Abstract
Mutations in PEX, a phosphate-regulating gene with homology to endopeptidase on the X chromosome, were recently identified in patients with X-linked hypophosphatemia (XLH), an inherited disorder of phosphate homeostasis characterized by growth retardation and rachitic and osteomalacic bone disease. To understand the mechanism by which loss of PEX function elicits the mutant phenotype, a study of its mRNA localization and ontogenesis was undertaken. Using the reverse transcriptase-nested polymerase chain reaction (RT-nested PCR) with polyA+ RNA purified from mouse testis, a 337-
Keywords
A
Human and mouse PEX/Pex cDNAs have now been cloned and sequenced (Du et al. 1996; Beck et al. 1997; Guo and Quarles 1997; Strom et al. 1997). Amino acid sequence comparisons have demonstrated structural homologies between PEX/Pex and members of the neutral endopeptidase family as previously observed in the partial sequence of the candidate gene (The HYP Consortium 1995). The peptidases of the neutral endopeptidase family are Type II integral membrane glycoproteins with a relatively short cytoplasmic N-terminal region, a single transmembrane domain, and a long extracytoplasmic domain, which contains the active site of the enzyme. Known members of the neutral endopeptidase family include neutral endopeptidase-24.11 (NEP), endothelin-converting enzymes (ECEs), and the erythrocyte cell surface protein KELL (for a review see Turner and Tanzawa 1997). NEP [also known as neprilysin, common acute lymphoblastic leukemia antigen (CALLA), CD10, or enkephalinase] is a widely distributed peptidase involved in the degradation of several bioactive peptides, such as the enkephalins, the atrial natriuretic peptides, and the endothelins (Turner and Tanzawa 1997). The ECEs are involved in the bioactivation of Big endothelins into endothelins, but no function has yet been attributed to KELL.

Emulsion autoradiography showing ISH pattern for Pex mRNA at the anatomic level in a sagittal section from a mouse embryo at e16. A significant concentration of Pex mRNA is seen in developing bones (arrows) including calvaria (C), mandible (M), and ribs (R). Bar = 1 cm.
The mechanism by which loss of PEX function elicits the bone and renal abnormalities observed in XLH patients is not clear. There are no data suggesting the presence of PEX/Pex mRNA in the kidney (Du et al. 1996; Beck et al. 1997; Grieff et al. 1997). In contrast, PEX/Pex mRNA was detected in bones by Northern blot hybridization and in other adult and fetal tissues, such as lungs, liver, muscles, and ovaries by RT-PCR and RNase protection assays (Du et al. 1996; Beck et al. 1997).
To identify a specific role for PEX/Pex, we were interested first in its tissue and cell distribution. For this reason, using ISH we examined Pex mRNA temporal and spatial patterns of expression on sagittal sections of embryonic mice from Day 13 to 19 and in newborn mice. At this period of development, most tissues are already formed and many are functional. For example, the ossification process starts on Day 14.5 post coitum (Rugh 1991). We demonstrate by ISH that Pex mRNA is expressed in osteoblasts and odontoblasts and suggest a specific role for this putative peptidase in bone and tooth development. Northern analysis was used to examine the presence of Pex mRNA in adult mouse tissues. This analysis revealed decreased concentrations of Pex mRNA in the adult bones and non-growing teeth.
Materials and Methods
Animals
For ISH, we used unfixed, frozen tissues from fetuses of CD1 time-pregnant female mice. The fetuses were grouped according to embryonic age—10, 11, 12, 13, 14, 15, 16, 17, 18 and 19 intrauterine days of life (e10, e11, etc.) and postnatal Day 3 and 7 (p3 and p7) and were prepared as described (Marcinkiewicz et al. 1993). Whole embryos and newborn mice were rapidly removed, cooled in ice-cold PBS, embedded in Tissue-Tek OCT compound (Miles; Elkhart, IN), frozen at -30C in isopentane, and cut into 10-μm sections. The sections were mounted on 0.5% gelatin-coated slides and stored at -80C.
Sites of Pex mRNA expression in primordium of mandibular bone (
Sites of Pex mRNA expression in developing mouse calvaria on e14 (
DNA Manipulations
All DNA manipulations, including the reverse transcription from RNA, PCR, and cloning, were done according to standard protocols (Sambrook et al. 1989; Ausubel et al. 1994). DNA sequencing was performed on double-stranded DNA (Tabor and Richardson 1987).
Cloning of a Mouse Partial Pex cDNA
To obtain a mouse Pex probe for ISH and Northern blot analysis, degenerate oligonucleotides corresponding to conserved regions among members of the neutral endopeptidase family were designed using the published human PEX gene sequence (The HYP Consortium 1995) and were used in an RT-nested PCR reaction with polyA+ RNA purified from testis of CD1 mice (Charles River; Montréal, Québec, Canada). The expected 337-
Preparation of cRNA Probes
The pCRII plasmid containing the Pex cDNA fragment was linearized with Xho1 and used as a template in an in vitro transcription assay to synthesize a single-stranded anti-sense RNA probe with SP6 RNA polymerase. For control, sense RNA probe was synthesized with T7 RNA polymerase after linearization of the plasmid with Kpn1. For ISH, Pex riboprobes were labeled with both [35S]-UTP and [35S]-CTP (1250 Ci/mmol; Amersham, Arlington Heights, IL) because very low mRNA levels were reported previously (The HYP Consortium 1995; Du et al. 1996; Beck et al. 1997). For Northern blot analysis, probes were labeled with [32P]-UTP (800 Ci/mmol; Dupont/NEN, Wilmington, DE). The 18S rRNA probe (a generous gift from Dr. M. Uhler) is of bovine origin and strongly cross-hybridizes to a number of different mammalian species.
In Situ Hybridization
ISH was undertaken using RNase-free solutions, starting with frozen cryostat tissue sections that were slowly immersed in cold formaldehyde in 0.1 M phosphate buffer (pH 7.2) and maintained in this solution for 45–60 min, then washed extensively with PBS. The tissues were treated for 10 min with acetic anhydride in 0.1 M TEA. After dehydration with alcohol, the tissue sections were dried and then incubated overnight at 55C with a hybridization solution consisting of 75% formamide, 10% polyethylene glycol, 3 X SSC (1 X SSC = 0.15 M NaCl, 0.015 M Na citrate, pH 7.2), 50 mM phosphate buffer, pH 7.2, 1 X Denhardt's (made from 50 X stock solution: 1% Ficoll, 1% polyvinylpyrrolidone, and 1% bovine serum albumin in water), 0.5 mg/ml yeast tRNA, and 0.1 mg/ml sonicated denatured salmon sperm DNA. To increase the signal/noise ratio, the dithiothreitol (DTT) concentration was set at 200 mM (Miller et al. 1993). After hybridization, the sections were washed sequentially in 2 X, 1 X, 0.5 X, and 0.1 X SSC containing 10 mM DTT for 10, 15, 20 and 60 min at 20C, 20C, 50C, and 55C, respectively. After the washing step in 1 X SSC, the sections were incubated with RNase A at 200 μg/ml for 30 min at 37C to remove unbound cRNAs. Sections were then dehydrated in a series of alcohol baths and dried. Hybridization was examined on X-ray film (exposure time 5 days), followed by autoradiography using NTB-2 emulsion (Kodak; Rochester, NY) for 30 days at 4C and development in D19 solution (Kodak). The sections were stained with hematoxylineosin and viewed under dark- and brightfield illumination. Some observations were done using Nomarski's attachment.
Alkaline Phosphatase Activity
Localization of alkaline phosphatase activity (Roach and Shearer 1989) was performed with an azo dye coupling method (Alkaline Phosphatase Substrate Kit, cat. # SK-5300 Vector Blue) as recommended by the manufacturer (Vector Laboratories; Burlingame, CA).
RNA Extraction and Northern Blot Analysis
Northern blot analysis was performed using total RNA from newborn and adult calvariae and teeth, and from newborn brain, lung, and liver. For this purpose, the frozen tissues were mixed with TRIzol Reagent (Life Technologies/Gibco-BRL; Burlington, Ontario, Canada) and disrupted with a Polytron. Total RNA was extracted according to Chomczynski (1993), as recommended by the manufacturer. As estimated by spectroscopy at 260 nm, 16 μg of total RNA samples was loaded per lane in 1% agarose gel containing 20 mM HEPES, pH 7.8, 1 mM EDTA, and 6% formaldehyde. After electrophoresis, the RNA was transferred from the gel to a nylon membrane and fixed to the filters by long-wave
Results
Pex ISH at Anatomic Resolution in Mouse Embryo
The Pex expression pattern was analyzed by ISH using anti-sense riboprobes on histological sections obtained from embryonic and postnatal mice (Figures 1–5). ISH at anatomic resolution was examined after emulsion autoradiography on embryonic Day 16 (e16) (Figure 1). Pex mRNA was readily detectable in regions of calvaria, mandible, and ribs. Although not apparent in Figure 1, the vertebrae and long bones also contained Pex mRNA on e16, whereas later, around birth, Pex mRNA could also be detected in developing teeth. Overall, Pex mRNA was localized within alkaline phosphatase territory (not shown). Controls were performed with sense riboprobes, which produced nonspecific background elevated in skin (see also Figures 3d and 4d) and skeletal muscles (not shown). Other tissues, including kidney, lung, liver, and brain, were negative.
Sites of Pex mRNA expression in mandible bone on e14 (
Pex mRNA in Osteoblasts
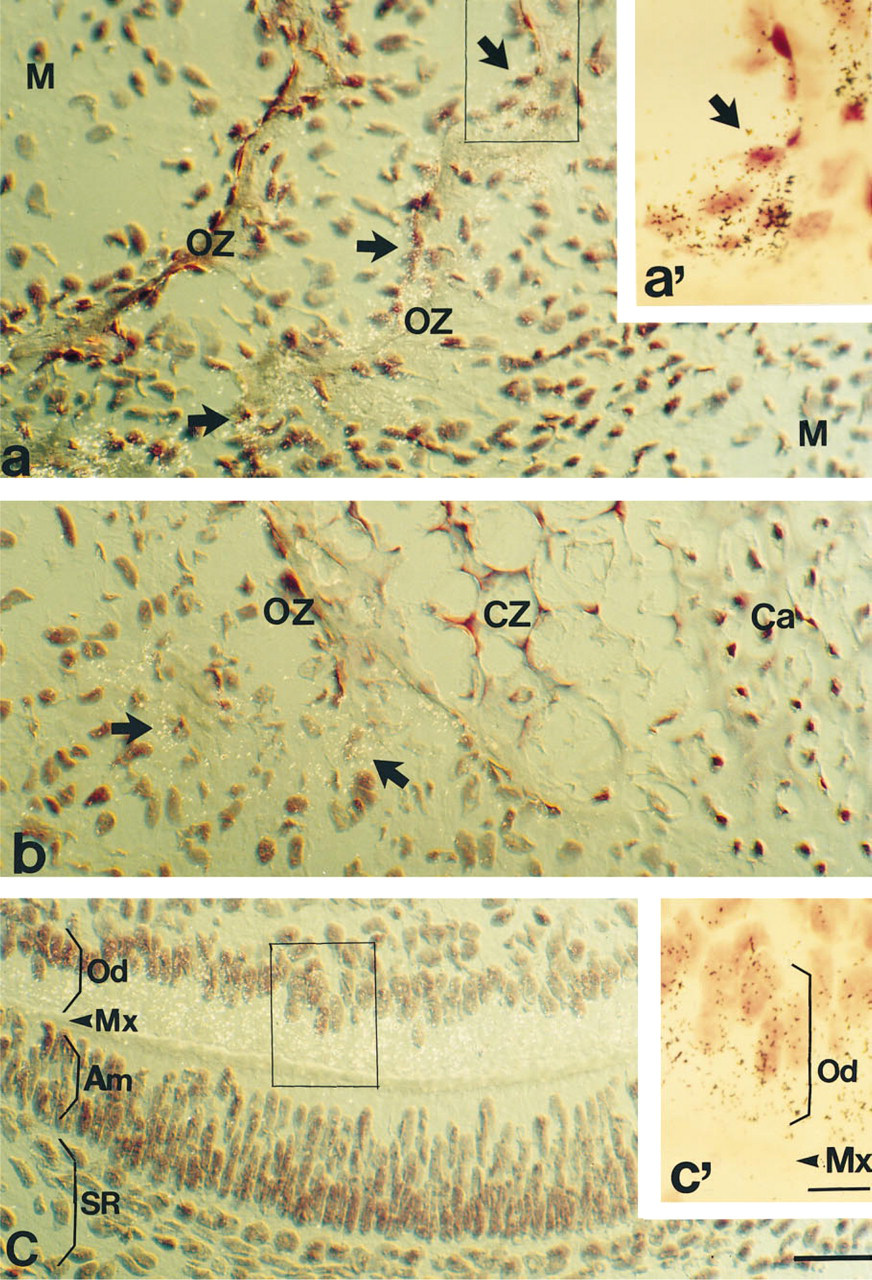
To identify cells synthesizing Pex mRNA, representative tissues were examined under higher microscopic magnification. Figures 2a and 2a', which are higher magnifications of the area shown in Figure 4c, demonstrate the presence of Pex mRNA on e16 in mandibular intramembraneous ossification centers. This topographically heterogeneous region is composed of an ossification zone (OZ) characterized by the presence of (a) a bone extracellular matrix with a dense cell population on the border zone and (b) an undifferentiated mesenchyme (M). ISH revealed the presence of Pex mRNA within cells bordering bone extracellular matrix. This topography suggests that Pex-expressing cells are osteoblasts. The majority of osteoblasts display Pex ISH labeling (Figure 2a').
Hybridization sites were also identified in vertebral endochondral ossification centers (Figure 2b). Pex mRNA was seen within an ossification zone (OZ) adjacent to an unlabeled calcified zone (CZ) and the cartilage (Ca). Bone extracellular matrix was well delineated within OZ, with a significant concentration of Pex-labeled cells on a border zone. There was no apparent hybridization labeling outside of OZ.
Pex mRNA in Odontoblasts
Within tooth, Pex mRNA was concentrated in the layer of odontoblasts (Od) (Figure 2c, which represents a higher magnification of the area shown in Figures 5b and 2c'). In contrast, the layer of ameloblasts (Am) and the stellate reticulum of the enamel organ (SR) were unlabeled. Because the dentino–enamel matrix separates the odontoblast epithelium from the ameloblast epithelial layer, the cell population expressing Pex mRNA is particularly well-defined in this tissue.
Pex mRNA Ontogeny
To determine temporal and tissue-specific patterns of Pex gene expression, the calvaria, mandible, and teeth were examined at the stages preceding and following the onset of chondrification (e11), ossification (e14.5), and odontogenesis (e14). ISH results are shown in Figures 3–5.
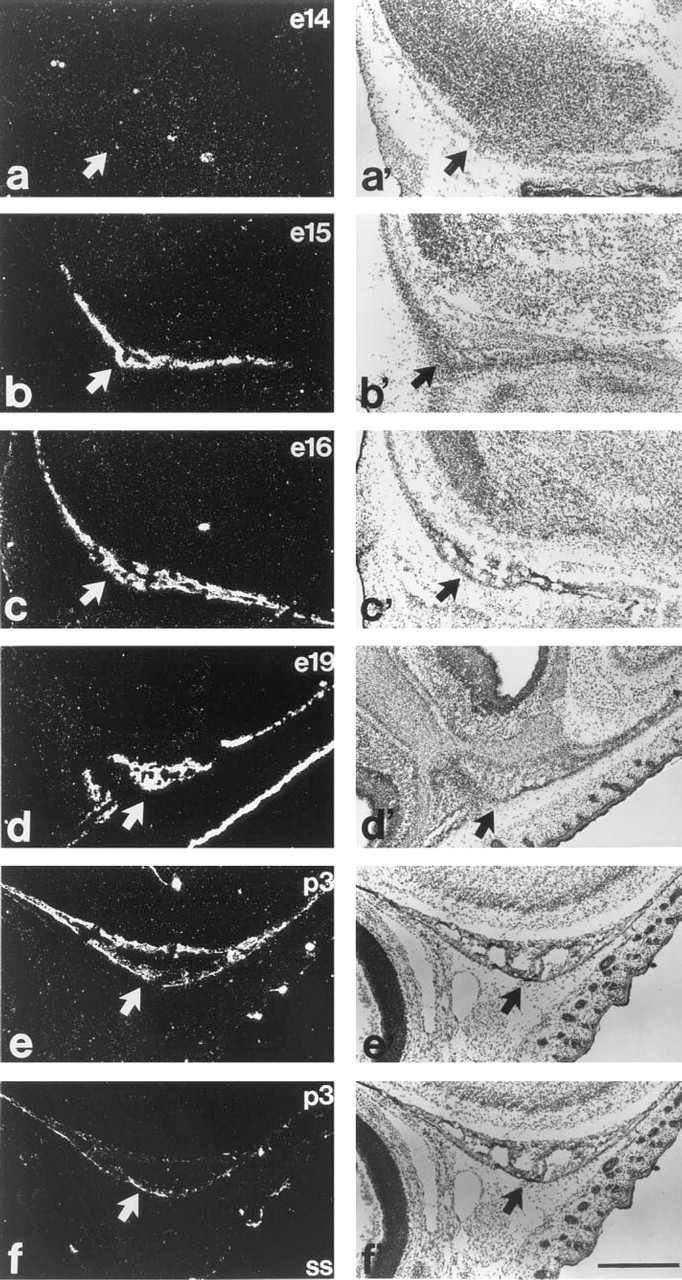
Figure 3 shows the presence of Pex mRNA in the calvaria from e15 to p3. Although rudimental calvaria is already visible on e14 (Figures 3a and 3a'), Pex mRNA is not detectable at this time but is evident thereafter. It is worth noting the presence of bone extracellular matrix within rudimentory calvaria at e15 but not e14. This matrix was seen as a deposition of acellular eosinophilic material along and within a space delineated by a front of osteoblasts (data not shown). Pex mRNA remains present in the calvaria during later gestation and early postnatal development.
Figure 4 depicts Pex mRNA distribution in the mandible from e15 to p3. Despite the absence of the bone extracellular matrix, which is not present early on, the rudiments of the mandible bone are evident by e14. Similar to calvaria, a dramatic elevation of Pex mRNA is evident on e15 (Figure 4b). Pex mRNA remains present until p3.
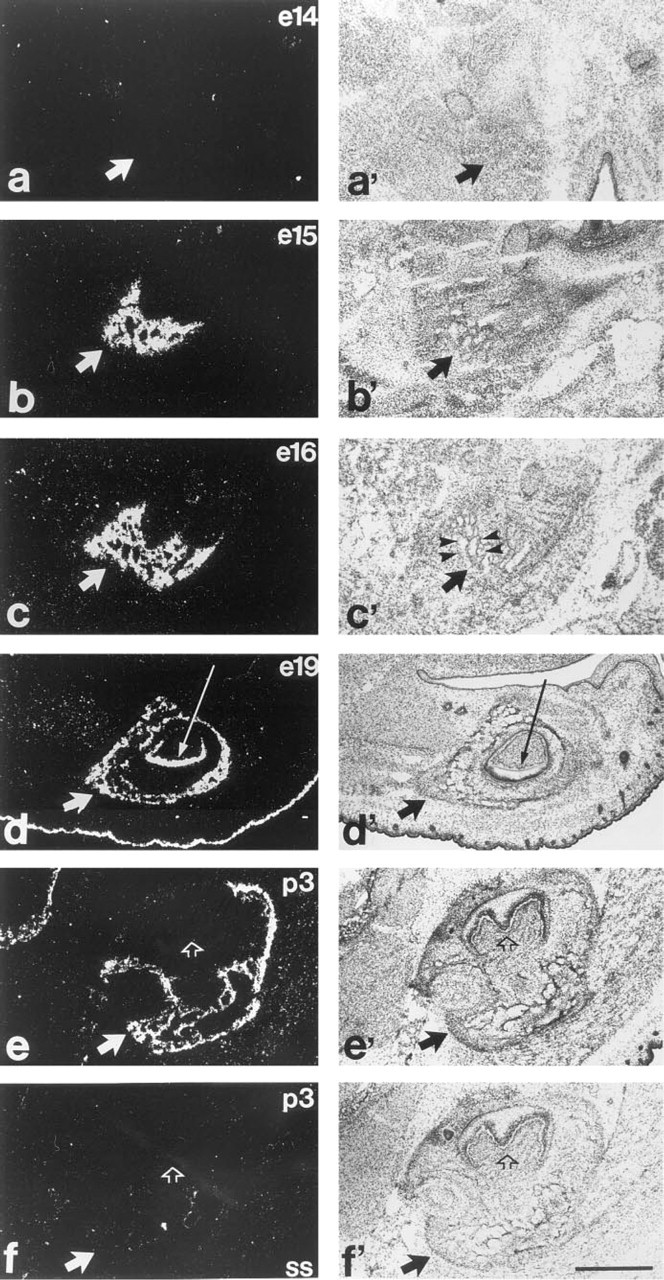
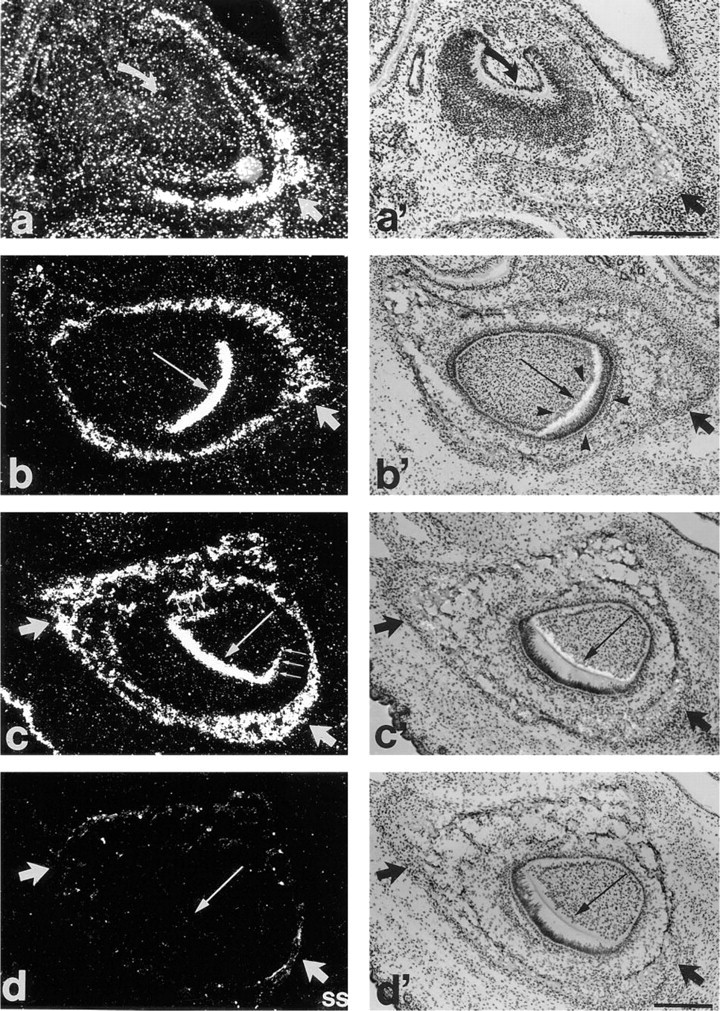
In addition to bones, Pex mRNA labeling can be observed in both incisors and molars, although the onset of expression varies with time of tooth development (Figures 4 and 5). All teeth shown in Figure 5 are from the same section of e19 mouse and include one molar (Figures 5a and 5a'), one inferior incisor (Figures 5b and 5b'), and one superior incisor (Figures 5c and 5c'). Incisors are strongly labeled, whereas rudimental molars not. The presence of dental extracellular matrix was noted within the two incisors but not in the molar. In addition to incisors, first molars showed PEX mRNA labeling on p3, whereas second molars were positive on p7 (results not shown).
Characterization of Pex mRNA in Adult Bone and Teeth
To verify the presence of Pex mRNA in adult mouse, Northern analysis was performed with total RNA obtained from p3 and adult tissues. A Pex transcript of approximately 7
Discussion
This report provides histochemical evidence for Pex mRNA expression in murine embryonic and postnatal bones and teeth. In these tissues, Pex mRNA was detected in osteoblasts and odontoblasts, respectively. With both ISH and Northern analysis, bones and teeth were the only tissues in which the presence of the Pex mRNA could be detected, suggesting that these two tissues are privileged sites for Pex expression in the developing mouse. A significant concentration of Pex mRNA was also detected in adult incisors, which grow continuously in rodents, whereas in adult calvariae and in nongrowing molars Pex mRNA expression appeared to be considerably lower. Taken together, these data suggest a role for Pex in the development of bones and teeth.
Pex mRNA present in developing teeth (long arrows) and mandibular bone (short arrows) in e19 mouse. Early (
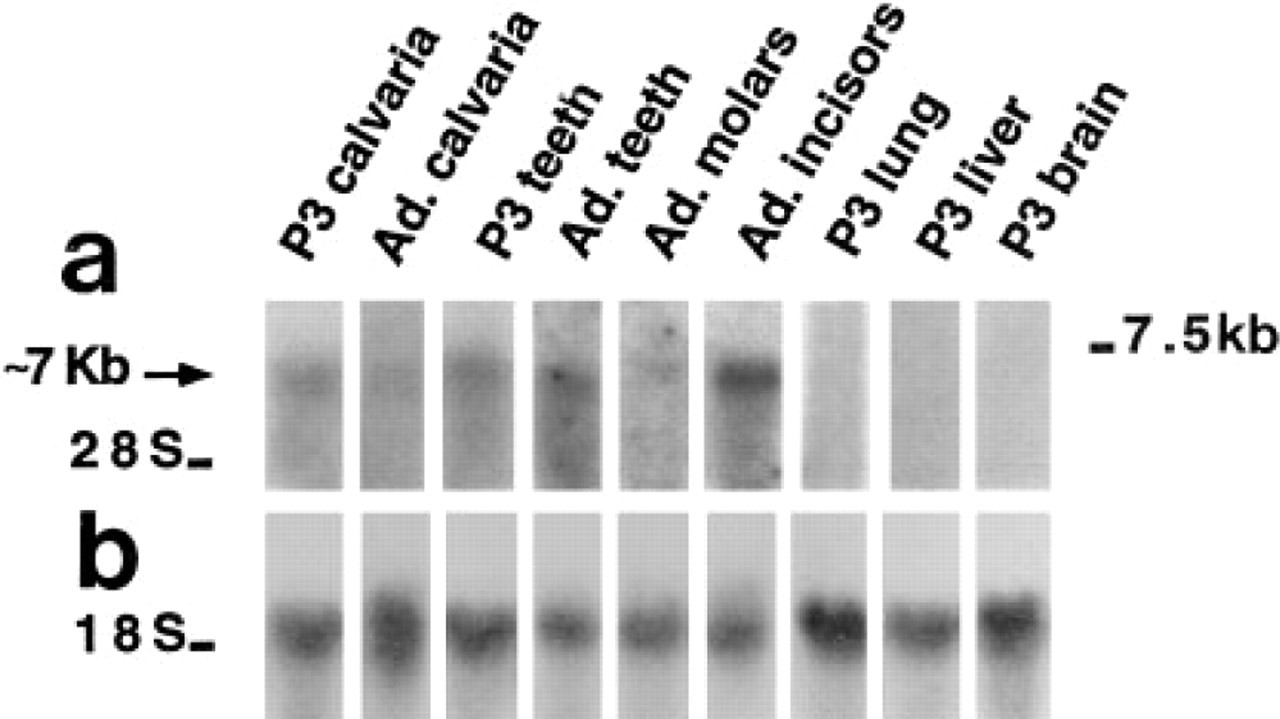
Northern blot analysis of Pex mRNA levels in mouse p3 and adult calvariae, teeth, adult molars, and incisors, p3 lung, p3 liver, and p3 brain. Data shown are from three different Northern blot experiments (Lanes 1–3, 4–6, and 7–9). (
Osteoblasts and chondroblasts/chondrocytes are important cell components of developing bone. Major products of osteoblasts and chondrocytes are the extracellular matrix of bone and cartilage, respectively. The bone extracellular matrix is composed of 90% collagen and 10% noncollagenous proteins (Desbois and Karsenty 1995; Nefussi et al 1997), including osteocalcin, osteopontin, bone sialoprotein, and osteonectin. Our ISH results provide evidence for the presence of Pex mRNA in osteoblasts, localized in close proximity (border zone) to the extracellular matrix, suggesting that Pex may play a role in matrix elaboration, deposition, and/or mineralization. In addition, we demonstrate a temporal relationship between the onset of Pex mRNA expression and the onset of matrix deposition. Although not identical to the situation observed during bone development, Pex mRNA expression in teeth also suggests a striking correlation with matrix deposition.
Our results are in concordance with the clinical features of Hyp and Gy mice, which harbor large deletions in the Pex gene (Beck et al. 1997; Strom et al. 1997). Both mutant animal models exhibit abnormalities in bone and tooth formation, including rickets, osteomalacia, and formation of an interglobular dentin (Eicher et al. 1976; Lyon et al. 1986; Abe et al. 1992). In Hyp mice, mineralization of bone extracellular matrix is delayed (Ecarot-Charrier et al. 1988; Ecarot et al. 1992). Therefore, loss of Pex function appears to be responsible for the bone mineralization defect in Hyp mice. The precise mechanism by which Pex may regulate mineralization of the extracellular matrix is unclear. However, the Pex protein structure suggests that it may function as a peptidase (The HYP Consortium 1995; Turner and Tanzawa 1997). Therefore, one can postulate that Pex is a key component controling the bioactivity of one or several peptides influencing osteoblast and odontoblast proliferation and/ or differentiation. It may also be involved in proteolysis of extracellular matrix proteins. In this regard, an interesting candidate is osteocalcin, a 46 amino-acid peptide produced and secreted exclusively by osteoblasts and odontoblasts (Desbois et al. 1994; Desbois and Karsenty 1995). Studies performed with knockout mice suggest that osteocalcin interferes with the mineralization process (Ducy et al. 1996), a finding compatible with the plasma levels of osteocalcin in Hyp mice relative to normal mice (Gundberg et al. 1992).
Like patients with XLH, both Hyp and Gy mutant mice develop hypophosphatemia secondary to impaired renal phosphate reabsorption (Rasmussen and Tenenhouse 1995). The relationship between the renal phosphate transport defect in XLH, Hyp, and Gy mice and the loss of Pex function is not clear. A possible role of PEX/Pex may be to metabolize a heretofore unidentified circulating peptide hormone involved in regulation of renal phosphate transport and phosphate homeostasis. A factor that inhibits phosphate uptake by normal mouse proximal tubule cells in primary culture was recently reported in Hyp mouse serum (Lajeunesse et al. 1996), a finding consistent with data from parabiosis experiments between normal and Hyp mice (Meyer et al. 1989) and kidney cross-transplantation studies in Hyp vs normal mice (Nesbitt et al. 1992). This hypothetical phosphaturic factor has not yet been identified and its source has not been established. We suggest that osteoblasts and odontoblasts may be involved in the inactivation of this hypothetical factor. Alternatively, Pex could be involved in the processing of an inactive precursor into a bioactive peptide that stimulates phosphate reabsorption by the kidney.
In conclusion, we demonstrate the presence of Pex mRNA in both embryonic and adult bones and teeth. Cells expressing Pex have been identified as osteoblasts and odontoblasts. Therefore, Pex could be a useful marker for these two cell types. In adult bones, the levels of Pex mRNA were lower than those in embryos. In adult teeth, Pex mRNA concentration remained elevated in incisors and was lower in molars. These results suggest that Pex is involved in the development of bone and tooth tissues.
Footnotes
Acknowledgements
Supported by grants from FCAR (to LDG, PC, and GB) and the Medical Research Council of Canada (MT-12686, to MM). AFR is supported by the Canadian International Development Agency (CIDA), Canada and by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Brazil.
We are grateful to Ms J. Marcinkiewicz for skillful technical help and Mr C. Charboneau for photographic work.
