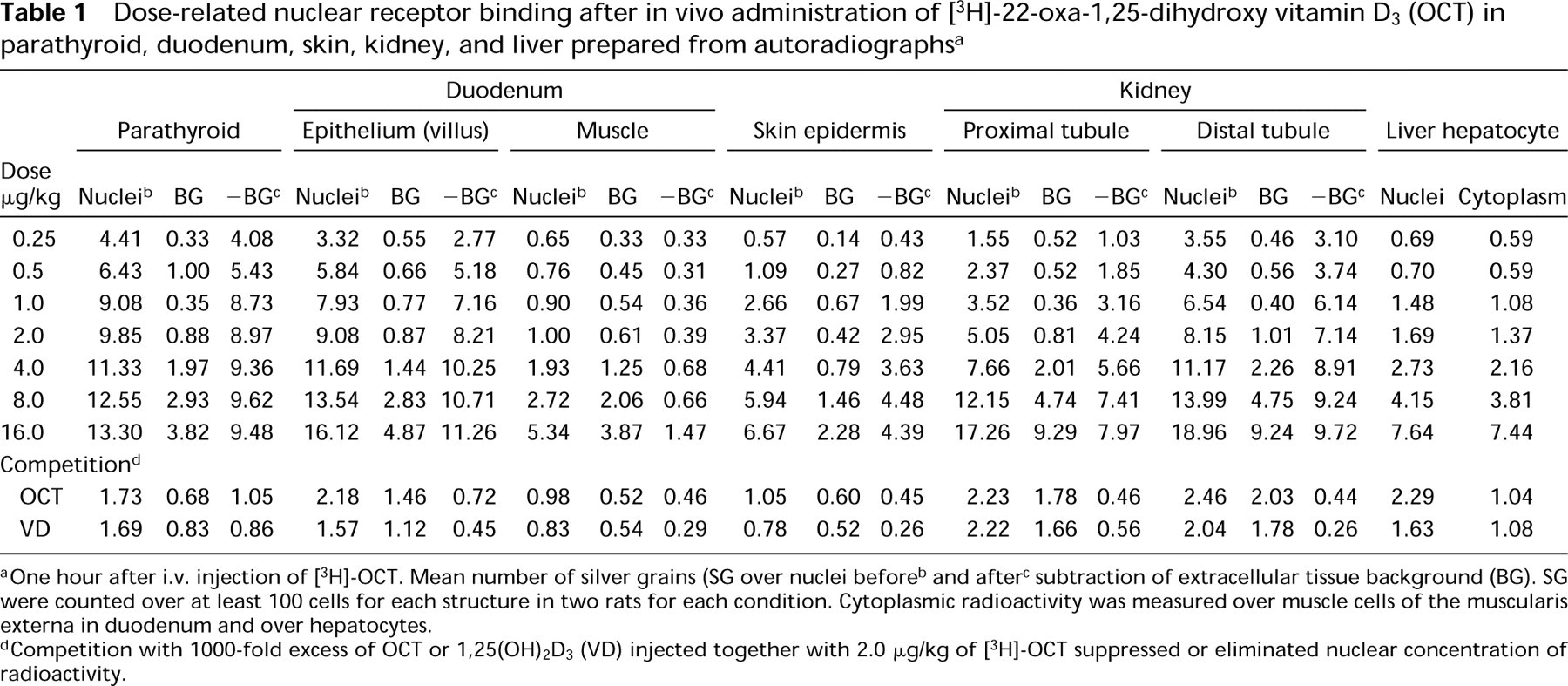
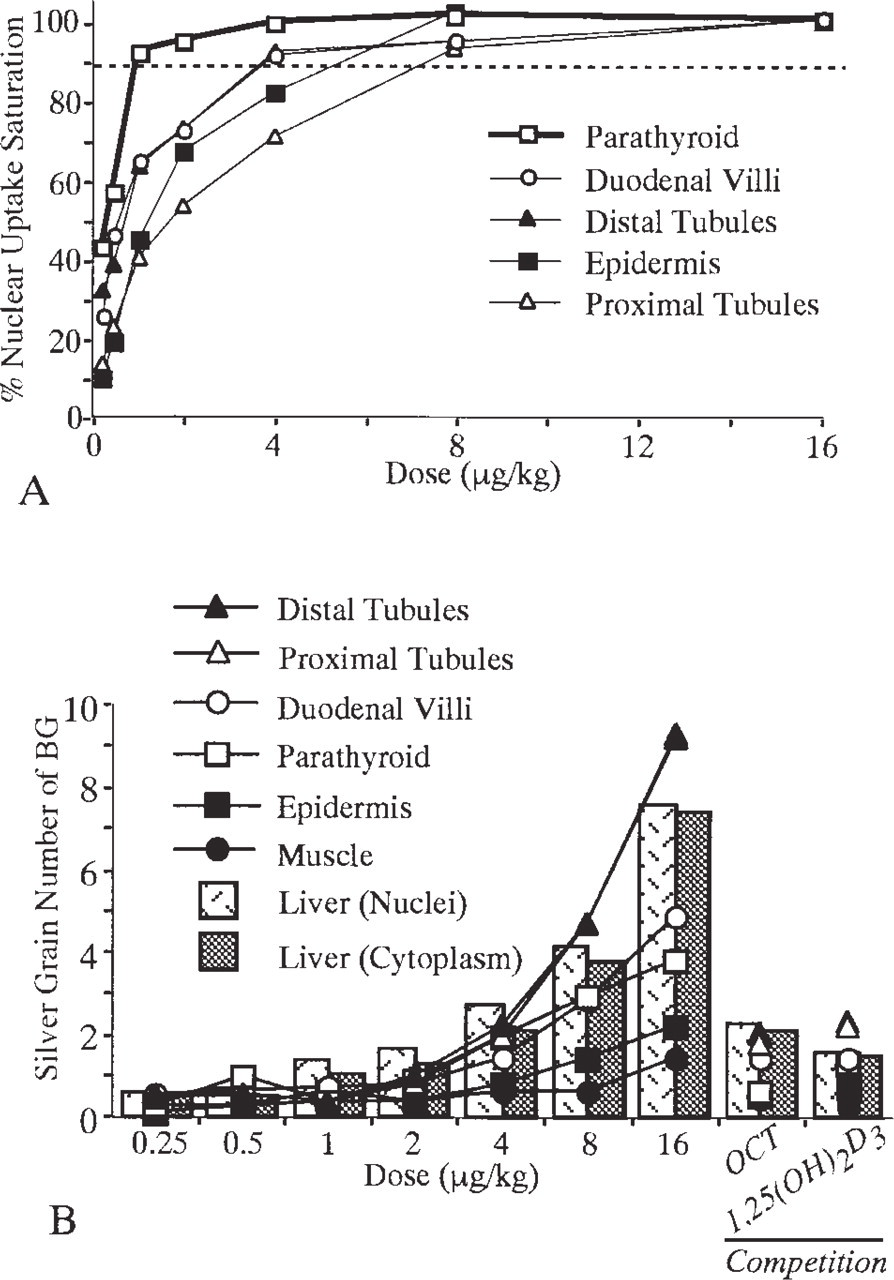
The number of silver grains in nuclei of each target cell type is listed in Table 1. The nuclear accumulation of radioactivity increased in a dose-related fashion towards a plateau. The beginning of the plateau was reached for parathyroid chief cells at the lowest dose level of approximately 1-2 μg/kg bw and at 4-6 μg/kg bw for the other tissues, except for the proximal tubule epithelium, which follows approximately at 8 μg/kg bw (Figure 5A). In contrast, extranuclear and extracellular radioactivity levels did not reach a plateau but increased almost linearly (Figure 5B). Nuclei and cytoplasm of hepatocytes of the liver (Figure 4A) and smooth muscle cells of the duodenum (Figure 4B) also showed a continuous increase of radioactivity without a plateau, although slightly higher nuclear radioactivity could be noted in hepatocytes when the nuclear density of silver grains was compared to that of the cytoplasm.
In the competition experiments with a 1000-fold excess of unlabeled 1,25(OH)2D3, nuclear concentration of radioactivity was abolished in parathyroid chief cells (Figures 1I and 2I), duodenal absorptive epithelium, kidney distal and proximal epithelium, and skin keratinocytes (Figure 3I). In contrast, silver grain densities over cytoplasm of target cells, extracellular compartments, nuclei and cytoplasm of hepatocytes, and smooth muscle cells of the duodenum showed few or no changes.
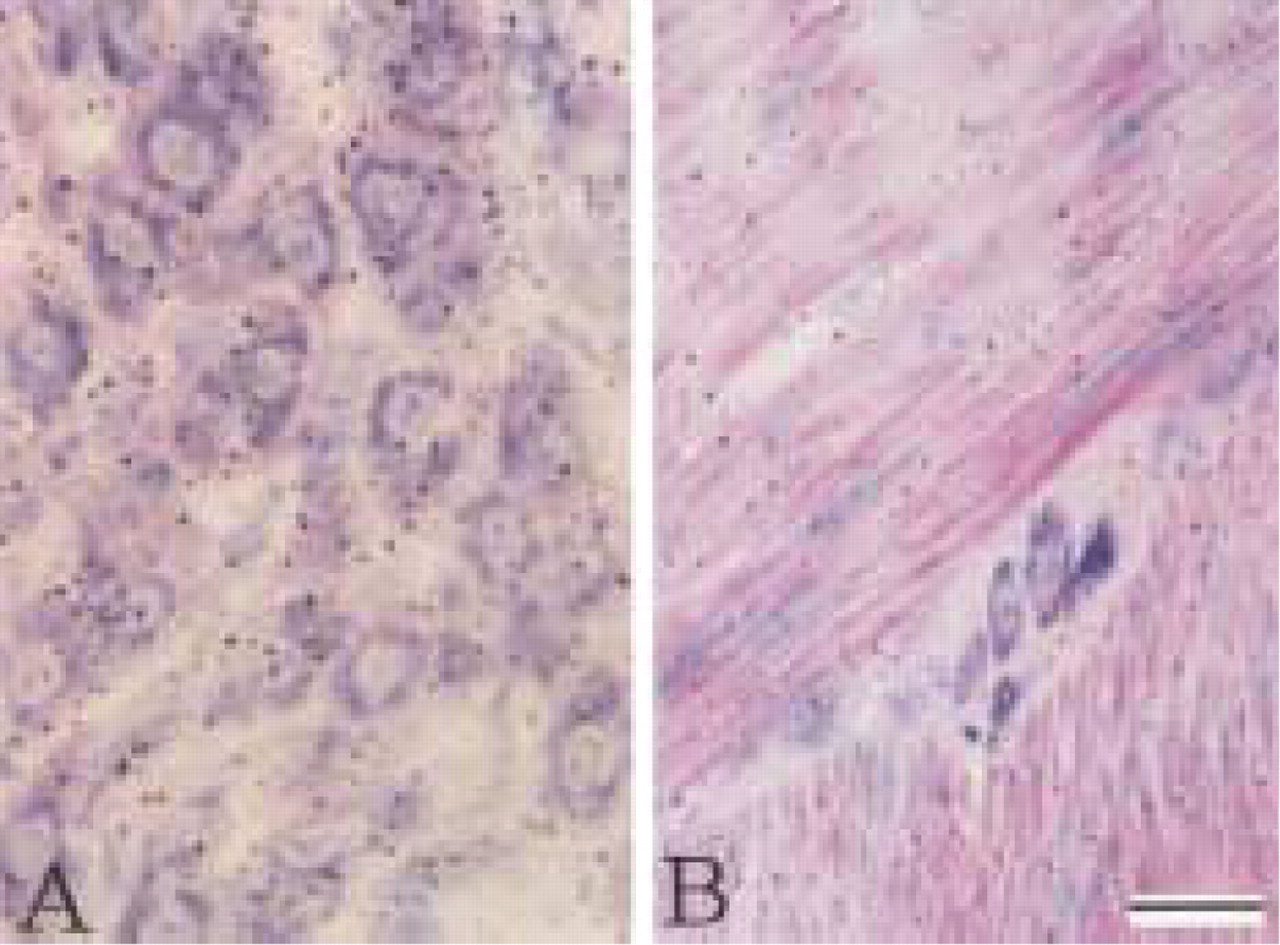
Autoradiographs of liver (
Discussion
In this study, quantitative receptor autoradiography was used to determine in vivo saturation doses for nuclear uptake of [26-3H]-OCT by individual cell types of the parathyroid, duodenal villi, kidney proximal and distal tubules, and skin epidermis.
Autoradiography has high resolution and sensitivity for demonstrating ligand-receptor binding at cell levels compared with biochemical approaches at tissue levels. By quantitative immunoradiometric assay with tissue extracts, tissue levels of 1,25(OH)2D3 receptor were found to be highest in proximal small intestine and colon, containing over 1000 fmol/mg total protein, where ileum and kidney contained one half and one fourth of this amount, and thyroid/parathyroid, bone, and skin moderate levels of receptor of 40-80 fmol/mg (Sandgren et al. 1991). In contrast, by quantitative receptor autoradiography, target cell levels of OCT binding to VDR were relatively different. Because unlabeled 1,25(OH)2D3 prevented [26-3H]-OCT from accumulating in nuclei, OCT may bind to the same receptor as 1,25(OH)2D3. At the maximal dose, the silver grain numbers over nuclei were highest in duodenal epithelium, high in kidney distal tubule epithelium and parathyroid chief cells, intermediate in kidney proximal tubule epithelium, and lowest in skin epidermis. The data obtained by the histochemical approach differ from those obtained by the biochemical immunoradiometric assay. Because the biochemical data are based on homogenized tissue extracts, different cell populations are lumped together and the ratio of target cell vs nontarget cell, such as parathyroid chief cells and thyroid follicle cells, may modulate or even obscure the desired information on receptor content. It is noteworthy that the parathyroid chief cells contain a relatively high amount of receptor-ligand binding among the target cells, when information is based on individual cell types and cell resolution.
Because OCT circulates mainly bound to LDL (Teramoto et al. 1995), the metabolic clearance from blood (plasma T1/2) is shorter than that of 1,25(OH)2D3 bound to DBP. The plasma T1/2 of OCT is 30 min after intravenous injection in rats (Dusso et al. 1991). However, it is noteworthy that nuclear receptor occupation remains at several hours (unpublished autoradiographic diographic data) despite low blood levels. This study was performed at one hour after intravenous injection, when near-peak concentration exists in target cell nuclei.
Dose-related nuclear receptor binding after in vivo administration of [3H]-22-oxa-1,25-dihydroxy vitamin D3 (OCT) in parathyroid, duodenum, skin, kidney, and liver prepared from autoradiographs a
One hour after i.v. injection of [3H]-OCT. Mean number of silver grains (SG over nuclei before
and after
subtraction of extracellular tissue background (BG). SG were counted over at least 100 cells for each structure in two rats for each condition. Cytoplasmic radioactivity was measured over muscle cells of the muscularis externa in duodenum and over hepatocytes.
Competition with 1000–fold excess of OCT or 1,25(OH)2D3 (VD) injected together with 2.0 μg/kg of [3H]-OCT suppressed or eliminated nuclear concentration of radioactivity.
In vivo nuclear receptor “saturation” (
Comparable histochemical or biochemical receptor saturation studies are not available in the literature for vitamin D, but exist for estradiol. When the data reported in this study are compared with those reported for estradiol, there is good correspondence regarding saturation dose range and differential of receptor saturability. Using a [3H]-estradiol exchange assay, Anderson et al. (1973) showed that uterine nuclear saturation occurs between 1.0 and 2.5 μg/rat. Using quantitative autoradiographic analysis of in situ [3H]-estrogen concentration and saturation by individual cell types, one hour after intravenous injection, in the uterus and vagina nuclear silver grain numbers for six cell types increased with dose from 1.0 to 27 μg/kg bw when they plateaued. In the pituitary, four target cell types plateaued at 9 μg/kg bw, but thyrotropes at 3 μg/kg bw. Both specific tissues, as well as their different cell types, reveal differences in saturation level and uptake capacity of [3H]-estrogen in the mouse (Holderegger et al. 1981).
The results with [26-3H]-OCT are in general agreement with those of [3H]-estradiol, indicating in vivo saturate levels of nuclear receptor binding between 2 to 8 μg/kg bw for OCT and 3 to 9 or 27 μg/kg bw for estradiol. These results together demonstrate a cell type-specific behavior of receptor-ligand binding, which is likely to be an important factor in the functional responses of specific target cell populations.
Parathyroid chief cells required the lowest receptor saturation dose of [26-3H]-OCT among the target cells. This low-dose receptor saturability indicates a high sensitivity of the parathyroid to treatment with OCT as well as 1,25(OH)2D3, because both compounds share the same receptor (Stumpf et al. 1994, 1995a, b). This sensitivity of the parathyroid to vitamin D and OCT compound is reflected in the results of applications reported in the literature. 1,25(OH)2D3 receptors are decreased in parathyroid glands in chronically uremic dogs (Brown et al. 1989a) and in patients with renal failure (Korkor 1987). OCT was able to prevent the decrease in vitamin D receptor content in parathyroid glands of uremic rats, even though its potency was less than that of 1,25(OH)2D3 (Denda et al. 1996). Secondary hyperparathyroidism in uremic patients was effectively suppressed by treatment with 1,25(OH)2D3 (Slatopolsky et al. 1984). Accordingly, the results of these clinical and experimental studies, together with the strong nuclear concentration of radiolabeled OCT in parenchymal cells of the parathyroid demonstrated in the present autoradiographic experiments, suggest a high therapeutic utility of OCT for the regulation of parathyroid function. OCT that binds to the same receptor as 1,25(OH)2D3, with an apparent high affinity for the parathyroid, has much fewer systemic effects on plasma calcium levels than 1,25(OH)2D3 and is therefore less toxic than the parent compound in the treatment of parathyroid dysfunctions.
