Abstract
Microscopic analysis in combination with cytochemistry and immunocytochemistry has revealed the presence of four cell types not previously described in the portal area and parenchyma of the liver from an experimental rodent hepatocarcinogenic rat model. Within the intrahepatic bile ductules, which proliferate after administration of chemical carcinogens and partial hepatectomy, small, undifferentiated nonpolarized, nonepithelial cells with a blast-like phenotype and polarized epithelial cells different from the polarized epithelial cells that typically line the walls of the bile ductules were found. In the connective tissue stroma surrounding the bile ductules, nonpolarized epithelial cells with hepatocyte phenotype were found. In the parenchyma, subpopulations of bile ductule epithelial cells that established ATPase-positive bile canalicular structures, including the formation of desmosomes and tight junctions, with parenchymal hepatocytes within the hepatic lobule were found. These observations raise the following questions in this model. Are there undifferentiated progenitor cells with stem cell-like properties within bile ductules? What are the interrelations of the newly described cell types with each other, with parenchymal hepatocytes, with preneoplastic nodules, and with hepatomas? Do the heterogeneous cell types within the bile ductules, in the surrounding connective tissue, and within the hepatic cords represent intermediate stages of single or multiple cell lineage pathways leading to hepatocyte differentiation, liver regeneration, and/or preneoplastic nodule formation?
Keywords
S
We report here additional studies on the portal region of rat liver after a carcinogenic protocol. We substantiate and extend our earlier findings that during the early stages of liver carcinogenesis a greater diversity of cell types is present within the proliferating bile ductule system, the connective tissue stroma surrounding the ductules, and the hepatic parenchyma. Our new studies demonstrate that the blast-like cells in the basal region of the bile ductules acquire the properties characteristic of bile ductule epithelial cell populations, parenchymal hepatocytes, and preneoplastic nodular cells. The ductal blast-like cells replicate and undergo a differentiation program that involves the development of polarity and specialized plasma membrane properties, including the formation of desmosomes, tight junctions, and apical microvilli. We also describe other cell types that have not been previously seen in this model, including the following: polarized transitional cells that have catalase-positive peroxisomes which migrate out of the bile ductule structures: nonpolarized hepatocyte-like cells in the connective tissue stroma; and subpopulations of polarized bile ductule epithelial cells that integrate within the hepatic cords. This latter cell type is in direct contact with parenchymal hepatocytes and forms ATPase-positive bile canalicular structures as well as desmosomes and tight junctions with hepatocytes. These studies suggested that the ductal blast-like cells may have multipotential stem cell-like properties and that diverse and multiple cell lineage pathways may be involved in hepatocyte differentiation, liver regeneration, and preneoplastic nodule formation.
Materials and Methods
Animals
Male Fisher 344 rats weighing 120–130 g were obtained from Charles River Laboratories (Wilmington, MA) and maintained in a central animal facility with a 12-hr light/dark cycle for 1 week before treatment with a carcinogen and partial hepatectomy protocol (Solt et al. 1977; Faris et al. 1991). The protocol was administered to 10 rats. It consists of the following: a single
Tissue Fixation
Liver slices (2–3 mm thick) were removed from the rats and fixed in the following fixatives by immersion: (a) cold 4% paraformaldehyde in 0.1 M PBS, pH 7.4, for 3 hr with shaking; (b) a cold mixture of 4% paraformaldehyde/2.5% glutaraldehyde in 0.1 M cacodylate buffer, pH 7.4, for 3 hr with shaking (Karnovsky 1965); (c) 4% osmium tetroxide, pH 7.4, for 1 hr after above Fixative a or b. Liver slices fixed in Fixatives a or b were cryoprotected by infusion with increasing concentrations of cold sucrose before sectioning.
Tissue Sectioning
Liver sections were prepared as follows: (a) aldehyde-fixed frozen sections (10–20 μm) with a Sartorius freezing microtome (Leitz; Gottingen, Germany); (b) aldehyde-fixed non-frozen sections (15–30 μm) using a Lancer vibratome, Series 1000 (Polysciences; Warrington, PA); (c) 1-μm and 50–80-nm Epon-embedded sections using an LKB ultramicrotome II (LKB; Stockholm, Sweden).
Enzyme Cytochemistry
Free-floating aldehyde-fixed (Fixative b, above) frozen and nonfrozen sections were incubated for the following enzymes: (a) catalase activity in peroxisomes and microperoxisomes, using a medium containing 3,3′diaminobenzidine tetrahydrochloride (DAB-HCl) (Sigma) as substrate at pH 9.7 (Novikoff et al. 1977) at 37C for 30–45 min; (b) adenosine triphosphatase (ATPase) activity in bile canaliculi using as substrate sodium salt of adenosine triphosphate at pH 7.4 at 37C for 30–45 min (Wachstein and Meisel 1957); (c) endogenous or exogenous peroxidase activity using a medium containing DAB-HCl, pH 7.4, at room temperature (RT) for 15–20 min (Graham and Karnovsky 1966). Controls for each enzyme included incubation in medium without substrate.
Immunocytochemistry
Free-floating aldehyde-fixed (Fixative a, above) frozen and nonfrozen sections were exposed to the following antibodies overnight at 0C: (a) cytokeratin 19 monoclonal antibody (Amersham; Arlington Heights, IL); (b) rabbit polyclonal anti-rat glutathione-S-transferase Yp (Biotrin International; Dublin, Ireland). Either a peroxidase-labeled second antibody method or a fluorescent-labeled (Cy 3 or Cy 5) second antibody method (Jackson Immunoresearch Laboratories; West Grove, PA) was employed to detect primary antibodies. Pretreatment of sections to inhibit endogenous peroxidase (with hydrogen peroxide; Sigma), inhibit background fluorescence (with sodium borohydride; Sigma), prevent nonspecific binding of primary antigens (with goat serum, bovine serum albumin; Sigma), and enhance immunoreagent penetration into tissue (Triton X-100, Tween; Sigma) were performed as previously described (Novikoff et al. 1991,1996a,b). Controls included exposure to all reagents except primary antibodies. Sections were also exposed to fluorescein (FITC)-labeled phalloidin (Molecular Probes; Eugene, OR) (1:20 dilution of stock solution (300 U/1.5 ml of cold methanol) for 60 min RT to detect actin filaments and to propidium iodide (Molecular Probes) (1 mg/50 ml of PBS) for 5 min at RT to detect nuclear and cytoplasmic nucleic acids. Sections were also sequentially stained for the co-localization of cytokeratin 19 using Cy 5-labeled second antibody, FITC-phalloidin, and propidium iodide.
Microscopy
Fluorescent-labeled liver sections were examined with an inverted Nikon fluorescent microscope using a X60 oil immersion lens with numerical aperture 1.4 Planapo objective. The microscope was attached to the Bio-Rad MRC 600 confocal laser imaging system (Bio-Rad Laboratories; Hercules, CA) equipped with a krypton/argon laser. Black level, gain and laser intensity, Kalman averaging, excitation intensity, pinhole aperture, and Z-series analysis of sections were performed as previously described (Novikoff et al. 1996a). Enzyme cytochemically labeled liver sections were examined and photographed with a Zeiss Ultraphot II (Carl Zeiss; Thornwood, NY). Ultrathin Epon-embedded sections were examined with a Philips EM 300 equipped with a goniometer stage (Philips Electronic Optics; Millersville, MD).
Results
Confocal Microscopic Observations
Nonpolarized and Polarized Bile Ductule Cells. Earlier light microscopic studies of sections of liver from rats treated with AAF-DEN 2–3 days post PH revealed the presence of small cells situated at the base of the ductules underneath the bile ductule epithelium (Novikoff et al. 1996b). The small basal cells did not appear to be in contact with the lumen of the ductule, suggesting that the small basal cells were not polarized. To determine whether the small basal cells had contact with the ductule lumen and whether these cells possessed a polarity similar to that of bile ductule epithelial cells, we performed confocal microscopic analysis of the basal cells. We used fluorescent-labeled phallodin to label the luminal aspect of the ductule cells and propidium iodide to label nuclei. Figure 1a and inset illustrate the presence of small basal cells located beneath bile ductule epithelial cells. Actin staining (green) is evident at the luminal surface of all the cells that comprise the bile ductule except the small basal cells. The small basal cells appear to have a spherical nucleus, a clear zone around them, do not have actin localized on any of their surfaces, and are beneath bile ductule epithelial cells that have actin on the apical surface in contact with the ductule lumen. Laser scanning of the liver sections through their entire depth at intervals of 0.5 μm revealed that actin staining was not detected at any level of the small basal cells whereas actin staining was present at all levels of the bile ductule epithelial cells that line the ductule (see Figure 2 in Novikoff et al. 1996b). Figure 1a, inset, illustrates the properties of the small basal cells within the ductules more clearly and also shows that ductule cells with this phenotype are not evident in the surrounding connective tissue stroma. Confocal microscopy therefore established that the basal cells were not in contact with the lumen and were not polarized like the bile ductule epithelial cells. Previous confocal analysis studies also showed that the basal cells did not express epithelial markers or lymphocyte markers (Novikoff et al. 1996b). Figures 1b and 1c show the tri-localization of actin, a polarization marker, of cytokeratin 19, a bile ductule epithelial marker, and of propidium iodide, a nuclear marker. Actin (yellow) is present at the luminal surface of the bile ductule epithelial cells and in bile canaliculi of contiguous hepatocytes (green). Cytokeratin 19-positive bile ductule cells (red) extend from the portal triad area into the hepatic parenchyma (Figure 1b). Single and groups of cytokeratin 19-positive cells are found within the hepatic cords interspersed between hepatocytes (Figure 1c). One of the cytokeratin 19-positive bile ductule cells exhibits an elongated profile and shows actin staining on two regions of its surface, one region in contact with the ductule lumen and the other in contact with a bile canaliculus of a parenchymal hepatocyte (Figure 1b). The nucleus of this elongated ductule cell is a considerable distance from the luminal surface. This ductule cell has changed its normal apical-basal polarity and has formed two surfaces that are actin-positive. Figure 1c reveals that a subpopulation of cytokeratin-19-pos-itive bile ductule cells integrate within the hepatic lobule and form bile canalicular structures, as determined by actin localization. These bile ductule cells do not appear to be attached to other bile ductule cells or to have a surface exposed to the ductule lumen, as illustrated in Figure 1b. Previous ultrastructural studies in a different carcinogenic rat model have shown that where bile ductule cells and hepatocytes contact, bile canalicular structures with characteristic microvilli, tight junctions, and desmosomes are formed. The newly formed bile canaliculi demonstrate activity of ATPase, an ectoenzyme present in normal hepatocyte bile canaliculi (Novikoff et al. 1991; Hixson et al. 1992), whereas the microvilli of the bile ductule cell facing the ductule lumen do not express ATPase activity. In the DEN-AF-PH rat model, we have also found a sub-population of bile ductule cells in contact with parenchymal hepatocytes. Figure 2a illustrates the formation of a bile canaliculus that expresses ATPase activity between a bile ductule cell and a hepatocyte. The ductule cell in contact with the hepatocyte is also in contact with the ductule lumen. No ATPase activity is associated with the ductule lumen plasma membrane. Microvilli are present on the plasma membrane bordering the ductule lumen and the plasma membrane of the bile canaliculus. These ultrastructural studies confirm the confocal microscopic observations (Figure 1b) that there are subpopulations of bile ductule cells with two luminal surfaces. Basal lamina is seen surrounding the bile ductule cells except for the bile ductule cell that is in contact with hepatocytes.
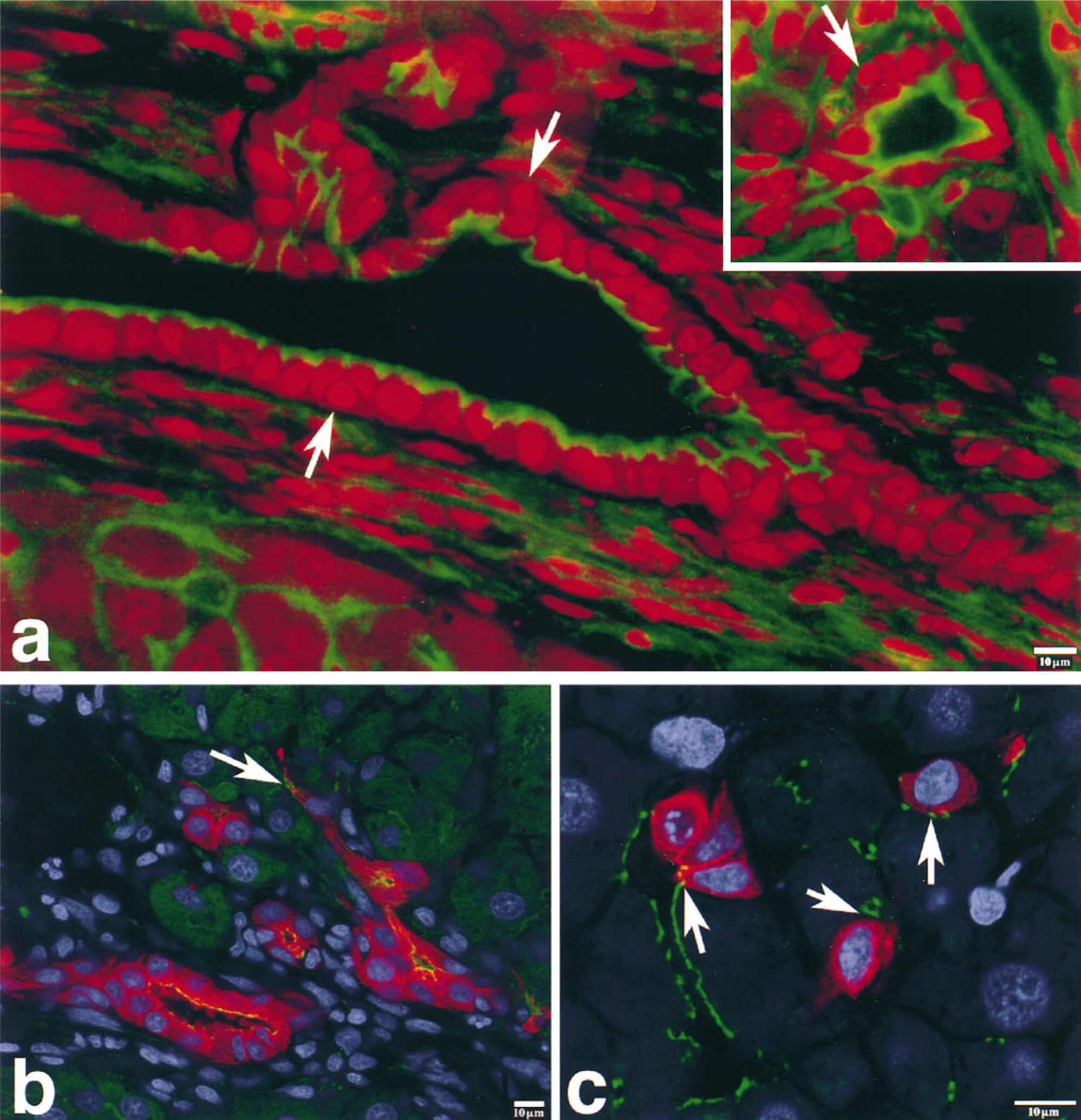
Confocal microscopic images of liver sections from a rat treated with DEN-AAF-PH 3-day protocol. (a and inset) Co-localization of actin (green) and propidium iodide (red). Actin is distributed at the apical surface (facing the lumen) of the bile ductule epithelial cells. Arrows indicate small basal cells within the bile ductules. The small basal cells do not show actin on their apical surface, are not in contact with the lumen, and are enveloped by bile ductule epithelial cells. The nucleus of the basal cell appears spherical. Note that branches of the bile ductule extend into the hepatic parenchyma. Actin is also localized in bile canaliculi of hepatocytes. (Inset) Cross-section of another bile ductule reveals a basal cell that does not have actin on its surface and is not in contact with the lumen. (b,c) Co-localization of actin (yellow, green), cytokeratin-19 (red), and propidium iodide (blue). Arrow in b indicates an elongated cytokeratin-19-positive bile ductule cell that shows actin localization at the ductule lumen (yellow) and at a bile canalicular structure (green) formed between the bile ductule cell and a hepatocyte. Arrows in c show actin-positive bile canalicular structures (green) that have formed between a subpopulation of cytokeratin-19-positive bile ductule cells (red) and parenchymal hepatocytes. The cytokeratin-19-positive cells appear integrated within the hepatic cork.
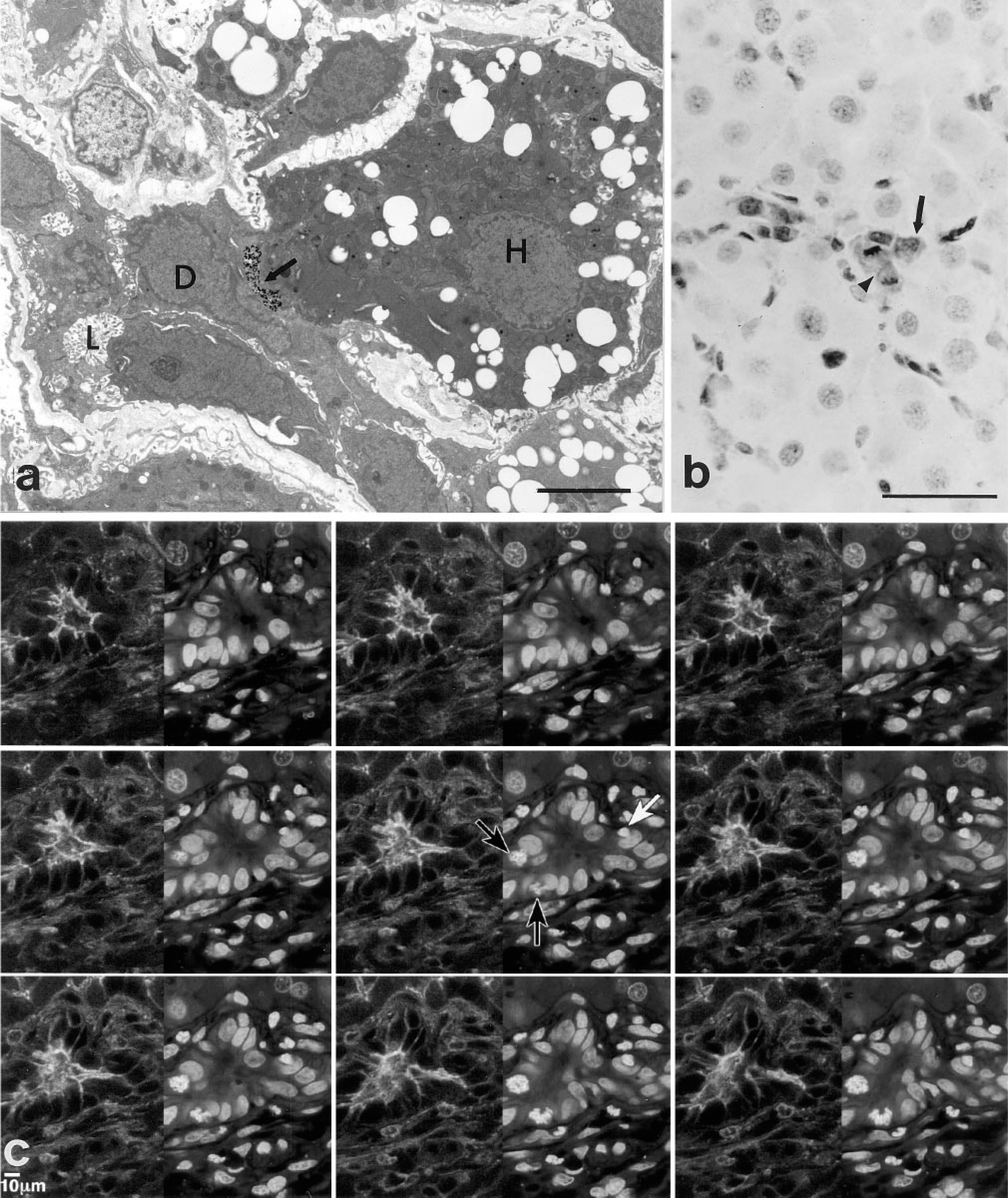
Localization of ATPase activity in a section of rat liver. A bile ductule is evident within the liver lobule. Arrow indicates an ATPase-positive bile canaliculus that has formed between a bile ductule cell (D) and a hepatocyte (H). The bile ductule cell (D) in contact with the hepatocyte (H) retains its exposure to the ductule lumen (L). There is no demonstrable ATPase activity in the ductule lumen and basal lamina is not present where the bile ductule cell and the hepatocyte contact. Bar = 5 μm. (
Bile Ductule-derived Cells Integrate and Proliferate into the Hepatic Cord. Confocal microscopic analysis also revealed the presence of subpopulations of bile ductule epithelial cells that appeared to be integrated within the hepatic cord. Single or multiple contiguous cytokeratin 19-positive cells are in contact with hepatocytes within the hepatic cords and form actin-positive bile canalicular structures (Figure 1c). One of the cytokeratin-19-positive cells is in mitosis, suggesting that replication of these cells occurs within the hepatic lobule. We also found GST-Yp-positive cells integrated within the hepatic cord, some of which are in mitosis (Figure 2b). These cells are also cytokeratin-19-positive, suggesting that they have a bile ductule phenotype. None of the parenchymal hepatocytes in Figure 2b expresses GST-Yp. We do not know the cell lineage relations between the bile ductule cells with the dual phenotype, i.e., both hepatocyte and bile ductule properties (Figures 1b and 2a) that are part of the ductule, and the cytokeratin-19-positive and GST-Yp-positive bile ductule cells (Figures 1c and 2b) that appear not to be part of the ductule but appear to be integrated within the hepatic cord and have formed actin-positive bile canaliculi with parenchymal hepatocytes. Whether these cells belong to the same cell lineage pathway or represent different subpopulations of bile ductule cells that are in the process of differentiating along separate cell lineage pathways remains to be determined. However, at this stage of their differentiation, we have only determined that they have acquired one hepatocyte phenotype, i.e., the formation of bile canaliculi.
Mitotic Images of Basal Cells Within Bile Ductiles. Previous studies showed that the basal cells express proliferating cell nuclear antigen and showed basal cells in mitosis, indicating that the basal cells have the capacity to proliferate (Novikoff et al. 1996b). We have encountered additional basal cells in mitosis within bile ductules by performing laser scanning of the liver sections. Figure 2c illustrates a scanned image of a liver section showing a bile ductule region stained for actin and propidium iodide. In the initial scan, basal cells were not evident. However, by performing a Z-series analysis, we detected two basal cells in mitosis at the basal aspect of the ductule as well as a nonmitotic basal cell. This scanned image also shows that the basal cells do not express actin on their apical sufaces at any level of the section, confirming the observation that the basal cells are not polarized.
Electron Microscopy
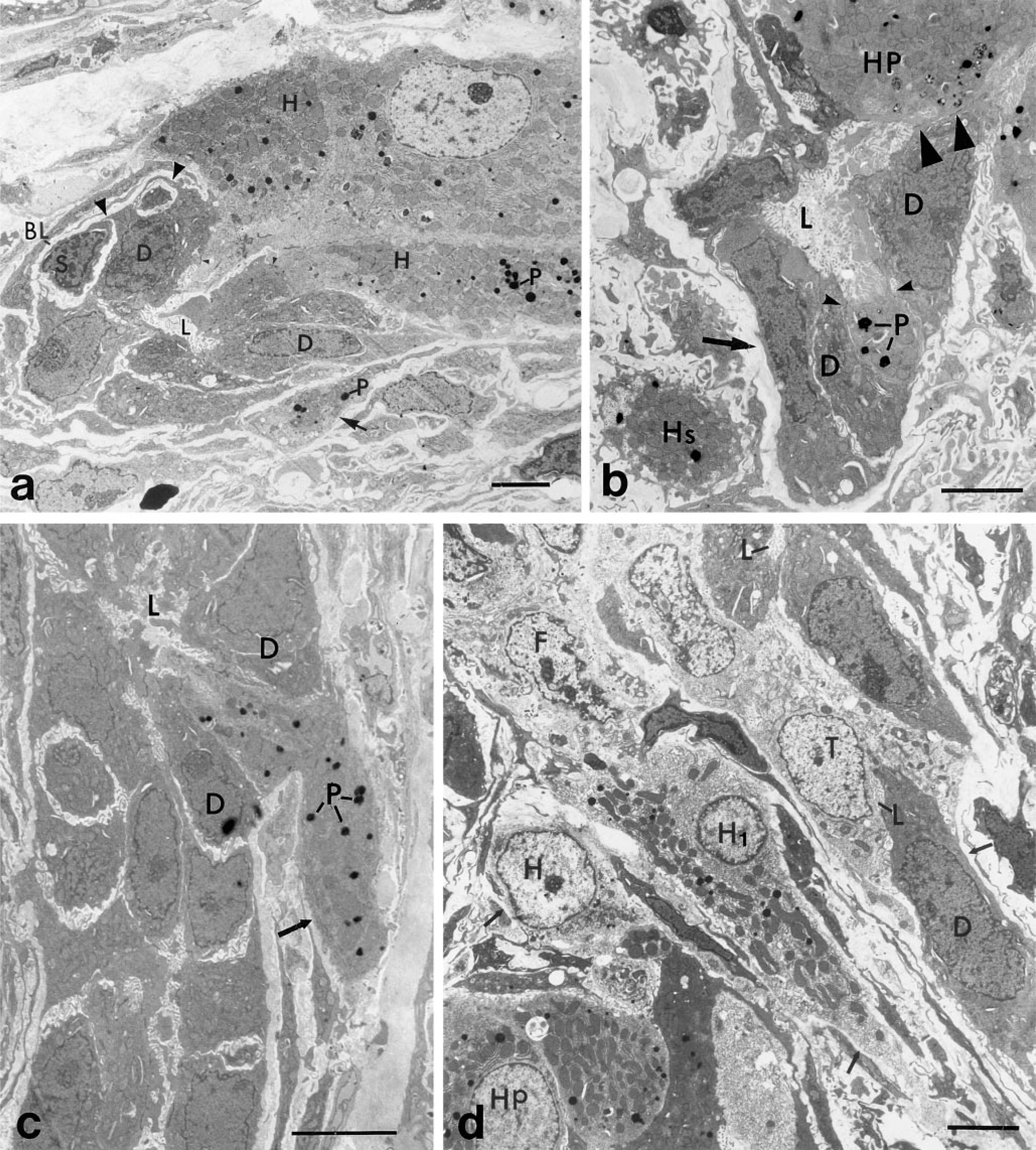
Heterogeneity of Cell Phenotypes Within Bile Ductules. Light microscopic studies of the livers of mice suggested that the duct of Hering, a specialized region of bile ductule cells contiguous with parenchymal hepatocytes, was the site of a stem-cell compartment because when toxins were used to destroy hepatocytes, bile ductule cells proliferated and differentiation of bile ductule cells into cells with hepatocyte phenotype was observed (Wilson and Leduc 1958). These observations led us to study the portal region of hepatocytes in the AAF-DEN-PH liver carcinogenic model to determine whether a stem-cell compartment is also present in this model. Ultrastructural analysis of regions of bile ductules analogous to the duct of Hering and in the connective tissue stroma surrounding the bile ductules revealed cell phenotypes not previously described (Figures 3a–3d). We found that the cell phenotypes within the bile ductule are heterogeneous and include polarized epithelial cells (Figures 3a–3d), some of which contain catalase-positive peroxisomes (Figures 3b and 3c) and small, less-differentiated cells (Figure 3a) at the base of the ductule that correspond to the basal cells we observed with the confocal microscope (Figures 1a, 1a inset, and 2c). We also found nonpolarized epithelial cells within the stroma that contained catalase-positive peroxisomes (Figures 3a, 3b, and 3d). Figures 3a and 3b show duct of Hering regions of the bile ductule in which parenchymal hepatocytes and bile ductule cells are contiguous and desmosomes are present between the adjacent bile ductule cells and hepatocytes. Catalase-positive peroxisomes are seen only in hepatocytes (Figures 3a–3c), in polarized cells within the ductule (Figures 3b and 3c), and in elongated nonpolarized cells within the connective tissue stroma (Figures 3a, 3b, and 3d). The less-differentiated basal cells we observed are not polarized like the bile ductule epithelial cells, i.e., their apical surface is not in contact with the ductule lumen and their basal surface does not rest on a basal lamina (Figure 3a). The basal cells are in close proximity to a subpopulation of bile ductule cells that extend thin cytoplasmic extensions around the basal cells and separate them from the basal lamina. Figure 4a illustrates more distinctly the ultrastructural features of basal cells and is a serial section of a previously published micrograph (Figure 6b in Novikoff et al. 1996b). The basal cells resemble the undifferentiated or blast-like phenotype expected of stem cells. The basal cells exhibit a nucleus that occupies most of its interior, has a thin rim of cytoplasm with no or few organelles, is without plasma membrane specializations, and appears to have a clear zone around them. Figure 4a also illustrates the different cell types within the bile ductule, two of which had not been previously described in this model. In addition to the undifferentiated basal cell, another cell with polarized properties of the more typical bile ductule epithelium is seen. However, this polarized cell has a lighter cytoplasm and a larger nucleus. Earlier studies from our laboratory have shown that ductule cells with this phenotype acquired a few catalase-positive peroxisomes and were termed ductule transitional hepatocyte-like cells (Novikoff et al. 1996b). The ultrastructural appearance of the ductule transitional cells resembles the ductal hepatocytes seen in bile ductule hyperplasia induced in rats by furan (Sirca et al. 1994). In the present study, we also found polarized ductule cells in the bile ductule that contained many catalase-positive peroxisomes (Figures 3b and 3c). Profiles of polarized bile ductule epithelial cells containing catalase-positive peroxisomes appear to migrate from the ductule into the surrounding basal lamina (Figure 3c). In addition to polarized bile ductule cells with peroxisomes, there are nonpolarized cells with catalase-positive peroxisomes in the surrounding connective tissue stroma, suggesting that these cells also have hepatocyte properties. Two of these cells are seen between bile ductules and parenchymal hepatocytes (Figure 3d). In addition to peroxisomes, they contain the characteristic smooth endoplasmic reticulum of differentiated parenchymal hepatocytes but do not exhibit the plasma membrane specialization of differentiated hepatocytes such as bile canaliculi. The origin of the cells in the stroma and their relation to bile ductule epithelial cells containing catalase-positive peroxisomes in bile ductules, to parenchymal hepatocytes, and to nodular cells remains to be determined.
Liver sections of bile ductules (duct of Hering region) incubated for catalase activity. (
Replication and Differentiation of Ductule Nonpolarized Basal Cells. Our ultrastructural studies have confirmed the confocal microscopic studies indicating that basal cells undergo mitosis. Figure 4b shows a basal cell in a metaphase stage of mitosis that is located beneath a bile ductule epithelial cell. The chromososmes of the dividing basal cells are distinct. The mitotic basal cell is not in contact with the ductule lumen but is associated with the basal lamina. We have also found basal cells at the end of mitosis. Figure 4c and 4d show the separation of two basal cells after the completion of mitosis. The two basal cells in Figure 4d may have resulted from the mitosis of a single basal cell because cytoplasmic connections are evident between them. These basal cells are not in contact with the basal lamina, in contrast to the mitotic basal cell in Figure 4b. We have also encountered basal cells within the ductules that appear more differentiated than the basal cell in Figure 4a or in Figure 6a of a previous publication (Novikoff et al. 1996b). The more differentiated basal cells are most likely derived from the less-differentiated basal cells. Figures 4c–4e illustrate more differentiated basal cells beneath bile ductule cells. They have more cytoplasm but their plasma membrane surface is without specializations. The basal cells in Figures 4c and 4d are not in contact with the ductule lumen or the basal lamina and are enclosed by cytoplasmic extensions of the adjacent bile ductule epithelial cells. The basal cells in Figures 4b and 4e are also not in contact with the ductule lumen but are in contact with the basal lamina. We frequently find fibroblasts in close proximity to the region of the bile ductule at which basal cells are located (Figures 4b, 4d, and 4e).
Section of a bile ductule within the hepatic lobule comparing the different morphologies of an undifferentiated basal cell (S), a ductule transitional cell (T), and bile ductule epithelial cells (D). The basal cell shows a high nucleus-to-cytoplasm ratio, has no cytoplasmic organelles or plasma membrane specializations, and is not exposed to the ductule lumen (L) or the basal lamina (arrow). The ductule transitional cell (T) and the bile ductule epithelial cells (D) show similar apical-basal polarity. However, the transitional cell has a larger nucleus and lighter cytoplasm. Desmosomes (arrowheads) are present between the polarized ductule transitional cell and bile ductule epithelial cells. No desmosomes are present on the basal cell. (
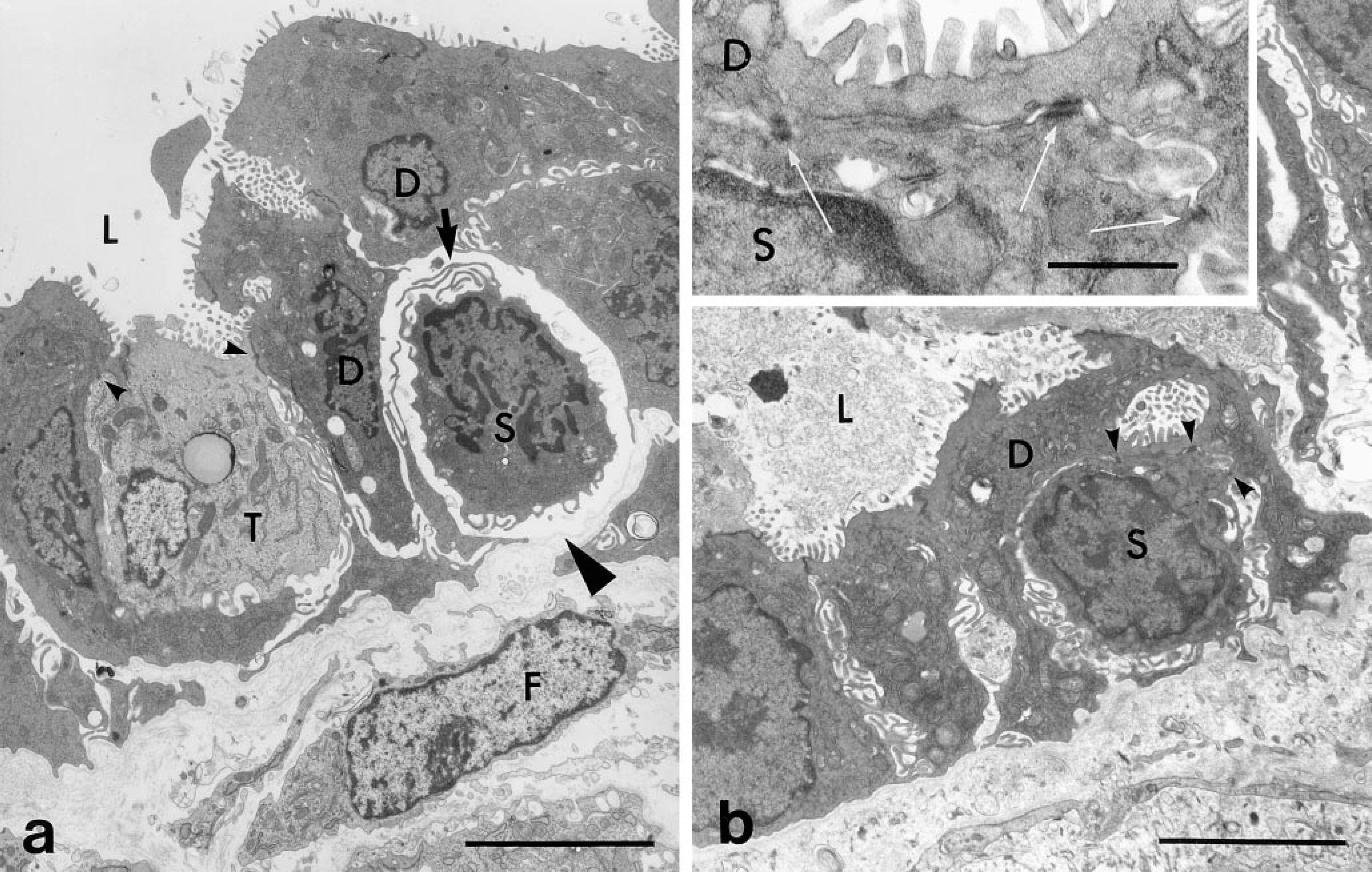
Section of bile ductule showing basal cell differentiation. (
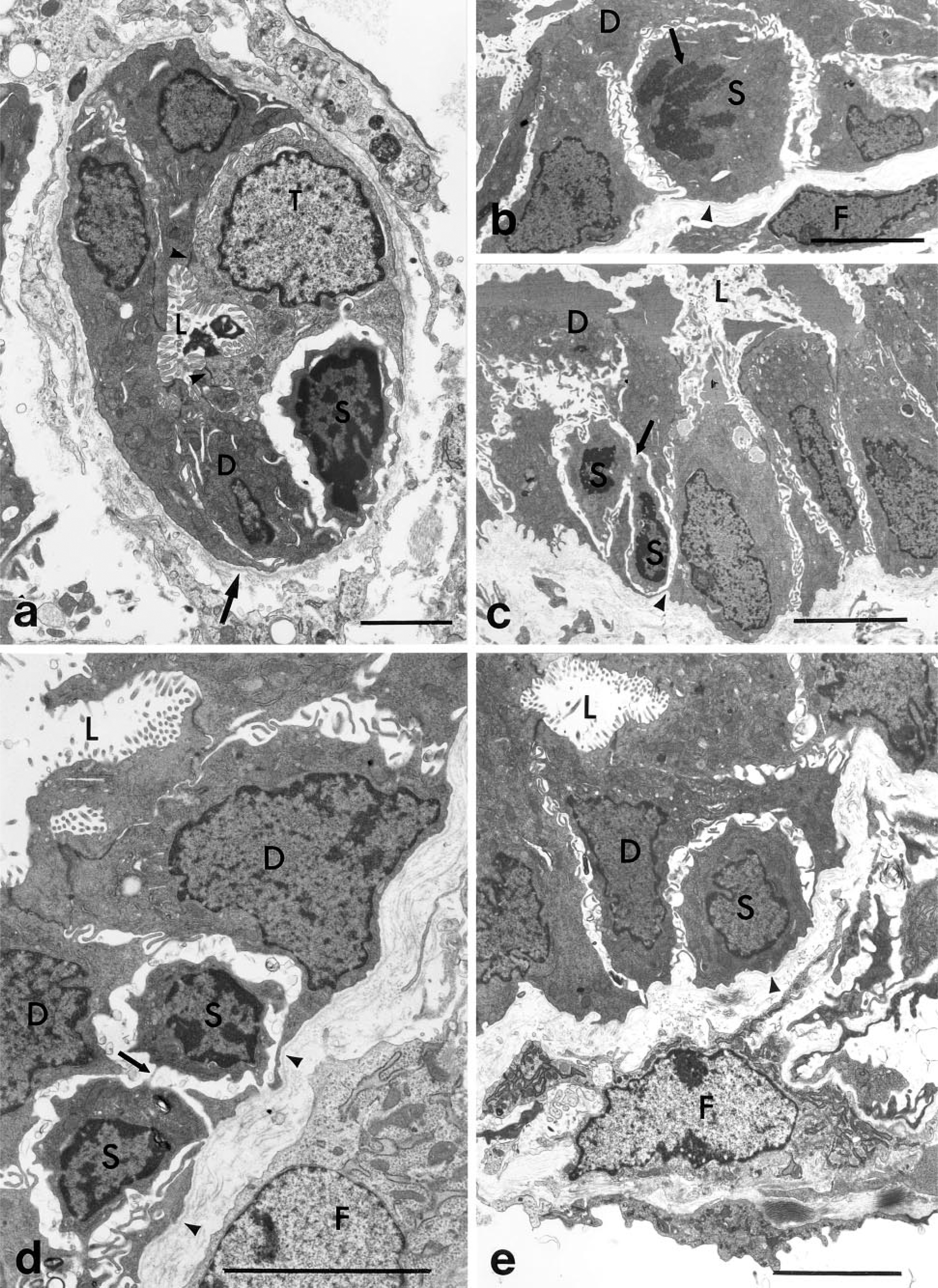
Basal Cells Develop Plasma Membrane Structures. The close proximity of basal cells and bile ductule epithelial cells is suggestive of a lineage relationship between them. To demonstrate whether the basal cells are progenitor cells to bile ductule cells or other hepatic cells, the basal cells have to be labeled with markers such as [3H]-thymidine or retroviral vectors so that more direct cell lineage studies can be performed. We have demonstrated that the basal cells divide, permitting such direct studies to be performed. Our laboratory is currently performing such studies. However, in the absence of more direct studies, we have been analyzing the basal cells for the acquisition of intrinsic structural plasma membrane markers characteristic of ductule epithelial cells, parenchymal hepatocytes, and preneoplastic nodular cells. These include apical microvilli, desmosomes, tight junctions, and gap junctions. These plasma membrane properties are detectable by electron microscopy and would provide strong but not unequivocal evidence that basal cells can differentiate into cells with an epithelial phenotype. Figure 5a shows a basal cell with a change in the structure of the plasma membrane that faces the ductule lumen. The plasma membrane has developed structures resembling microvilli. There is also a change in the lateral plasma membrane of the basal cell, suggesting possible development towards the establishment of cell polarity. We have also found that the basal cells develop another plasma membrane specialization, i.e., desmosomes. Figure 5b illustrates the presence of three small desmosomes that have developed between the plasma membrane of the basal cell and the bile ductule epithelial cell. The characteristic ultrastructure of the desmosomes is evident in the enlargement in the inset. Therefore, by an analysis of the ultrastructural properties of the plasma membrane of basal cells, we have suggestive evidence that the basal cells have the potential to differentiate into polarized ductule epithelial cells, parenchymal hepatocytes, and preneoplastic cells.
Discussion
Relation of Ductule Basal Cells to Stem Cells
Previous studies have suggested a variety of epithelial cell types as candidates for liver stem cells or progenitor cells. These included oval cells, bile ductule cells, periductular cells, transition duct cells, embryonic bipotential cells, and rat liver-derived epithelial (RLE) cell lines (Grisham and Porta 1964; Lombardi 1982; Sell and Salman 1984; Tsao and Grisham 1987; Marceau et al. 1989; Fausto 1990; Coleman et al. 1993). We reported in a rat liver carcinogenic model the presence of nonepithelial ductule basal cells that exhibited an undifferentiated phenotype and differed from other previously reported putative stem or progenitor cells in that they are not polarized, as determined by actin localization in apical microvilli, and not epithelial, as determined by the absence of the epithelial marker cytokeratin-19 (Novikoff et al. 1996b). The undifferentiated ductule basal cells have a blastlike nuclear and cytoplasmic morphology resembling the phenotype of stem or progenitor cells of the hematopoietic system (Van Bekkum 1977). Our previous studies showed that the undifferentiated ductule basal cells do not have the morphology of lymphocytes and do not express T- and B-lymphocyte antigens (Novikoff et al. 1996b). The location of the ductule basal cells at the basal aspect of the ductule parallels that of stem cells from other tissues, e. g., intestinal crypt stem cells, deep rete epidermal stem cells, and limbal region corneal stem cells (LeBlond 1981; Lavker and Sun 1983; Cotsarelis et al. 1989). The enclosure of the ductule basal cells by bile ductule epithelial cells provides a niche or protective environment similar to that observed in stem cells from renewing tissues (Potten and Loeffler 1990; Alterman 1992). We have also found that the undifferentiated ductule basal cells also replicate and progressively develop a more differentiated phenotype. It appears that the ductule basal cells undergo a differentiation program. They increase in size, acquire more cytoplasm, and they or their progeny exhibit changes in plasma membrane ultrastructure. The plasma membrane at the apical surface facing the ductule lumen develops surface microvilli and desmosomes and the entire plasma membrane profile of the cells appears polarized. The factors that control proliferation of basal cells and differentiation of basal cells from a nonpolarized phenotype to a polarized phenotype are not known. Of interest is the consistent observation of the close proximity of fibroblasts and collagen fibers to the region of the bile ductules at which the basal cells are located. There is evidence that growth factors and connective tissue components, e.g., collagen and laminin, may be involved in cell proliferation and differentiation. Other investigators have observed fibroblasts in close proximity to bile ductules during carcinogenesis that expressed the growth factor TGF-α and desmin (Evarts et al. 1993). Whether the undifferentiated ductule basal cells we have found are putative liver stem cells, stem-cell progeny, or non-stem-cell progeny remains to be determined. It is known that for stem cells to be maintained they must undergo cell division to replenish themselves and produce non-stem-cell progeny that differentiate into mature cells (Potten and Loeffler 1990). It is not known whether the ductule basal cells we have observed in mitosis belong to a true stem-cell compartment or to an amplifying transit cell compartment committed to differentiate. We have observed ductule basal cells larger than the smaller, undifferentiated ductule basal cells. However, even the largest of the ductule basal cells did not express epithelial markers at the early time points during liver carcinogenesis that we studied. However, studies of the liver at later times in the carcinogenic protocol may reveal that ductule basal cells have acquired other phenotypes of hepatic epithelial cells. This is expected because we have found some ductule cells that have developed plasma membrane specializations, including desmosomes and apical plasma membrane microvilli, at early stages of the protocol. Immunocytochemical studies are in progress to determine whether the plasma membrane specializations express proteins associated with plasma membrane structural components, e.g., connexins in gap junctions, desmoplakin in desmosomes, and actin in microvilli. These studies would provide further evidence that the ductule basal cells have the capacity to differentiate and may be progenitors of bile ductule epithelial cells, hepatocytes, and/or preneoplastic nodules. Related to our observations is the concept that stem cells or transit cells may be transformed or become malignant during replication. One view is that because stem cells or their progeny are long-lived and proliferate, they are more likely to be transformed by carcinogens and progress to carcinoma (Potten and Morris 1988). Another variant of this view considers tumor development as a process that characterizes normal tissue development and that carcinomas arise from malignant stem cells that can differentiate but do not because they are perturbed or arrested and, as a result, an expanding population of proliferating tumor cells with various potentials for differentiation is produced (Pierce and Speers 1988; Sell and Pierce 1994).
Relation of Ductule Basal Cells to Preneoplastic Nodules
At present we have no direct evidence that the ductule basal cells enter a cell lineage pathway that gives rise to preneoplastic nodules in the DEN-AAF-PH rat model. Because the ductule basal cells replicate, it may be possible to label these cells with retroviral vectors and to follow their fate over time to determine their migration and differentiation into preneoplastic nodules. Because the hepatocytes are inhibited from proliferating in this model and the only cells that appear to proliferate are cells within the bile ductules, we may be able to provide direct evidence that a subpopulation of bile ductule cells contributes to the development of preneoplastic nodules. Several investigators (Williams 1980; Cameron 1989; Farber 1992; Gerlyng et al. 1994; Lin et al. 1995) have reported that the progenitor cells responsible for the formation of nodules and hepatomas are carcinogen-altered parenchymal hepatocytes. It is also possible that there are multiple cell lineage pathways for nodular development operating simultaneously in the liver during carcinogenesis that involve altered hepatocytes as well as sub-populations of bile ductule cells.
Fate of Ductule Transitional Cells
In addition to the ductule nonpolarized basal cells, we have also observed, within the proliferating bile ductule cells, polarized transitional ductule cells that exhibit a cytoplasmic hepatocyte phenotype, i.e., catalase-positive peroxisomes. These cells within the ductules have properties of both bile ductule epithelial cells and of parenchymal hepatocytes. They have actin-positive microvilli on the plasma membrane surface facing the lumen and some are seen in contact with the basal lamina. Most of the transitional cells are cytokeratin-19-positive, although we have seen some that did not express this antigen. However, the ductule transitional cells differ from the bile ductule epithelial cells in that they exhibit a larger and rounder nucleus, a lighter cytoplasm, and few to many peroxisomes. The ductule transitional cells are most likely a later differentiation stage of a subpopulation of bile ductule epithelial cells. Undifferentiated basal cells are frequently seen in close association with transitional cells and with bile ductule epithelial cells, indicating a possible cell lineage relationship. We do not know whether the transitional cells derive from the undifferentiated ductule basal cells or from bile ductule epithelial cells. Several investigators have reported the differentiation of transitional epithelial cells from a proliferative population of bile ductule cells, termed oval cells in carcinogenic and non-carcinogenic protocols (Grisham and Porta 1964; Evarts et al. 1987; Marceau et al. 1989; Fausto 1990; Hixson et al. 1992; Sell 1993; Factor et al. 1994; Sarraf et al. 1994; Sirca et al. 1994). However, these studies were performed before it was known that there was heterogeneity in the cell types within the bile ductules and in the surrounding connective tissue stroma. We have not determined the fate of the ductule transitional cells containing catalase-positive peroxisomes. Ultrastructural observations indicate that ductule transitional cells migrate out of the bile ductule into the connective tissue stroma. We have also observed nonpolarized cells in the connective tissue stroma that exhibit a hepatocyte phenotype. Some of these cells are located between the bile ductules and the parenchymal hepatocytes. These observations suggest that these cells represent intermediate stages in a cell lineage pathway that begins in the proliferating bile ductules. We would like to determine the fate of the ductule transitional cells and the connective tissue stroma nonpolarized hepatocyte-like cells. We would like to know whether they are involved in the repopulation of the parenchymal hepatocytes or whether they contribute to the development of the preneoplastic nodule. We have not determined whether the ductule transitional cells have the functional capacity of bile ductule cells or hepatocytes. Studies are in progress to determine whether hepatocyte-specific receptors are expressed in the transitional cells.
Integration of Bile Ductule Cells Within the Hepatic Parenchyma
Our observations that subpopulations of cytokeratin-19-positive and GST-positive bile ductule cells integrate within the hepatic parenchyma by the formation of bile canaliculi and desmosome structures suggest that there are other bile ductule cells that can be categorized as transitional cells. The integrated bile ductule transitional cells have plasma membrane properties and cytoplasmic organelle phenotype different from those of the transitional cells described within the ductule and in the connective tissue stroma. One population of integrated transitional bile ductule cells has a dual polarity i.e., they are simultaneously attached to parenchymal hepatocytes and to other bile ductule cells organized around the ductule lumen, and they exhibit microvilli on two regions of their plasma membrane, one defined by the lumen held in common with other bile ductule cells and the other part of a canalicular structure with parenchymal hepatocytes. Another population of integrated transitional bile ductule cells has become fully integrated among the parenchymal hepatocytes within the hepatic cord and is not associated with bile ductule cells organized around a ductule lumen. Each subpopulation of integrated transitional bile ductule cells develops bile canalicular structures with hepatocytes that have similar properties to the bile canaliculi present between contiguous hepatocytes, i.e., the bile canaliculi are ATPase- and actin-positive and desmosomal structures are evident. However, in the dual polarized transitional bile ductule cells, ATPase is not detected in the ductule lumen. We have also observed that, at the site of attachment of the dual polarized transitional bile ductule cells with hepatocytes, there is a loss of the basal lamina. However, the basal lamina is evident surrounding the other bile ductule epithelial cells organized around the lumen. The factors that determine which bile ductule cells become attached to hepatocytes are not known. Studies are in progress to determine whether the integrated cells develop other hepatocyte phenotypes in addition to bile canaliculi. Confocal and electron microscopy in combination with enzyme cytochemistry and immunocytochemistry has been critical in determining the dual phenotype of the transitional bile ductule cells and their incorporation within the hepatic lobule. The integrated transitional bile ductule cells could easily have been interpreted as small differentiated hepatocytes. Previous studies in the DEN-AAF-PH rat model have reported GST-Yp-positive cells within the hepatic parenchyma in the same early stages of the protocol we have studied and have suggested that these cells were small, carcinogen-altered hepatocytes that will develop into preneoplastic nodules (Cameron 1989; Farber 1992). Studies are in progress to determine whether the ATPase- and GST-positive transitional cells we observed within the hepatic cord are similar to the GST-positive cells observed by other investigators. If indeed they are the same cells, this suggests that the preneoplastic nodule cells are in part derived from a subpopulation of bile ductule epithelial cells rather than from carcinogen-altered parenchymal hepatocytes. The integrated transitional cells have the capacity to replicate, thereby increasing their numbers within the hepatic lobule. These observations suggest that multiple cell lineage and sublineage pathways may be involved in liver restoration after partial hepatectomy and/or nodular development and that the pathways include a direct integration of subpopulations of transitional bile ductule cells and their replication within the hepatic cord. Nontransformed and neoplastically transformed RLE cells were reported to integrate into hepatic cords after intrahepatic transplantation (Coleman et al. 1993,1997). The integrated transitional bile ductule cells we describe and the integrated RLE cells may be derived from similar hepatic precursor cells located within the bile ductule system.
In summary, the most significant observation we report is the initiation of the differentiation of an undifferentiated ductule basal cell compartment that has a nonpolarized and nonepithelial phenotype within proliferating bile ductules in a liver carcinogenic rat model. The development of polarity, microvilli, and desmosomes, phenotypic differentiation markers similar to those of hepatic epithelial cells, on the plasma membrane surface of undifferentiated ductule basal cells provides convincing evidence that the ductule basal cells have entered a cell lineage pathway leading towards epithelial differentiation. Of interest is that the markers we have found associated with ductule basal cells are intrinsic markers of the plasma membrane of bile ductule epithelial cells, hepatocytes, and nodular cells. We propose that the nonpolarized, undifferentiated ductule basal cells that have a blast-like phenotype, replicate, and differentiate constitute a stem cell-like or progenitor cell compartment that gives rise to bile ductule cells, and possibly to hepatocytes and preneoplastic nodules, via multiple lineage and sublineage pathways. The other cell types we have observed, i.e., the polarized transitional ductule cells within the bile ductules, the nonpolarized transitional cells within the connective tissue stroma, and the polarized transitional bile ductule cells within the hepatic cords, most likely represent different stages of the multiple cell lineage pathways that are involved in the regeneration of the liver and/or the development of hepatomas. Studies are in progress to sort out the pathways and to determine the cellular origin of the preneoplastic nodules, as well as to determine whether there are multiple cell compartments with stem-cell properties.
Footnotes
Acknowledgements
Supported by NIH grant CA06576.
We thank Michael Cammer, Analytical Imaging Facility, Albert Einstein College of Medicine, for expert assistance with the confocal microscopic studies.
