Abstract
Using antibodies against the 31-kD and 70-kD subunits of vacuolar type H+-ATPase (V-ATPase) and light microscopic immunocytochemistry, we have demonstrated the presence of this V-ATPase in rat submandibular gland. We have also investigated the adaptive changes of this transporter during acid-base disturbances such as acute and chronic metabolic acidosis or alkalosis. Our results show intracellularly distributed V-ATPase in striated, granular, and main excretory duct cells in controls, but no V-ATPase immunoreaction in acinar cells. Both acute and chronic metabolic acidosis caused a shift in V-ATPase away from diffuse distribution towards apical localization in striated and granular duct cells, suggesting that a V-ATPase could be involved in the regulation of acid–base homeostasis. In contrast, during acidosis the main excretory duct cells showed no changes in the V-ATPase distribution compared to controls. With acute and chronic metabolic alkalosis, no changes in the V-ATPase distribution occurred.
The two-stage hypothesis of salivary secretion, first described by Thaysen et al. (1954), is now well established. The primary saliva is an isotonic plasma-like fluid, which is secreted from acinar cells and, in most species, is subsequently modified in the duct system by secretion of K+ and HCO3 - and absorption of Na+ and Cl- (Young et al. 1967,1970; Young 1968; Knauf et al. 1982; Paulais et al. 1994). Current models of HCO3 - secretion in the main excretory duct of the rat submandibular gland propose the presence of luminally located Na+/H+-, K+/H+-, and Cl-/HCO3 - exchangers together with Na+ and Cl- channels and a basolaterally located Na+ pump, a K+ channel, and an additional Na+/H+ exchanger (Young et al. 1967, 1970; Young 1968; Schneyer 1968,1969; Zhao et al. 1995).
Studies of secretory mechanisms in the duct system were originally performed in the main excretory duct because of its accessibility for microperfusion studies (Young 1968; Schneyer 1968,1969; Young et al. 1967, 1970; Knauf et al. 1982). However, the main excretory duct represents only a small portion of the salivary duct tissue, the majority being made up of intralobular ducts. In the rat submandibular gland, intralobular ducts can be morphologically divided into intercalated, granular, and striated ducts (Tamarin and Sreebny 1965; Shackleford and Schneyer 1971; Young and Van Lennep 1978). The intercalated ducts are short, narrow duct segments that connect the acini to the larger striated ducts. The striated ducts exhibit extensive infoldings of the basal cell membrane surrounded by many mitochondria. This suggests that they might represent the major site of the duct system in which re-absorption of NaCl takes place. The proximal segments of the striated ducts form granular ducts. They have few basal infoldings, are characterized by an abundance of secretory granules, and exhibit a pronounced cellular heterogeneity. Three different cell types are present: wide dark granular cells, light granular cells, and narrow agranular cells. These duct segments are particularly well developed in the submandibular glands of male rodents (Flon et al. 1970; Dorey and Bhoola 1972; Cutler and Chaudhry 1973). On the basis of their morphological appearance, it has been speculated that granular ducts might have a reduced ion transport capacity compared to striated ducts. However, functional studies in isolated intralobular granular ducts have demonstrated similar electrolyte transport properties in main, granular, and striated duct segments (Dehaye and Turner 1991; Dinudom et al. 1993a,b).
The salivary duct epithelium responds to acid–base changes in the blood by marked changes in secretion of HCO3 -. During metabolic acidosis, ductal HCO3 -secretion is abolished, whereas during metabolic alkalosis HCO3 - secretion is enhanced (Knauf et al. 1975).
In the kidney the cellular mechanisms involved in the regulation of acid–base homeostasis have been studied in more detail (Gluck et al. 1982; Madsen and Tisher 1984; Bastani et al. 1991; Verlander et al. 1991; Sabolic et al. 1997). Morphological studies have provided evidence for two types of intercalated cells in the cortical collecting tubule. The HCO3 - secreting B-cells possess a Cl-/HCO3 - exchanger in the apical membrane and a vacuolar H+-ATPase in endocytotic vesicles that can fuse with the basolateral membrane. H+ secretion occurs in A-cells, which have a vacuolar H+-ATPase (V-ATPase) in the apical and a Cl-/HCO3 -exchanger in the basolateral membrane (Brown et al. 1988a,b; Alper et al. 1989). Schwartz et al. (1985) have suggested that acidosis induces HCO3 --secreting cells (B-cells) to change the location of these transporters to the opposite membranes. However, other studies have proposed adaptive mechanisms that do not necessitate conversion of intercalated cells to cells with opposing polarity of H+ transporters. Immunohistochemical distribution of V-ATPase in normal rat kidney revealed the existence of several morphologically different types of intercalated cells in the cortical collecting duct (Brown et al. 1988b), including a population of cells exhibiting V-ATPases in both apical and basolateral membranes (Bastani et al. 1991). According to this model, adaptive response to acid–base changes should occur by changing the distribution of V-ATPases between intracellular and apical or basolateral membrane location.
A vacuolar proton pump that is physiologically and structurally identical to the kidney V-ATPase has also been found in osteoclasts, which appears to be responsible for creation of the acidic microenvironment fundamental to bone resorption (Baron et al. 1985; Blair et al. 1989; Väänänen et al. 1990). Recently, it has also been suggested that chronic acidosis increases V-ATPase expression in osteoclasts (Nordström et al. 1997).
In the present study, we have determined the localization of the V-ATPase in the secretory endpieces and segments of the duct system in rat submandibular gland by immunohistochemistry. Furthermore, we have assessed the distribution of this enzyme during acute and chronic metabolic acidosis or alkalosis.
Our results demonstrate the presence of an intracellularly distributed V-ATPase in the entire duct system but its absence in acinar cells of the rat submandibular gland. Metabolic acidosis induces a redistribution of the H+-ATPase to the apical side in striated and granular duct cells but not in main excretory duct cells. In contrast, no adaptive change of the distribution of V-ATPase was found during metabolic alkalosis.
Materials and Methods
Materials
Picric acid, formaldehyde, acetic acid, Triton X-100, and Tween 20 were obtained from Merck (Darmstadt, Germany). Gelatin, 3,3-diaminobenzidine, naphthol AS-MX phosphate, and 4-benzoylamino-2,5-dimethoxy-benzenediazonium chloride hemi [zinc chloride]salt (Fast Blue RR Salt) were from Sigma (Deisenhofen, Germany). Dimethylformamide was obtained from Serva (Heidelberg, Germany). Vogel Histo-Comp was from Vogel (Giessen, Germany). Enhanced chemiluminescence reagents were from Amersham–Buchler (Braunschweig, Germany). Nonfat dry milk and prestained protein standards were obtained from Bio-Rad (Munich, Germany). Polyvinylidene difluoride (PVDF) membranes and X-ray films were from Dupont (Bad Homburg, Germany). Pefabloc SC was obtained from Boehringer (Mannheim, Germany). All other reagents were of analytical grade.
Methods
Thirty-six male Wistar rats (200–300 g), six rats for each experimental group, were used. Acute metabolic acidosis and alkalosis were induced by gastric administration of 1.5 M NH4Cl (2.4 ml/100 g bw) or of 0.976 M NaHCO3 (5 ml/100 g bw), respectively. Control animals were treated with 1.37 M NaCl (2.4 ml/100 g bw). The animals were sacrificed 4–6 hr later. Chronic acidosis and alkalosis were induced according to Wright and Knepper (1990), with additional administration of 0.28 M NH4Cl or 0.28 M NaHCO3, respectively, in the drinking water for 6–9 days. Control animals received 0.28 M NaCl in the drinking water.
The animals were anesthetized with pentobarbital. After collection of arterial blood from the abdominal aorta and urine from the urinary bladder, pH, pCO2, HCO3 -, base excess, and %O2 saturation were determined in blood and urine. Subsequently, the animals were sacrificed and the submandibular glands were removed. Glandular tissue was fixed by immersion in Bouin's fixative (water-saturated 71.4% picric acid, 23.8% formaldehyde, and 4.76% acetic acid) for 48 hr at 4C, dehydrated through graded series of isopropanol (80%, 96%, and 100%) and methylbenzoate (100%), and embedded in paraffin. The paraffin blocks were cut in serial 5-μm sections.
Immunocytochemistry
For localization of the vacuolar-type H+-ATPase, indirect enzyme immunocytochemistry was applied. Antibodies against the A- and E-subunits of the bovine H+-ATPase were produced by immunizing rabbits with synthetic peptides coupled to hemocyanin, containing the amino acid sequence of the C-terminus of bovine H+-ATPase A- and E-subunits, DMQNAFRSLED and CGANANRKFLD, respectively, similarly as described by Gluck and Caldwell (1987), Hemken et al. (1992), Hirsch et al. (1988), and Marushack et al. (1992). Goat-anti rabbit alkaline phosphatase- or horseradish peroxidase-conjugated secondary IgG antibodies were from Dako (Hamburg, Germany).
After deparaffinization the sections were rinsed with water for 20 min and washed three times for 5 min with 0.05 M Tris buffer, pH 7.4. The slides were incubated with the primary antibody (1:200 dilution) at room temperature (RT) overnight in 0.05 M Tris buffer containing 0.04% sodium azide, 0.25% gelatin, and 0.06% Triton X-100 to reduce nonspecific background staining. Slides were washed three times for 5 min in 0.05 M Tris buffer and incubated with the secondary antibody (1:50 dilution) for 2 hr at RT in 0.05 M Tris buffer containing 0.25% gelatin and 0.06% Triton X-100. Finally, the sections were rinsed three times for 5 min in 0.05 M Tris buffer, and peroxidase activity was visualized with diaminobenzidine and hydrogen peroxide. Alkaline phosphatase was developed using freshly made solutions of 200 mg Fast Blue RR Salt in 200 ml 0.05 M Tris and addition of 20 mg naphthol AS-MX Phosphate in 500 μl dimethylformamide.
The same immunocytochemistry protocol was applied in six rats from each of the six groups (i.e., acute control, acute acidosis, acute alkalosis, chronic control, chronic acidosis, and chronic alkalosis) and six randomly selected sections from each animal were taken for one analysis. This procedure was repeated on different days at least 10 times. In total, at least 2160 sections were analyzed. Additional procedures were also performed using serial sections from adjacent tissue. Antibodies to the 31-kD and 70-kD subunits of the vacuolar H+-ATPase gave identical immunolabeling patterns. Control incubations without primary antibody, with preimmune serum, or preincubation with 0.5 mM synthetic peptide against which the antibodies had been raised gave no detectable staining. Sections of rat kidney were used as positive controls.
Labeled sections were examined with a light microscope (Olympus; Tokyo, Japan) and photographed. For cell counting, only cells with a distinct nucleus were included. The distribution of the immunoreactivity was classified into absent, apical, basolateral, or diffuse localization. At least 500 cells were counted in each section. Granular duct cells were sub-classified with respect to their morphology into wide dark granular, light granular, and narrow agranular cells.
All values were expressed as mean ± SD. Unpaired Student's t-test was used for statistical analysis. p<0.05 was considered statistically significant.
Preparation of Cortical Brush Border Membranes, Endocytotic Vesicles, and Submandibular Gland Homogenate
Renal cortical brush border membrane vesicles (BBMVs) were isolated by the Mg2+/EGTA precipitation method described by Biber et al. (1981). Briefly, rat kidney cortex slices were homogenized in an iso-osmotic medium containing 5 mmol/liter EGTA. Brush border membranes were purified after two precipitations with MgCl2 (12 mmol/liter) and differential centrifugation.
Endocytotic vesicles from renal cortical homogenates were isolated by differential and Percoll density gradient centrifugation as described by Sabolic and Burckhardt (1990). Submandibular gland homogenate was prepared by grinding tissue with 50 strokes of a motor-driven Potter homogenizer in 10 ml of ice-cold homogenizing buffer containing (in mM) 280 mannitol, 10 HEPES, 10 KCl, 1 MgCl2, and 0.1 Pefabloc SC. The homogenate was centrifuged at 50 × g for 5 min and the supernatant was collected. The pellet containing unbroken cells was resuspended in 10 ml of the same buffer and homogenized once more. After centrifugation at 50 × g for 5 min, both supernatants were combined. Homogenate and membrane vesicles were stored at −20C until use. Protein concentration was assayed as described by Bradford (1976), using bovine serum albumin as a standard.
SDS-PAGE and Western Blotting
Electrophoresis and blotting procedures were performed essentially as described earlier (Thévenod et al. 1994). Briefly, proteins were separated by SDS-PAGE on 9% acrylamide Laemmli (1970) minigels and transferred onto polyvinylidene difluoride (PVDF) membranes. The efficiency of protein transfer was monitored with prestained protein standards. Blots were blocked with 3% nonfat dry milk in Tris-buffered saline containing 0.05% Tween-20 for 8 hr and incubated with primary antibodies (1:2500 dilution) against the 31-kD or the 70-kD H+-ATPase subunit overnight. After incubation with horseradish peroxidase-conjugated secondary antibody (1:6000 dilution), blots were developed in enhanced chemiluminescence reagents and signals were visualized on X-ray films.
Results
Systemic Acid–Base Parameters
NH4Cl administration produced acute metabolic acidosis, with a blood pH of 6.95, a base excess of −24.50 mM, and a urine pH of <6.0, compared to control values of 7.39, −2.13, and 6.46, respectively. NaHCO3 administration produced acute metabolic alkalosis with a blood pH of 7.47, a base excess of 12.48 mM, and a urine pH of 7.95. Chronic acidotic animals had a blood pH of 7.10, a base excess of −15.80 mM, and a urine pH of <6.0, compared to control values of 7.31, −3.90, and 6.30, respectively. Chronic alkalotic animals had a blood pH of 7.37, a base excess of −0.79 mM, and a urine pH of 7.98.
Figure 1 shows a Western blot analysis of endosomal vesicles, cortical brush border membrane vesicles (BBMV), and submandibular gland homogenate (Ho) with the polyclonal antibodies against the 31-kD and 70-kD H+ pump subunits raised as described in Materials and Methods. Similarly to endosomal and renal cortical brush border membranes, salivary gland homogenate showed immunoreactivity to V-ATPase sub-units. However, reactivity of submandibular gland homogenate was less intense than in endosomal and brush border membranes.
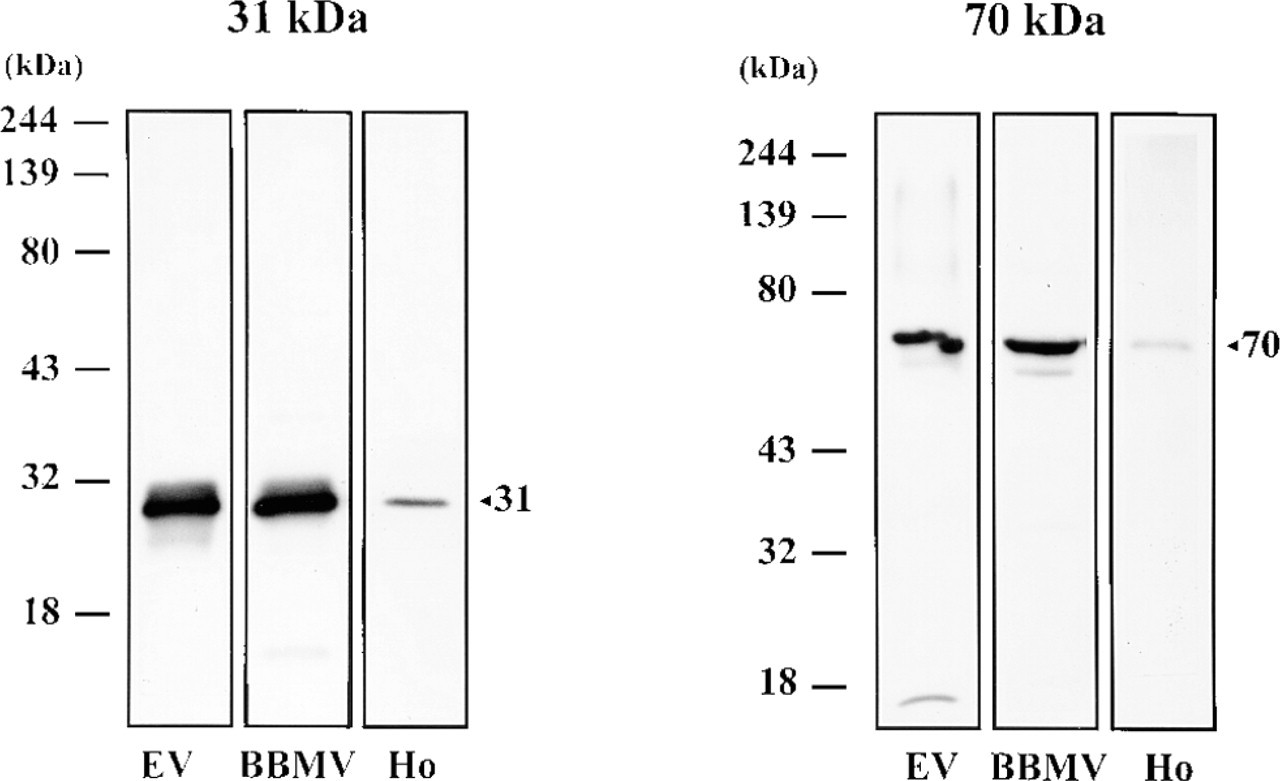
Western blot of V-ATPase subunits (31-kD and 70-kD) in endosomal vesicles (EV), rat renal cortical brush border membrane vesicles (BBMV), and submandibular gland homogenate (Ho). Each lane contained 20 μg of protein. The positions of the molecular weight standards are indicated.
When immunocytochemistry was carried out, both 31-kD and 70-kD subunits could be localized in rat submandibular ducts but not in acinar cells. However, the pattern and the intensity of labeling varied among different duct segments.
V-ATPase Distribution in Controls
Figures 2–4 show the labeling pattern of V-ATPase in the different morphological segments of rat submandibular gland. A quantitative analysis of the results is provided in Tables 1 and 2.
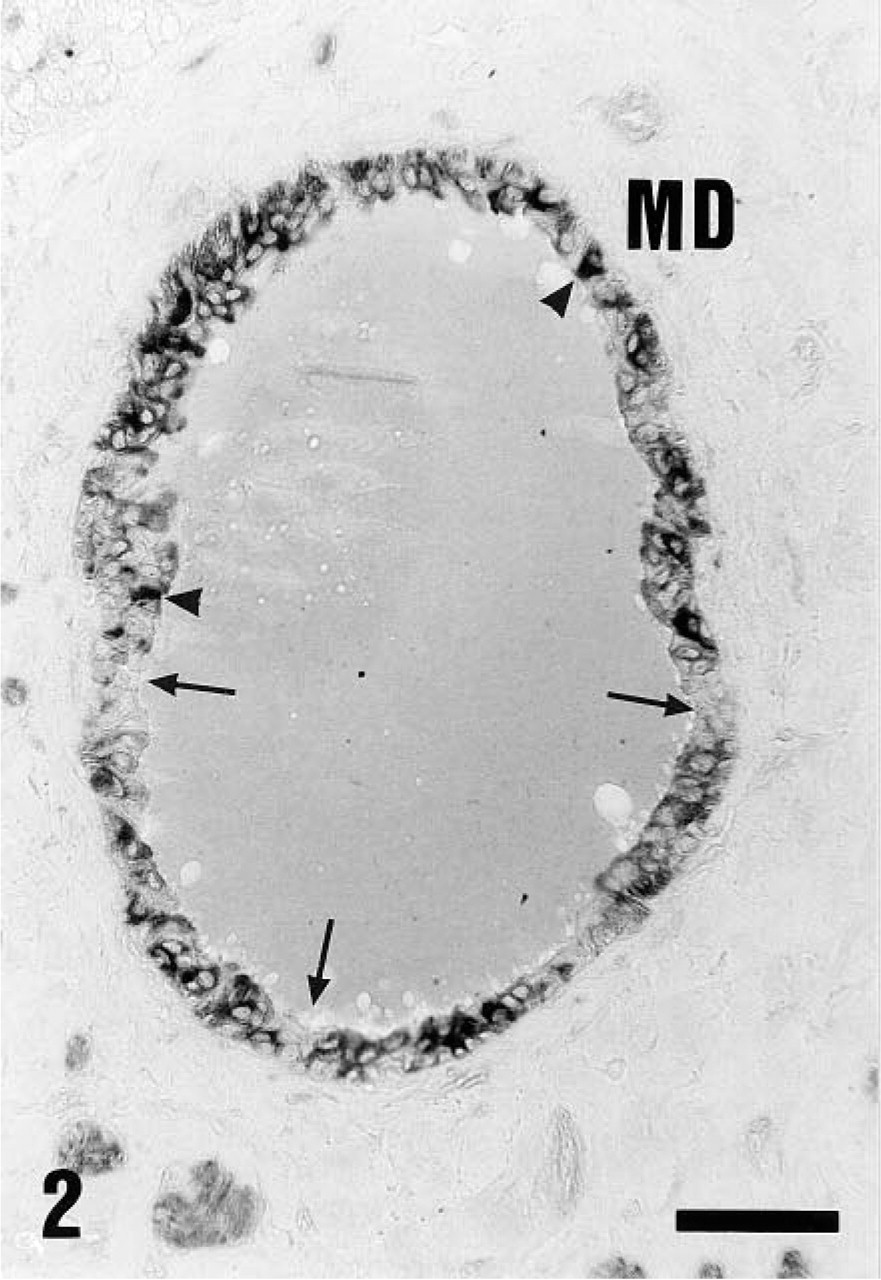
Distribution of V-ATPase (70-kD subunit, 1:200 dilution) in control animals using alkaline phosphatase labeling. Main excretory duct cells (MD) showed a diffuse cytoplasmic distribution. Certain cells were unstained (arrows), and some cells were luminally stained (arrowheads). Similar immunolocalization was observed using the antibody against the 31-kD V-ATPase subunit (not shown). Bar = 30 μm.
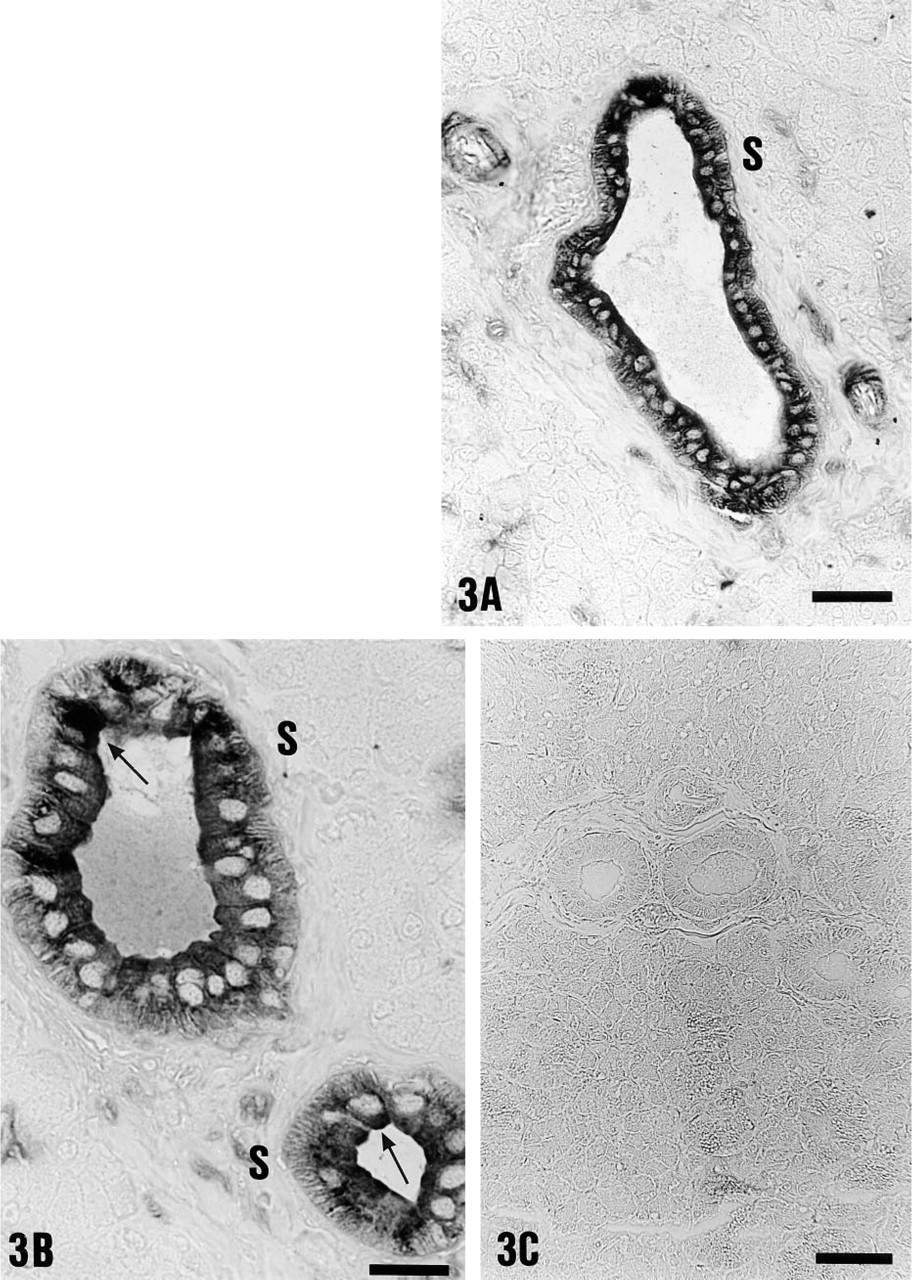
Immunolocalization of the 70-kD V-ATPase subunit (1:200 dilution) in submandibular striated duct (S) cells of controls using alkaline phosphatase staining. (
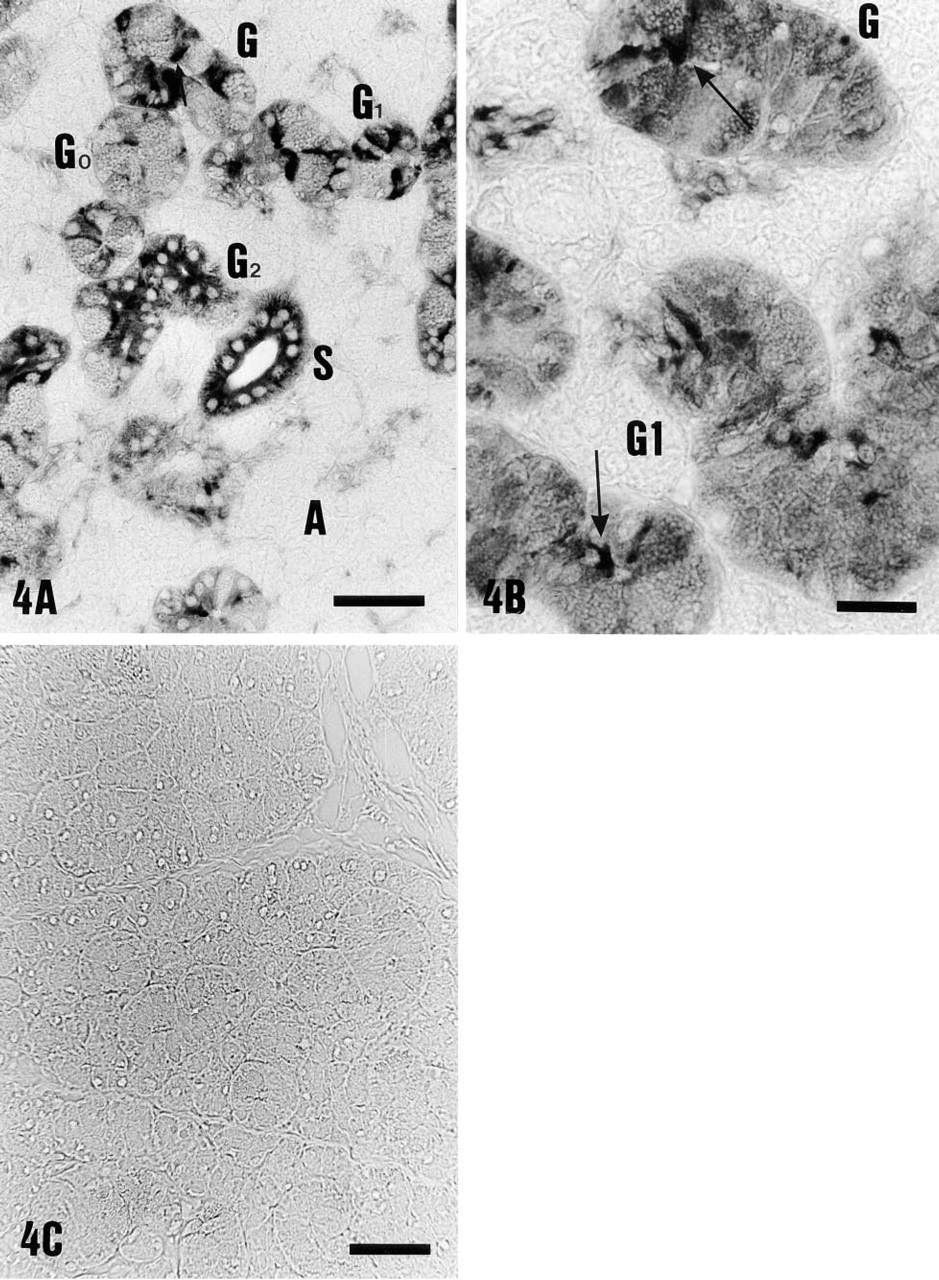
Light microscopic immunoperoxidase staining of striated (S) and granular (G) duct cells of rat submandibular gland with the antibody against the 31-kD V-ATPase subunit. (
V-ATPase immunostaining of rat submandibular duct cells in controls, acute acidosis, and acute alkalosis a
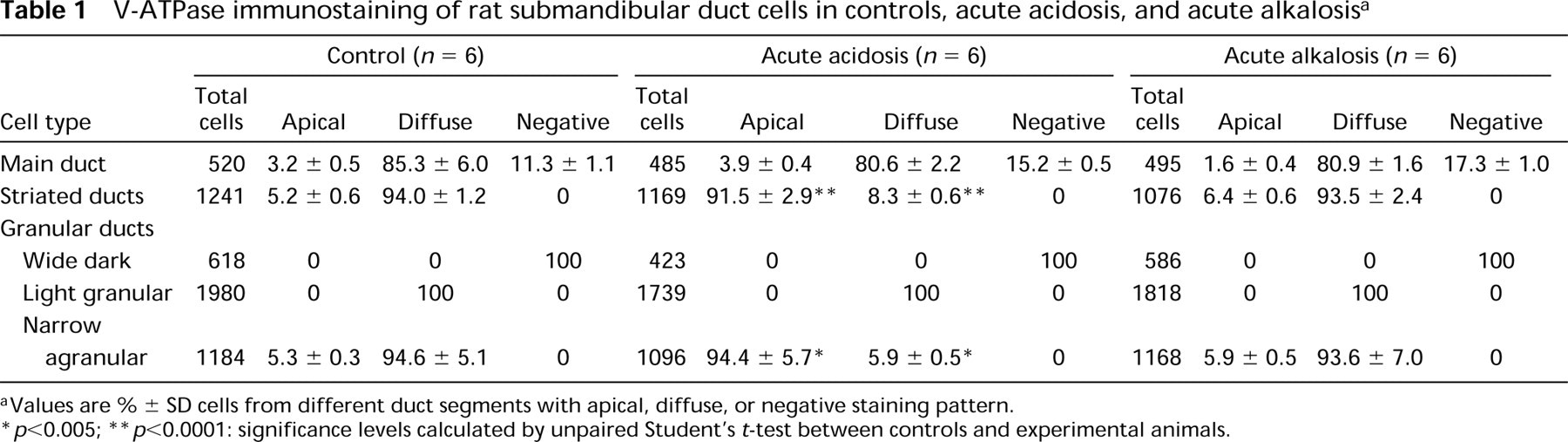
Values are % ± SD cells from different duct segments with apical, diffuse, or negative staining pattern.
p<0.005;
p<0.0001: significance levels calculated by unpaired Student's t-test between controls and experimental animals.
V-ATPase immunostaining of rat submandibular duct cells in controls, chronic acidosis, and chronic alkalosis a
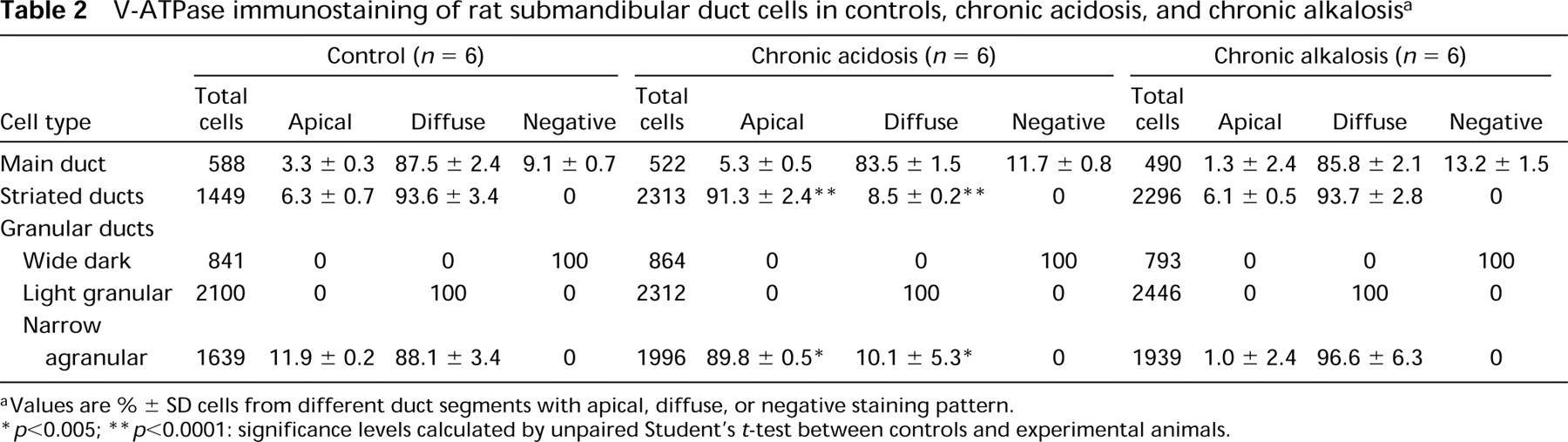
Values are % ± SD cells from different duct segments with apical, diffuse, or negative staining pattern.
p<0.005;
p<0.0001: significance levels calculated by unpaired Student's t-test between controls and experimental animals.
In the main excretory duct cells (MD) of control animals, most cells showed a diffuse intracellular staining of weak to moderate intensity (Figure 2). Apical labeling was also observed in certain cells (Figure 2, arrowheads), representing approximately 3% of main duct cells (Tables 1 and 2). In addition, V-ATPase-containing cells were separated from each other by nonreactive cells (Figure 2; arrows, Tables 1 and 2).
In contrast to main excretory duct cells, V-ATPase immunoreactivity was present in all cells of the striated ducts (S) in control animals (Figures 3A and 3B). Striated ducts showed homogeneous, diffuse intracellular labeling of moderate to strong intensity (Figures 3A and 3B). Apical V-ATPase distribution was also detected in 5–6% of striated duct cells (Figure 3B, arrows; and Tables 1 and 2). Control incubations using preimmune serum gave no detectable staining (Figure 3C).
Granular ducts, which are believed to be modified striated ducts and form the proximal segment of striated ducts, showed a complex pattern of V-ATPase immunoreactivity (G, Figures 4A and 4B). Granular duct cells without any V-ATPase immunoreactivity (G0, Figure 4A) as well as granular duct cells showing diffuse labeling of weak intensity (G1, Figure 4A) were observed in the same section. Moreover, as can be seen from Figure 4A, close to the weakly stained segments, segments with intense diffuse (G2) or apical staining (arrow) were found. This variability was present in different granular ducts in all animals and also within the same duct, which reflects the morphological heterogeneity of these ducts. In wide dark granular cells (G0, Figure 4A), no V-ATPase immunoreactivity was found. In light granular cells, in which the granules occupy less total volume than in dark granular cells, diffuse intracellular staining of weak intensity was observed (G1, Figure 4B). In contrast, agranular cells (Figure 4B) always showed V-ATPase immunolocalization of moderate to strong intensity. In 88–95% of narrow agranular cells V-ATPase distribution was diffuse intracellularly, with 5–10% of cells being apically stained (Figure 4B, arrows). Staining was eliminated by incubation of the antiserum with 0.5 mM peptide antigen (Figure 4C).
Intercalated ducts, which represent the most proximal part of the duct tree, connect the secretory end-pieces with the granular ducts. Intercalated ducts showed no detectable H+-ATPase immunoreactivity (data not shown).
V-ATPase Distribution During Acidosis
During acute and chronic metabolic acidosis, no major changes in the pattern of H+-ATPase labeling or staining intensity in main excretory duct cells were observed, compared to controls (Tables 1 and 2). As an example, Figure 5A shows V-ATPase distribution in main excretory duct cells during acute metabolic acidosis. In contrast, both acute and chronic metabolic acidosis affected the distribution of V-ATPase in striated duct cells (Figures 5B-5D). In >90% striated duct cells (S), V-ATPase was apically located, suggesting that a redistribution of V-ATPase from the interior of the cell to the apical cell side had occurred. Statistical analysis of the data revealed significant differences between control and acidotic animals (Tables 1 and 2; p<0.0001). The intensity of immunoreactivity was moderate in acute and strong in chronic metabolic acidosis. Granular duct cells showed a selective sensitivity to acidosis (G, Figures 5D and 5E). In narrow agranular cells of the granular ducts, a redistribution of V-ATPase similar to that in striated ducts occurred (Figures 5D and 5E, arrows), away from diffuse distribution towards apical localization (p<0.002; Tables 1 and 2). In contrast, no changes in the staining pattern were observed in wide dark and light granular cells compared to controls. Wide dark granular cells remained unstained (Figure 5D, arrowhead), whereas light granular cells showed a diffuse cytoplasmic V-ATPase distribution (Tables 1 and 2; Figure 5E, arrowhead).
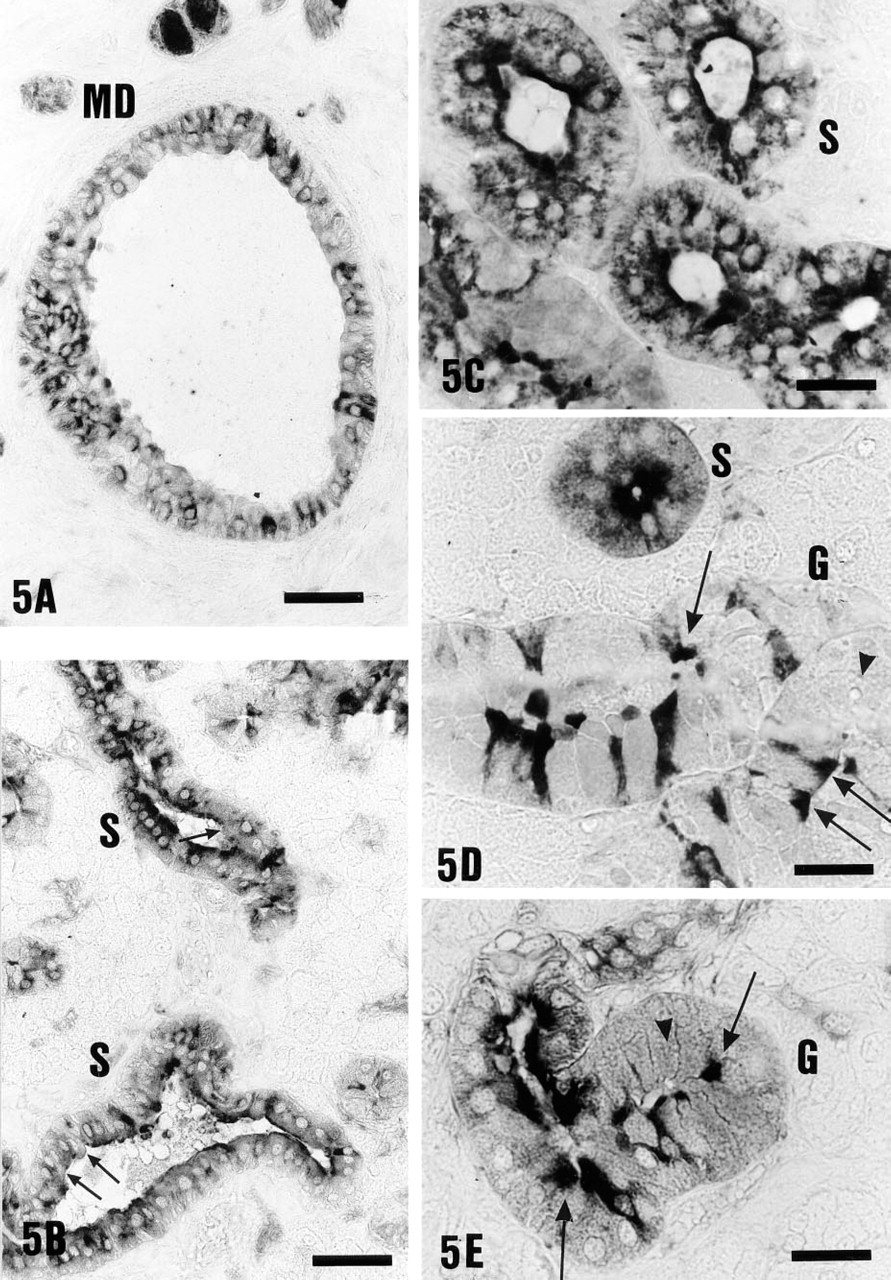
V-ATPase distribution (31-kD or 70-kD) during acute and chronic metabolic acidosis. (
V-ATPase Distribution in Alkalosis
During acute and chronic metabolic alkalosis, no significant changes in V-ATPase immunoreactivity were observed compared to controls (see Tables 1 and 2).
Discussion
In the present morphological study a vacuolar H+-ATPase (V-ATPase) was detected in rat submandibular gland. Immunocytochemical methods were applied by which localization of the V-ATPase could be determined in different duct segments. Using antibodies against cytoplasmic domains of the 31-kD and 70-kD subunits of V-ATPase, we were able to find immunoreactivity in striated, granular, and main excretory duct cells. The distribution of V-ATPase was found to be diffuse, i.e., intracellularly located, in all duct segments. In contrast, secretory endpieces showed no detectable V-ATPase. In addition, we investigated the distribution of V-ATPase in acute and chronic metabolic acidosis or alkalosis to see if body acid–base disturbances might influence the distribution of this H+ transporter, similarly as reported in the kidney (Bastani et al. 1991; Sabolic et al. 1997). Alkalosis induced no detectable changes in the distribution of V-ATPase in all duct segments. Acidosis caused a distribution of V-ATPase to the apical cell side in striated and granular duct cells but not in the main excretory duct cells. This finding suggests that V-ATPase could be involved in the regulation of acid–base homeostasis by allowing a vectorial H+ transport from the cell into the lumen of the duct system. In acidosis, the parallel operation of a H+ pump and a luminally located Cl-/HCO3 - exchanger (Young et al. 1970; Knauf et al. 1975) could lead to H+ secretion and HCO3 - reabsorption through a recently described, basolaterally located Cl-/HCO3 -exchanger (Xu et al. 1995; Zhao et al. 1995).
From studies of ion transport mechanisms (Turner 1993), it appeared reasonable to assume that the main excretory duct of the rat submandibular gland is representative of the entire duct tree. Perfusion studies in the main excretory duct, as well as studies on isolated intralobular ducts, have shown similar electrolyte transport in both duct segments (Dehaye and Turner 1991; Dinudom et al. 1993a,b; Paulais et al. 1994). Our results demonstrate that, in contrast to the main excretory duct, the striated and granular duct segments of the rat submandibular gland show a distinct sensitivity to acidosis. Striated duct cells appear to undergo an adaptation process in acidosis and to translocate the H+ pump to the luminal cell side, which suggests that they could become H+-secreting cells. Immunolabeling of the V-ATPase, however, only indicates its presence whithout providing information about its biological activity or function.
In the main excretory duct, electron microscopy has shown the presence of three morphologically different cell types: dark cells, light cells, and basal cells (Tamarin and Sreebny 1965; Shackleford and Schneyer 1971). Knauf et al. (1975) suggested that the HCO3 -secretory and the HCO3 - reabsorptive mechanisms are localized in different cell types. Dark cells were assumed to be H+-secreting cells and light cells the HCO3 - secreting cells. During metabolic acidosis, the proportion of dark cells increased significantly (Knauf et al. 1975). In the present study, using light microscopy, it was not possible to detect any changes in distribution between the cells with and without V-ATPase in the main excretory duct. Furthermore, no morphological differences could be detected between the striated duct cells of acidotic animals and controls. To address this point, electron microscopy will be necessary.
The kidney plays a vital role in acid–base homeostasis and several studies have established the involvement of a vacuolar H+-ATPase in acid–base regulation (Madsen and Tisher 1984; Bastani et al. 1991; Sabolic et al. 1997). Profound adaptive changes in the immunocytochemical distribution of H+-ATPases in the medullary collecting duct have been observed after acidosis. H+-ATPase labeling in acidotic rats was shifted from cytoplasmic vesicles to apical plasma membranes, whereas in alkalotic rats cytoplasmic vesicle staining was enhanced and labeling of the plasma membrane disappeared (Bastani et al. 1991). A significant finding of the present study is the demonstration that similar adaptive processes could also occur during acidosis in other epithelia, such as the submandibular gland.
