Abstract
In biological tissues, specific carbohydrate moieties of the oligosaccharide chains of glycoproteins can be localized by lectin binding. Such carbohydrate moieties are among the factors that mediate cell-cell or cell-matrix interactions during pre- and postimplantation embryonic development. Binding sites for the lectins RCA I, WGA, and LTA were localized in preimplantation mouse embryos at the ultrastructural level with the help of postembedding lectin gold cytochemistry. WGA and RCA I binding sites, but no LTA binding sites, were present in the zona pellucida. WGA and RCA I binding sites were found at cell surfaces of morulae and in cells of the inner cell mass of blastocysts, suggesting that N-acetylglucosamine-, terminal β-galactosyl-, and N-acetylgalactosamine-rich glycoproteins might be involved in cell-cell and cell-matrix interactions. WGA binding sites were found predominantly in electron lucid vesicles of the blastomeres, whereas RCA I was detected in electron dense vesicles of the compacted morula and later in the polar trophoblast cells. This allows early identification of blastomere cells that later differentiate into the polar trophoblast.
G
Lectin binding sites have been localized in preimplantation embryos, mainly to elucidate the role of carbohydrates in the fertilization and implantation process. Chavez and Enders (1981) found RCA I bound to Day 5 mouse embryos. Wu et al. (1984) detected RCA I and WGA binding sites in the zona pellucida of mouse ova.
Most researchers, have thus far relied on pre-embedding lectin histochemistry on whole embryos at the light microscopic level: WGA, RCA I, RCA II, and ConA have been demonstrated to bind to preimplantation embryos (Brownell 1977). Binding of the lectins recognizing 5-N-acetylgalactosamine, e.g., SJA, HPA, and DBA, has also been shown (Sato and Muramatsu 1985). Few investigators have localized lectin binding sites in preimplantation embryos at the ultrastructural level.
Nicholson et al. (1975) localized WGA and RCA I binding sites in mammalian ova applying the pre-embedding ferritin method. Konwinski et al. (1977) localized ConA binding sites with the help of peroxi-dase-labeled ConA at the ultrastructural level. These ultrastructural studies were performed with pre-embedding techniques which, however, have a slight disadvantage. Although pre-embedding can allow the localization of lectin binding sites in the zona pellucida by penetration, labeling of intracellular structures can be seen only when the probe is internalized. In contrast, postembedding lectin-gold cytochemistry enables a reliable localization of lectin binding sites within cells and cell organelles.
Although some authors have localized glycoconjugates in the zona pellucida of postovolatory oocytes in the hamster (Roux and Kan 1991) and the rat (Avilés et al. 1994) this method has not been previously applied to investigation of lectin binding sites within pre-implantation embryos, i.e., morulae and blastocysts, to study intracellular structures. We therefore localized WGA, LTA, and RCA I binding sites in the pre-implantation mouse embryo at the ultrastructural level by use of the postembedding technique.
Materials and Methods
Animals
Female NMRI mice were kept on a normal day/night cycle and received Altromin commercial food and water ad libitum. The day on which a vaginal plug was detected at 1100 hr after a mating period of 3 hr was designated Day 0 of gestation.
Tissue Preparation
On Days 2–4 of gestation (from the four-cell stage to blastocyst stages), the mice were sacrificed by cervical dislocation and the uteri with the oviducts were removed. These were transferred to 0.1 M PBS, pH 7.2, at 4C, the oviducts were sealed with a clamp, and buffer solution was injected into the cavum uteri from the vaginal side. When pressure had built up, the clamp was quickly opened and the embryos flushed out through the oviduct into the buffer solution.
LR Gold Embedding
The embryos were next fixed for 15 min in 4% formaldehyde and 0.5% glutaraldehyde in 0.1 M PBS. After incubation for 30 min in 10 mM ammonium chloride, the embryos were dehydrated in a series of 30%, 50%, and 70% ethanol and transferred to gelatin capsules containing LR Gold (London Resin; Reading, UK) supplemented with 0.8% of the light-sensitive accelerator benzil. The gelatin capsules containing the embryos were sealed air-tight and the LR Gold was then polymerized at −25C with the light of a halogen lamp (15 V, 150 W).
Lectin-Gold Cytochemistry
For this study, five morulae of the eight-cell stage, five morulae of the 16-cell stage, five blastocysts with a zona pellucida, and five blastocysts with no zona pellucida were employed.
For orientation purposes and for determination of the developmental stages of the embryos, 1 -μm-thick sections were cut from the embedded embryos and stained with toluidine blue. Thereafter, ultrathin sections were cut with a Reichert ultramicrotome and collected on formvar-coated nickel grids. The nickel grids were preincubated for 10 min at room temperature (RT) with 0.01 M PBS (pH 7.2). The sections were then incubated for 1 hr at RT with 30-nm gold-labeled RCA I, 15-nm gold-labeled WGA, or with 20-nm gold-labeled LTA (all from EY Laboratories; San Mateo, CA) diluted 1:20 in PBS buffer. After having been rinsed in PBS, the sections were stained with 2% osmium tetroxide in 0.01 M PBS for 10 min, 1% uranyl acetate for 15 min, and 1% lead citrate for 5 min and were examined with a Zeiss EM 109 electron microscope.
Controls
The control for the specificity of the staining reactions was carried out on sections incubated for 1 hr at RT with the gold-labeled lectins, the carbohydrate binding sites of which had been blocked by preincubation with the corresponding inhibiting sugars for 1 hr at RT.
WGA was incubated with N,N'-diacetylchitobiose (0.1 mM), N,N',N”-triacetylchitotriose (0.1 mM), and N-acetyl-
Results
Morula Stage
WGA. Strongest staining for WGA binding sites was seen in the zona pellucida (Figure 1) and on all cell surfaces of the blastomeres (Figure 2). In the cytoplasm, many labeled membrane vesicles were seen. Strong gold labeling was also observed in association with the limiting membrane of vesicles of the blastomeres, which appeared to contain electron-lucid material in their interior. These stained vesicles were seen in all blastomeres of both early and compacted morula. In the cytoplasm of the flat blastomeres located in the outer zone of the compacted morula, a few solitary gold-labeled electron-dense vesicles were present (Figure 3).

Early morula after staining for WGA binding sites. Arrows, strong staining of the zona pellucida; triangle, staining of vesicles located in the cytoplasm of a blastomere. Bar = 25 nm.

WGA binding sites. Arrows, staining at the cell surface of two adjacent blastomeres (B) of an early morula. Bar = 25 nm.
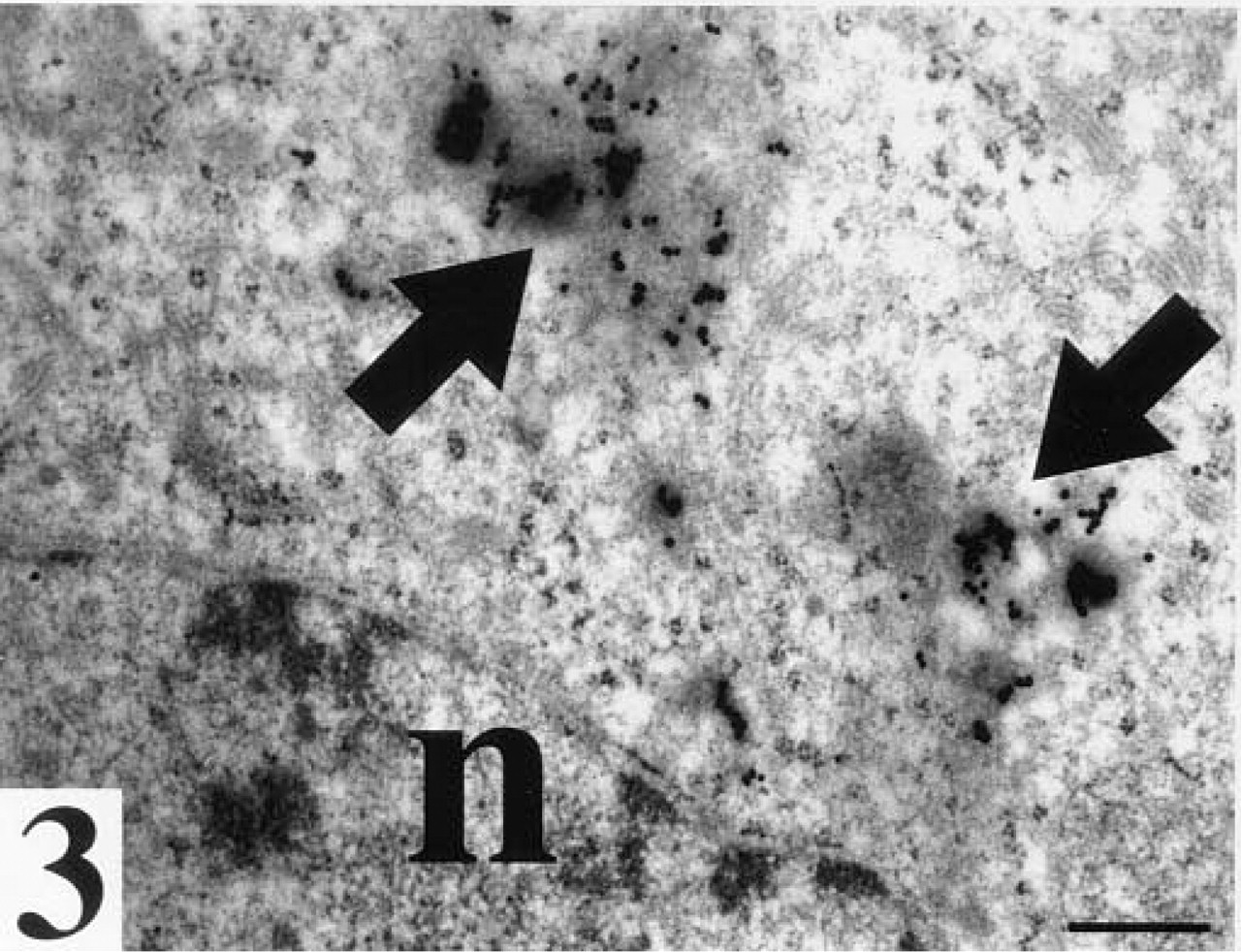
WGA binding sites. Arrows, staining in electron dense vesicles in the cytoplasm of a flat blastomere located in the outer zone of a compacted morula; n, nucleus. Bar = 25 nm.
Early morula after staining for RCA I binding sites. Arrows, staining of the zona pellucida. Bar = 25 nm.

RCA I binding sites. Arrows, staining at the cell surface of two adjacent blastomeres (B) of a compacted morula; triangle, staining of an electron dense vesicle in a flat blastomere. Bar = 25 nm.
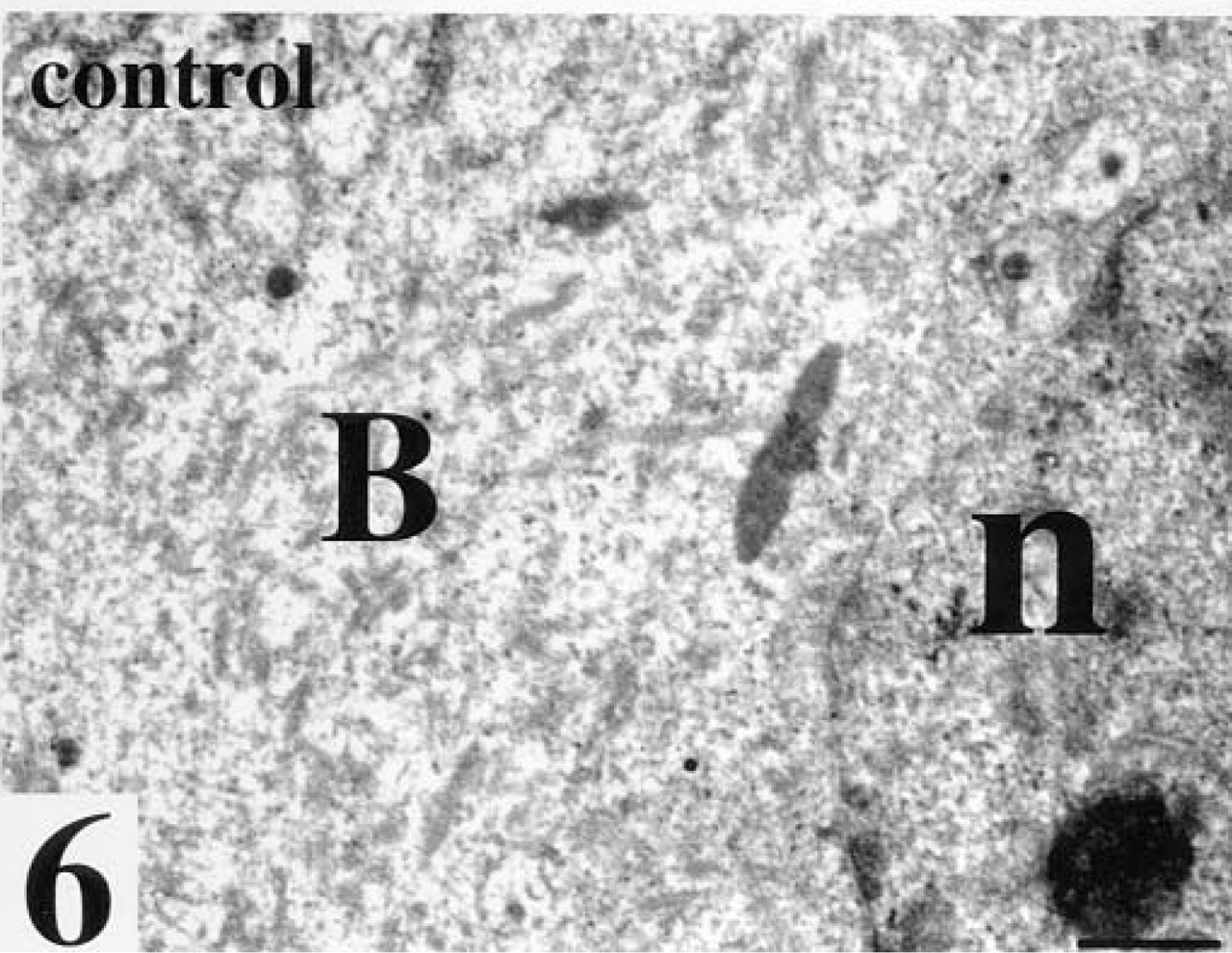
Compacted morula after staining for WGA incubated with its inhibiting sugars. No gold labeling is seen, B, blastomere; n, nucleus. Bar = 25 nm.
Blastula Stage
In the mural trophoblast, only a few gold-labeled dense vesicles were seen in the cytoplasm. In contrast, many stained vesicles with electron-lucid material in their interior, again with preference for the limiting membrane, and stained vacuoles were observed (Figure 8). In some but not all microvilli oriented towards the exterior, as well as in microvilli oriented towards the inner cavity of the blastocysts, WGA binding sites were seen.
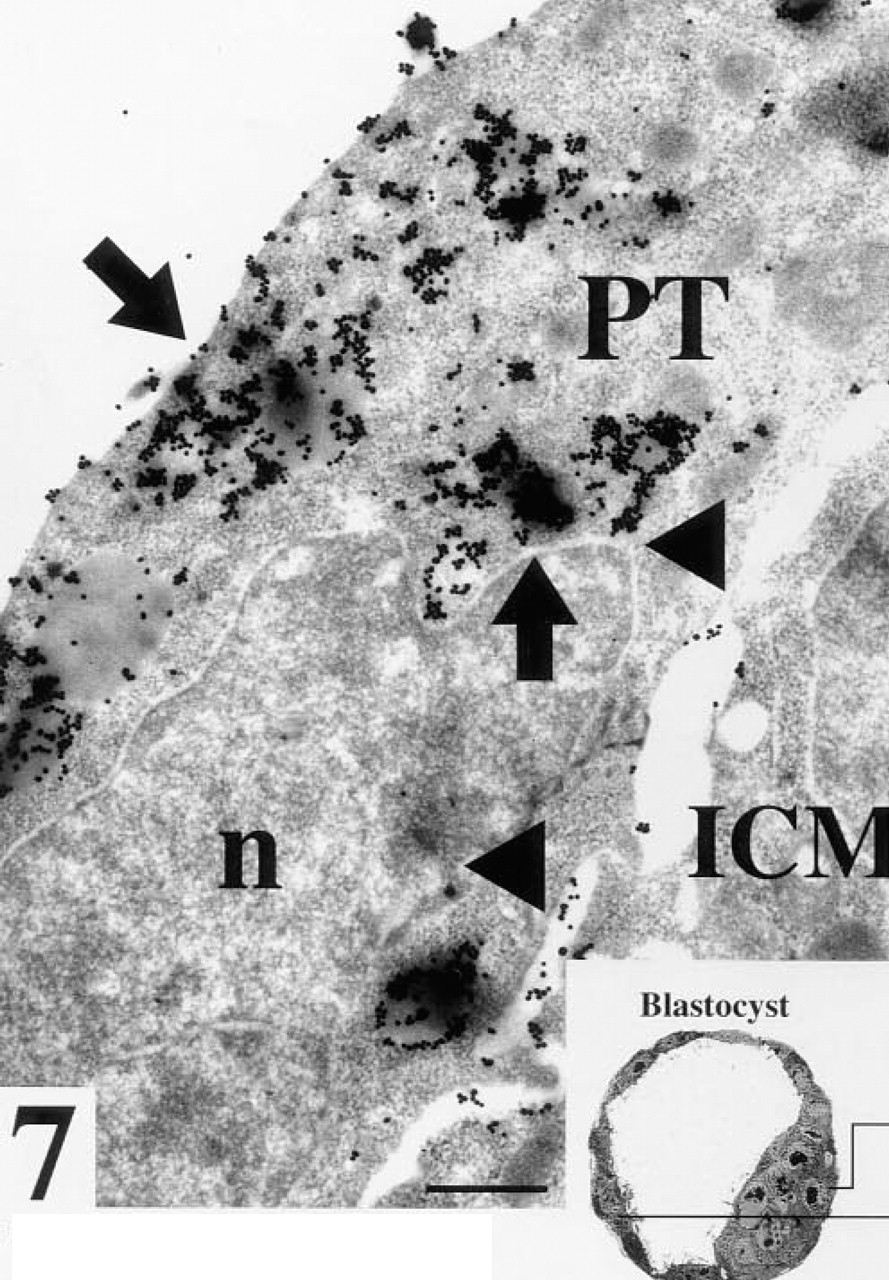
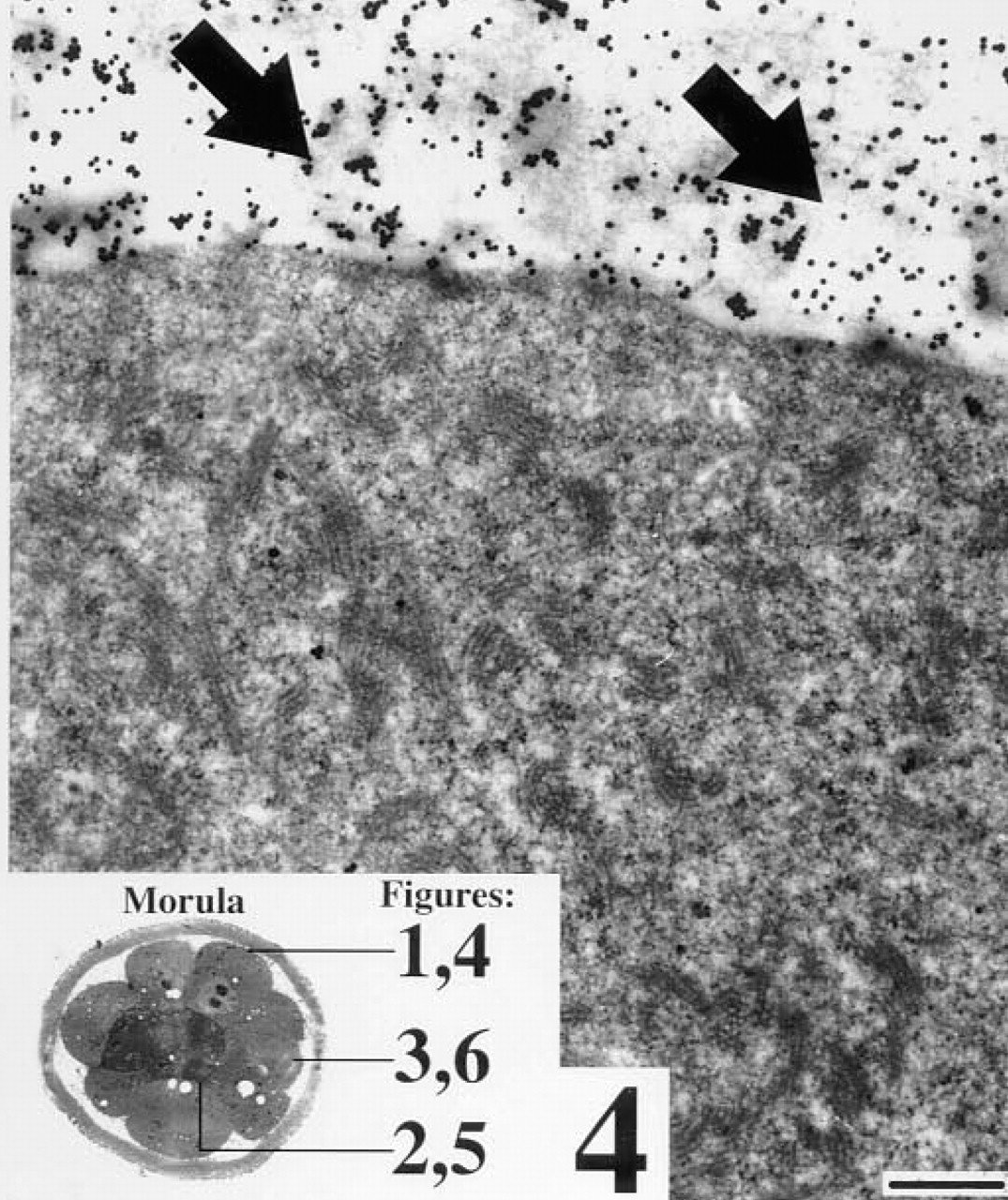
Blastocyst with no zona pellucida after staining for WGA binding sites. Arrows, strong staining of an electron dense vesicle in the cytoplasm of a polar trophoblast cell (PT); ICM, cells of the inner cell mass; triangles, staining between a trophoblast and a cell of the inner cell mass; n, nucleus. Bar = 25 nm.
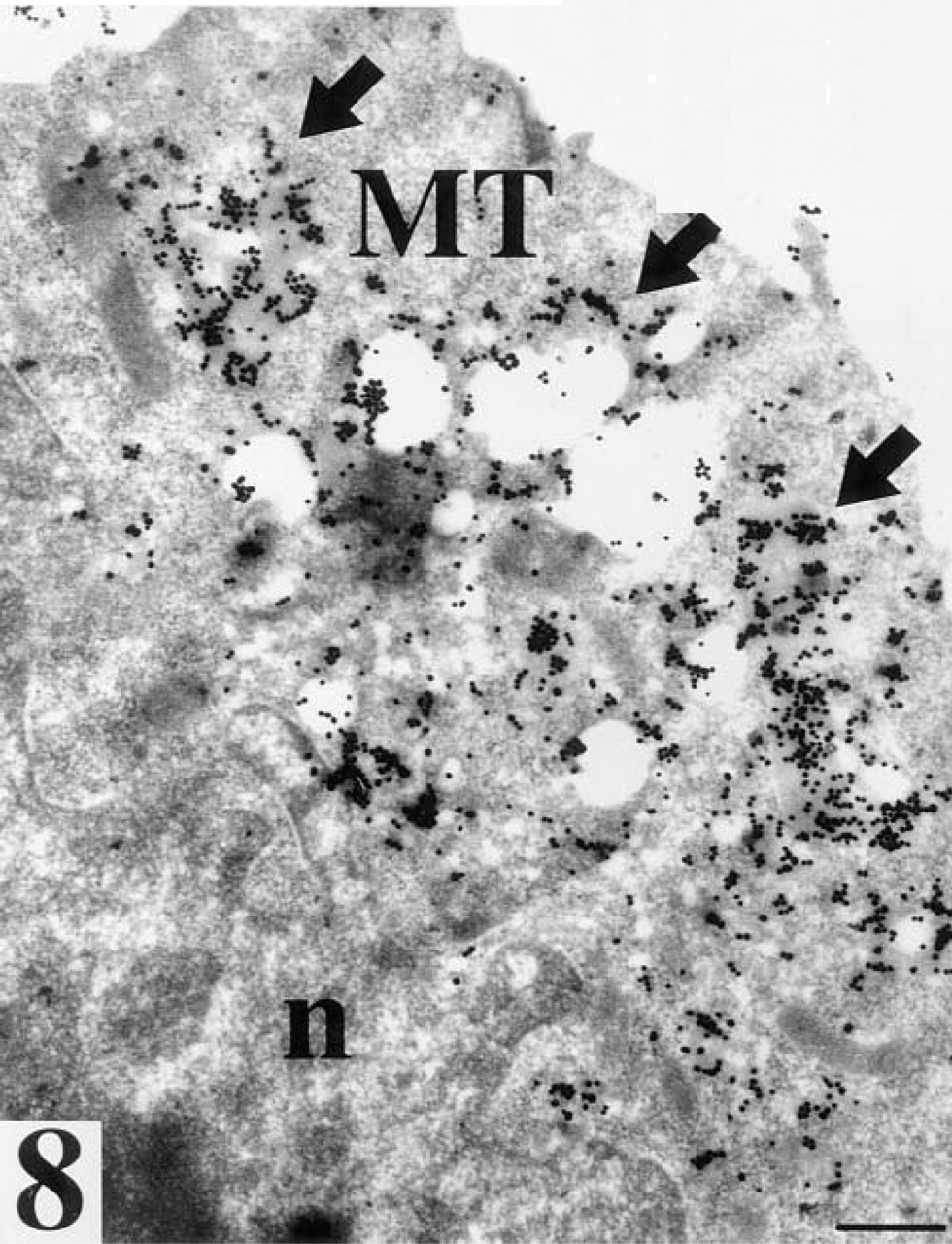
Mural trophoblast (MT) of a blastocyst with no zona pellucida after staining for WGA binding sites. Arrows, strong staining of vesicles located in the cytoplasm oriented towards the exterior of the trophoblast cell; n, nucleus. Bar = 25 nm.
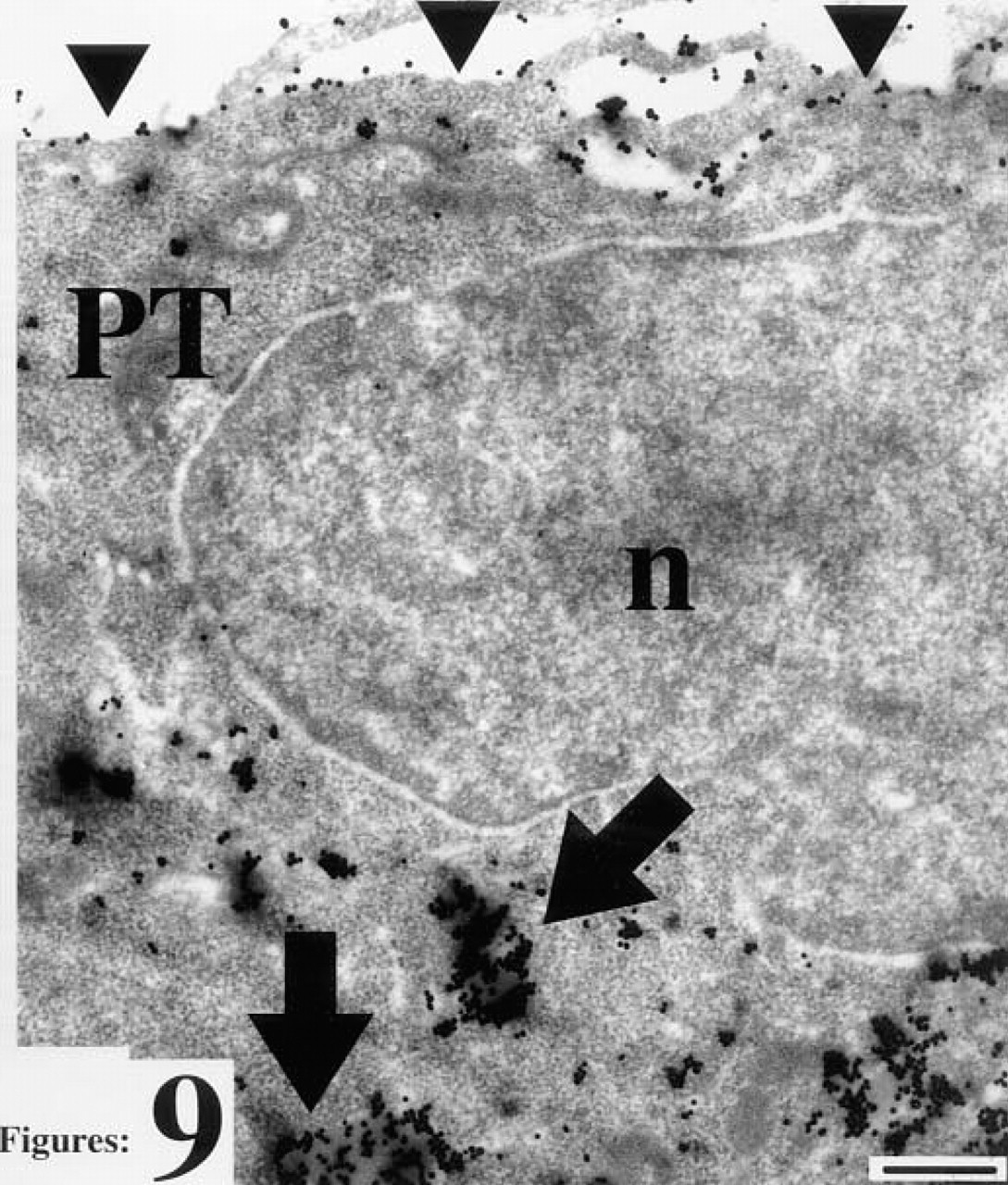
Polar trophoblast cell (PT) after staining for RCA I binding sites. Arrows, staining of vesicles, triangles, staining of the cell surface oriented towards the ICM; n, nucleus. Bar = 25 nm.

Polar trophoblast cells (PT) of a blastocyst with no zona pellucida after staining for RCA I binding sites. Arrows, staining of interdigitate processes of polar trophoblast cells. Bar = 28 nm.
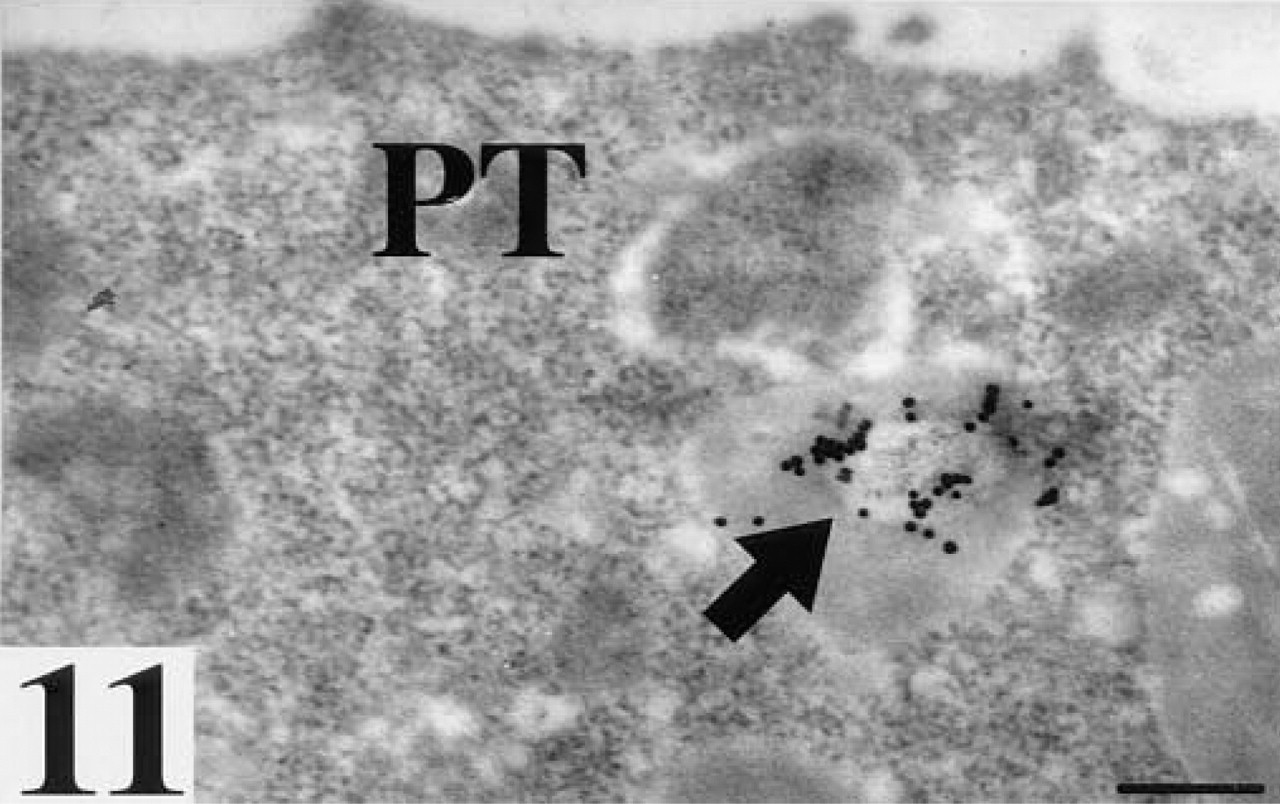
Polar trophoblast cell (PT) after staining for LTA binding sites. Arrow, staining of solitary vesicle. Bar = 25 nm.
Discussion
Lectins are elegant markers for detection of sugar moieties of glycoproteins or glycolipids. With their help, it is possible to recognize different carbohydrates in the various developmental stages of embryogenesis. In the work presented here, we used the postembedding lectin-gold method on LR Gold embedded embryos.
Embedding in the hydrophilic resin LR Gold (Newman et al. 1983; Herken et al. 1988) ensures reasonably good preservation of the morphology along with preservation of the reactivity of lectin binding sites. This technique has already been well established for the ultrastructural localization of lectin binding sites in postimplantation embryos (Salamat et al. 1993; Herken et al. 1990).
The preimplantation embryos investigated showed WGA and RCA I binding sites in the zona pellucida. This was previously shown by Roux and Kan (1991) for RCA I binding sites of the zona pellucida of hamster ova or by Avilés et al. (1994) for WGA and RCA I of rat preimplantation embryos. Avilés et al. (1994) found no LTA binding sites in the zona pellucida.
Our postembedding approach allows the localization of lectin binding sites also within the intracellular structures of the embryos. Strong staining of the cell surfaces of the cells of the blastomeres and of the cells of the ICM of the blastocysts was seen. Therefore, the sugars represented by WGA, i.e., mainly N-acetylglucosamine (GlcNAc) moieties and by RCA I, i.e., mainly terminal β-
Brownell (1977) could not find LTA binding sites in the early embryo at the light microscopic level in the blastocyst. However, we found LTA binding sites in a few cell-cell contact zones in the morula at the ultrastructural level, which can be easily overlooked in a light microscopic investigation. Therefore, it appears that the sugar entities detected by LTA, i.e., mainly fucose (Spicer and Schults 1992; Damjanov, 1987), might play a role only in early developmental steps in cell contact processes.
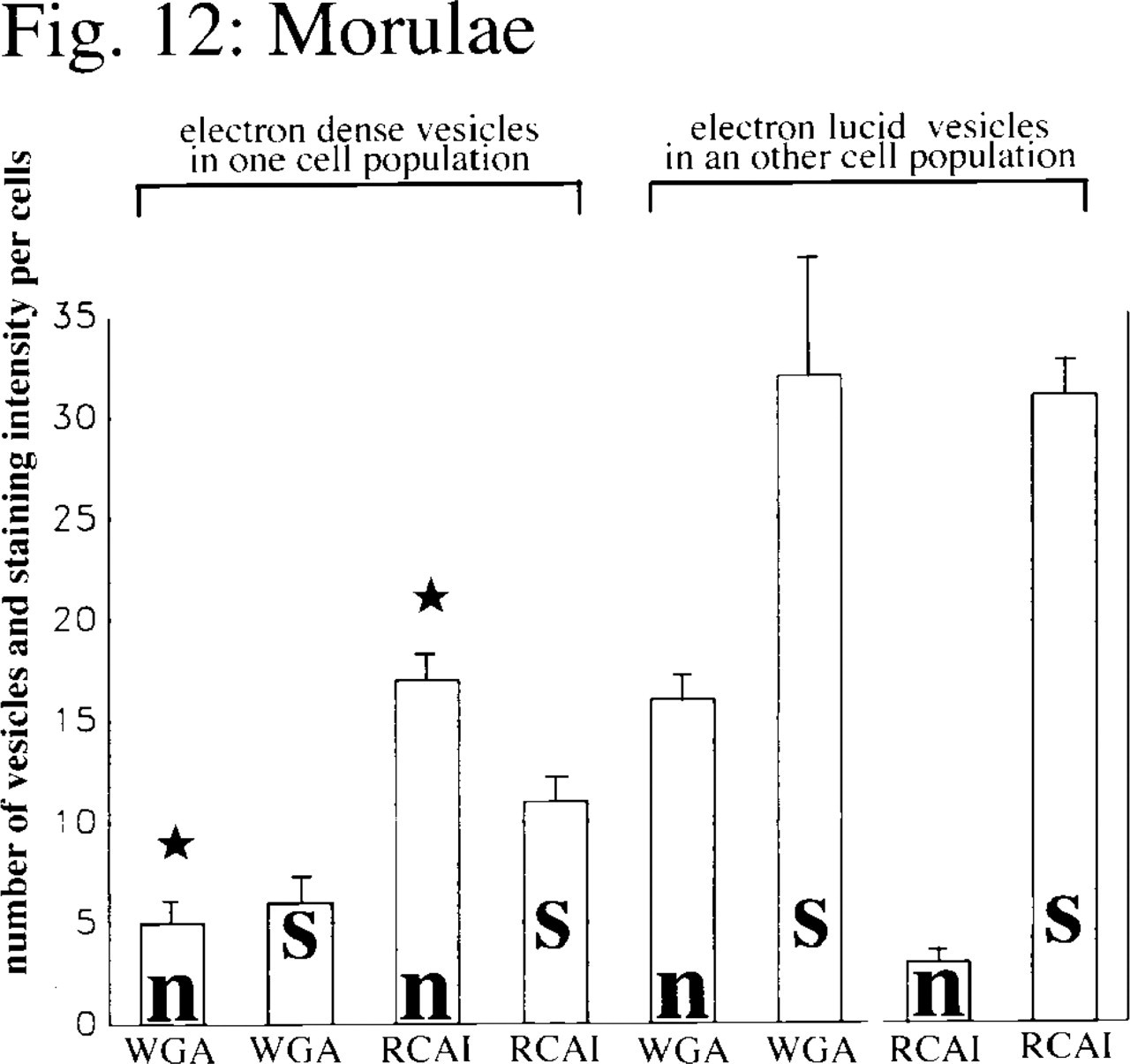
Mean values with SEM (bars) of the numbers of gold particles per cell after WGA or RCA I staining (s) and the numbers of dense or lucid vesicles (n) stained with WGA or RCA I for different blastomere populations within the morulae (n = 12; star, p≤0.01; significant differences were noted for p-values using the Wilcoxon-Mann-Whitney test for unpaired samples).
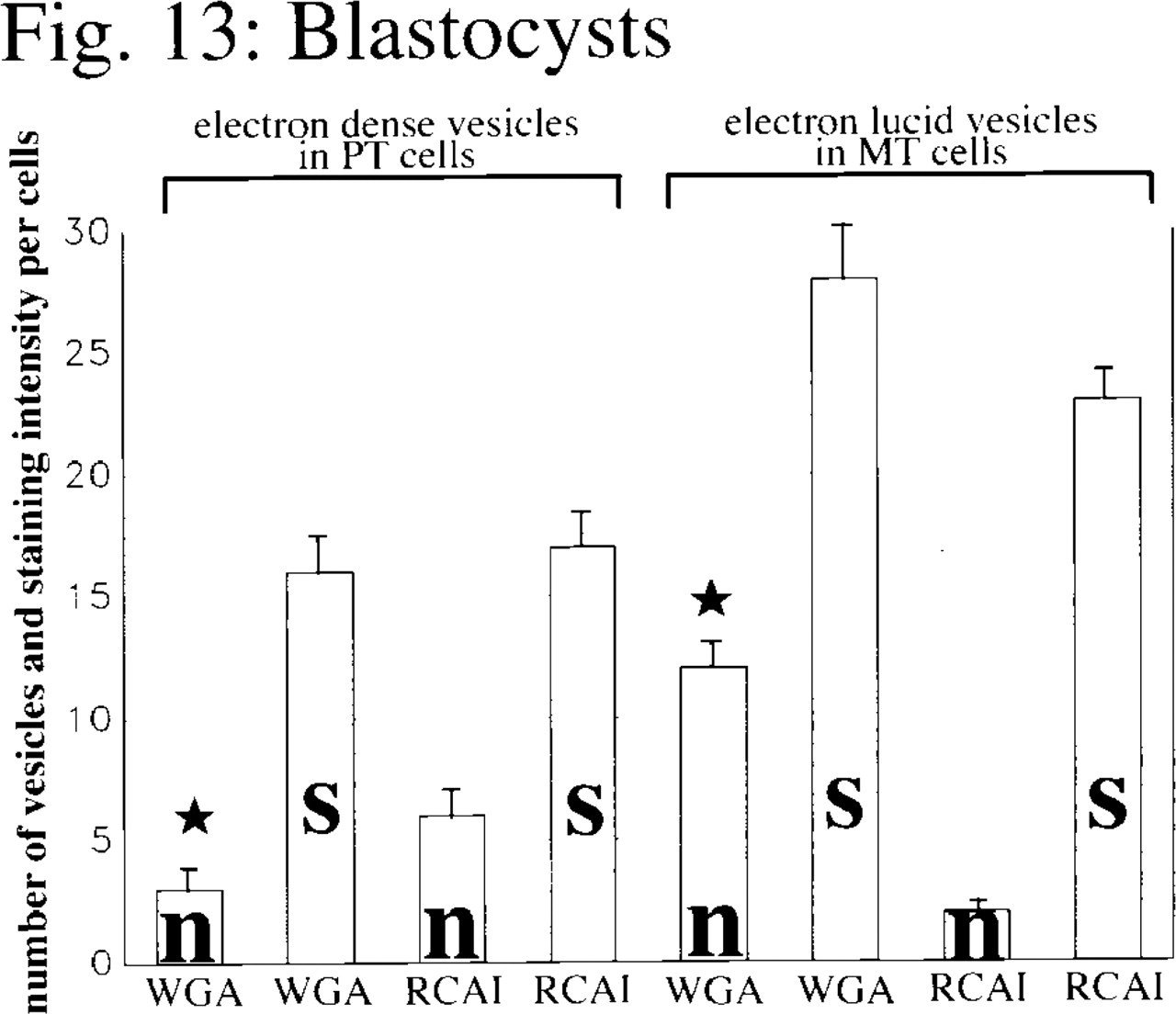
Mean values and SEM (bars) of the numbers of gold particles per cell after WGA or RCA I staining (s) and the number of dense or lucid vesicles (n) stained with WGA or RCA I for either polar or mural trophoblast cells within the blastocysts (n = 10; star, p≤0.01; significant differences were noted for p-values using the Wilcoxon-Mann-Whitney test for unpaired samples).
The staining of contact zones between the polar trophoblast and the ICM with WGA and RCA I hints at the involvement of GlcNAc- and β-Gal-rich glycoproteins in cell recognition processes between polar trophoblast cells and the adjacent ICM cells. Furthermore, morulae and blastocysts exhibit quite a number of intracytoplasmic vesicles, some of them electron-lucid and others with electron-dense material in their interior. We found significant differences (see Figures 12 and 13) in the number and staining pattern of these vesicles. One population of blastomeres within the morulae contained large numbers of electron-dense vesicles strongly stained for RCA I, as well as a few dense vesicles stained for WGA binding sites. Corresponding to the pattern described above, cells of the polar trophoblast of the blastocysts also showed electron-dense vesicles stained for WGA and RCA I binding sites. In contrast, another population of blastomeres in the morulae exhibited electron-lucid vesicles stained for WGA. Such vesicles stained for WGA were also found in cells of the mural trophoblast.
Our results therefore show that even as early as the morula stage one can identify blastomeres that will later develop into polar trophoblast cells by the presence of large numbers of electron-dense vesicles stained with RCA I, as well as a few dense vesicles stained for WGA binding sites. The cells of the polar trophoblast later differentiate into cells of the extraembryonic ectoderm (Gardner 1982). In contrast, cells that are destined to develop into mural trophoblast cells or cells of the ICM exhibit large numbers of vesicles containing electron-lucid material that stains strongly with WGA. Although these vesicles can also be found in the mural trophoblast of the blastocysts, they were no longer present in the cells of the ICM.
In a previous investigation we found developing basement membranes in the blastocyst at the interior side of the mural trophoblast (Salamat et al. 1995), an area in which Reichert's membrane later develops. It has been shown that the well-developed, multilayered Reichert's membrane possesses extensive RCA I binding sites (Salamat et al. 1993), and that these RCA I binding sites derive from glycoconjucates synthesized by trophoblast cells. We therefore expected the presence of RCA I binding sites at the interior side of the mural trophoblast. However, in the present investigation we could not find any basement membrane-like structures exhibiting RCA I binding sites in this part of the blastocyst. These results indicate that RCA I binding sites do not occur in the early unilayered basement membrane of the trophoblast, which is the basis of the future Reichert's membrane, but only in postimplantation stages, when this membrane has already fully developed into the multilayered Reichert's membrane. This phenomenon awaits further elucidation, but the occurrence of RCA I binding sites later in development suggests a possible change in the glycosylation pattern of the proteins of the developing Reichert's membrane.
Footnotes
Acknowledgements
Acknowledgment
We would like to thank Cyrilla Maelicke, B.Sc., for editing the manuscript.
