Abstract
Transforming growth factor-β1 (TGF-β1) is implicated in prostate development, and elevated expression of TGF-β1 has been correlated with prostate carcinogenesis. In this study, cell type specificity of TGF-β1 and TGF-β receptor Type II (RcII) protein expression was determined by immunocytochemistry in human normal prostate and compared to prostate carcinoma tissues. Heterogeneous localization patterns of LAP-TGF-β1 (TGF-β1 precursor) and RcII were observed in both epithelial and mesenchymal cells in fetal prostate, with LAP-TGF-β1 localizing to more basal epithelial cells. Homogeneity of LAP-TGF-β1 staining was increased in neonatal, prepubertal, and adult prostate, with elevated immunoreac-tivity noted in epithelial acini relative to stromal tissue for both LAP-TGF-β1 and RcII proteins. In stromal tissues, RcII cell localization exhibited staining patterns nearly identical to smooth muscle α-actin. In prostate carcinoma, LAP-TGF-β1 localized to carcinoma cells with an increased staining heterogeneity relative to normal prostate. In contrast to normal epithelial cells, carcinoma epithelial cells exhibited low to nondetectable RcII staining. Stromal cell staining patterns for LAP-TGF-β1 and RcII in carcinoma, however, were identical to those of normal prostate stromal cells. These studies implicate both epithelial and stromal cells as sites of TGF-β1 synthesis and RcII localization in the developing and adult normal human prostate. In addition, these data indicate a loss of epithelial expression of RcII concurrent with altered LAP-TGF-β1 expression in human prostate carcinoma cells.
TGF-β1
TGF-β1 has been implicated in several aspects of carcinoma progression, including an elevated metastatic potential and masking of tumor cells from immune surveillance (Welch et al. 1990; Steiner and Barrack 1992; Chang 1995; Pierce et al. 1995). Expression of TGF-β1 message and protein is elevated in many transformed cell lines and carcinomas, including prostate cancer, and elevated expression has been correlated with metastatic progression (Merz et al. 1991; Thompson et al. 1992; Eklov et al. 1993; Truong et al. 1993; Steiner et al. 1994; Van Laethem et al. 1995). However, many carcinoma cell lines exhibit a decreased response to TGF-β1 (Huggett et al. 1991; Chang et al. 1993; Rodeck et al. 1994; Herman and Katzenellen-bogen 1994), shown in some cases to result from defects in the TGF-β receptor Type II (Rc II) (Park et al. 1994). Hence, it has been postulated that paracrine effects of carcinoma-derived TGF-β1 on adjacent stroma could lead to an increased metastatic capability through regulation of stromal cell-mediated events (Pierce et al. 1995). Accordingly, the cell type localization of TGF-β1 and TGF-β receptors is important to understand TGF-β1 mechanisms in normal development and cancer progression.
Localization of TGF-β1 mRNA and protein in the rodent has shown epithelial cell expression in both normal prostate gland and the Dunning R3327 PAP tumor (Lindstrom et al. 1996). In human subjects, previous studies to assess the cell-type specificity of TGF-β1 protein expression in prostate carcinoma have met with equivocal results. The use of the LC antibody to TGF-β1 (Carruba et al. 1994) has shown intracellular staining in prostate carcinoma epithelial and stromal cells (Truong et al. 1993; Steiner et al. 1994). However, this antibody may recognize only cells positive for cell-bound or intracellularly activated TGF-β1 (Barcellos-Hoff et al. 1994). Use of the CC antibody, which recognizes extracellular TGF-β1 (Carruba et al. 1994), has shown extracellular localization primarily in stromal regions adjacent to carcinoma acini. Because TGF-β1 is secreted as a latent form, these studies have not yet established the cell type(s) synthesizing TGF-β1 in human prostate carcinoma. However, the development of an antibody to the LAP region of human TGF-β1 precursor (Barcellos-Hoff et al. 1994) makes possible the definitive localization of cells synthesizing TGF-β1 protein.
Similar studies to examine expression of TGF-β receptors in prostate carcinoma has been recently reported by several groups (Kim et al. 1996; Williams et al. 1996; Guo et al. 1997). These studies correlate a loss of receptor expression coordinate with carcinoma progression. An alteration in receptor expression pattern in carcinoma relative to receptor and TGF-β1 expression patterns in developing human fetal and neonatal prostate and in normal adult prostate, however, have not been examined in a comprehensive manner.
The present study was conducted to determine cell-type specificity and differential staining patterns of cellular LAP-TGF-β1 precursor and RcII protein in developing and normal adult human prostate tissue in comparison with prostate carcinoma. The results presented here characterize fetal to adult expression patterns and show alterations in LAP-TGF-β1 and RcII staining patterns in carcinoma cells relative to normal prostate epithelial cells, with no apparent alteration in stromal cell staining patterns for either protein.
Materials and Methods
Tissue Preparation
Carcinoma tissue was removed from patients undergoing prostatectomy for treatment of prostate cancer. Prostate tissues were provided by the Scott Department of Urology and the Department of Pathology, The Methodist Hospital, Baylor College of Medicine. Normal adult prostate glands were removed from cadaver donors. Consent and tissue processing procedures followed human tissue use protocols established in the SPORE (Specialized Program of Research Excellence) in prostate cancer at Baylor College of Medicine. Fetal and neonatal prostate tissues were processed and provided by Dr. Edith Hawkins (Texas Children's Hospital, Houston, TX). After removal, whole adult glands were reproducibly sectioned in samples, regions P1–P15 as previously described (Wheeler and Lebovitz 1994), and all tissues were fixed in 10% neutral buffered formalin at room temperature (RT) overnight, processed through dehydration, and embedded in paraffin blocks according to routine histological procedures (Bratthauer 1994). Serial sections (5 μm) were made by routine microtomy from multiple regions of an individual sample, applied to poly-
Immunohistochemistry
Antibody to human LAP-TGF-β1 (no. AB-246-PB) was obtained from R & D Systems (Minneapolis, MN). This antiserum was prepared to recombinant human LAP as immunogen and is monospecific for the human LAP pro-segment TGF-β1 precursor with no crossreactivity to LAP-TGF-β2 precursor or other cytokines or proteins (Barcellos-Hoff et al. 1994). This antibody has been shown to specifically recognize intracellular LAP-TGF-β1 precursor in human cells by immunohistochemistry and Western analysis (Barcellos-Hoff et al. 1994). Antibody specific to human RcII (no. 06–227) was obtained from Upstate Biotechnology (Lake Placid, NY). This antibody was made to the first 28 amino-acid residues of human RcII, using a synthetic peptide as immunogen, and is monospecific for human p75 type II TGF-β receptor isoform with no crossreactivity to other receptors (Lin et al. 1992). This antibody has been used to localize RcII receptor protein in a cell type-specific manner in human uterine tissue (Chegini et al. 1994). Anti-smooth muscle α-actin antibody (Boehringer Mannheim, Mannheim, Germany; clone asm-1) was made to the first 10 residues with specificity for human, murine, and chicken smooth muscle α-actin with no crossreactivity with skeletal or cardiac α-actin. Secondary antibodies conjugated to biotin and specific to either goat, mouse, or rabbit IgG, were obtained from either Cappel (anti-goat; Cochranville, PA) or Sigma (anti-mouse and anti-rabbit; St Louis, MO).
Tissue sections on slides were deparaffinized in Hemo-De clearing agent (xylene substitute; Fisher Scientific; Fair Lawn, NJ) for 15 min at RT and step-rehydrated through a graded series of ethanol. Tissue sections were equilibrated in PBS (pH 7.4) and permeabilized with 0.1% Triton X-100 in PBS for 5 min. For RcII immunolocalization, the microwave fixation antigen retrieval method (Shi et al. 1991) was found to provide optimal and most consistent staining. For LAP-TGF-β1, the antigen retrieval method produced identical results to standard procedures. For the microwave antigen retrieval procedure, slides were rehydrated as above, placed in a 2–L glass beaker containing 0.01 M citrate buffer (pH 5.9), and microwaved at full power (800 W) for 30 min before cooling and equilibration in PBS (Shi et al. 1991). To neutralize endogenous peroxidase activity, slides were pretreated with 3% hydrogen peroxide for 5 min. Primary antibodies were applied at the following concentrations: anti-human LAP-TGF-β1 (0.5 μg/ml); anti-human RcII (5 μg/ml); and anti-smooth muscle α-actin (5 μg/ml), per manufacturer's recommended concentrations in PBS plus 1% BSA (PBS-BSA buffer, 40 μl per section under a plastic coverslip) for 1 hr in a humidified chamber at 37C. Tissue sections were washed with agitation consecutively in PBS followed by PBS/0.1% Triton X-100, and PBS for 10 min each. Immunoreactivity was detected using biotinylated secondary antibodies (1:500 rabbit anti-goat and 1:1000 dilution goat anti-rabbit in PBS-BSA buffer) incubated for 45 min as above, followed by a 30-min incubation with ExtrAvidin–peroxidase (Sigma, 1:500 dilution in PBS-BSA buffer) and visualized by a 7-min incubation with 0.7 mg/ml 3,3'diaminobenzidene tetrahydrochlo-ride as the chromagen (Sigma, in 1.6 mg/ml urea hydrogen peroxide, 60 mM Tris buffer, pH 7.6, RT). Control parallel slides were prepared either lacking primary antibody or lacking primary and secondary antibodies, or stained with normal sera to control for background reactivity. The color reaction was terminated by immersion in PBS for 5 min, followed by rinsing in water. Coverslips were mounted on slides with 40 μl Biomedia gel mount (Fisher) and slides analyzed by light microscopy using a Nikon Labophot-2 system fitted with a Nikon N6000 camera and photographed with Ektachrome ASA 400 slide film.
Results
Analysis of LAP-TGF-β1 and RcII protein localization was performed in normal human prostate glands including fetal, neonatal, prepubertal, postpubertal, and adult prostate glands (n = 12). Donor ages were 37-week fetal, 6.5-month-old, 11.5-month-old, 10.5-year-old, 14-year-old, 19-year-old, 29-year-old, 38-year-old, and one adult donor of unknown age. Each was cancer-free (histopathology analysis) and multiple regions of each sample were analyzed for localization studies.
Human fetal prostate was typified by cords of rudimentary epithelial islands and glandular acini lined by two or three layers of cuboidal cells, as shown in Figure 1A. Immunolocalization of LAP-TGF-β1 protein exhibited a spotty, focal distribution, restricted to the cuboidal epithelial cells of acini (Figure 1B) and developing ducts (data not shown). Low to nondetectable immunoreactivity was observed in adjacent stromal mesenchymal cells. Epithelial cells exhibited a heterogeneous staining pattern, with apparent LAP-TGF-β1-negative cells adjacent to LAP-TGF-β1-positive cells, which tended to be oriented more towards the basal surface of the ducts or acini. No extracellular immunoreactivity was observed. Control sections in all cases showed no specific immunoreactivity, as shown in Figure 1C.
In contrast to LAP-TGF-β1, localization of RcII protein was observed in both epithelial and stromal cells in fetal prostate, as shown in Figures 1D and 1E. Islands of epithelial cell clusters and distinct acini were positive for RcII immunoreactivity, with some cells exhibiting differential staining intensity relative to adjacent cells. Stromal cells in close apposition to epithelial acini exhibited a comparable or higher staining intensity for RcII. The stromal cell staining pattern for RcII (Figure 1E) was almost identical to the staining pattern for smooth muscle α-actin, as shown in serial sections (Figure 1F). To further illustrate this co-localization, Figure 1G shows the localization of RcII in a stromal region void of epithelial acini. Figure 1H is a direct serial section stained for smooth muscle α-actin. Antibody positive staining for RcII was seen in smooth muscle tunica media of a centrally located muscular artery (Figure 1G) and in other stromal cells positive for smooth muscle α-actin. The similarity of stromal RcII localization with α-actin localization in serial sections was a pattern observed in all sets of tissues examined from fetal to adult carcinoma.
Localization of LAP-TGF-β1 and RcII in neonatal tissue exhibited a more homogeneous staining pattern relative to fetal tissue. Both LAP-TGF-β1 and RcII exhibited an increase in staining intensity in epithelial cells of glandular acini, as shown in Figures 2A and 2B from a 6.5-month-old neonate. Compared with fetal tissue, epithelial cells were more uniformly positive for LAP-TGF-β1. Basal cells exhibited an elevated LAP-TGF-β1 staining intensity relative to columnar acini cells, as shown in Figures 2A and 2C. LAP-TGF-β1 was present in stromal cells but was weaker in immunoreactivity compared with epithelial cells and showed a more focal and heterogeneous pattern. RcII protein appeared to be uniformly expressed in all epithelial cells and in isolated stromal cells, as shown in Figure 2B. Localization patterns in epithelial cells were similar in an 11.5-month-old neonatal prostate (Figures 2C and 2D). Stromal regions, however, showed an apparent increase in LAP-TGF-β1-positive stromal cells (Figure 2C). Epithelial acini were uniformly RcII-positive, as were isolated stromal cells (Figure 2D). Connective tissue trabeculae, observed between regions of glandular acini, exhibited lower immunoreactivity for RcII, whereas stromal cells immediately adjacent to epithelial acini were RcII-positive, as shown in Figure 2D, where stromal cells appear in cross-section. LAP-TGF-β1 and RcII localizations in the prostate of a 10.5-year-old were similar to those in neonatal tissue (data not shown).
Figures 2E and 2F show representative regions of expression patterns in normal disease-free adult tissues (29-year-old). These prostate glands contained a more extensive development of glandular acini, exhibiting a more well-developed, mature secretory phenotype typical of the normal adult human prostate gland. LAP-TGF-β1 (Figure 2E) was uniformly localized in epithelial acini and exhibited a more heterogeneous staining pattern in stromal cells. RcII (Figure 2F) staining patterns were uniformly homogeneous in epithelial acini in all tissues. RcII exhibited a spotty pattern in stromal cells and, as in fetal tissue, showed a staining distribution identical to that of α-actin (data not shown). Staining patterns for both LAP-TGF-β1 and RcII were essentially identical in prostate tissue samples from a 14-, 19-, and a 38-year-old (data not shown) compared with the 29-year-old (Figures 2E and 2F). A comparison of fetal with adult tissues showed that immunoreactivity for RcII in stromal cells increased with age from neonatal to adult. In all samples analyzed, the staining patterns and relative staining intensity per cell for both LAP-TGF-β1 and RcII showed no apparent differences in regions of transition zone compared with peripheral zone of the mature prostate gland. In addition, the localization pattern for LAP-TGF-β1 and RcII in normal regions of prostate carcinoma specimens (n = 23) were identical to patterns observed in disease-free normal adult prostate tissue as described above.
LAP-TGF-β1 and RcII protein expression in human prostate carcinoma tissues exhibited fundamental changes in staining intensity and patterns of cell type localization. Multiple regions of excised whole prostate gland were examined from 23 patients with peripheral zone prostatic carcinoma in regions ranging from well to more poorly differentiated (Gleason 3–4) in each of 23 patients. Figure 3A shows LAP-TGF-β1 localization in a well- to moderately differentiated Gleason 3 region. LAP-TGF-β1 staining intensity was high in carcinoma epithelial acini but exhibited a more variable pattern of cell-to-cell staining relative to normal glandular tissue. Some acini exhibited an obvious increase in staining intensity relative to normal prostate. Figure 3B shows a representative staining profile of a region of a less moderately differentiated carcinoma (Gleason 3–4). The staining patterns of LAP-TGF-β1 in smaller acini consistently exhibited a more variable staining intensity relative to normal prostate gland. This pattern of variable staining intensity was observed in all Gleason 3–4 carcinoma samples examined. In addition, individual cells within an acinus exhibited differential staining intensities. Figure 3C shows a representative LAP-TGF-β1 staining pattern from a more poorly differentiated region composed of small, angular acini invading the stroma. As shown here, small acini typically exhibited an apparent increased staining intensity relative to larger fused glands. Very few stromal cells were positive for LAP-TGF-β1 in carcinoma tissue, regardless of differentiation status or Gleason score and, in general, showed an apparent lower staining intensity relative to carcinoma epithelial cells (Figures 3A-3C).
In contrast to LAP-TGF-β1 localization patterns, carcinoma epithelial acini exhibited low to nondetectable RcII staining. Figure 3D shows a representative RcII staining pattern in a well-differentiated carcinoma (Gleason grade 3). An apparent lack of specific RcII staining in carcinoma epithelial cells was noted in acini, and a relatively strong staining intensity was observed in adjacent stromal cells. Figure 3E shows a representative staining pattern observed in regions of smaller, closely packed carcinoma acini (moderately differentiated, Gleason 3–4). Epithelial cells exhibited low RcII staining intensity, whereas individual stromal cells between acini showed high RcII staining intensity. Notable RcII staining was observed in some small carcinoma acini, as shown in Figure 3F (arrow) and in Figure 3G, whereas in larger acini RcII was not detectable (see Figure 3F, acini in left side). In stromal regions containing arteries, the tunica media smooth muscle layer was consistently positive for RcII staining, as shown in Figure 3G (arrow), whereas the outer tunica adventitia (fibroblasts) was negative.
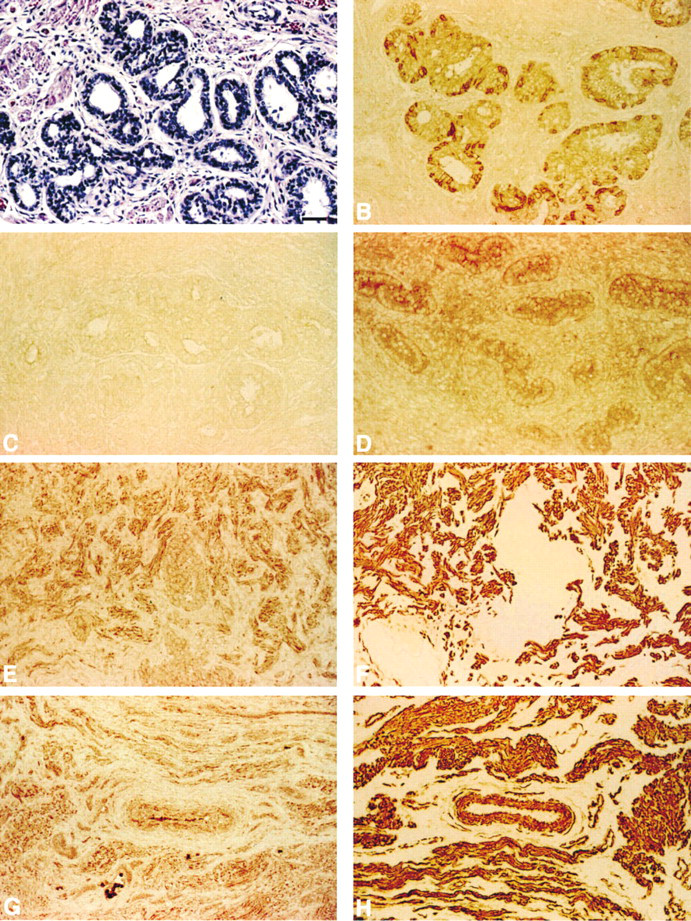
Localization of LAP-TGF-β1 and RcII in human fetal prostate. (
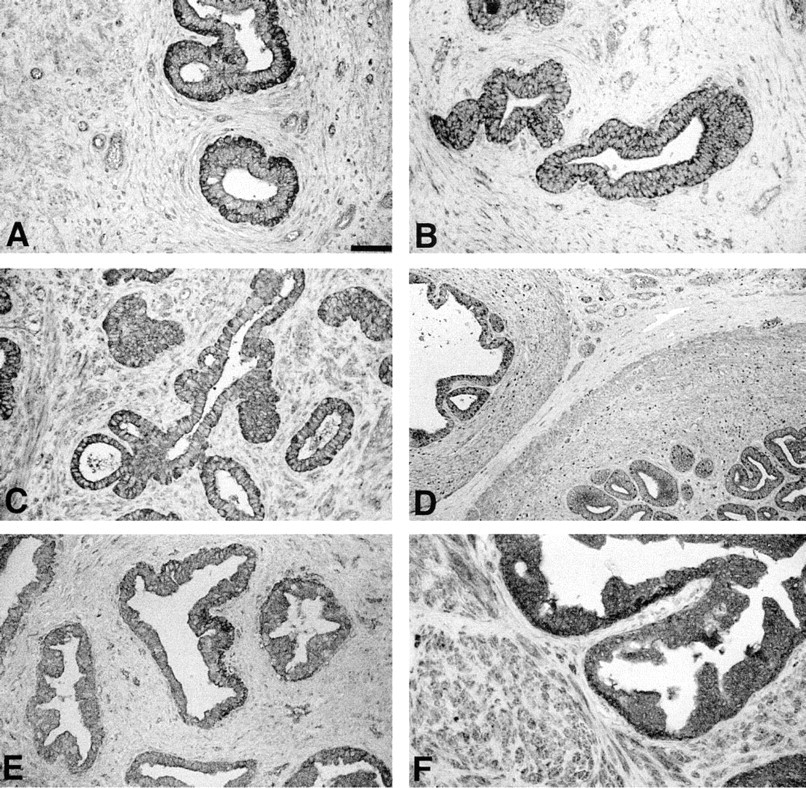
Localization of LAP-TGF-β1 and RcII in neonatal through normal adult prostate gland. (
Discussion
This study used immunocytochemistry to localize the LAP-TGF-β1 precursor and the RcII receptor to determine the spatial and cell-type specificity of synthesis patterns in developing normal human prostate for comparison with prostate cancer tissue. These studies show localization of LAP-TGF-β1 primarily in isolated basal epithelial cells during development, and less frequently in stromal cells, whereas RcII is present in both epithelial and stromal cells. In fetal tissue, localization of LAP-TGF-β1 was observed in basal cells or in the second layer of cells in rudimentary acini. Stromal cells in fetal prostates were essentially negative for LAP-TGF-β1 protein, implicating a subset of epithelial cells as a site of TGF-β1 synthesis during human prostate development. In contrast, stromal cells in fetal prostate stained positive for RcII, compared with more moderate staining For RcII in epithelial cells. Comparison of neonate with adult showed an increase in the apparent number of positive cells and overall staining homogeneity for both proteins. However, no major apparent changes were noted in postpubertal tissue compared with prepubertal tissues.
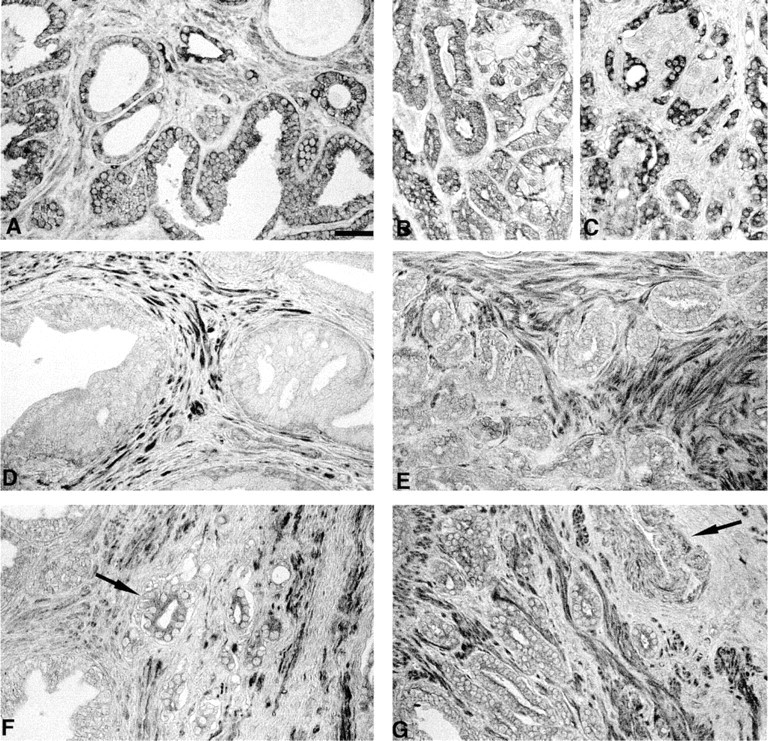
Localization of LAP-TGF-β1 and TGF-β receptor type II in prostatic carcinoma. (
Neonatal, prepubertal, and adult human prostate glands showed localization of LAP-TGF-β1 and RcII in both epithelial acini and adjacent stromal cells. Epithelial cells exhibited an apparent increased staining relative to stromal cells. Notably, the stromal cell localization of both LAP-TGF-β1 and RcII was almost identical to the pattern of smooth muscle α-actin localization. These data are in close agreement with the preferential localization of androgen receptor in α-actin positive cells (Prins et al. 1991; Prins and Birch 1993) and is consistent with hypotheses as to the central role of α-actin positive cells (likely smooth muscle) in stromal–epithelial interactions in prostate development (Prins et al. 1991; Prins and Birch 1995). Such stromal–epithelial interactions are likely important in carcinoma progression as well.
Of fundamental significance were the altered patterns of LAP-TGF-β1 and RcII staining in prostate carcinoma. Carcinoma cells were heterogeneous in LAP-TGF-β1 staining intensity, with heavy staining noted in many cells relative to normal prostate epithelial cells. Our studies are consistent with data in other reports indicating that elevated TGF-β1 message and extracellular protein is associated with prostate carcinoma progression, albeit localized in stroma (Thompson et al. 1992; Truong et al. 1993; Steiner et al. 1994). Stromal cell staining intensity and the pattern of LAP-TGF-β1 and RcII were essentially identical in both carcinoma and normal tissues in our study. These data suggest that the elevated extracellular accumulation of TGF-β1 is primarily the result of elevated expression by carcinoma epithelial cells as opposed to stromal cells.
Significantly, RcII protein staining in prostate carcinoma epithelial cells was almost undetectable relative to normal epithelial cells. However, stromal cell staining for RcII was not altered in carcinoma relative to normal tissue stomal cells. These data suggest that a decrease in RcII in epithelium is associated with human prostate carcinoma progression. Three recent studies have reported that prostate carcinoma epithelial cells exhibit a decreased expression of TGF-β RcI and RcII receptors (both mRNA and protein) (Kim et al. 1996; Williams et al. 1996; Guo et al. 1997). The present study corroborates and extends these previous observations in describing a maintained stromal cell expression of RcII in carcinoma progression relative to normal prostate. This finding was not reported in the previous studies. A second observation unique to this study was the expression of RcII in select small carcinoma acini in moderately differentiated regions. The significance of this observation relative to carcinoma progression is not yet known. The present study utilized the antigen retrieval method for the RcII localization, which exhibited a significant increase in the sensitivity of antibody-specific reactivity. Moreover, differing sources of antibodies were used in the respective studies. The precise role of TGF-β1 in prostate carcinoma progression and the biological significance of altered RcII staining in carcinoma cells remain to be determined. However, our data and those of others must be interpreted with caution owing to the non-quantitative nature of immunocytochemistry, and alternative explanations may exist. Differential staining intensities may relate to altered protein turnover or secretion rates. Hence, it is difficult to conclude with certainty that one cell type necessarily synthesizes more protein than another, using immunocytochemistry. Although descriptive by nature, immunocytochemistry remains the only method to study native protein localization in specific cell types. Therefore, immunocytochemistry is of value to determine cell types synthesizing the protein of interest and apparent pattern changes associated with disease progression.
An overexpression of TGF-β1 concomitant with altered RcII expression and altered cell response appears to be a general observation in many neoplasias and derived cell lines. Melanoma cell lines, spontaneous transformants of high-passage liver cells, E1A-trans-formed 293 cells, breast carcinoma cells, and variants of Meth A sarcoma cells all expressed TGF-β1 at elevated levels and demonstrated enhanced tumorigene-sis in vivo (Huggett et al. 1991; Arrick et al. 1992; Chang et al. 1993; Herman and Katzenellenbogen 1994; Rodeck et al. 1994). Consistently, these lines were resistant to TGF-β1 downstream responses, typically measured through inhibition of cell growth. These studies together suggest that resistance to TGF-β1 may be an integral step in spontaneous transformation leading to tumorigenesis. Several human gastric cancer cell lines resistant to TGF-β1 exhibit genetic alterations in RcII, including truncated message or no detectable mRNA expression (Park et al. 1994). These studies theorized that escape from proliferation control by TGF-β1 during carcinogenesis could be the result of changes in RcII expression.
In this regard, an overproduction of TGF-β1 in prostate cancer, concurrent with a variable resistance of carcinoma cell response through RcII alteration, could result in several stroma-associated alterations leading to a more favorable local environment for carcinoma progression. Angiogenesis, for example, is directed by the stroma, is induced by TGF-β1, and is necessary for tumor progression (Pepper et al. 1990; Yang and Moses 1990). This is consistent with TGF-β1-induced effects in experimental prostate carcinomas (Steiner and Barrack 1992). Moreover, because TGF-β1 action alters extracellular matrix (ECM) remodeling (Chang 1995), alterations in stroma-derived ECM remodeling may also enhance carcinoma progression patterns, as is typically seen in breast and prostate metastases (Welch et al. 1990; Steiner and Barrack 1992). Moreover, it is well established that different carcinomas induce different patterns of “fibroblastic response” in vivo (Lieubeau et al. 1994) and that stromal cell responses, including expression of growth factors, are mediated by paracrine-acting factors derived from carcinoma cells (Singer et al. 1995).
Normal prostate development as well as carcinoma initiation and progression in the human prostate gland likely involves a complex interplay between androgens, growth factors, and stromal–epithelial interactions. In addition to other factors, TGF-β1 is known to affect normal cell proliferation and differentiation and influences carcinoma phenotype and malignant progression. Further studies to address the specific role of TGF-β1 action in prostate stromal and epithelial cells will allow a more comprehensive understanding of the role of this growth factor in prostate development and carcinoma progression.
Footnotes
Acknowledgements
Supported by NIH DK45909, CA58093, CA58204, and by a grant from Sheffield Biomedical Technologies Inc.
We wish to thank Dr Tom Wheeler, Department of Pathology, Baylor College of Medicine and Dr Edith Hawkins, Texas Children's Hospital, for providing fixed tissue specimens and pathological analysis of tissue sections, and the SPORE in Prostate Cancer Research at Baylor College of Medicine for providing fixed tissue samples. We also wish to thank Ms Liz Hopkins for microtomy.
