Abstract
Using antibodies highly specific for L-arginine and L-citrulline, we localized these amino acids in rat kidney with immunohistochemical methods. Highest levels of arginine immunoreactivity were observed in epithelial cells of proximal tubules in the outer stripe of the outer medulla and the collecting ducts in the cortex. Staining intensity of proximal convoluted tubules in the outer stripe decreased from the inner side to the outer side. In the inner medulla, collecting ducts were labeled with moderate intensity. Staining within the cortex was apparent only with collecting ducts. Citrulline immunoreactivity was localized in the epithelial cells of collecting ducts both in the cortex and medulla. Immunoreactivity was also found in glomerular podocytes and in the epithelial cells of proximal convoluted tubules in the outer medulla. These localizations were different from those of other amino acids previously reported. The precise cellular distribution of arginine and citrulline in rat kidney was determined for the first time by an immunohistochemical method in the present study.
B
Materials and Methods
Animals
Experiments were performed using 2-month-old male rats of the Sprague-Dawley strain that were bred in our laboratory. They were maintained in an animal room at a temperature of 24 ± 2C with a relative humidity of 55 ± 10% and a light/dark cycle of 12 hr. Rats were allowed free access to a commercial diet (NMF; Oriental Yeast, Tokyo, Japan) and tapwater.
Tissue Preparation
For the immunohistochemical demonstration of L-arginine and L-citrulline, the rats were perfused via the heart with a mixture of 1% GAL, 4% paraformaldehyde, 0.2% picric acid, and 2% sucrose in 0.1 M sodium acetate buffer, pH 6.0 (Schmechel et al. 1980). After perfusion, kidneys were removed and immersed in the same fixative for 4–5 hr. Then they were rinsed several times with 50 mM Tris-HC1 buffer, pH 7.4, dehydrated with a graded alcohol series, and embedded in paraffin. Seven-μm-thick sections were mounted on albumin-coated slides.
Antibodies
The preparation and specificity of anti-L-arginine and anti-L-citrulline antibodies were the same as reported previously (Aoki et al. 1991). In brief, L-arginine and L-citrulline were coupled with guinea pig serum albumin and rabbit serum albumin, respectively, via glutaraldehyde. Each amino acid conjugate was emulsified with an equal volume of complete Freund's adjuvant and repeatedly injected intracutaneously into multiple sites on the backs of guinea pigs or rabbits. Each antibody was purified by affinity chromatography with the respective amino acid immobilized on formyl cellulofine (Seikagaku Kogyo; Tokyo, Japan). Specificities of the purified antibodies were examined by a dot immunobinding assay (Hawkes et al. 1982). Reactivity of the antibodies was studied against L-citrulline-, L-arginine-, L-ornithine-, L-aspar-tate-, L-glutamate-, GABA-, L-glycine-, L-taurine-, L-leucine-, L-isoleucine-, L-valine-, and β-alanine-albumin complexes produced as described previously (Aoki et al. 1991). The purified anti-arginine and anti-citrulline antibodies were found to be specific to the L-arginine- and L-citrulline-albumin complex, respectively (Figure 1).
Immunohistochemical Processing
Sections were deparaffinized in xylene, dehydrated through a graded alcohol series, and washed twice in 50 mM Tris-HCl buffer containing 500 mM NaCl, pH 7.6. Sections were incubated with the anti-arginine (0.5 μg/ml) or anti-citrulline (0.2 μg/ml) antibody, left overnight at room temperature, and followed by an immunohistochemical procedure using the peroxidase-anti-peroxidase method (Sternberger 1974). 3,3' Diaminobenzidine tetrahydrochloride (Dojindo Chemical Institute; Kumamoto, Japan) was used as chromogen, which had been freshly prepared as a solution of 20 mg in 100 ml Tris buffer (50 mM, pH 7.6) that contained 0.01% H2O2. Control sections incubated with non-immune serum showed no positive staining.
Results
L-Arginine Immunoreactivity
Low-power magnification of the section demonstrated that L-arginine immunoreactivity is predominantly located in the outer stripe of the outer medulla (Figure 2A). Higher magnification revealed it to be localized in the cells of the proximal tubules and the collecting ducts (Figure 2C). Staining intensity of proximal convoluted tubules in the outer stripe of the outer medulla decreased from the inner side to the outer side (Figures 2A and 2C). Staining within the cortex was apparent exclusively in collecting ducts. Immunopositive cells of these collecting ducts exhibited a characteristic mosaic-like pattern, with some of the epithelial cells being immunoreactive for arginine whereas other cells were immunonegative (Figure 2B). Glomeruli and proximal and distal tubules in the cortex were not stained. Interestingly, proximal straight tubules and collecting ducts were not stained in the inner stripe of the outer medulla (Figure 2A). In the inner medulla, collecting ducts were labeled with moderate intensity (Figure 2D).
L-Citrulline Immunoreactivity
L-citrulline immunoreactivity was present in both the cortex and the medulla of the kidney (Figure 3A). In the cortex, the immunoreactivity was clearly localized in podocytes of the glomeruli, endothelial cells near the vascular pole, and endothelial cells of the collecting ducts (Figures 3B-3D). Immunostaining of the collecting ducts was more intense in about half of the cells than in the other half. Cells of proximal and distal tubules were not stained. In the outer and inner stripes of the outer medulla, immunoreactivity was observed in the medullary ray, epithelial cells of proximal convoluted and straight tubules, and collecting ducts (Figures 3E and 3F). Staining intensity in the collecting ducts was greater than that of proximal tubules. In the inner medulla, all cells of collecting ducts were strongly labeled (Figure 3G). Cells of the pelvic wall were also labeled intensely (Figure 3A).
Discussion
Immunohistochemical localization of some neutral and acidic amino acids (GABA, taurine, aspartate, and glutamate) were studied in the rat kidney (Ma et al. 1994; Trachtman et al. 1993; Párducz et al. 1992), and none of the distribution patterns of these amino acids was identical to any other. Therefore, it was of interest to determine how arginine and citrulline, basic amino acids, are distributed in rat kidney. Arginine and citrulline were found to be highly concentrated in the proximal tubules, in which none of the four amino acids studied previously had been proved. Consequently, the distribution of the individual amino acids examined to date appears to be specific for each amino acid.

Dot immunobinding assay of anti-arginine and anti-citrulline antibodies. Specificity of the antibodies was studied by a dot immunobinding assay on a nitrocellulose membrane. Rabbit or guinea pig serum albumin conjugates of various amino acids, including L-arginine (Arg), L-citrulline (Cit), L-ornithine (Orn), β-alanine (β-Ala), L-glutamate (Glu), L-aspartate (Asp), taurine (Tau), GABA, L-glycine (Gly), L-valine (Val), L-leucine (Leu), and L-isoleucine (Iso-leu) were applied to a nitrocellulose membrane and incubated with an affinity-purified antibody at a dilution of 1:1000. Antigen-antibody reactions were visualized by the peroxidase-anti-peroxidase method. Purified antibody gave a strong immunostaining only on the spot of the specific amino acid against which the antiserum had been raised (arrows).
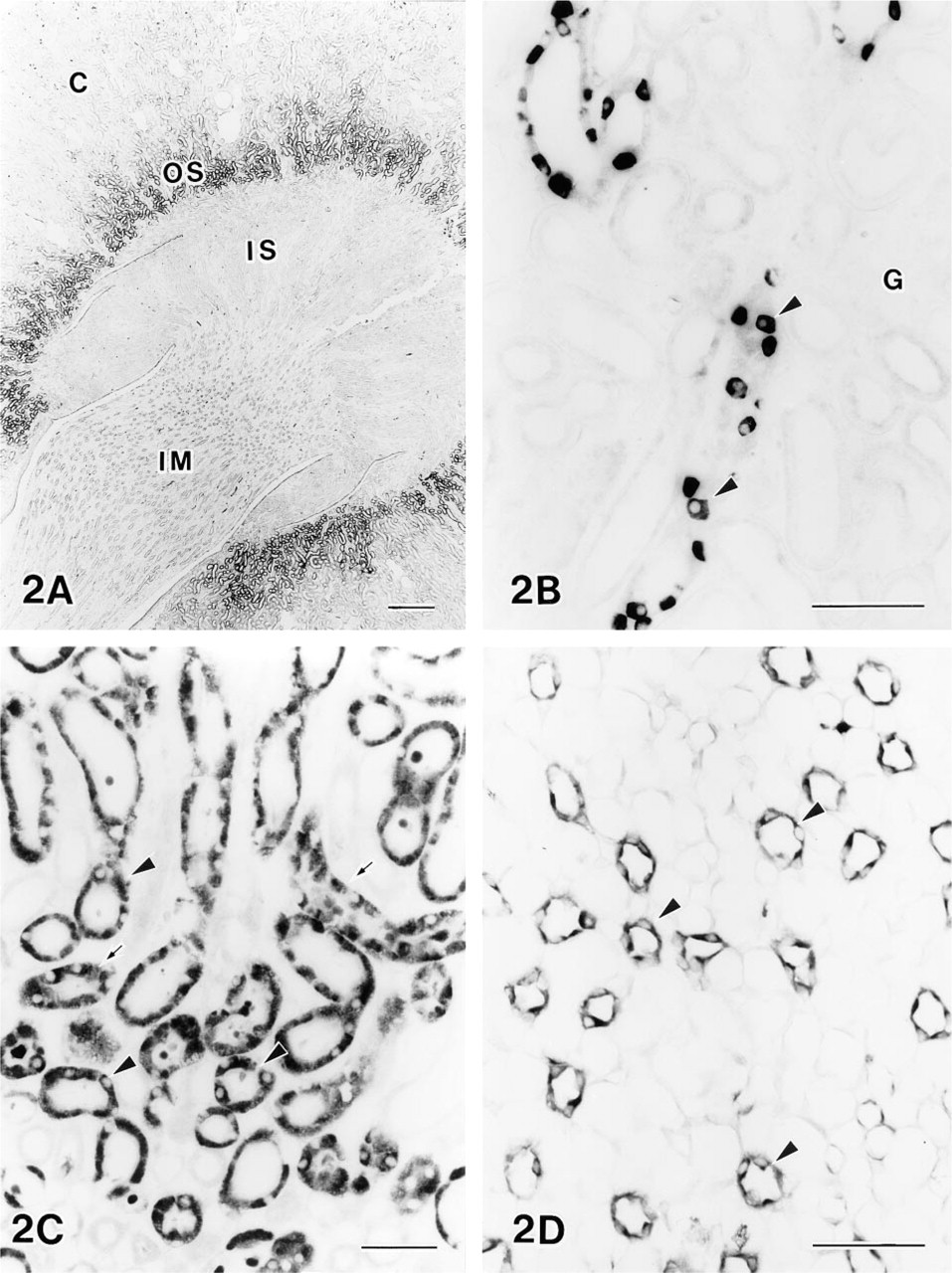
Photomicrographs showing the distribution of L-arginine immunoreactivity.
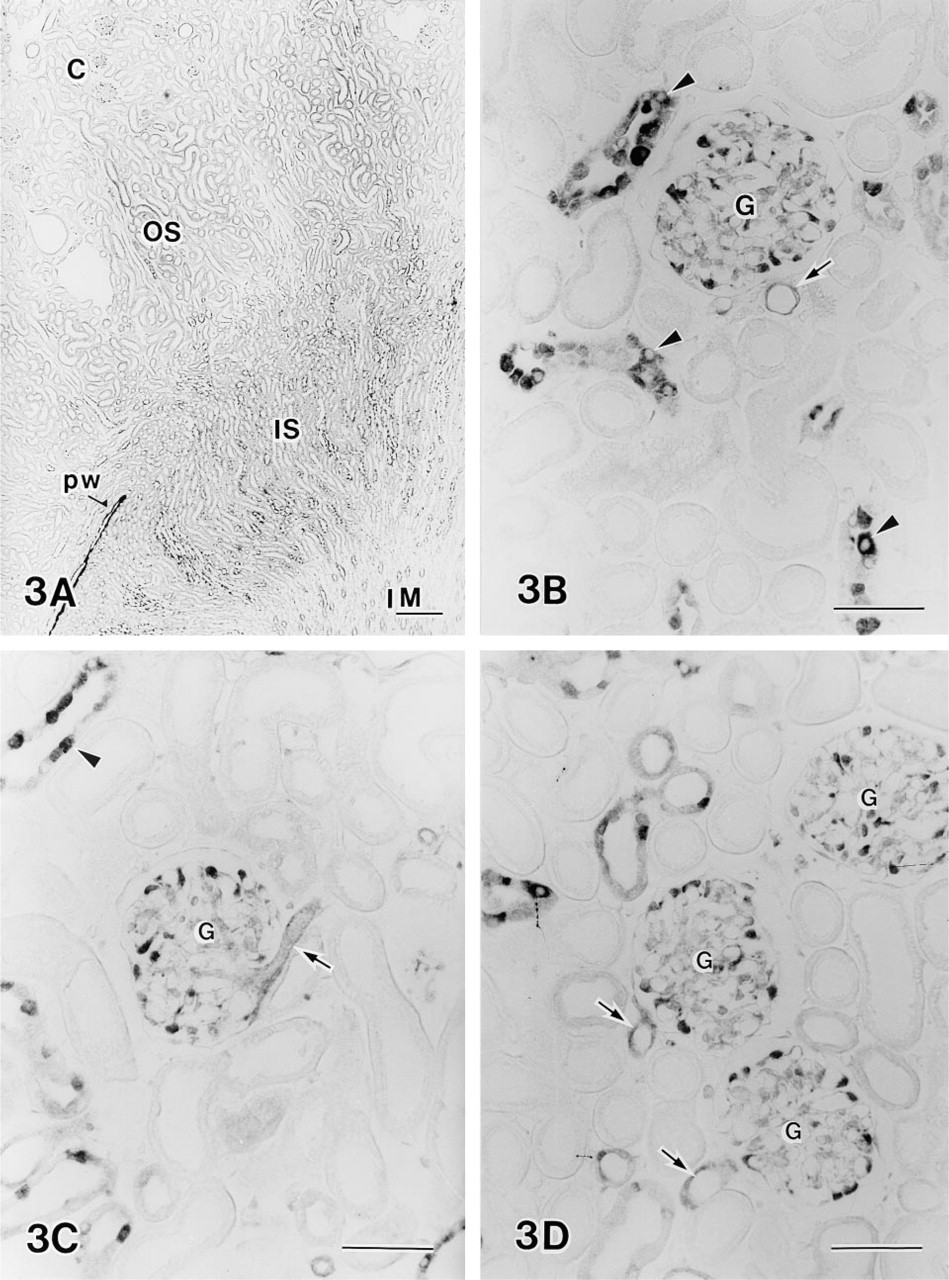
Immunohistochemical demonstration of L-citrulline in the rat kidney.
The kidney synthesizes a considerably large portion of endogenous arginine, and the proximal convoluted tubules have been suggested as the predominant site for arginine synthesis (Levillain et al. 1990,1993). By immunohistochemical and in situ hybridization studies, arginine-synthesizing enzymes and their mRNAs were also shown to be localized in proximal tubules (Morris et al. 1991). The localization of arginine immunoreactivity in proximal tubules, as revealed in the present study, is in good agreement with these reports. Arginine synthesized in the kidney is largely released into the renal blood flow and a portion of it is used within the kidney (Levillain et al. 1990). Dantzler and Silbernagl (1988) suggested that arginine is concentrated and recycled in the medulla. In the epithelium of the collecting duct, several amino acids were accumulated and their physiological significance in increasing the cellular osmotic pressure by acting as organic osmolytes has been proposed (Trachtman et al. 1988). Therefore, arginine in the collecting duct may be accumulated from the bloodstream or tubule fluid and may contribute to increasing the cellular osmotic pressure. Arginine in the collecting duct may also be used as the substrate for an enzyme that catabolizes arginine, NO synthase, as suggested by several reports (Markewitz et al. 1993; Stoos et al. 1992; Terada et al. 1992).
Citrulline immunoreactivity was localized in cortical and medullary collecting duct cells, proximal tubules in outer medulla, and glomerular podocytes. Arginase and ornithine aminotransferase, enzymes of the urea cycle that catabolize arginine to citrulline, were localized in the cortex and outer medulla (Levillain et al. 1993). Therefore, the localization of these citrulline-synthesizing enzymes is similar to that of the citrulline immunoreactivity revealed in the present study. Citrulline is also produced as the co-product of NO formation, and NO plays a role in activating guanylate cyclase. It has been shown that NO regulates solute transport in the cortical and inner medullary collecting duct (Stoos et al. 1992), and mRNAs for NO synthase and guanylate cyclase are expressed in both cortical and medullary collecting ducts (Markewitz et al. 1993; Terada et al. 1992). These reports are consistent with our results showing that citrulline immunoreactivity is present in collecting ducts.
In the collecting ducts, about half of the epithelial cells in the cortex and most cells in the inner medulla were immunoreactive to both anti-arginine and anti-citrulline antibodies. Two types of cells, principal cells and intercalated cells, are known in the collecting ducts, and the latter cells gradually decrease from about 35% of the cell population in the ducts of the outer medulla to 10% in the inner medulla (Fawcett 1994). Therefore, the immunopositive cells are considered to be the principal cells.
Immunohistochemical demonstration of free amino acids was first achieved by Storm-Mathisen et al. (1983). Since that study, many amino acids have been successfully localized in various tissues by many laboratories (Ma et al. 1994; Storm-Mathisen and Ottersen 1990; Aoki et al. 1987,1988). In those studies, free amino acids in the tissue were fixed on tissue proteins by glutaraldehyde contained in the fixative and the antiserum against each amino acid was raised by immunizing animals with amino acid-glutaraldehyde-serum albumin complex. Antibody thus raised was reactive with the respective amino acid conjugated with glutaraldehyde but was not reactive to amino acid contained in the peptide chains of the serum albumin or other proteins. This was also the case in the present study, and although guinea pig serum albumin contains a lot of arginine in its peptide chain (in bovine serum albumin, 23 arginine residues are contained within 585 amino acids), a dot of the serum albumin was not immunostained with our anti-arginine antibody. Consequently, the antibody against arginine was considered to be reactive with a special form of arginine that is free in vivo and is immobilized by glutaraldehyde on proteins.
