Abstract
We describe an enzyme histochemical technique for the simultaneous demonstration of acetylcholinesterase (AChE) and monoamine oxidase (MAO) (Types A, B, or A+B) in fixed-frozen sections. Several regions in the mesencephalon and brainstem were examined for both somatic and neuropil labeling. The results obtained are equivalent or superior to those obtained using previous methods for the individual localization of these enzymes. The simultaneous visualization of AChE and MAO in the same section allows the relationship of the two enzymes to be easily assessed with brightfield microscopy.
Keywords
B
We describe here a comparatively simple enzyme histochemical method with which to co-localize AChE and monoamine oxidase (MAO). Of significant value is the ability of this method to distinguish among serotonergic (MAO-B-positive) and noradrenergic (MAO-A-positive) nuclei and to enable simultaneous viewing of the distribution of AChE and MAO with brightfield microscopy.
Materials and Methods
Tissue was obtained from adult male Long-Evans hooded rats, Dutch-belted rabbits, ferrets, and mongrel cats, in accordance with Institutional Animal Care and Use Committee (IACUC) guidelines. After a lethal injection of sodium pento-barbital (125 mg/kg IP), rats were perfused transcardially with 100 ml room-temperature PBS (pH 7.4) for 2 min, 100 ml cold (3–5C) 1% paraformaldehyde-1% glutaraldehyde fixative in 100 mM sodium phosphate buffer, pH 7.4 (referred to here as phosphate buffer) for 2 min, 400 ml cold fixative at a slower rate for 20 min, and finally with 100 ml cold phosphate-buffered sucrose (15% w/v, pH 7.4) for 2 min. Perfusion volumes were adjusted appropriately for the other species. The brains were immediately removed, blocked, and immersed overnight in phosphate-buffered sucrose with 0.2% paraformaldehyde (pH 7.4) at 3–5C. Serial sections were cut at 50 μm on a freezing microtome, collected in cold phosphate buffer, and processed immediately after cutting while floating freely in compartmentalized staining nets.
Enzyme Histochemistry
To determine the possible effects of one enzyme histochemical procedure on the reaction product of the other, sections processed for either AChE or MAO alone were compared with a third set of sections processed for double labeling. Typically, sections received the following treatment. (a) Two sets of alternate sections were first processed for MAO activity by the nickel-intensified/coupled-peroxidase method (Kitahama et al. 1984, 1986) in the same media. (b) One set of MAO-labeled sections was then transferred to the compartmentalized staining net containing the third, unlabeled set of sections. (c) These two sets of sections (one for double labeling and one for AChE only) were then processed for AChE activity by the post-coloring copper ferricyanide version of Koelle's method (Koelle and Friedenwald 1949) developed by Tsuji (1974) for electron microscopy. By incubating sections together, the single-labeled sections served as controls for the double-labeled sections. Additional controls were also performed using inhibitors for the various enzymes.
Method for MAO Localization
Tissues processed for total MAO activity (Types A+B) received the following treatment: (a) sections were rinsed three times for 1 min each in phosphate buffer, (b) preincubated for 15 min in 50 mM Tris-HCl buffer at pH 7.6, (c) incubated for 30 min to 9 hr at 22–25C in darkness with agitation at 1-hr intervals (note: sections must not be allowed to overlap or become folded because this significantly reduces contact of the section with the incubation medium), (d) rinsed three times for 1 min each in phosphate buffer, and (e) mounted the following day from phosphate buffer onto chromalum-gelatin-subbed slides and allowed to air-dry overnight. The MAO incubation medium consisted of 0.02% tyramine HCl (Sigma Chemical; St Louis, MO, except where otherwise noted), 0.005% diaminobenzidine-HCl, 0.05% horseradish peroxidase (Type II), 0.065% sodium azide (Al-drich Chemical; Milwaukee, WI), and 0.3% nickel sulfamate (Aldrich) in 50 mM Tris-HCl buffer, pH 7.6. Sections labeled for MAO-A activity received 0.1 μM (1/2500 v/v of a 250-μM stock solution) L(-)-deprenyl HCl (a selective inhibitor of MAO-B; Fowler et al. 1981), and those labeled for MAO-B activity received 0.1 μM (1/2500 v/v of a 250-μM stock solution) clorgyline-HCl (a selective inhibitor of MAO-A; Fowler et al. 1981) added to the preincubation and incubation media. Control sections were treated identically except for the addition of both deprenyl and clorgyline to the preincubation and incubation media. MAO inhibitors were obtained from Research Biochemicals (Natick, MA).
Method for AChE Localization
Tissues processed for AChE activity received the following treatment: (a) sections were rinsed seven times for 1 min each in distilled deionized water, (b) preincubated for 15 min in 100 mM sodium acetate buffer, pH 5.2 (referred to here as acetate buffer) with 20 μM tetraisopropyl pyrophosphoramide (iso-OMPA), (c) incubated for 30 min to 2 hr at 22–25C or 37C with constant agitation, (d) rinsed seven times for 1 min each in distilled deionized water, (e) postincubated for 15 min in coloring solution, (f) rinsed seven times for 1 min each in distilled deionized water, and (g) mounted the following day from water or gelatin-alcohol solution onto chromalum-gelatin-subbed slides and allowed to air-dry overnight. The AChE incubation medium consisted of 4 mM acetylthiocholine iodide (ASChI), 2 mM copper, 10 mM glycine, and 20 μM iso-OMPA (also called tetra[monoisopropyl] pyrophosphortetramide, a selective inhibitor of non-acetyl cholinesterases) (Koelle et al. 1974; Aldridge 1953; Austin and Berry 1953) in acetate buffer. The incubation medium was prepared no more than 30 min before use, usually during the preincubation period, by combining a copper/glycine solution (cupric sulfate and glycine free base in 1/4 of the final volume of acetate buffer) and a solution containing the ASChI and iso-OMPA (1/2500 v/v from a 50-mM stock solution) in 3/4 of the final volume of acetate buffer. The incubation medium was then preheated to 37C (22–25C incubations excluded) and the final pH adjusted to 5.2. The postincubation coloring solution consisted of 3% potassium ferricyanide in acetate buffer. Control sections were treated identically except for the addition of 100 μM (1/2500 v/v 250 mM stock solution) 1,5-bis[4-allyldimethylammoniumphenyl]pentan-3-one dibromide [BW284c51 (also called BW297c50), a selective inhibitor of AChE] (Koelle 1955; Fulton and Mogey 1954; Austin and Berry 1953) to the preincubation and incubation media. Because non-somatic AChE staining was very intense in some neural regions, it was necessary in some cases to pretreat the animal with 0.6 mg/kg IM di-isopropyl fluorophosphate (DFP, an inhibitor of acetyl and non-acetyl cholinesterases) (Hawkins and Mendel 1947) 5.5 hours before sacrifice to reduce non-somatic AChE staining (Butcher 1978). Therefore, pretreatment with DFP facilitated visualization of AChE-positive somata that would normally have been obscured by intense neuropil staining. In addition, DFP pretreatment allows those AChE-positive somata to be classified as rapidly synthesizing AChE, a criterion that has been used in the identification of cholinergic somata (Mizukawa et al. 1986; Butcher and Woolf 1984; Mesulam et al. 1984; Satoh et al. 1983).
All mounted sections were dehydrated in graded alcohols (15 min each in 70%, 90%, 100%, 100%), cleared in xylene (three times for 30 min), and coverslips applied with DPX mountant (BDH, Poole, UK, imported by Gallard-Schlesinger Industries; Carle Place, NY).
Results
The method was evaluated for its suitability in several experimental applications: (a) the simultaneous visualization of AChE and MAO in neuronal somata, (b) the determination of MAO-A- and MAO-B-positive cell populations paired with the simultaneous demonstration of AChE, and (c) the simultaneous visualization of non-somatal AChE and MAO staining. Appropriate controls were also performed to evaluate the potential effects on one another of the two staining reactions. Because the MAO procedure was always performed before AChE histochemistry, MAO-stained sections were evaluated for the possible loss of staining product due to the subsequent AChE procedures. As shown in Figures 1A and 1B, MAO staining was not affected by subsequent reactions required for visualization of AChE activity. In addition, AChE-stained sections were evaluated for the possible interference in staining by the preceding MAO procedure. As shown in Figures 1D and 1E, AChE staining was not affected by the prior reactions required to visualize MAO. Furthermore, single-labeled control sections treated with MAO (i.e., deprenyl and clorgyline) or cholinesterase inhibitors (i.e., iso-OMPA and BW284c51), as well as the double-labeled control sections treated with all four inhibitors, were completely devoid of staining (Figure 1C). This suggests that the staining for both MAO and AChE activity was specific when performed either individually or in combination.
Double Labeling
Simultaneous visualization of AChE and MAO labeling of somata and neuropil (in the same section), using the double-labeling protocols outlined in Materials and Methods, is shown in Figure 2. Because the copper ferrocyanide reaction product of similar AChE procedures has been shown to have peroxidase-like activity (Tago et al. 1986), there was the possibility that the copper ferricyanide reaction product could cause nonspecific staining if it was performed before the MAO procedure. Therefore, the MAO procedure was always performed first in the double-labeling experiments. The low-power photomicrograph of a section through the dorsolateral aspect of the pontine tegmentum of the rat shown in Figure 2A reveals the distribution of AChE- and MAO-A-positive somata within the locus coeruleus and the adjacent mesencephalic nucleus of the trigeminal nerve. The presence of somatal profiles containing a reddish-brown, homogeneously distributed precipitate indicates AChE-labeled neurons, whereas the black granular deposits reveal the presence of MAO-A neurons. Both the color and the nature of the reaction products produced by this double-labeling technique allow each marker to be simultaneously co-localized within individual neurons. A prominent delineation is also evident between those neurons double labeled for AChE and MAO-A in the locus coeruleus and those that are single labeled for AChE in the mesencephalic nucleus of the trigeminal nerve. The region outlined in Figure 2A, which is shown at higher magnification in Figure 2A', shows that double-labeled somata can be distinguished within the same section.
An adjacent section of the dorsolateral pontine tegmentum, shown in Figure 2B, illustrates the absence of MAO-B (i.e., no black, granular reaction product) in locus coeruleus sections double labeled for AChE and MAO-B. However, the distribution of reddish-brown AChE-positive somata in locus coeruleus is similar to the MAO-A/AChE distribution seen in Figure 2A. Note that several black MAO-B-positive somata can be seen to the left of the locus coeruleus. Such somata were usually in close approximation to blood vessels.
Simultaneous visualization of AChE and MAO labeling was also observed in other brainstem loci (Figures 2C and 2D) in tissue obtained from the same animal and reacted at the same time as that shown in Figures 2A and 2B. Low-power photomicrographs of the region around the dorsal raphe and paradorsal raphe nucleus reacted for AChE and MAO-B activity (Figure 2D) reveal many somatal profiles containing both the reddish-brown AChE precipitate and the black granular deposits of MAO-B. The area outlined in Figure 2D, which is shown at higher magnification in Figure 2D', illustrates double- and single-labeled somata within the same section.
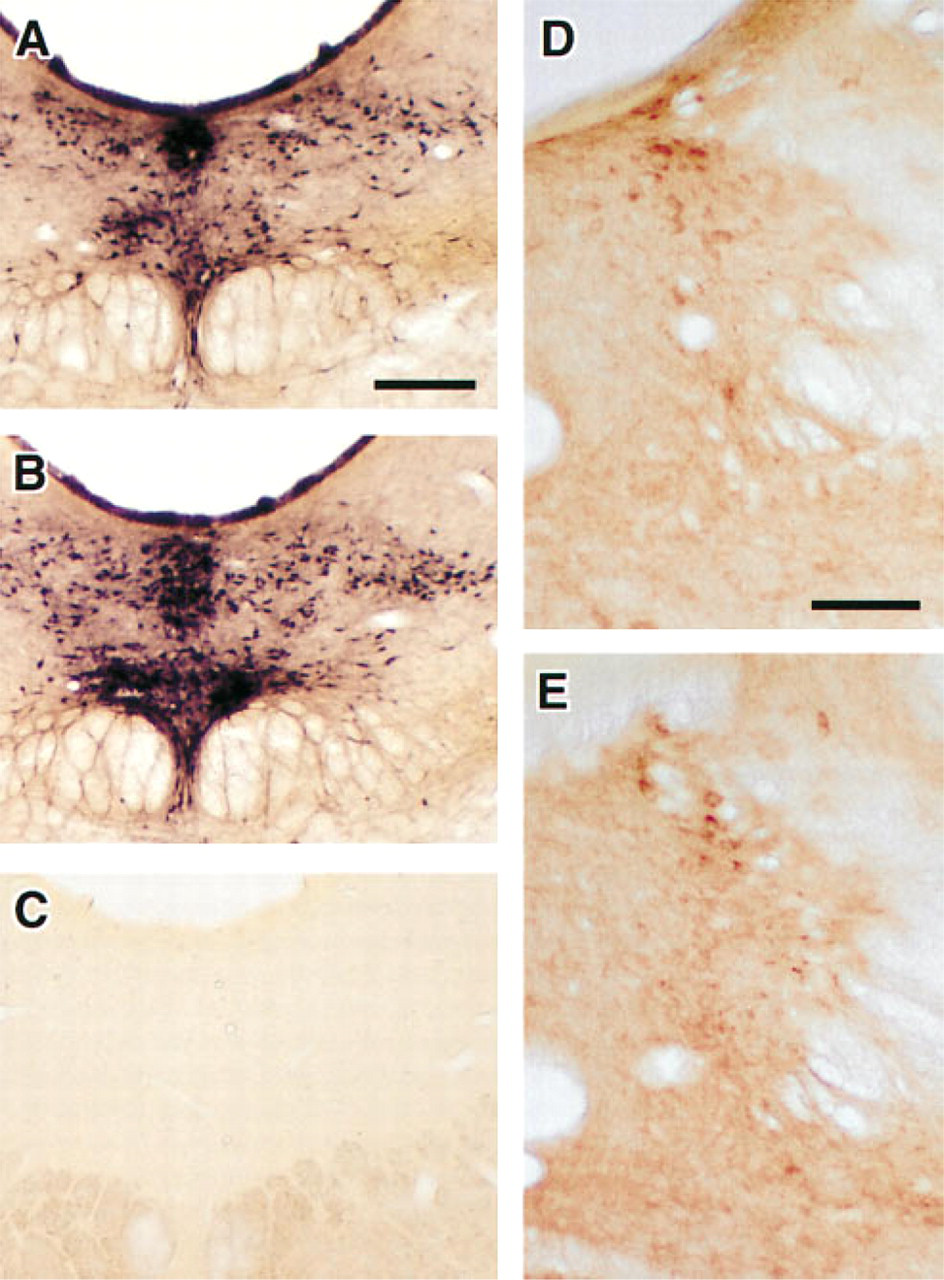
Absence of crossreactivity in MAO and AChE histochemical reactions.
In addition to the somatal staining described above, it was also feasible with the present technique to simultaneously visualize neuropil labeling. In the deep layers of the superior colliculus, where afferents, efferents, and various neurochemical markers are organized as a mosaic of discontinuous patchy domains (Graybiel and Illing 1994; Illing and Graybiel 1984, 1994; Huerta and Harting 1984), regions containing dense neuropil labeling for AChE and MAO were readily apparent (Figure 2E). In this case, the MAO visualized was for total MAO (Types A+B), and AChE staining was performed without DFP pretreatment. Some domains positive for AChE (closed arrowheads) and MAO (open arrowheads) were complementary, whereas others appeared to be overlapping (open/closed arrowheads). Observations of this material at higher magnifications (not shown) suggest that it is possible to resolve individual axonal and/or dendritic profiles.
Discussion
The results obtained here using a single-step double-labeling method revealed that many neurons in locus coeruleus that reacted positively for MAO-A also stained positively for AChE. These results are consistent with those previously obtained with the two-step double-labeling localization process (Albanese and Butcher 1979; de la Torre and Surgeon 1976) and with previous studies of the distribution of AChE by enzyme histochemical techniques (Caffe 1994; Ennis and Shipley 1992; Mizukawa et al. 1986; Paxinos and Watson 1986; Butcher and Woolf 1984; Mesulam et al. 1984; Satoh et al. 1983; Albanese and Butcher 1979; Lewis and Schon 1975), and of MAO-A by enzyme histochemical (Kitahama et al. 1986, 1994; Konradi et al. 1989; Willoughby et al. 1988; Arai et al. 1986) and immunohistochemical (Kitahama et al. 1994; Konradi et al. 1988; Thorpe et al. 1987; Westlund et al. 1985, 1988, 1993) techniques. The distinct color and nature of the reaction products produced by the present double-labeling technique allow both markers to be simultaneously co-localized within individual neurons. To our knowledge, only one other study has been done using similar techniques to co-localize AChE and MAO. However, this study (see Nakamura et al. 1993) did not produce the clarity of staining possible with the present technique.
The clarity and specificity of simultaneous AChE/MAO labeling are approximately equivalent to those seen when each of these procedures was used independently. Consequently, despite the extensive use of heavy metals and phosphates in the MAO procedure and the observation that these significantly impair AChE staining (van Ooteghem and Shipley 1984), no degradation of AChE labeling was observed. Although it is not immediately evident why the expected interference in AChE labeling was not apparent, one possibility is that the metals are rendered inactive by binding to the reaction products in the MAO procedure and that a sufficient amount of phosphate is removed by the washes to eliminate any inhibitory effects they might have on the subsequent AChE procedure. Regardless of the specific factors that allow the combined AChE/MAO-labeling paradigm to work, it has proved to be an extremely effective means for examining the distribution of these enzymes simultaneously in the neuropil and among individual somata. Furthermore, the results indicate that the method is applicable in different structures and species.
There are several obvious advantages of the technique presented. One is its usefulness in discriminating between MAO-A and MAO-B. As noted earlier, this ability can be utilized to distinguish noradrenergic cell populations that exhibit enhanced MAO-A activity from serotonergic cell populations that exhibit enhanced MAO-B activity. In addition, with regard to neuropil labeling, the ability to localize each of the MAO subtypes may prove useful for distinguishing putative monoaminergic terminal from non-terminal labeling. Because MAO-A is believed to be neuronal in origin (Westlund et al. 1985, 1993; Student and Edwards 1977), whereas MAO-B is more likely extraneuronal (e.g., glial) (Konradi et al. 1989; Westlund et al. 1985; Levitt et al. 1982; Student and Edwards 1977) except in serotonergic nuclei, the present method could be used to distinguish between these elements in the neuropil. Moreover, because both AChE and MAO reaction products are electron-dense, they can be viewed by electron microscopy (Maeda et al. 1987; Tsuji and Fournier 1984; Tsuji 1974). Another advantage of the method is its ease of use. It does not require animal decapitation or fresh-frozen sections, and the reaction products are non-labile and can be viewed simultaneously with brightfield microscopy. Because of the low cost and ease of use of the method, it is well suited to the routine processing of archival materials and/or large-scale comparative studies.
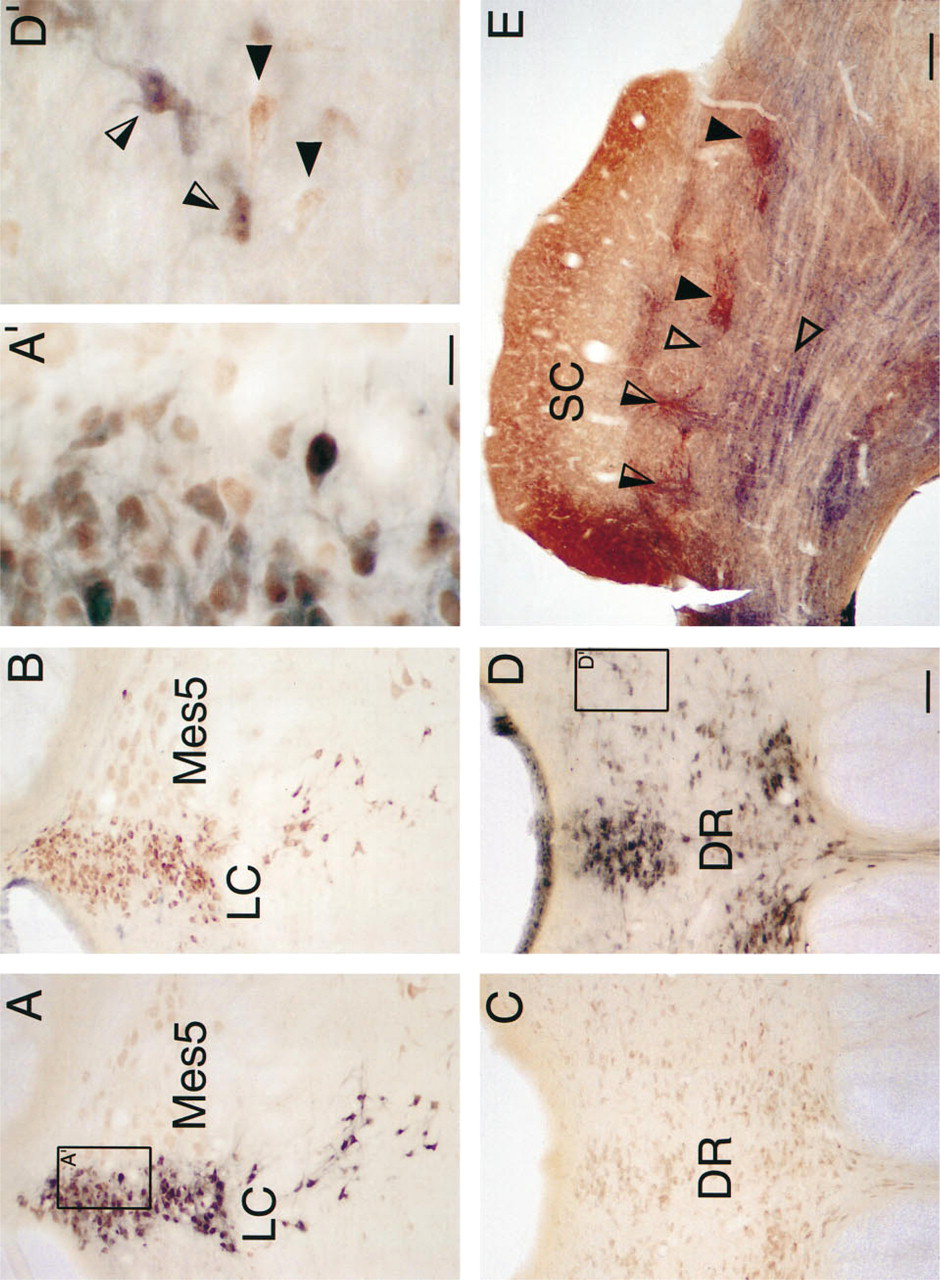
Simultaneous visualization of AChE and MAO labeling in various regions of the rat CNS.
A negative aspect of this technique for co-localization is that the staining products for the two enzymes may obscure each other. This can sometimes lead to the inability to unequivocally identify double-labeled somata. Immunohistochemical methods tend to give more discrete localization and are therefore more likely to give unequivocal identification of double-labeled somata. Because antibodies for AChE, MAO-A, and MAO-B have recently become available from a variety of sources (e.g., Accurate Chemical & Scientific, Westbury, NY, and Chemicon International, Temecula, CA), immunohistochemical double labeling should be possible. However, enzyme-labeled immunohistochemical techniques suffer from the same diffusion artifact problem that enzyme histochemical techniques do, and immunofluorescent techniques would still be required to achieve unequivocal identification of double-labeled somata. The need for specialized equipment to view double-labeled immunofluorescence and the increased expense of immunohistochemical reagents needed to achieve this level of accuracy for identification of double-labeled somata must therefore be considered. Another shortcoming of the present method is that when DFP pretreatment is not possible (e.g., human postmortem specimens), visualization of AChE-positive somata may be obscured by non-somatal staining. Under these circumstances, immunohistochemical (Mesulam et al. 1994) or alternative enzymatic methods (Kujat et al. 1993; Schatz et al. 1992; Kugler 1987; Tago et al. 1986; Hedreen et al. 1985) for AChE staining may be preferable when coupled with other methods of MAO staining (e.g., Nakos and Gossrau 1993; Gossrau and Richter 1992).
The results of this study are in agreement with previous studies showing that AChE is found in cholinergic and non-cholinergic neurons (Mizukawa et al. 1986; Butcher and Woolf 1984; Mesulam et al. 1984; Eckenstain and Sofroniew 1983; Satoh et al. 1983). In recent years, AChE has been associated with non-cholinergic functions within the central nervous system, e.g., degradation of amides (Checler et al. 1994), neuronal differentiation and development (Coleman and Taylor 1996; Jones et al. 1995; Layer and Willbold, 1994, 1995), and interactions with excitatory amino acid (Appleyard 1994) and monoaminergic (Abo et al. 1992; Ennis and Shipley 1992) neurotransmitters. The relationship of non-cholinergic AChE to monoamines has been best studied in the dopaminergic neurons of the substantia nigra (Greenfield 1991, 1995), and the method described here may prove particularly helpful in examining such functions in noradrenergic and serotonergic nuclei.
Footnotes
Acknowledgements
Supported by NIH grants NS 22543, EY 06562, and NS 35008.
