Abstract
Immunohistochemistry (IHC) has provided major insights about the classification of brain tumors by identifying cellular markers of phenotype and about tumor growth potential with nuclear markers of proliferation. In situ hybridization (ISH) research shows promise for diagnostic applications in tumor classification. The avidin-biotin conjugate IHC procedure is highlighted for diagnostic use on routinely processed clinical specimens. The immunophenotypes of brain tumors are tabulated in reference to their common IHC markers. Tumors that have been correctly classified by their IHC phenotypes include the giant-cell glioblastoma, primary brain lymphoma, and central neurocytoma. Phenotypes that may be more definitively detected by ISH, such as pituitary hormone, immunoglobulin light chain, and collagen messages are described. IHC of nuclear proliferation markers correlates with grade of malignancy, predicts tumor growth potential, and is prognostic for patient survival. The incorporation of bromodeoxyuridine, the expression of proliferating cell nuclear antigen, and the expression of Ki-67 antigen detected by MIB-1 antibody are compared in regard to their cell cycle activity and labeling index determinations. Fluorescence in situ hybridization (FISH) of brain tumor interphase nuclei and chromosomes is described. Abnormal FISH signals of specific chromosomes are associated with different types of brain tumors, with different grades of malignancy, and with mesenchymal drift of glioma cells in culture.
B
The neuropathological interpretation of brain tumors begins with their classification by their histological and cytological features that resemble elements of the nervous system (McKeever et al. 1997a). Thus, gliomas are tumors with glial features, neuronal tumors resemble neurons, and neuroembryonal tumors resemble parts of the developing brain. These are major different categories of primary brain tumors. Immunochemistry (IHC) distinguishes these and other categories by their molecular phenotypes (Cáccamo and Rubinstein 1997; McKeever 1997). Moreover, IHC has startled observers, revealing unsuspected molecular phenotypes that define major changes in neurodiagnostic categories of certain tumors. These are highlighted in this review.
Determination of grade of malignancy concludes the neuropathological interpretation of a brain tumor (Kleihues et al. 1993). A critical aspect of this is assessment of the proliferative capacity of the tumor. The standard histological method of estimating proliferative capacity of a neoplasm has been its mitotic index. However, many brain tumors contain so few mitoses that an accurate mitotic index is impossible in biopsy specimens. This dilemma is ameliorated by nuclear IHC markers that provide a more complete assessment of proliferative capacity. These nuclear markers of proliferation include the incorporation of bromodeoxyuridine (BUDR), the expression of proliferating cell nuclear antigen (PCNA), and the expression of Ki-67 antigen detected by MIB-1 antibody.
In the terminology of “translational research,” i.e., the clinical utilization of basic science, IHC has been translated to regular clinical use for brain tumor diagnosis. Concurrently, new IHC phenotypic markers continue to be found, and the best of these will translate into diagnostic use. Nuclear IHC markers of proliferation are presently being translated into the diagnostic arena. Their ultimate diagnostic utility will depend in large part on the extent of their power to predict patient survival, which appears to be substantial for astrocytomas (McKeever et al. 1997b). On the other hand, in situ hybridization (ISH) is largely a research method at this time, a method with substantial promise for providing new insights about brain tumors.
Immunohistochemistry of Phenotypic Markers
What cell type does a brain tumor resemble? Phenotypic analyses lay the foundation for classification of brain tumors into specific diagnostic categories (Russell and Rubinstein 1989; Burger and Scheithauer 1994). Classic methods of phenotypic analysis are based on structural features that correlate with a known type of normal cell (McKeever and Blaivas 1994). For example, cells like stars (astro-) compose a tumor (-oma) to form an astrocytoma. Such analyses commonly begin with tissue sections stained with a general stain for nuclei and cytoplasm, such as hematoxylin and eosin, supplemented by conventional histochemical stains for collagen and reticulin, as needed (McKeever and Balentine 1987). This approach has worked for many brain tumors. However, major miscalculations have also occurred.
Figure 1 is an H&E-stained section of a brain tumor with remarkably large malignant cells. Glycosylated collagen between these malignant cells (Figure 2) led people to believe it to be a sarcoma. For decades it was considered to be a “monstrocellular sarcoma” (Zülch 1979; Kleihues et al. 1993). However, the IHC stain for glial fibrillary acidic protein (GFAP) clarified its true nature (Figure 3). Its large malignant cells are GFAP-positive, indicating the diagnosis of giant-cell glioblastoma. GFAP is the most specific marker for gliomas, especially astrocytomas and ependymomas, available today.
Another tumor originally mistaken for a reticulum cell sarcoma or microglioma is the primary brain lymphoma (Rubinstein 1972; Zülch 1979; Figure 4). IHC for immunoglobulins revealed their true phenotype (Garvin et al. 1976). Today there are specific B-lymphocyte cell surface IHC markers, such as L26 (CD20), that are more reliable for diagnosis on paraffin sections than IHC with anti-immunoglobulin antibodies (Figure 5; Table 1). The expression of L26 and the lack of expression of T-cell markers indicate that the tumor is a B-cell lymphoma. In addition, ISH for immunoglobulin mRNA may provide a more specific alternative to anti-immunoglobulin antibodies on paraffin sections (see below; Weiss et al. 1990).
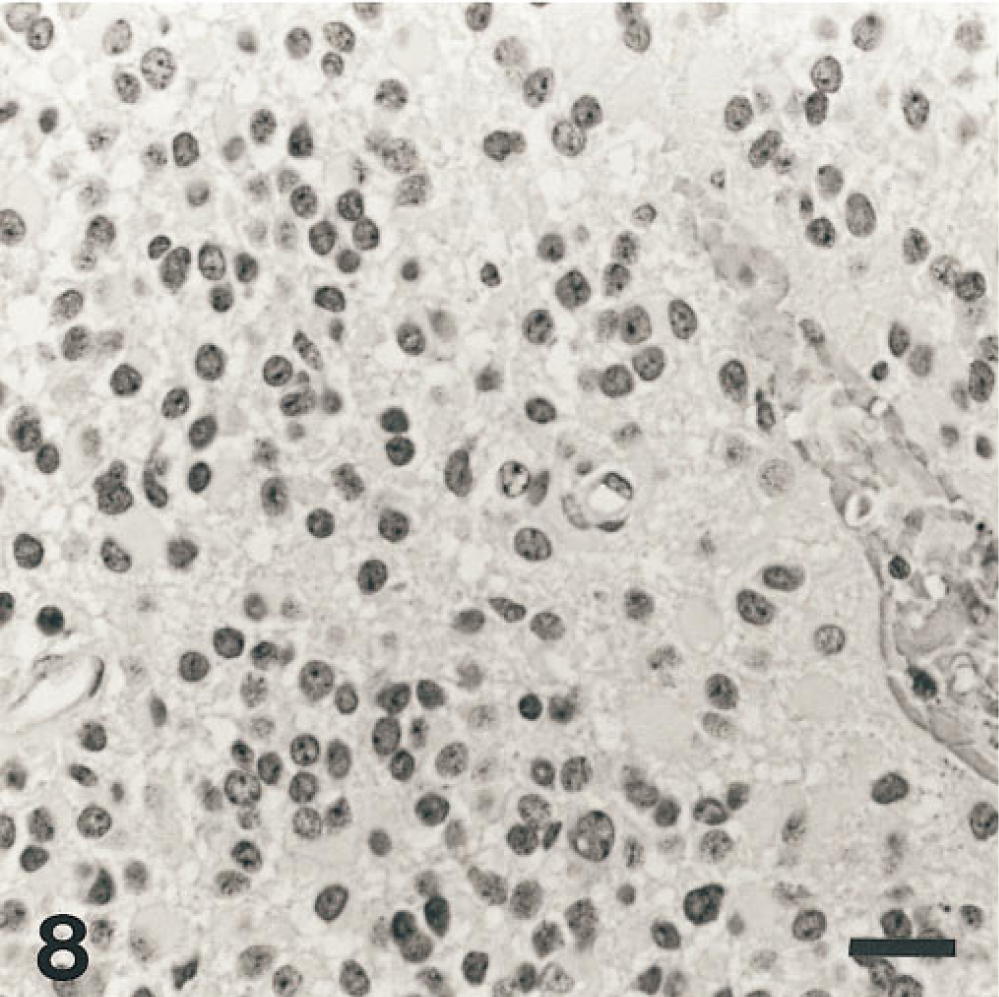
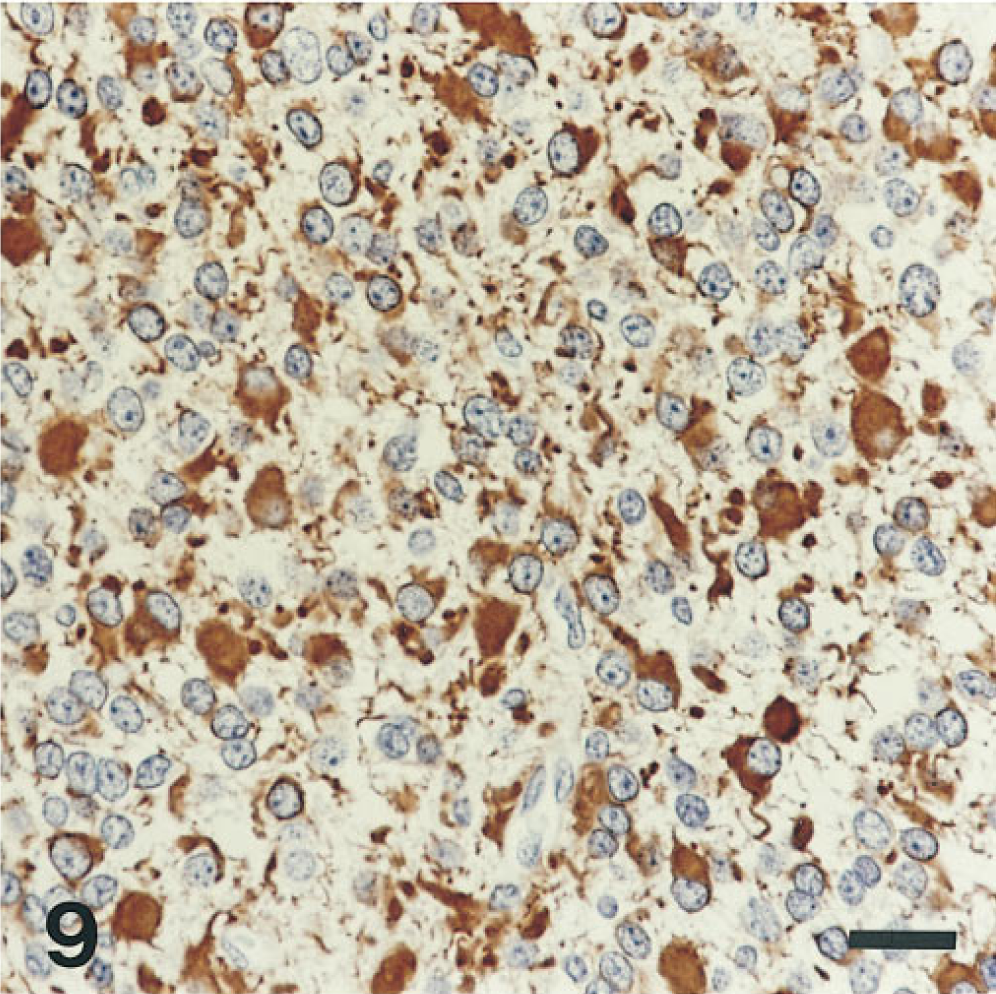
Until a few years ago, most central neurocytomas were interpreted to be gliomas (McKeever and Blaivas 1994). As a consequence their benign nature was not recognized and some were seriously overtreated. Today these neuronal tumors (Figures 6 and Figures 7; Table 1) can be recognized by IHC for molecules expressed by neurons such as synaptophysin, which distinguish them from gliomas (Figures 8 and Figures 9; Table 1).
Table 1 lists alphabetically the IHC stains commonly used for evaluating tumors at the University of Michigan at this time. The majority use monoclonal antibodies, but GFAP and S-100 are detected with polyclonal antibodies. Polyclonal antibodies tend to be quite sensitive, useful in detecting low levels of antigens, but are not as specific as monoclonal antibodies.
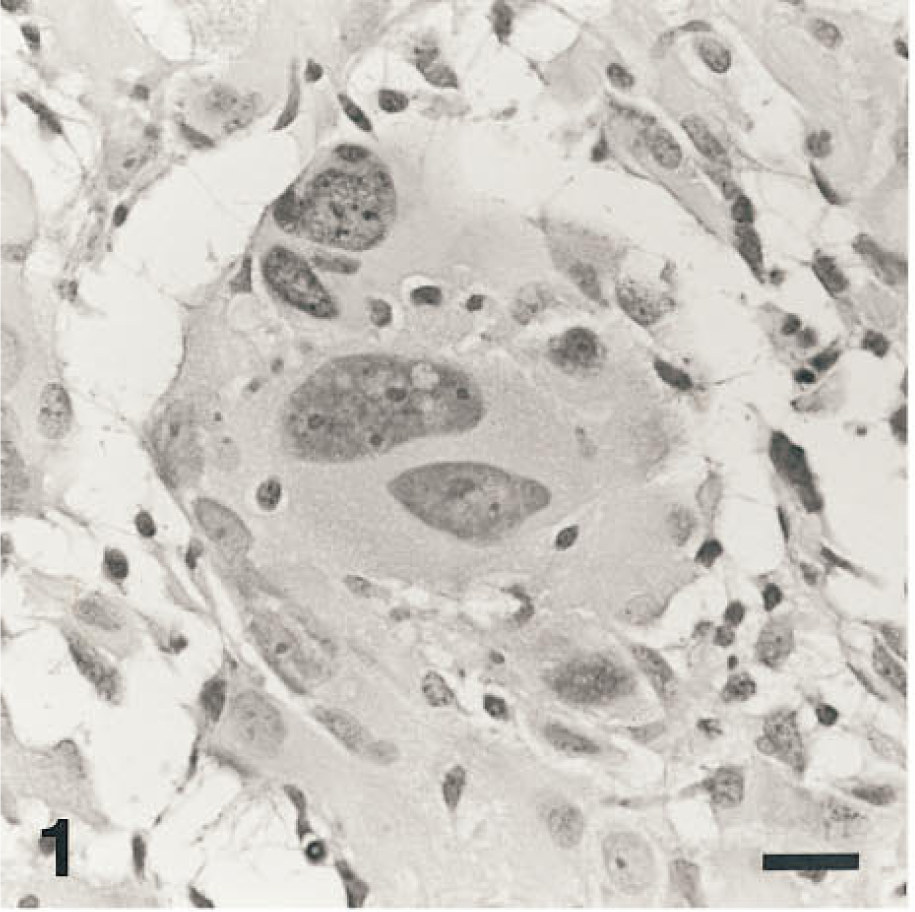
Gigantic cells contain multiple nuclei with pleomorphic nucleoli. Hematoxylin and eosin stain.
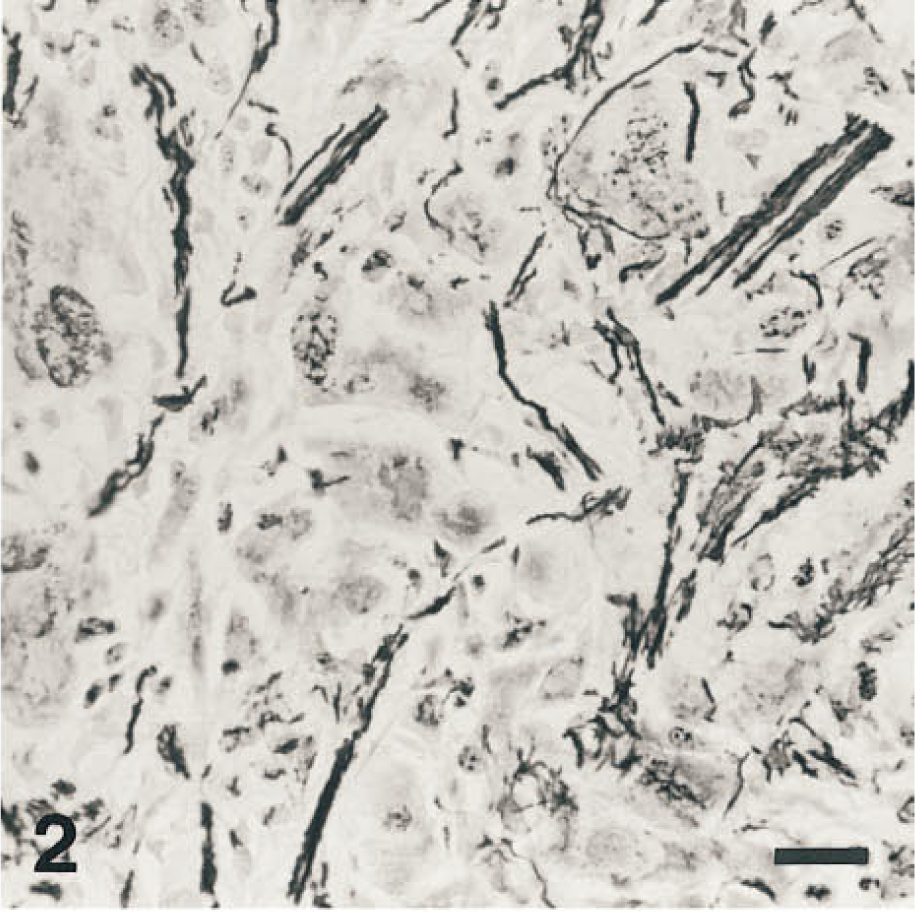
Reticulin stain darkly colors glycosylated collagen with silver between the gigantic cells and suggests that it is a sarcoma.
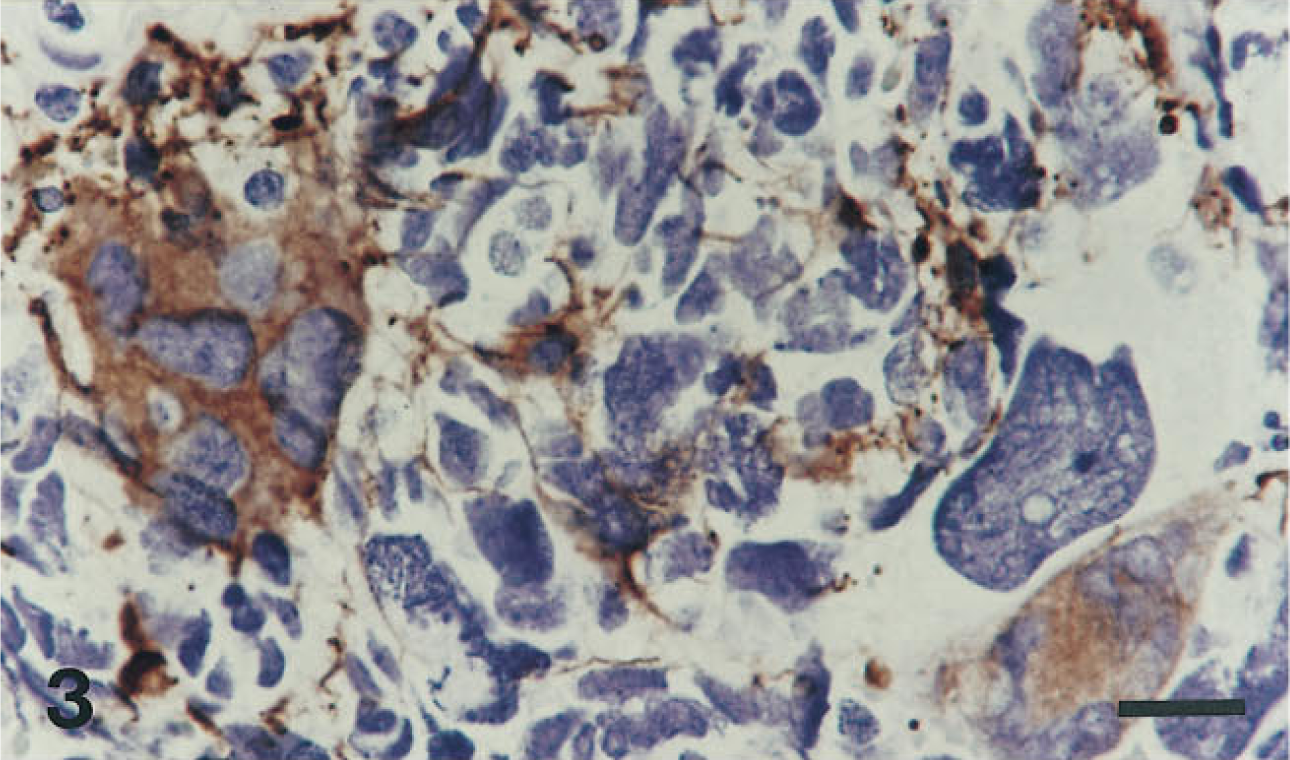
Immunohistochemistry stains cytoplasmic glial fibrillary acidic protein (GFAP) brown, showing that this “monstrocellular sarcoma” is actually a glioblastoma. Hematoxylin counterstain.
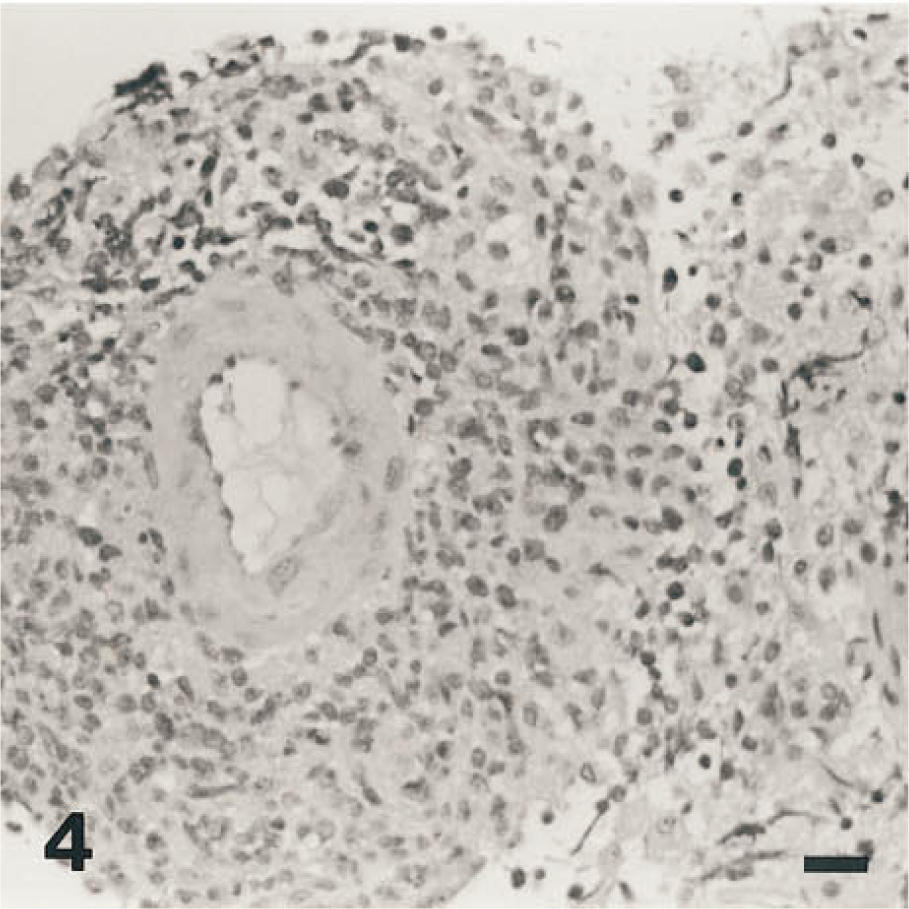
The malignant cells grow in and around vessels. Hematoxylin and eosin stain.
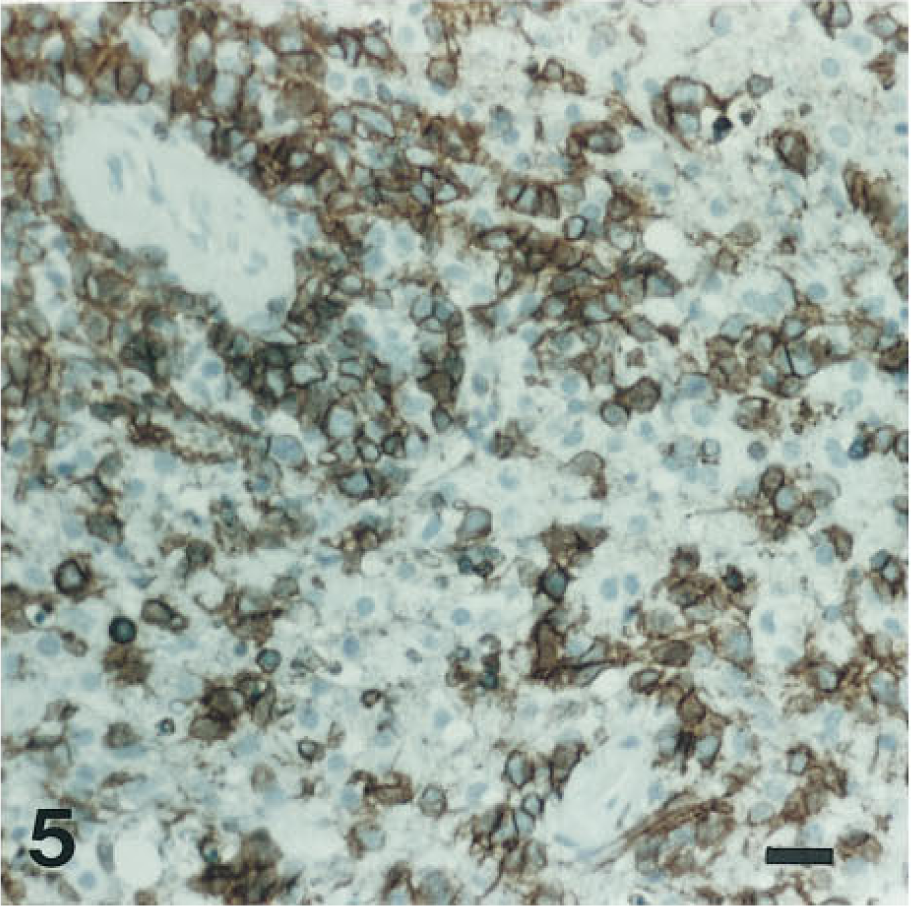
Immunohistochemistry reveals that the neoplastic cells of this “reticulum cell sarcoma” express the B-lymphocyte cell surface marker L26. Hematoxylin counterstain.
The source of many brain tumor antigens to which IHC antibodies were raised was a differentiated cell of specific lineage. Although the neoplastic counterparts of these cell lineages often express the expected antigens, this oncological fidelity is not universal. Neoplasms of a high grade of malignancy are prone to express unusual antigens. IHC of the neuroembryonal tumors has produced startling examples of this (Table 1). Primitive neuroectodermal tumors, including cerebellar medulloblastomas common in childhood, may express intermediate filaments commonly found in carcinoma, muscle tumors, gliomas, and neuronal neoplasms (Gould et al. 1990). Certain glioblastomas and gliosarcomas produce focal cellular subpopulations positive for cytokeratin (Mörk et al. 1988). These examples underscore the remarkable diversity of differentiation that has been revealed by IHC on highly malignant brain tumors.
Selected useful immunohistochemical stains for phenotypic analysis of central nervous system (CNS) tumors a
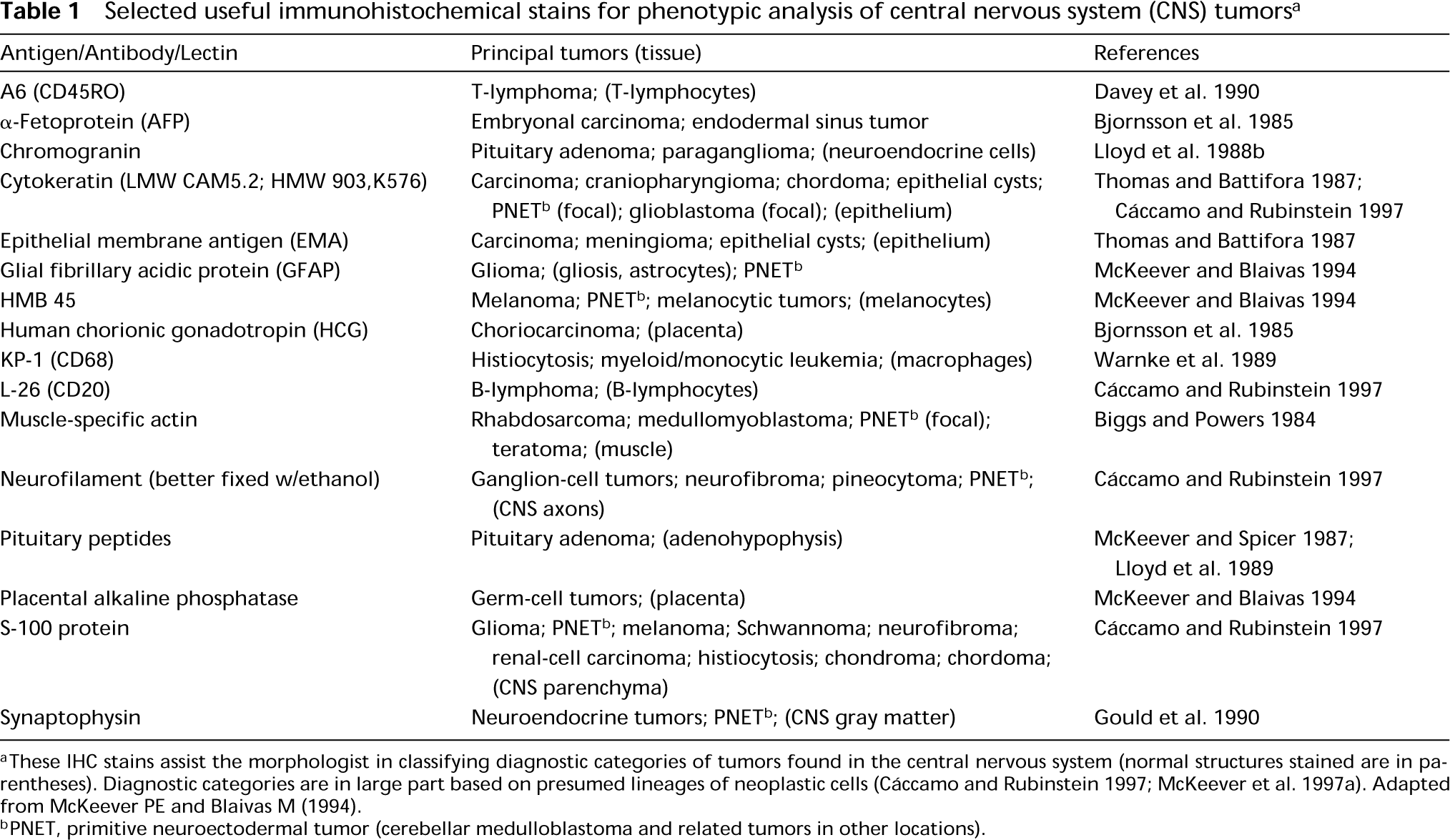
aThese IHC stains assist the morphologist in classifying diagnostic categories of tumors found in the central nervous system (normal structures stained are in parentheses). Diagnostic categories are in large part based on presumed lineages of neoplastic cells (Cáccamo and Rubinstein 1997; McKeever et al. 1997a). Adapted from McKeever PE and Blaivas M (1994).
bPNET, primitive neuroectodermal tumor (cerebellar medulloblastoma and related tumors in other locations).
Immunohistochemistry of Nuclear Markers
Brain tumors are given a histopathological grade of malignancy (relying mainly on features in H&E-stained slides) to estimate the patient's prognosis. The histopathologically defined grades of gliomas include noninvasive juvenile astrocytomas as the lowest grade and invasive, mitotically active glioblastoma as the highest (Kleihues et al. 1993). In essence, the grade of malignancy is an attempt to estimate the proliferative and invasive capacity of the tumor. Its invasive capacity is judged simply by looking at the margin of the tumor with brain (McKeever et al. 1997a,b). The standard histopathological method of estimating its proliferative capacity has been its mitotic index. However, brain tumors of low and intermediate grade contain so few mitoses that an accurate mitotic index is impossible in biopsy specimens. Even in mitotically active tumors, the mitotic index is not necessarily a reliable indicator of the entire pool of proliferating cells because of individual variations in length of the various cell cycle phases (Germano et al. 1989). Nuclear IHC markers of proliferation provide alternative assessments of proliferation capacity. These markers include the incorporation of bromodeoxyuridine (BUDR), the expression of proliferating cell nuclear antigen (PCNA), and the expression of Ki-67 detected with MIB-1 antibody, which identify different proliferative phases of the cell cycle.
New methods of assessment of cell cycle parameters with proliferation markers are ways of quantitating a larger and more representative portion of proliferating cells than is possible by counting mitoses (Figures 10 and Figures 11). These methods are compared below.
BUDR is a halopyrimidine analogue of thymidine that is incorporated into DNA during its synthesis in the S-phase of the cell cycle. Cells that have incorporated BUDR are detected with a specific monoclonal antibody after ethanol fixation and heat and/or acid treatment of tissue sections to expose the BUDR epitopes (Shi et al. 1997). The fraction of tumor cells that have incorporated BUDR during the interval of drug exposure are counted and expressed as a proliferation index or labeling index (LI). The LI of BUDR or any of the proliferation markers is the number of antigen-positive nuclei divided by the total number of nuclei in sampled microscopic regions of the tumor. Regions are sampled either randomly or by selecting regions that visually show the most labeling (Mukhopadhyay et al. 1990; Sallinen et al. 1994). This method has now virtually replaced radiolabeled thymidine for studies on patients.
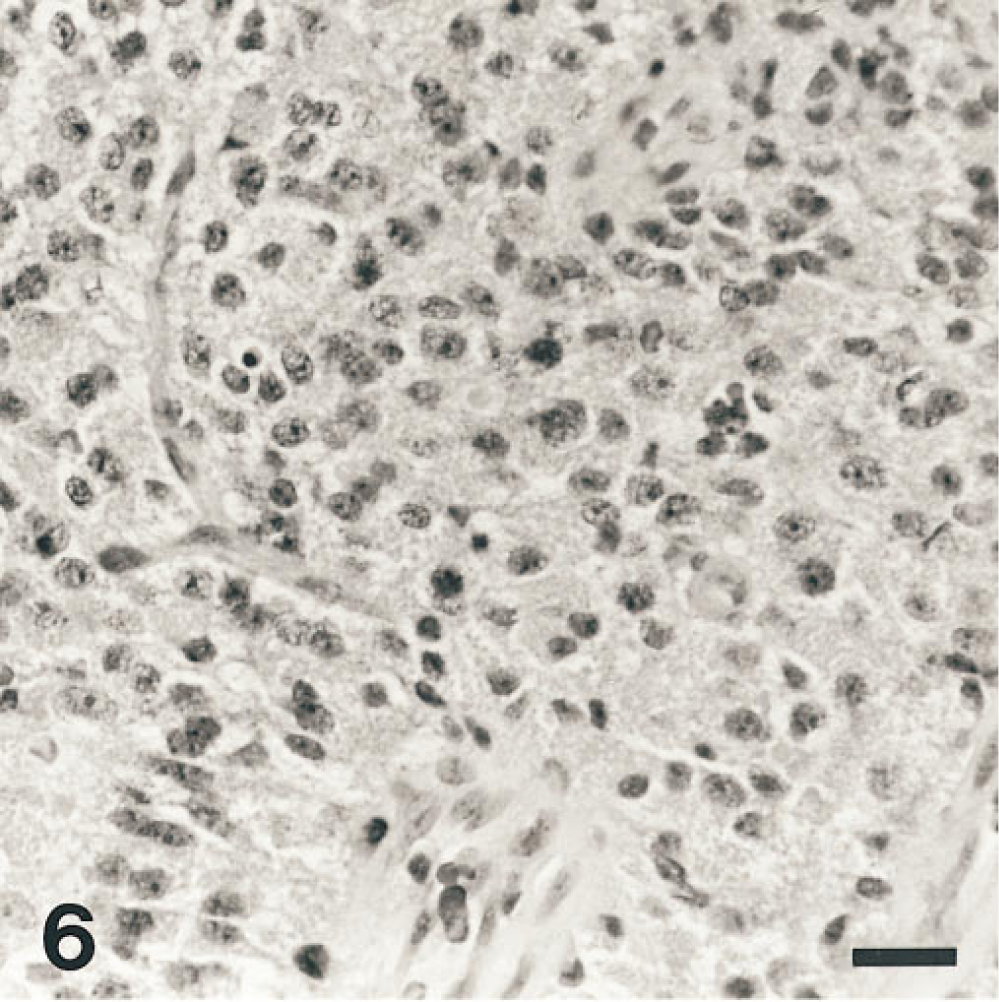
Epithelioid cells have round nuclei and perinuclear clear cytoplasmic zones. Hematoxylin and eosin stain.
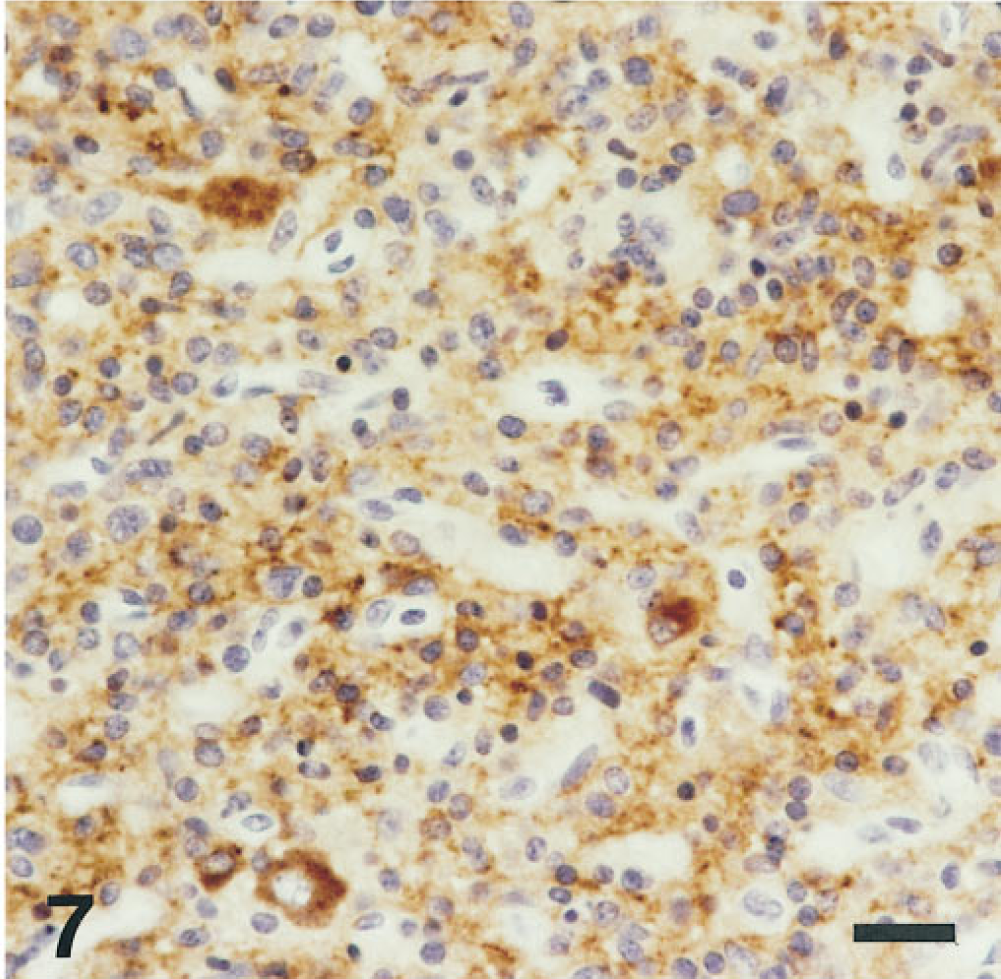
Tumor shown in Figure 6 expresses synaptophysin. This central neurocytoma was negative for GFAP. Hematoxylin counterstain.
Epithelioid cells have round nuclei and perinuclear clear cytoplasmic zones. Hematoxylin and eosin stain.
Tumor shown in Figure 8 contains a mixture of GFAP-positive and -negative cells. It was completely negative for synaptophysin. Oligodendroglioma hematoxylin counterstain.
Malignancy is associated with high BUDR labeling indices. In 38 patients with various brain tumors resected after a 1-hr pulse of in situ BUDR, high histopathological grades of malignancy were associated with a high BUDR LI (Hoshino et al. 1986). The highest average LIs were found among the most malignant tumors, including carcinomas, glioblastomas, and primitive neuroectodermal tumors. Subsequent observations confirm these seminal findings (McKeever et al. 1997b). Although this correlation is interesting, it leaves unanswered the question of whether BUDR LI can provide more information than histopathology, rather than simply paralleling the prognostic information provided by histopathology.
The answer to this question comes from studying tumors of the same histopathological grade that look similar to the pathologist but nevertheless are associated with variable patient survival. Applied to histopathologically indistinguishable low-grade astrocytomas, BUDR LI distinguished two groups with different prognoses. Patients with astrocytomas having an LI of less than 1% had better survival than the group with LIs greater than 1% (Hoshino et al. 1988). These important insights about proliferative capacity predicting patient survival have not seen widespread clinical application because of problems inherent in giving patients BUDR before surgery. In vitro labeling of fresh tumor tissue with BUDR obviates patient exposure, but I have found it difficult to achieve reliable results with in vitro labeling.
The proliferation antigens represented by PCNA, Ki-67 antibody, and MIB-1 antibody are attractive alternatives to BUDR. Unlike BUDR, they occur naturally in tissue (Garcia et al. 1989; McCormick et al. 1993) and do not require injecting patients or incubating living tissue with a drug that insinuates within DNA. This facilitates their use in hospital laboratories.
PCNA/cyclin is an auxiliary protein to DNA polymerase. It is synthesized in late G1- and S-phases of the cell cycle (Galand and Degraef 1989). Similar to BUDR, PCNA LI increases with tumor grade (Revesz et al. 1993). However, the question of whether its prognostic value simply parallels or actually exceeds the prognostic value of H&E histopathology alone has not been answered as clearly for PCNA as for BUDR. Multivariate analysis of clinicopathological indices and PCNA LIs of 45 gliomas showed that only PCNA and histopathological diagnosis independently predicted survival (Korkolopoulou et al. 1994). Other studies have shown PCNA predictive to a limited extent, but not independent of other factors by multivariate analysis (Ang et al. 1994; McKeever et al. 1997b).
The prognostic value of PCNA among brain tumors has yet to be agreed upon. One impediment is variation in LI among laboratories and even among IHC batches in the same laboratory (Khoshyomn et al. 1993; Sallinen et al. 1994; Hoyt et al. 1995; McKeever et al. 1997b). Many intermediate staining intensities confound the counting of PCNA-reactive nuclei (McKeever et al. 1997b; Figure 10). Innovations that distinguish proliferation from DNA repair may improve future studies with PCNA (Coltrera and Gown 1991; Aboussekhra and Wood 1995).
The MIB-1 and Ki-67 monoclonal antibodies recognize Ki-67 epitopes on a nuclear protein present in a portion of the cells in G1-, S-, G2-, and M-phases of the cell cycle (Gerdes et al. 1983). The original Ki-67 antibody was promising. One study of 36 patients revealed that the Ki-67 LI was more significantly related to survival than the modified Ringertz histopathological grading system of astrocytic gliomas (Montine et al. 1994). The mean Ki-67 LI of low-grade astrocytoma is significantly less than that of both anaplastic astrocytoma and glioblastoma (Raghavan et al. 1990; Montine et al. 1994). However, the original Ki-67 antibody bound only labile epitopes that did not withstand routine histological processing in formalin and paraffin (Montine et al. 1994).
The advent of an MIB-1 monoclonal antibody opens a new era in the application of proliferation markers because the MIB-1 antibody detects Ki-67 epitopes in paraffin sections of formalin-fixed tissue (McCormick et al. 1993; Figure 11). LIs from MIB-1 antibody are comparable to those of BUDR, but MIB-1 does not require drug exposure (McKeever et al. 1997b). Positive MIB-1 nuclei are more easily detected and counted than PCNA-positive nuclei (McKeever et al. 1997b). Evaluations of the prognostic value of MIB-1 labeling indices are scarce because the antibody is new, but results are promising (Karamitopoulou et al. 1994; Prayson et al. 1995; McKeever et al. 1997b). In one multivariate analysis, MIB LI was the only independent predictor of survival in the model (Sallinen et al. 1994). Most importantly, among only low-grade astrocytoma patients, MIB-1 distinguishes by their low labeling indices tumors that will allow a good prognosis (Mikhail et al. 1996).
In Situ Hybridization of Phenotypic Markers
The detection of mRNA codes is theoretically attractive for the diagnosis of neoplasms with little or no expression of the suspected protein antigen or with protein markers that lack sensitive and specific antibodies for their detection. Thus far, ISH is primarily a research method, but it has provided insights into the molecular pathology of pituitary adenoma and glioma. Three different examples follow.
ISH can detect a latent phenotype in specific cases where immunohistochemical markers are only partially effective. The mRNA messages detected by ISH correspond with their encoded peptide hormones localized immunohistochemically in pituitary adenomas. More importantly, ISH detected the messages for two hormones in some adenomas that expressed only a single hormone detectable by IHC (Lloyd et al. 1989). Thus, ISH can reveal potentially bihormonal or plurihormonal adenomas in cases in which the adenoma may either secrete a second immunoreactive hormone immediately on production, translate no second hormone from its mRNA, or produce hormone with low immunoreactivity.
ISH can detect a latent phenotype where specific immunohistochemical markers are inadequate. The individual detection of κ- and λ-light chains of immunoglobulins provides particular power in the assessment of neoplastic vs inflammatory lymphocyte proliferation. Neoplastic lymphocytes express either κ- or λ-light chains, but not both as do lymphocytes in inflammation. This diagnostic discriminator of lymphoma is called light-chain restriction. Unfortunately, immunoglobulin light chains lack sensitive and specific antibodies for their detection in paraffin sections, confounding the interpretation of light-chain reaction. ISH for immunoglobulin mRNA has been proposed as a more specific alternative to these anti-immunoglobulins on paraffin sections (Weiss et al. 1990). Thus far, this is not commonly done in hospital laboratories, but it should become more attractive as immunofluorescent and immunohistochemical detection of mRNA replaces autoradiography (Stewart et al. 1996).
ISH can detect the cell of origin for extracellular protein markers. Unlike the rest of the body, normal brain contains very little collagen. Nevertheless, certain glioblastomas contain abnormal bands of collagen and spindled cells that resemble sarcoma mixed with more typical malignant glioma cells (McKeever et al. 1984). To determine whether the glioma cells or the spindled cells actually produce this collagen, ISH for Type IV collagen mRNA was employed (Lloyd et al. 1988a). Results indicated that the spindled cells produce the collagen, rather than the glioma cells (McKeever et al. 1993).
In Situ Hybridization of Nuclear Markers
Fluorescence in situ hybridization (FISH) can be used to localize specific DNA sequences either in nuclei or on chromosomes. FISH can locate repetitive sequences specific for individual chromosomes, whole chromosomes, and specific genes when a long enough gene probe is available (McKeever et al. 1996). FISH has a light microscopic counterpart localized by IHC. Thus far, FISH has mainly been used in the research rather than the diagnostic laboratory.
FISH can reveal chromosomal marker signals in cells cultured from gliomas (Figure 12). A long unanswered question in glioma cell biology has been the source of phenotypic alterations in cultured glioma cells. These alterations have been termed “mesenchy-mal drift” because the cultured glioma cells tend to lose glial and gain mesenchymal features, including collagen production (McKeever et al. 1991). FISH of chromosomal markers in these cells reveal three patterns (McKeever et al. 1995): (a) cells with aneuploid chromosomal signals of neoplastic glia that retain the phenotypic marker GFAP but in addition make Type IV collagen; (b) cells with aneuploid chromosomal signals of neoplastic cells in a nonglial pattern that have many mesenchymal phenotypic markers and lack GFAP; and (c) diploid mesenchymal cells that may be non-neoplastic cells from vascular walls.
Applied to metaphase spreads of brain tumor chromosomes, FISH provides new insights about cytogenetic observations. For example, elongation of a glioblastoma chromosome 17 by what appears to be the insertion of a single small band on GTG-banded chromosomes was shown by microdissection and FISH to be the insertion of DNA from three different chromosomes (McKeever et al. 1996).
Comparative genomic hybridization (CGH) is a different type of FISH in which the tumor DNA is a labeled probe rather than DNA fixed on a slide. It utilizes two-color FISH to compare this brain tumor DNA with an internal standard of normal DNA. Tumor DNA is labeled with one molecular tag, such as biotin. Normal DNA is labeled with another tag, such as digoxigenin. The binding of these two probes to metaphase spreads of normal human chromosomes is then compared. The result has been a plethora of observations of losses and gains of chromosomal DNA in gliomas (Schröck et al. 1994). The oncological significance of most of these findings remains to be elucidated.
The diagnostic applications of FISH and CGH have not been fully explored but will be in the next few years. The repetitive sequences that identify specific chromosomes have been identified in nonmitosing (interphase) nuclei with these ISH techniques. Astrocytic tumor progression to higher grades of malignancy is associated with increased variation in such signals for chromosomes 7 and 17 (McKeever and Wang 1993).
ISH has demonstrated that isochromosome 17q (one chromosome composed of two long arms of chromosome 17) is a constant finding in medulloblastoma (Giordana et al. 1997). If this is confirmed to be a specific marker of medulloblastoma, it could be very important in distinguishing this pleomorphic primitive neuroectodermal tumor from other tumors it resembles.
Summary
Immunohistochemistry has been embraced by diagnosticians seeking improved classification of brain tumors and has become the diagnostic standard for many tumors. IHC of nuclear proliferation markers is beginning to assume an important role in estimating prognosis, and in situ hybridization promises future insights. The conspicuous value of these procedures to the diagnosis and management of brain tumor patients encourages those who struggle to innovate and improve these markers to continue their worthy efforts.
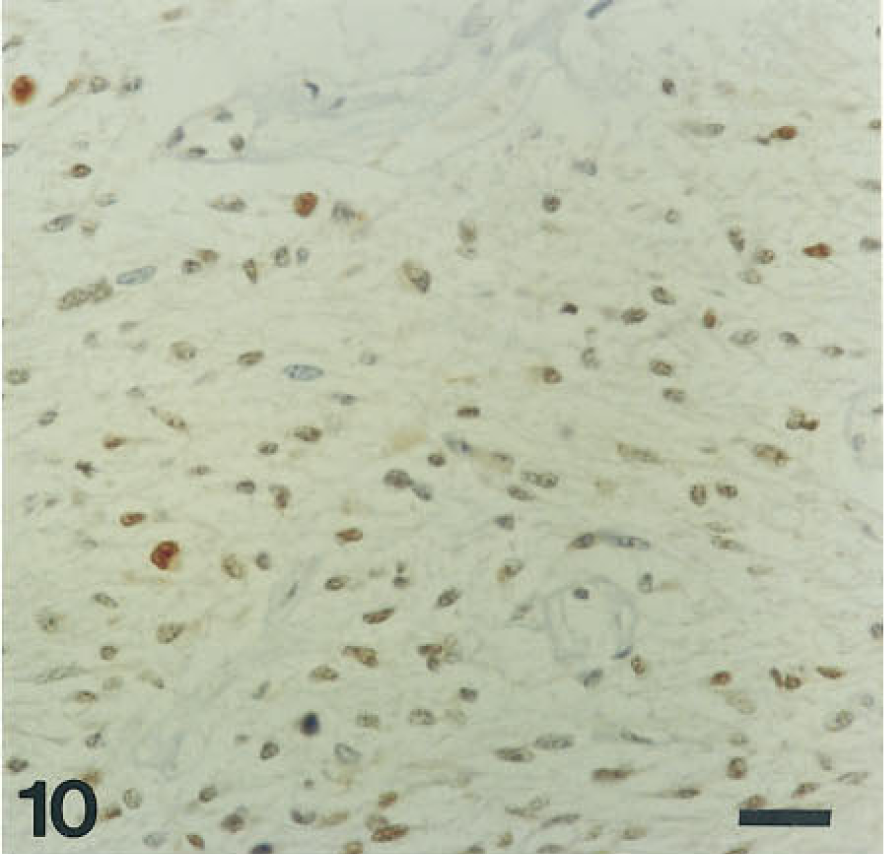
IHC stain of a glioma for proliferating cell nuclear antigen with hematoxylin counterstain. Bar = 25 μm.
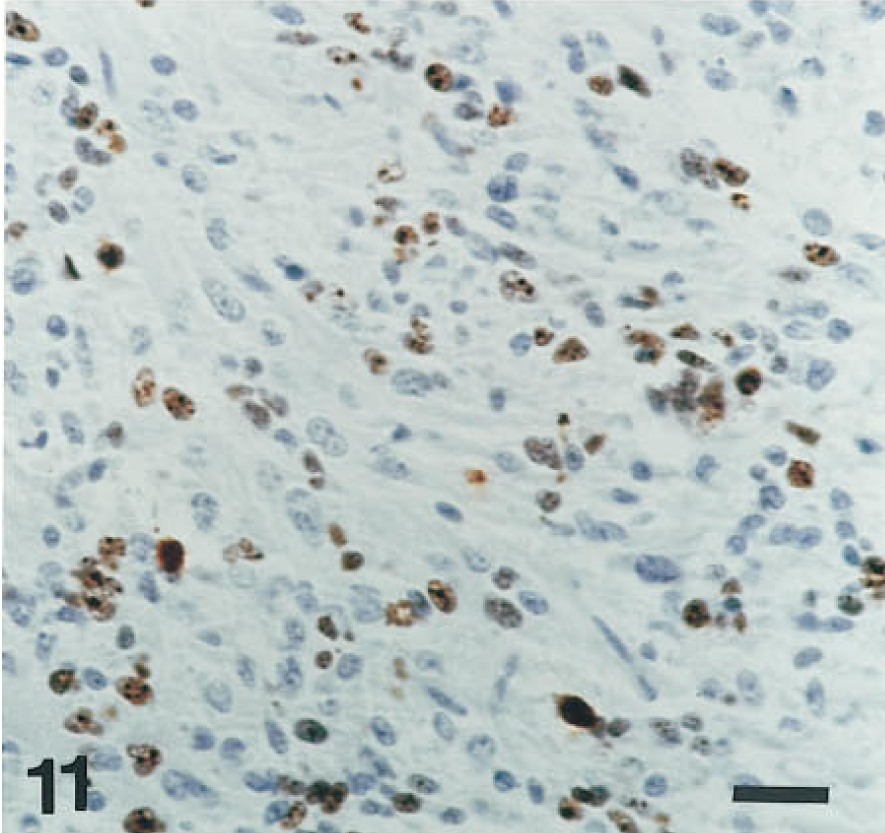
IHC stain with MIB-1 antibody reveals a high LI in this glioblastoma. Bar = 25 μm.
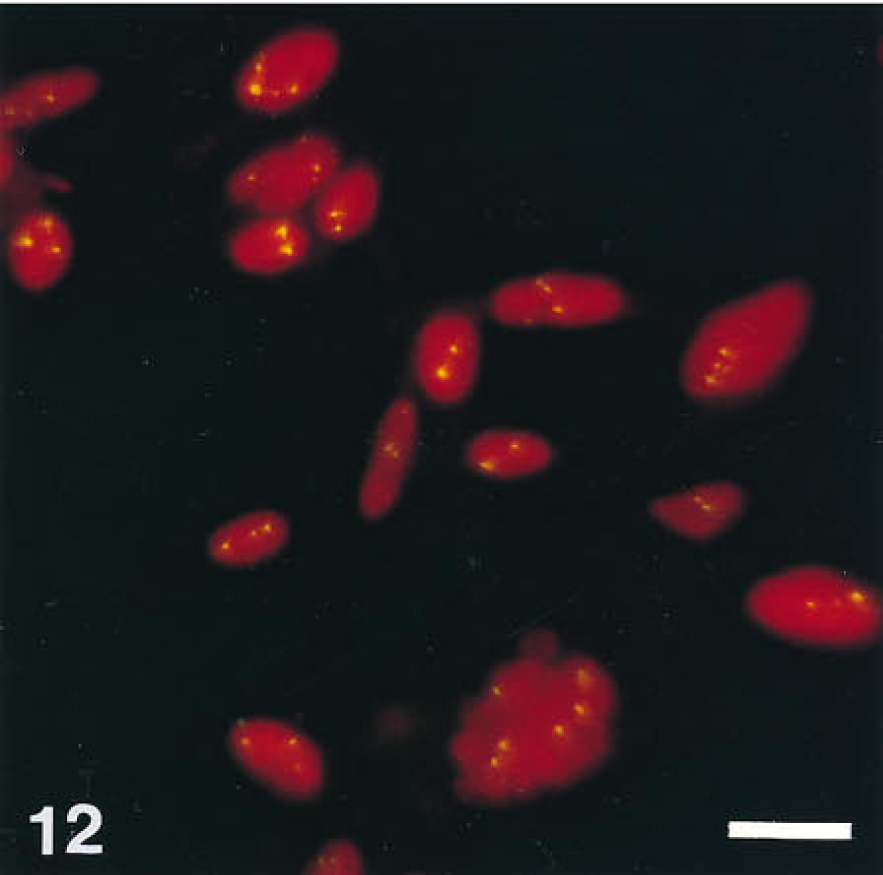
FISH for chromosome 7 reveals more than two signals per nucleus. Hyperdiploid chromosome 7 is a numerical alteration of glioma cells. Bar = 25 μm. From McKeever PE et al. (1995), with permission.
Footnotes
Acknowledgements
Supported in part by National Institutes of Health Grants (P03) CA 46592, CA 68545, and CA 47558 from the National Cancer Institute.
The skilled editorial assistance of Ms Dianna Banka is gratefully acknowledged.
