Abstract
Co-localization of chromogranin (Cg) A, B, and C has been studied in different neuroendocrine cell types in histologically normal mucosa from human gastrointestinal tract (corpus, antrum, duodenum, ileum, and colon) using single-, double-, and triple-immunofluorescence stainings. Virtually all enterochromaffin (EC) cells contained CgA, and those in the luminal two thirds of the antral mucosa and villi of small intestine often also contained CgB. A few EC cells in the duodenal crypts contained CgC. Most gastrin cells harbored both CgB and CgA, although rather more CgB than CgA, but some gastrin cells contained all three types, i.e., also CgC. Some CCK cells also contained all three chromogranins. Enteroglucagon cells in the duodenal villi contained CgA and some CgB. CgA (but not B or C) was found in some secretin, GIP, enteroglucagon/peptide YY, and neurotensin cells. A few somatostatin cells contained CgA but neither CgB nor CgC. CgA and C were found mainly in the basal cell region, whereas CgB occurred more diffusely throughout the cytoplasm. This varying distribution suggests that not all secretory granules contain CgA, or that CgB may occur in a nongranular form. The varying composition of the different chromogranins may reflect their complex functional roles in the widespread neuroendocrine system.
T
The purpose of the present study was, by applying double- or triple-immunofluorescence staining of the same section, to ascertain the extent to which the three main chromogranins (A, B, and C) can occur in different endocrine cell types and to determine their intracellular distribution in relationship to the various neuroendocrine hormones in different parts of the human gastrointestinal tract.
Materials and Methods
Tissue specimens from adult human gastric corpus and antrum, proximal duodenum, distal ileum, and sigmoid colon were obtained from surgical samples removed at operations for adenocarcinoma. The specimens examined were taken at least 3 to 5 cm from the neoplasm in macroscopically normal mucosa. All were histologically normal.
Fixation of the specimens was either in neutral picric acid-formaldehyde (Stefanini et al. 1967) or in 10% buffered neutral formalin, for 18–20 hr at room temperature (RT), followed by embedding in paraffin. Sections 5 μm thick were cut and attached to poly-
The sections were stained with hematoxylin-eosin or were immunostained by different methods to demonstrate various secretory granule products. The sensitive streptavidin-biotin complex (ABC) technique (Hsu et al. 1981), with diaminobenzidine as chromogen, was applied as a single immunostain mainly to reveal the distribution of endocrine cell types in the respective gastrointestinal regions, as well as to perform the control stainings specified below.
In the co-localization studies, immunofluorescence methods were used in single, double, or triple staining. In triple-immunofluorescence staining, the procedure was as follows. The sections were incubated with a cocktail of antibodies [one monoclonal + two polyclonal (anti-rabbit and anti-guinea pig)], overnight incubation at RT → biotinylated goat anti-guinea-pig IgG, 30 min at RT → a mixture of fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit IgG + Texas Red (TXRD)-conjugated goat anti-mouse IgG + aminomethyl coumarin acetic acid (AMCA)-conjugated streptavidin, 30 min at RT. Double immunostaining was performed in the same way except for exclusion of one of the primary antibodies and corresponding secondary antibodies. Before applying the respective primary antibodies, the sections were incubated with nonimmune sera from the animal species producing the secondary antibodies, at a dilution of 1:10.
If two primary polyclonal antisera raised in the same species (rabbit) had to be used, the staining technique was modified as follows: first, primary anti-rabbit antiserum was applied overnight at RT → monovalent FITC-conjugated goat Fab anti-rabbit IgG (0.2 mg/ml), overnight at RT → second primary anti-rabbit antiserum, overnight at RT → biotinylated swine anti-rabbit IgG, 30 min at RT → TXRD-labeled streptavidin, 30 min at RT (adapted from Negoescu et al. 1994).
The Fab concentration of 0.2 mg/ml overnight was found to saturate the epitopes of the first-step anti-rabbit antibody.
The primary antibodies are characterized in Table 1.
Antisera used in this study
a Antisera raised in rabbit.
b Antisera raised in mouse (monoclonal).
c Antisera raised in guinea pig.
d Antisera raised in rat (monoclonal).
Polyclonal antibodies to CgA were raised against a fragment covering amino acids 116–439 of human CgA (Stridsberg et al. 1993). The CgA fragment was purified from urine from a patient with a carcinoid tumor. CgB and CgC were raised in rabbits against synthetic peptides (Stridsberg et al. 1995). The sequences were selected to be specific for the respective proteins. The selected amino acid sequence for CgB was amino acids 312–331 (Benedum et al. 1987) and for CgC, 223–249 (Gerdes et al. 1989), with an additional tyrosine residue at the N-terminal. The CgB and CgC peptides were both amidated at the C-terminal. The peptides were coupled to bovine serum albumin (BSA) with glutaraldehyde and injected into rabbits to produce polyclonal antibodies.
Gastrin antibodies raised against the C-terminal portion were used in antrum and N-terminal gastrin antibodies in the duodenum. The latter antibodies did not crossreact with CCK. The CCK antibodies used did not crossreact with gastrin.
The labeled secondary antisera were biotinylated swine anti-rabbit IgG (Dakopatts; Glostrup, Denmark), biotinylated goat anti-guinea pig IgG, TXRD- and AMCA-labeled streptavidin (Vector Laboratories; Burlingame, CA), TXRD-conjugated goat anti-mouse IgG (Southern Biotechnology Associates; Birmingham, AL), FITC-conjugated goat anti-rabbit IgG, both the monovalent Fab fragment and the whole molecule (Sigma Chemical; St Louis, MO).
The control stainings included (a) omission of the primary antisera, (b) replacement of the first layer of antibody by nonimmune serum diluted 1:10 and by the diluent alone, (c) preincubation (24 hr) of primary antiserum with the relevant antigen (0.5, 2.5, and 7 mmol per ml diluted antibody solution, respectively) before application to the sections. These control tests were performed with both immunofluo-rescence and ABC techniques.
The sections were examined in a Vanox AHBS3 fluorescence microscope (Olympus; Tokyo, Japan) equipped with filters (Olympus) giving excitation at a wavelength of 475–555 nm for TXRD (filter no. 32821, dichroic mirror BH2-DMG), 453–488 nm for FITC (no. 32822, BH2-DMIB), and 340–375 nm for AMCA (no. 32817, BH2-DMU), and a double-band filter set (no. 39538, BH2-DFC5) for simultaneous visualization of TXRD- and FITC-labeled cells was also used (excitation at 550–570 nm and 480–495 nm, respectively). Photographs were taken with Fujicolor 400 film; the triple stainings were photographed by double-exposing the film first through the double band and then with the AMCA filter sets.
Co-localization of two or three of the fluorochromes was revealed by the resulting additive colors: the co-localization of green and red is perceived as yellow, blue (AMCA) and red as magenta, blue and green as cyan, and all three fluorochromes as white.
Results
Distinctly immunoreactive (IR) cells were revealed by all tested antisera in the relevant regions of the gastrointestinal tract when ABC and immunofluorescence methods were used with both fixatives. In double- and triple-immunofluorescence stainings, omission of one or two of the primary antibodies gave a staining pattern corresponding to the remaining primary antibody or antibodies. The other staining controls were all negative.
Distribution of CgA, B, and C in the Gastrointestinal Tract
CgA-IR cells were observed in all parts of the gastrointestinal tract and were more numerous than CgB-and CgC-IR cells. They were seen at all levels of the mucosa, although mainly in the middle third portion. The staining intensity of the CgA-IR cells was strong and even in all regions except for the antrum, where it varied from strong to very weak; the weak IR cells predominated in the antrum.
CgB-IR cells were most numerous in the antrum, where they occurred at almost the same frequency as CgA-IR cells. In duodenum they were sparse, in the ileum they were sporadic, and in the corpus and colon they were virtually nonexistent. In the antral mucosa, CgB-IR cells were observed mainly in the midportion, in the regions of the small intestine in the various levels of the villi, but only occasionally in the crypts, where they were seen in the upper half. A few CgB-IR cells were also observed in Brunner's glands.
CgC-IR cells were few in number, being present mainly in the antrum, where they were seen in patches in the middle third of the mucosa. A few cells were present in the duodenum (mainly in the crypts, occasionally in the villi) but were completely absent in Brunner's glands and the other gastrointestinal regions.
Co-localization of CgA, B, and C in Endocrine Cells
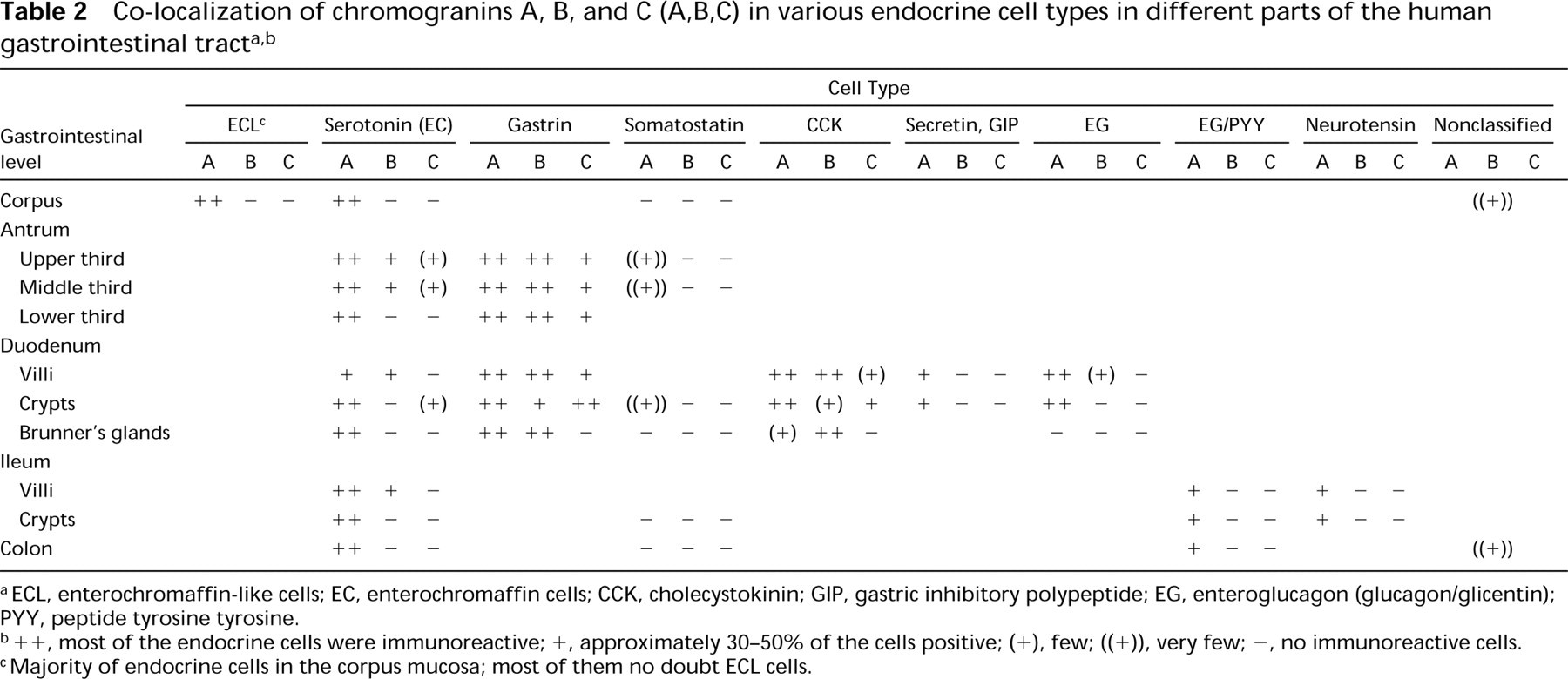
The results are summarized in Table 2.
Co-localization of chromogranins A, B, and C (A,B,C) in various endocrine cell types in different parts of the human gastrointestinal tracta,b
a ECL, enterochromaffin-like cells; EC, enterochromaffin cells; CCK, cholecystokinin; GIP, gastric inhibitory polypeptide; EG, enteroglucagon (glucagon/glicentin); PYY, peptide tyrosine tyrosine.
b ++, most of the endocrine cells were immunoreactive; +, approximately 30–50% of the cells positive; (+), few; ((+)), very few; −, no immunoreactive cells.
cMajority of endocrine cells in the corpus mucosa; most of them no doubt ECL cells.
Corpus. Serotonin (enterochromaffin, EC) cells but not somatostatin cells displayed CgA immunoreactivity. Most of the CgA-IR cells were probably entero-chromaffin-like (ECL) cells, a type predominating in this region but which could not be identified immunocytochemically because available histamine antibodies are unsuitable for use with formalin-fixed tissue.
Antrum. All EC cells were CgA-immunoreactive, and a subpopulation thereof also contained either CgB or CgC. Those cells harboring CgB and the few with CgC were located in the middle and upper parts of the mucosa. Only occasionally did the antral EC cells display immunoreactivity for all three chromogranins.
Most gastrin cells were immunoreactive for CgB and a fraction of them also for CgA, but a few contained all three chromogranins. CgA immunoreactivity in gastrin cells varied in intensity, being weak in most cells and sometimes virtually absent.
Immunoreactivity for CgB was stronger than for CgA or C (Figures 1A-1D and 2). A few gastrin cells were also serotonin-reactive and somatostatin cells were occasionally CgA immunoreactive (Figure 3).
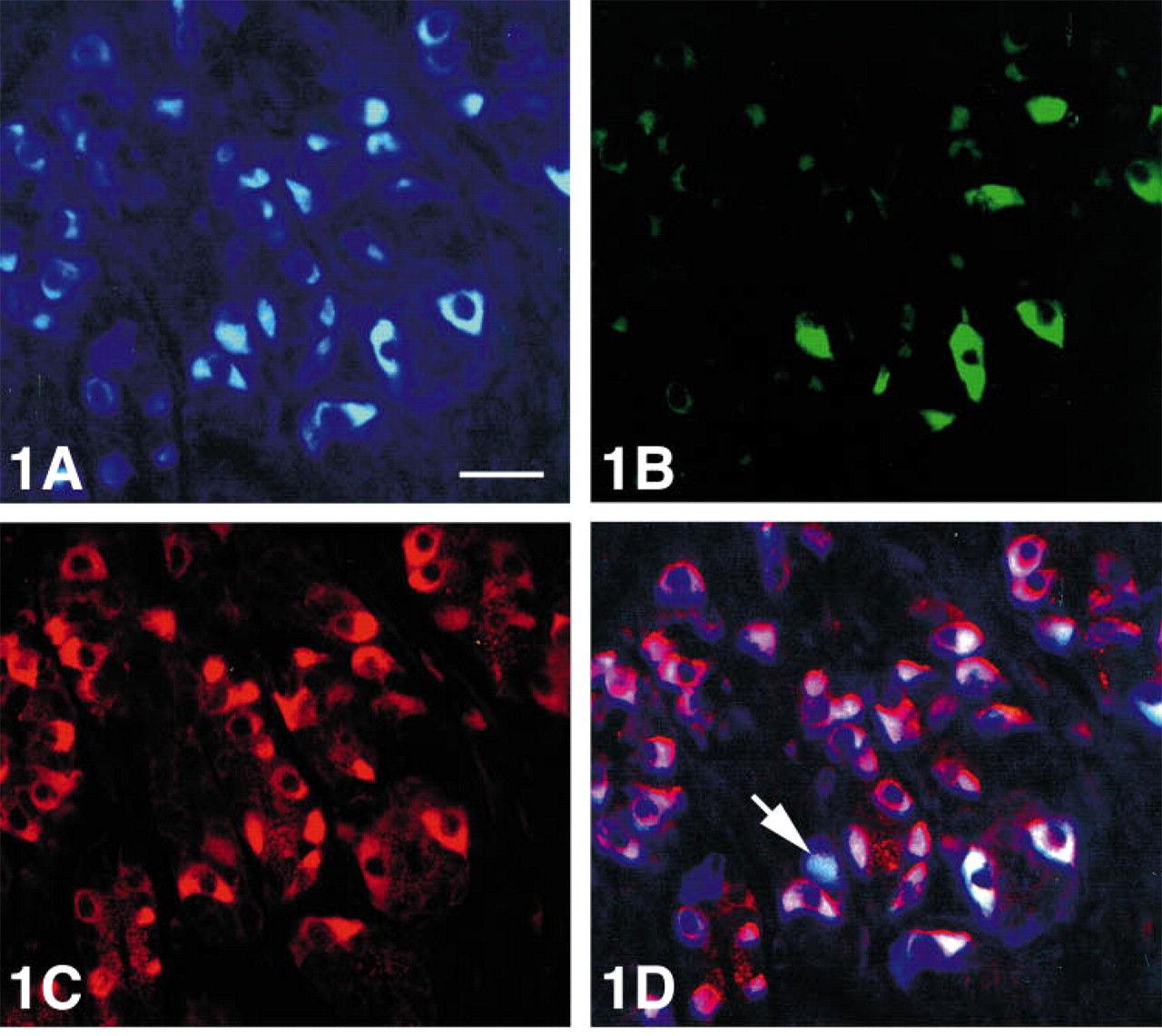
Human antral mucosa triple stained for (
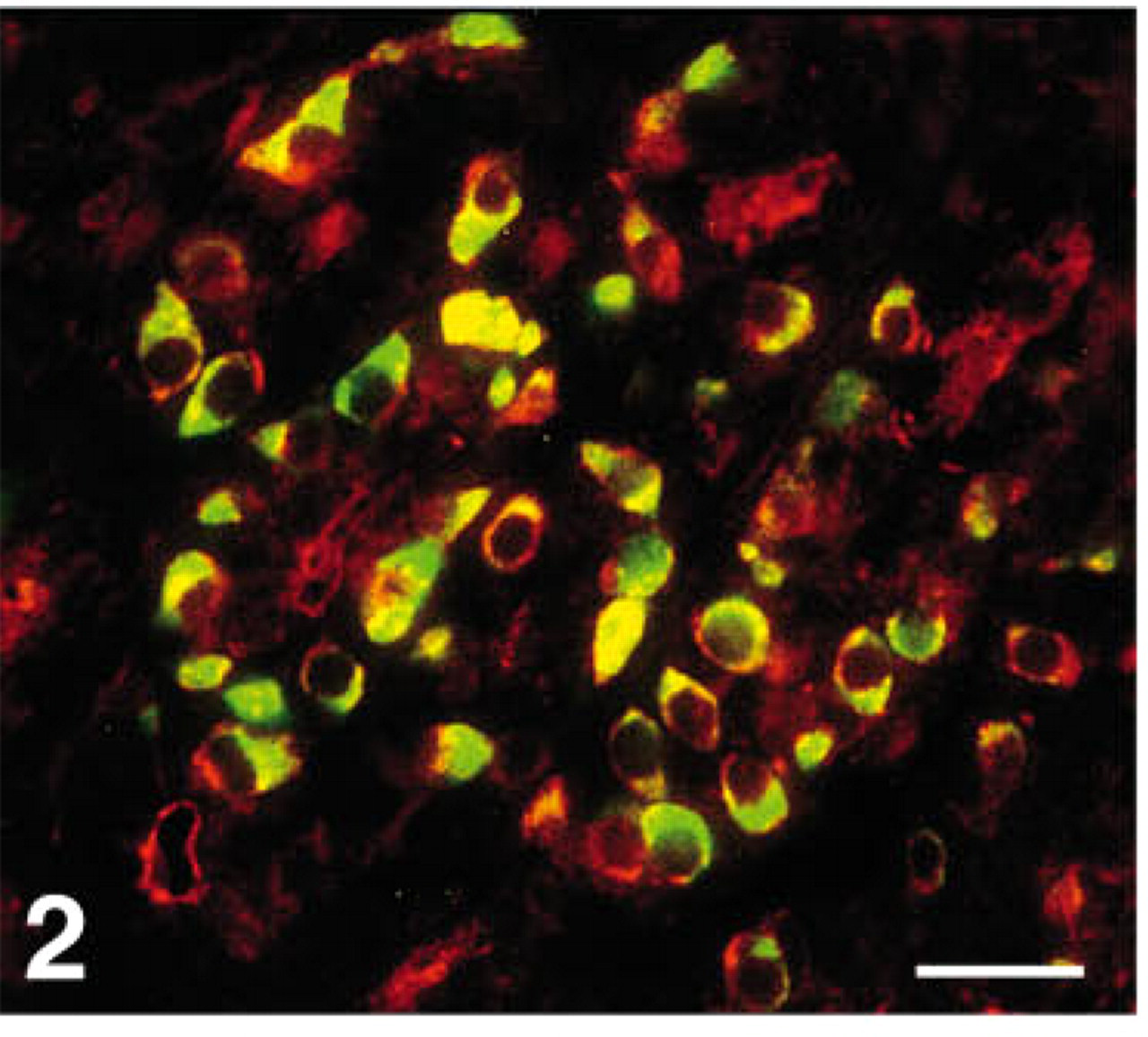
Human antral mucosa double stained for gastrin (FITC) and chromogranin C (Texas red). Most of the immunostained cells show co-localization of these two substances (yellow). This co-localization is mainly seen in the basal region of the gastrin cells, whereas only gastrin immunoreactivity (green) occurs in the entire cytoplasm. In a minority of the gastrin cell population, chromogranin C immunoreactivity (red) appears to occur alone in parts of the cytoplasm. Bar = 27 μm.
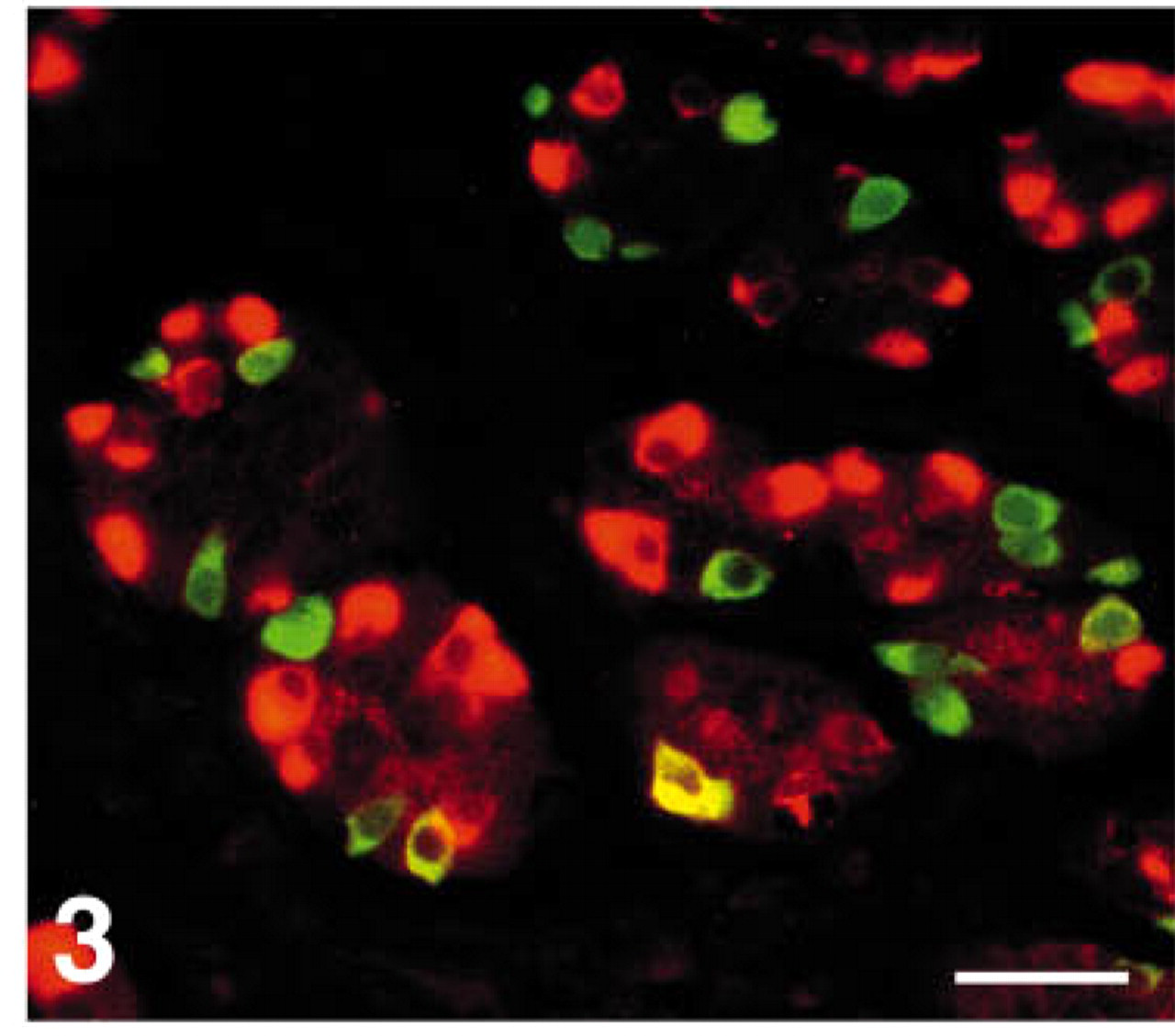
Human antral mucosa double stained for somatostatin (FITC) and chromogranin A (Texas red), showing distinct co-localization of these two substances in only one cell (yellow). Bar = 27 μm.
Duodenum—Mucosa. The EC cells located in the crypts were CgA-IR, but only some were located in the villi. CgB immunoreactivity was observed in a subpopulation of EC cells in the villi, but not in EC cells in the crypts (Figures 4A and 4B); occasional EC cells in the crypts contained CgC. Gastrin and CCK cells reacted to CgB and CgA antibodies, and some also to CgC. Some secretin and GIP cells and virtually all enteroglucagon cells were also immunoreactive to CgA, but somatostatin cells only occasionally so. A few enteroglucagon cells in this region were also CgB-immunoreactive.
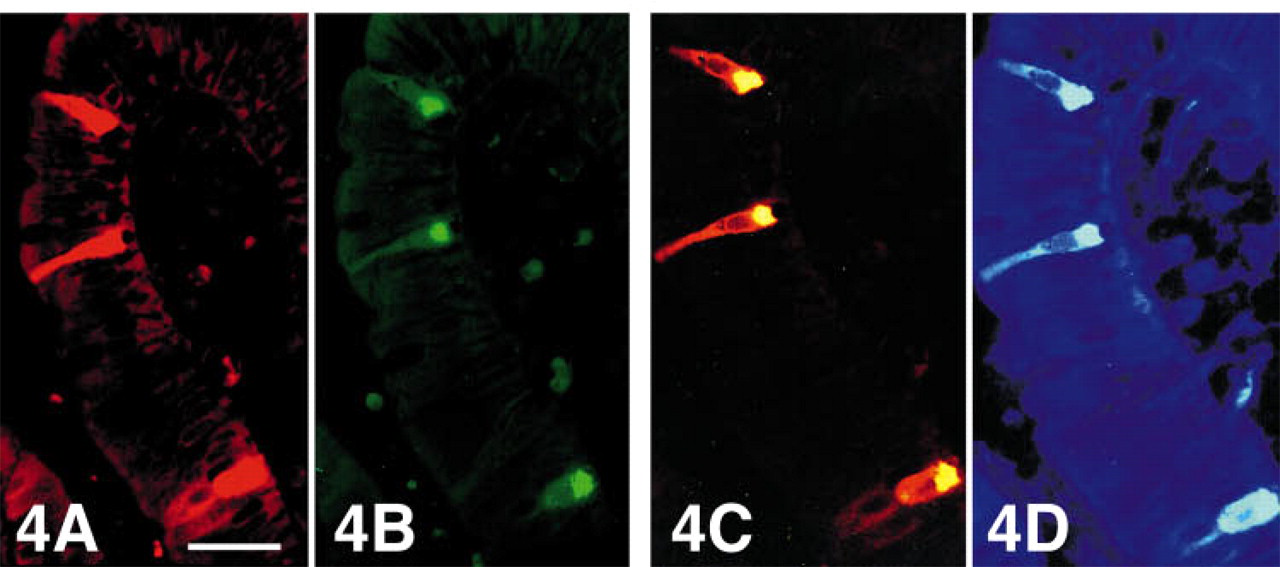
Tip of a villus of human duodenum triple stained for serotonin (Texas red), chromogranin A (FITC), and chromogranin B (AMCA). Serotonin (
Brunner's Glands. The identified EC, gastrin, and CCK cells (but not somatostatin or enteroglucagon cells) were CgA-IR. Gastrin and CCK cells also contained CgB.
Ileum. The EC cells were CgA-positive and some of those located in the villi also contained CgB. The somatostatin cells proved negative for all three chromogranins. Some enteroglucagon/PYY and neurotensin cells were CgA-IR, whereas CgB and C staining was absent.
Colon. The EC cells and a subpopulation of EG/PYY cells were found immunoreactive to CgA, but not to B or C. The somatostatin cells reacted negatively to all the Cg antibodies. The few CgB cells visualized could not be correlated to any of the endocrine cell types identified.
Intracellular Localization of Chromogranins and Corresponding Hormones
CgA and C were concentrated mostly in the basal (infranuclear) region of the cells, whereas CgB was more diffusely spread in the cytoplasm, as was serotonin immunoreactivity, involving the cell processes occasionally seen in some of these cells. CgA immunoreactivity in antral EC cells in the lower third of the mucosa was located mainly adjacent to the basal cell membrane. In gastrin cells, CgA was found mainly in the infranuclear region, although occasionally occupying the entire cytoplasm. CgA immunoreactivity was mainly basal in CCK, secretin, GIP, enteroglucagon, enteroglucagon/PYY, and neurotensin cells, whereas the entire cytoplasm was stained in the (few) CgA-IR somatostatin cells.
Discussion
For co-localization studies, the present multiple immunostaining technique using fluorochromes proved superior to single immunostaining in consecutive sections, because the substances could be determined more exactly in the individual cells. A similar technique—but with chromogens for light microscopy—is excellent if the substances occur in different cells, but difficulties may arise in determining the resulting color if they are located in the same cells. Immunoelectron microscopy can also be used in co-localization studies by using various sizes of gold particles, although the more complicated staining steps are a disadvantage compared with the present fluorescence methods. Furthermore, only a small number of cells can be studied at one time. With single-band filter sets, the different substances can be located individually. With the present technique, using a double-band filter, co-localization of two of these substances can be studied simultaneously, and with a further single-band filter and double exposure of the film it was possible to identify a third substance at the same time and even to determine its location in relationship to the other two. A triple-band filter for the three fluorochromes has also been tested, but it did not help us assess the stainings.
With double or triple immunostaining, crossreactivity of the antibodies must be avoided. In our investigation we used both monoclonal and polyclonal antibodies raised in different animal species. Only exceptionally did we use antibodies raised in the same animal species, and in these cases we blocked the epitopes of the first antibody by using monovalent Fab fragments of IgG. Appropriate control stainings, necessary to evaluate the staining specificity, were carried out. Furthermore, staining specificity was evaluated for single immunostaining, not only with fluorescence but also with the ABC technique.
The distribution of chromogranin immunoreactivity along the gastrointestinal tract is fairly similar to that demonstrated in previous reports (Varndell et al. 1985; Rindi et al. 1986; Bishop et al. 1989; Buffa et al. 1989; Schmid et al. 1989; Pelagi et al. 1992). CgA-IR cells predominated in all gastrointestinal regions studied, indicating the conventional wisdom of using CgA as a panendocrine marker. However, not all cells displayed CgA immunoreactivity. CgB-IR cells, were slightly fewer in the antrum compared with CgA-IR cells, and were sparse in duodenum and almost nonexistent in the corpus and colon. CgC was less common than the other members of the Cg family, and was present only in the antrum and duodenum.
As mentioned earlier, the ECL cells were indirectly identified, because the commercially available histamine antibodies tested were ineffective in our tissue. These antibodies need a special fixative containing carbodiimide. ECL cells predominate in the corpus and their frequency and distribution are as for the CgA cells, i.e., at least the majority of ECL cells must be CgA-positive, which would be consistent with other reports (Buffa et al. 1989), although they are both CgB- and C-negative.
EC cells displayed CgA immunoreactivity in all gastrointestinal regions examined, as in other reports (Varndell et al. 1989; Rindi et al. 1986; Buffa et al. 1985). CgA was found mainly in the infranuclear region, whereas CgB immunoreactivity, occurring in some EC cells in antrum, duodenum, and ileum, had a cytoplasmic distribution similar to that of serotonin, i.e., throughout the cytoplasm. This difference in the immunostainings suggests that either CgA is not stored in all secretory granules or else occurs in quantities too small to be detected by our immunostaining techniques, or that serotonin and CgB may occur in a nongranular form in the supranuclear cell region. CgB was demonstrated in a small subpopulation of EC cells in duodenum and ileum, located mainly in the villi. Earlier studies have produced various results regarding the occurrence of CgB in EC cells in the intestinal tract. Buffa et al. (1989) reported CgB-IR and non-CgB-IR EC cells, but they did not mention whether these subpopulations were located in the villi or in the crypts. Rindi et al. (1986) did not find CgB in EC cells, and Bishop et al. (1989), using electron microscopy, reported both CgB and GAWK (a fragment of CgB) in this cell type. Two subpopulations of EC cells have previously been identified, on the basis of their content of motilin and substance P, respectively (Pearse et al. 1974; Heitz et al. 1976; Heitz et al. 1978). These subpopulations have different distributions in the gastrointestinal tract, the former in the proximal small intestine, the latter in the distal part. However, the localization of these two subpopulations cannot be related to the presence or absence of CgB-IR cells because the latter occur in both proximal and distal small intestine, and mostly in the villi. This difference in the content of CgB in EC cells may suggest different cellular functions in the villi and in the crypts. Our finding of CgC-IR EC cells in the antrum and in duodenal crypts has not been reported earlier, possibly due to the quality of the antibodies and/or the fact that these cells are so sparse.
The antral gastrin cells were immunoreactive for both CgA and B, and some also for CgC. The latter glycoprotein has not been reported earlier in this cell type in humans, but was found in guinea pig (Cetin and Grube 1991; Cetin et al. 1992). It is less probable that these CgC-IR gastrin cells correspond to the cell population expressing both gastrin and serotonin; the former were more numerous. In the duodenum, few gastrin-IR cells reacted to CgA antibodies.
With very few exceptions, somatostatin cells were the only cell type in which we were unable to find CgA. The reason for this is unclear, but one possible explanation may be that CgA is present at such a low concentration that it cannot be detected with the immunostaining used, although Wiedenmann et al. (1988) reported similar results using the more sensitive PAP stain. Other explanations could be that CgA may be masked by the granule-related proteins, or is proteolytically fragmented in such a way that it does not react with our antibodies, or that with few exceptions it does not exist in these cell types. We have not succeeded in demonstrating any CgB or C in this cell type.
Regarding the secretin cells and CgA, our results differ somewhat from those reported by Buffa et al. (1989). Unlike these authors, we found CgA in only some of the secretin cells.
The co-localization of enteroglucagon and CgA varied in the different intestinal regions. In the duodenal mucosa, enteroglucagon cells reacted to CgA, which agrees with the results reported by Varndell et al. (1985). Surprisingly, this cell type did not react to CgA in Brunner's glands. In ileum and colon, where enteroglucagon is to a large extent co-localized with PYY, CgA appeared in only some of these cells. Neurotensin cells also had a variable CgA content.
It has been demonstrated ultrastructurally that CgA and B are present in secretory granules in the gastrointestinal tract (Varndell et al. 1985; Bishop et al. 1989). CgC has been found in secretory granules in the pituitary gland (Rosa et al. 1992), and may also have the same location in antrum and duodenum. In antral gastrin cells the distribution of chromogranins suggests at least two subpopulations of secretory granules, one in the infranuclear region, which may contain all the three chromogranins, the other in the supranuclear region containing only CgB. It remains to be shown whether or not the different chromogranins are co-localized in the same secretory granules, or whether chromogranin B may also exist in a nongranular form.
Our knowledge of the physiological functions of chromogranins is limited. Most studies have concentrated on CgA and its proteolytic fragments, which may have several biological roles, among others an intracellular hormone-binding function, as precursors to various smaller fragments that may be biologically active, and/or an inhibitory effect on the secretion of different hormones belonging to the neuroendocrine system, e.g., parathyroid hormone, parathyroid hormone-related protein, insulin, calcitonin, and catecholamines (cf. Winkler and Fischer-Colbrie 1992; O'Connor et al. 1994).
The function of CgB and CgC is still virtually unknown. In addition to having a role as precursors of smaller peptides (Huttner et al. 1991), CgB has been reported to bind calcium (Gorr et al. 1989), a function also attributed to CgA, and to have a trophic action in neurons (Chen et al. 1992). It has been suggested that CgC is associated with secretory granule maturation (Tooze et al. 1994), but this would contradict the finding of this glycoprotein in only a limited number of endocrine cell types.
Immunoelectron microscopic studies have shown that serotonin (Fujimiya et al. 1995; Okumiya et al. 1996a) and gastrin (Okumiya et al. 1996b) may exist in a nongranular form in the supranuclear cytoplasm of EC and gastrin cells, respectively. Our findings of an association between the intracellular distribution of the immunoreactivity of serotonin and gastrin with CgB, particularly in the luminal parts of the cells, may suggest a functional role for CgB as a carrier protein, at least for serotonin and gastrin. A surprising finding was the variation in CgA, B, and C immunoreactivity within the different cell types. There is still no plausible explanation for the varying extent and composition of the different chromogranins, but this may be a reflection of their complex roles in the neuroendocrine system related to their biological activities as hormone-binding substances, carrier proteins, precursors to smaller fragments, and proteins contributing to secretory granule maturation and hormone release. A further interesting question about their interrelationship is whether or not the chromogranins may be present in the same secretory granules. Such a co-localization study at the ultrastuctural level could indicate if different granule populations exist, and thereby provide further knowledge concerning the functions of the different members of the chromogranin family.
Footnotes
Acknowledgements
Supported by a grant from the Swedish Medical Research Council (project no. 102), by the Lion's Foundation, Uppsala, and by Hässle Läkemedel AB, Mölndal, Sweden.
We thank Ms Birgitta Vagnhammar for excellent technical assistance.
Parts of this work were presented at the 11th International Symposium on Regulatory Peptides, Copenhagen, Denmark, 3–7 September, 1996.
