Abstract
Prostaglandin E1 (PGE1) is commonly used in therapy for obstructive diseases, including ischemic retinopathy, in which pathogenetic reactive oxygen intermediates are responsible. However, the mechanism(s) of PGE1 in reducing tissue damage is still unclear. Adult T-cell leukemia-derived factor/human thioredoxin (ADF) is induced by oxidative stresses and has protective activity against oxidative cellular injury. To evaluate the possible involvement of ADF in the tissue-protective effect of PGE1, we analyzed ADF expression immunohistochemically using a rat transient retinal ischemia model. Rats were treated orally with 300 μg/kg/day OP-1206 α-cyclodextrin clathrate (OP-1206), a stable PGE1 analogue, for 14 days after photodynamic retinal vascular thrombosis by rose Bengal. Rats without any OP-1206 treatment were used as controls. In the OP-1206-treated rats, minimal retinal atrophy due to ischemia/reperfusion was observed histologically up to 14 days, whereas in the non-treated rats the inner layer of the retina became markedly atrophic. In parallel with the histological change, after 14 days following thrombosis ADF immunoreactivity was preserved on retinal pigment epithelial cells in the OP-1206-treated rats, whereas it was diminished in the non-treated rats. These findings suggest an important role for ADF in the OP-1206-dependent suppression of retinal tissue damage caused by oxidative insult.
Keywords
R
Prostaglandin E1 (PGE1) has a variety of biological activities, including potent anti-platelet and vasodilating activity (Gorman 1978; Elkeles et al. 1969). Because of these biological activities, PGE1 and some of its analogues have been reported effective for treatment of obstructive diseases in various organs, including ischemic retinopathy (Heffelfinger et al. 1987; Zygulska-Mach et al. 1980; Carlson and Olsson 1976). Recent studies have shown that some types of prostaglandins, including PGE1, have cytoprotective activity against various noxious agents, ethanol, and taulocholic acid in cultured gastric cells (Cherner et al. 1989; Tarnawski et al. 1988; Lancaster and Robert 1978). On the basis of these findings, we hypothesized that, in ischemic retinopathy, PGE1 may function as a tissue protector, accompanied by induction of ADF.
To test this hypothesis, we analyzed immunohistochemically the expression of ADF in the rat retina after induction of photodynamic retinal vascular thrombosis with or without treatment with a PGE1 analogue, OP-1206 α-CD. In addition, the possible mechanism(s) of PGE1 in reducing retinal damage is discussed.
Materials and Methods
All rats used in each experiment were maintained in an animal care facility in accordance with National Institutes of Health guidelines. A total of 36 male Sprague-Dawley albino rats, weighing 200–250 g, were housed in temperature-and humidity-controlled rooms (at 24 ± 2C with humidity of 55 ± 10%) with 12-hr light:12-hr dark cycles for 1 week before the administration of the test reagent. The rats were given laboratory chow (MF or NMF; Oriental Yeast, Tokyo, Japan) and water ad libitum. They were allocated to the following: Group 1 received OP-1206 treatment after thrombosis (n = 9); Group 2 did not receive OP-1206 treatment after thrombosis (n = 9); Group 3 received OP-1206 treatment without thrombosis (n = 9); and Group 4 received neither OP-1206 treatment nor thrombosis (n = 9).
Model of Vascular Thrombosis
These 18 rats were anesthetized during all procedures by an
Administration of Drug
In this study we used an oral PGE1 analogue, OP-1206 α-CD (Dainippon Pharmacology; Osaka, Japan). This compound, which has the same activity as PGE1 itself, is chemically stable at high temperatures and under intense light because it has a more physiologically compatible structure, consisting only of carbon, hydrogen, and oxygen atoms, and can be delivered orally (Adaikan and Karim 1981; Ohno et al. 1978). Moreover, this compound has its active site at the 15-OH base of the 6’ carbon, which is hardly affected by ROIs (Adaikan and Karim 1981; Ohno et al. 1978).
Each rat in Groups 1 and 3 was treated with a daily oral dose of 300 μg/kg of OP-1206 on each experimental day subsequent to the first day of light exposure, followed by vein occlusion. This concentration of OP-1206 was found in a preliminary study to be most effective in this experimental model (data not shown). OP-1206 was dissolved in normal saline and was delivered orally once a day (1700 hr). Each rat in Groups 2 and 4 received an equal volume of saline.
Immunohistochemistry
The rats were perfused through the left ventricle of the heart with 0.01 M PBS containing potassium (KPBS), pH 7.4. They were then perfused with freshly prepared 2% paraformaldehyde containing 0.1% glutaraldehyde in 0.1 M phosphate buffer, pH 7.4, at 4C, to wash out the blood before fixation. The eyes were then removed. All tissues were fixed in the same fixative as above for 6 hr at 4C, embedded in paraffin with a melting point of 56C, and cut into 5-μm sagittal sections. As a landmark, a slit incision was placed at the ora serrata of the temporal side (at the insertion of the lateral rectus muscle) of the eye. Tissue sections were collected on poly-
The tissue sections were incubated overnight at 4C with an anti-ADF or a control antibody dissolved in KPBS with 1% bovine serum albumin (BSA). At this stage we used an affinity-purified rabbit polyclonal anti-ADF antibody for which was previously confirmed the absence of extraneous crossreactions (Gauntt et al. 1994; Ohira et al. 1994). The sections were then rinsed with cold KPBS, incubated with horseradish peroxidase-conjugated donkey anti-rabbit IgG (Amersham International, Amersham, UK; NA 934, Batch 58) for 60 min and then rinsed again. The antigen-antibody complexes were visualized for 2 min in 0.3% H2O2 and freshly prepared 3,3'diaminobenzidine tetrahydrochloride (DAB, Dotite; Dojindo, Tokyo, Japan) in Tris buffer (pH 7.6). The sections were finally counterstained with Mayer's hematoxylin and observed by light microscopy.
We used IgG-purified, non-immune rabbit serum and KPBS with 1% BSA as a negative control. We also prepared positive controls in an identical manner on sections of normal rat brain and liver.
Quantitative Histological Changes by Statistical Analysis
Previously, we found in a pilot study that atrophy of retinal tissue was observed mainly in the inner retinal layer in this experimental model (Gauntt et al. 1994; Ohira et al. 1994). We paid attention to changes in the cell numbers of inner nuclear layer as an index of tissue damage and compared it between retinas with and without OP-1206 treatment. To delete errors stemming from individual variations in retinal thickness, we calculated the “damage index,” which was calculated by [(the number of nuclei in the inner nuclear layer/cm2)/the thickness of the whole retina] on each photo (X 200) and compared it statistically. The number of nuclei and the thickness for each eye was measured at the same point in sagittal sections (10 sections) near the optic nerve (n = 3). Statistical analyses were done using the Student's t-test. A probability of less than 5% was considered significant.
Results
The mean duration of occlusion of the retinal vein was 3.2 ± 0.4 days for the PGE1 analogue-treated groups and 3.3 ± 0.5 days in the non-treated groups. There was no difference between the PGE1 analogue-treated and the non-treated groups in the duration of venous occlusion. The area of direct light exposure exhibited retinal and choroidal damage. Adjacent areas showed no photodynamic damage. The surrounding areas of the retina displayed degenerative changes consistent with ischemia.
Analysis of Histological Change
Table 1 shows the quantitative evaluation of the retinal degeneration after thrombosis, with or without OP-1206 treatment. The damage to inner nuclear layer was compared between the retinas with and without OP-1206 treatment at each time point. There was no significant difference, with the degeneration of inner nucleus at 3 days after the thrombosis. However, at 7 days (p<0.01) and 14 days (p<0.001), cell survival in the inner nuclear layer in the OP-1206-treated retina was apparently preserved compared with the non-OP-1206-treated retina.
Evaluation of retinal tissue damage by ischemia/reperfusion with OP-1206 treatment a
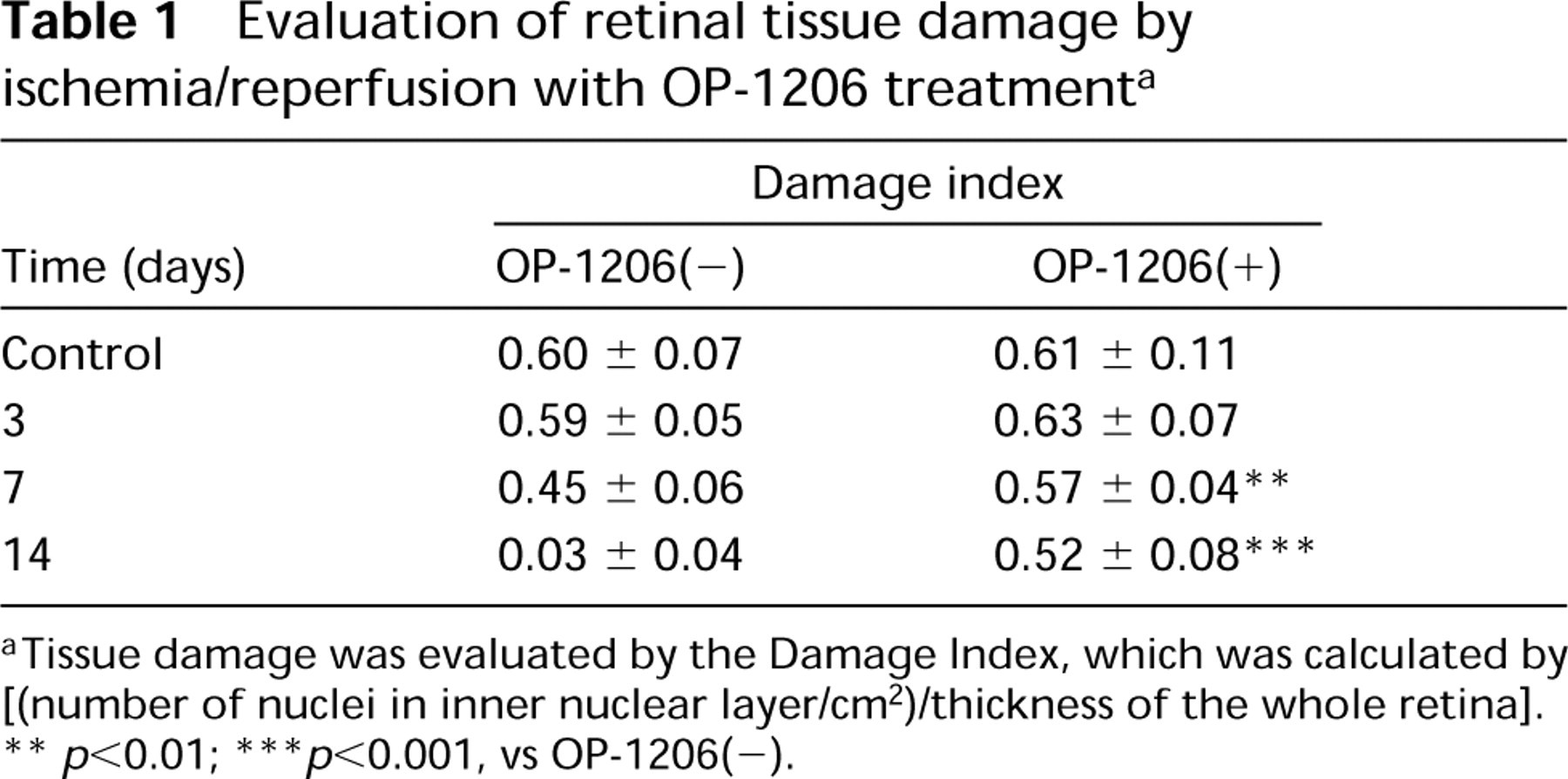
a Tissue damage was evaluated by the Damage Index, which was calculated by [(number of nuclei in inner nuclear layer/cm2)/thickness of the whole retina].
∗∗ p<0.01; ∗∗∗ p <0.001, vs OP-1206(-).
Immunohistochemistry
ADF Immunoreactivity in OP-1206-treated Rats. Representative retinal micrographs for each group with OP-1206 treatment (Group 1) are shown in Figure 1. At 3 days after venous occlusion, ADF immunoreactivity was observed in the internal limiting membrane, ganglion cell layer, inner and outer plexiform layers, and photoreceptor outer segments. Sparse and partial ADF immunoreactivity was observed from the outer nuclear layer to the photoreceptor inner segments. ADF immunoreactivity was markedly increased in the retinal pigment epithelial cell layer (Figure 1A). At 7 days after venous occlusion, weak ADF immunoreactivity was observed from the internal limiting membrane to the inner plexiform layers. Little ADF immunoreactivity was present from the inner nuclear layer to the photoreceptor outer segments, whereas relatively strong ADF immunoreactivity was detectable in the retinal pigment epithelial cells (Figure 1B). At 14 days after venous occlusion, ADF immunoreactivity was observed from the internal limiting membrane to the outer plexiform layers. ADF immunoreactivity was also clearly observed in the photoreceptor inner segments and the retinal pigment epithelial cells. Although focal edema was seen in the ganglion cell layer and in the inner and outer nuclear layers, no significant retinal atrophy was observed (Figure 1C).
ADF Immunoreactivity in Non-OP-1206-treated Rats. Representative retinal micrographs for each group without OP-1206 treatment (Group 2) are shown in Figure 2. At 3 days after venous occlusion, the ADF immunoreactivity was similar to that in the OP-1206-treated rats, except for the photoreceptor outer segments, which exhibited a positive reaction in the OP-1206-treated rats (Figure 2A). In retinas obtained 7 days after thrombosis, the pattern of ADF immunoreactivity was similar to that of the OP-1206-treated retina (Group 1), except for the retinal pigment epithelial cells, which exhibited a positive reaction after the third day after thrombosis (Figure 2B). At 14 days after venous occlusion, little ADF immunoreactivity was seen in the neural retina. Very weak ADF immunoreactivity was found in the retinal pigment epithelial cells. Severe atrophic change could be seen in the inner retinal layers (Figure 2C).
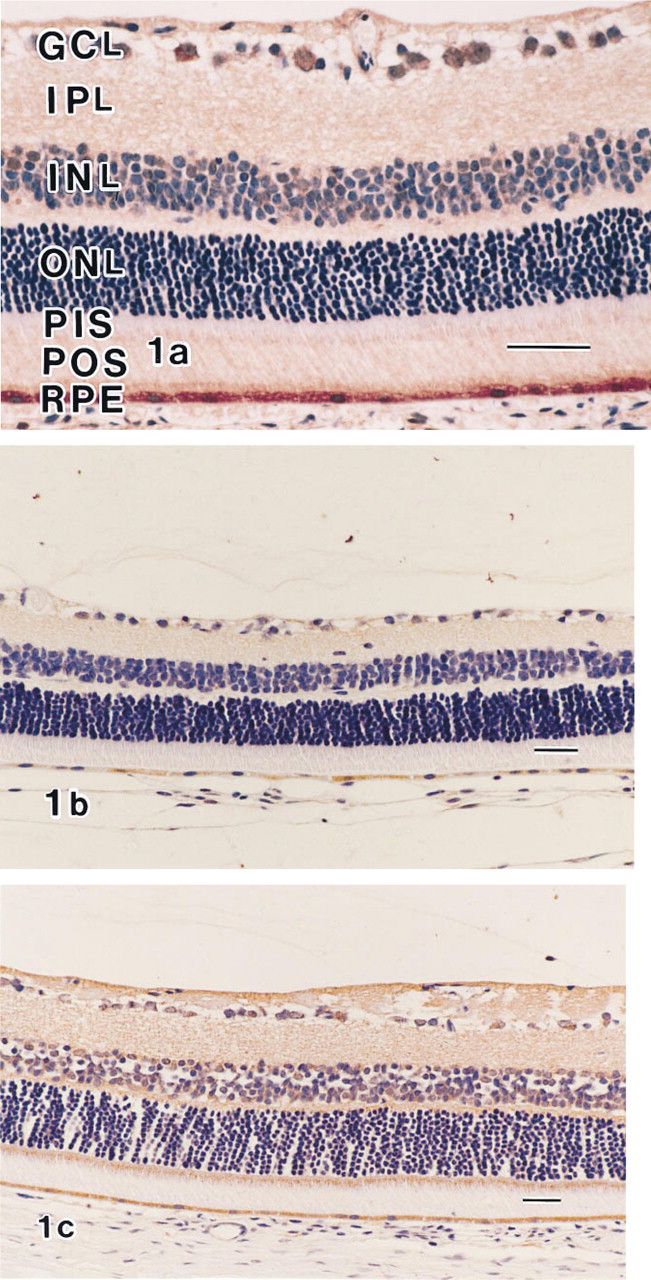
ADF immunolocalization in the retinas of OP-1206-treated rats (Group 1). (
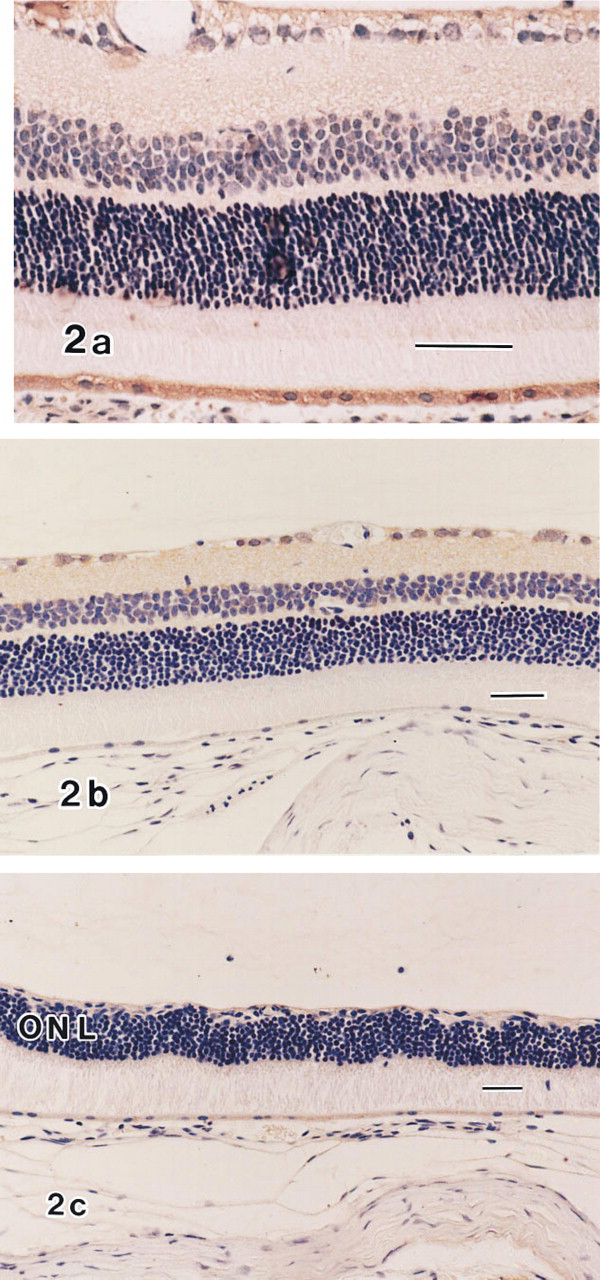
ADF immunolocalization in the retinas of non-OP-1206-treated rats. (
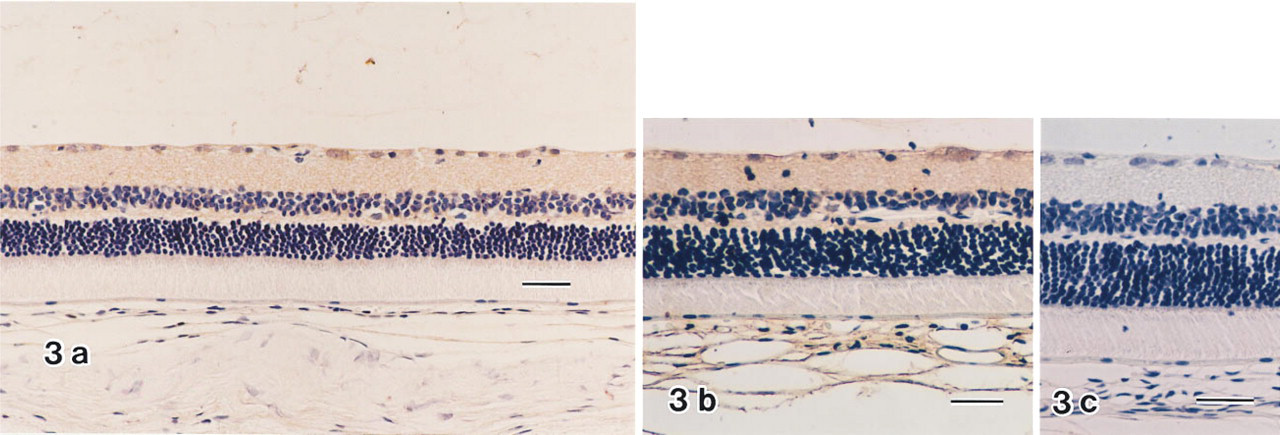
(
ADF induction in retinal pigment epithelial cells after photodynamic vascular thrombosis with or without OP-1206 a
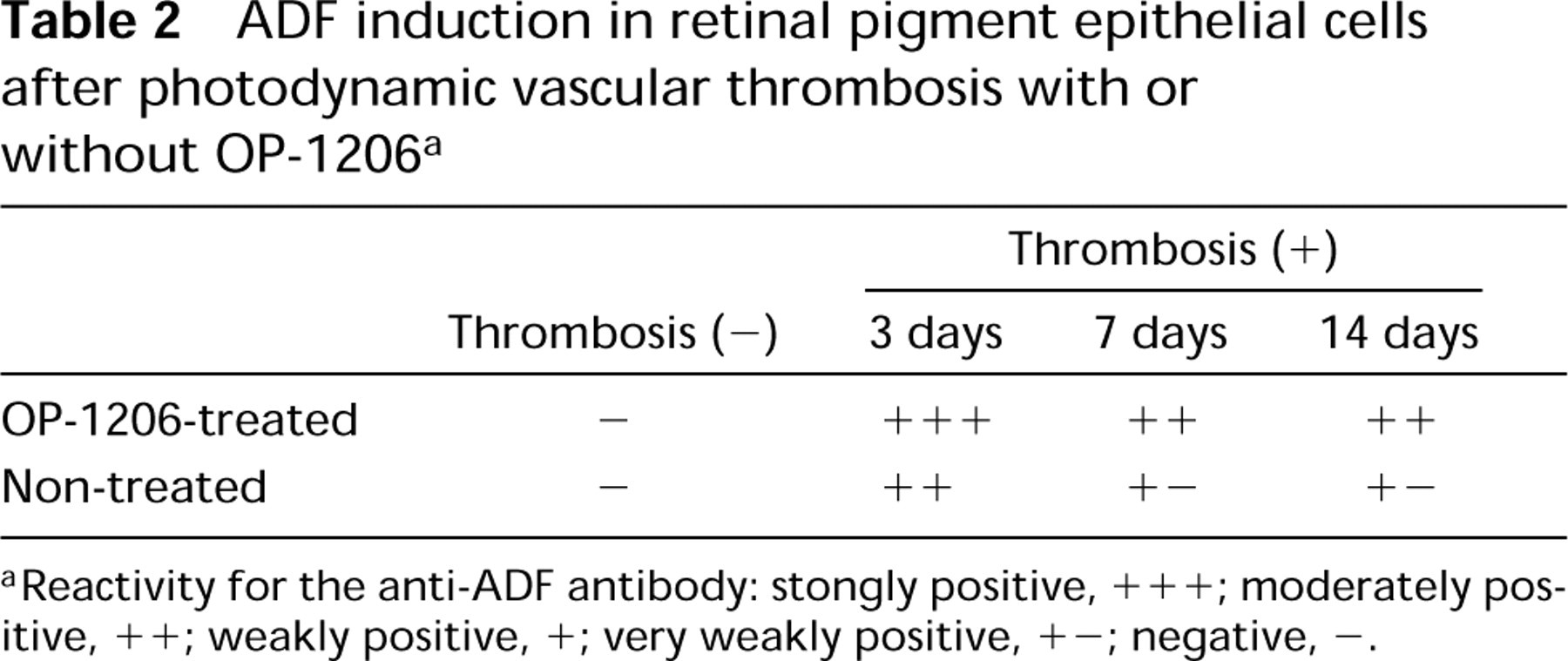
aReactivity for the anti-ADF antibody: stongly positive, +++; moderately positive, ++; weakly positive, +; very weakly positive, +-; negative, -.
Table 2 presents the grading of ADF immunoreactivity in the retinal pigment epithelial cells in the groups with photodynamic vascular thrombosis with or without OP-1206 treatment. This grading was done by a blinded observer.
ADF Immunoreactivity in Control Rats. In the OP-1206-treated retina without photodynamic retinal thrombosis (Group 3; Figure 3A), weak ADF immunoreactivity was localized to the internal limiting membrane, ganglion cell layer, and inner and outer plexiform layers. Little ADF immunoreactivity was observed from the inner nuclear layer to the retinal pigment epithelial cells. This localization was similar to that in the normal retina of Group 4 (Figure 3B). Moreover, no atrophic change could be observed in the retinas of any rats in these groups (Groups 3 and 4).
ADF immunoreactivity was not observed in sections incubated with non-immune IgG instead of the primary antibody (Figure 3C) or in sections incubated with KPBS in 1% BSA.
Discussion
In this study, we demonstrated that OP-1206 treatment suppresses the retinal damage induced by transient retinal ischemia and that ADF immunoreactivity was subsequently augmented in the retinal pigment epithelial cells of the OP-1206-treated rats. Previously, we found that ADF was strongly induced in these cells both under oxidative insult caused by ischemia/reperfusion in the rat and in culture when exposed to H2O2 (Gauntt et al. 1994; Ohira et al. 1994). ADF induction is speculated to be one of the defense reactions that protects the affected tissues (Iwai et al. 1992; Nakamura et al. 1992; Wakita et al. 1992; Yodoi and Uchiyama 1992; Fujii et al. 1991; Kusama et al. 1991; Yodoi and Turtz 1991; Tagaya et al. 1989). There are various reports regarding observations of the unique protective functions of ADF (Tomimoto et al. 1993; Matsuda et al. 1991). Moreover, the sensitivity of cultured retinal pigment epithelial cells against H2O2 was effectively reduced by preincubation with recombinant ADF (Gauntt et al. 1994). These findings support our hypothesis that ADF expression in the retinal pigment epithelial cells might be a part of the retinal autoprotective response against the oxidative stress caused by transient ischemia. In the OP-1206-treated rats, the correlation of the suppression of the retinal damage and the prolonged and augmented localization of ADF suggest that ADF may contribute to the suppression of retinal damage.
Although we have not confirmed in this study our hypothesis that the suppression of retinal damage by OP-1206 treatment depends on some unique cytoprotective function of OP-1206 that is involved in ADF induction, there are certain findings that support our hypothesis. In this study, we observed that there was no difference between the OP-1206-treated and non-treated groups in the duration of venous occlusion. This finding may be inconsistent with the presumption that some biological activities that PGE1 has been reported to possess, such as vasodilatation and anti-platelet functions, mainly contribute to suppression of retinal damage in this model. Moreover, this presumption cannot explain the phenomenon in which ADF, which has been reported to be induced by various oxidative stresses, was augmented in the OP-1206-treated retina.
On the other hand, we also found that ADF immunoreactivity was not augmented without oxidative insult despite treatment with OP-1206. This finding does not appear to support the hypothesis that OP-1206 may have a function of direct ADF induction. However, we found in in vitro study, using cultured retinal pigment epithelial cells, that the intracellular ADF induction by PGE1 requires the stimulation of a hyper-oxidant, H2O2 (personal observations). This finding suggests that the mechanism of ADF induction by PGE1 may be regulated by oxidative stimulation, although the mechanism has not been confirmed. Disablement of ADF induction in the rat retina by PGE1 alone may be due to this regulating mechanism. However, the finding that PGE1 has an ADF-inducible function only in pathogenetic conditions may more strongly support the hypothesis that PGE1 has a cytoprotective function involving ADF induction.
The retinal pigment epithelial cell has many functions through which it assists the photoreceptors. It phagocytizes and digests degenerating outer rod segments of photoreceptor cells. The outer segments of the photoreceptor cells contain high concentrations of polyunsaturated fatty acids, which are vulnerable to radicals and to reactive oxygen intermediates (Organisciak et al. 1985; Anderson et al. 1984; Fliesler and Anderson 1983). It is reasonable to assume that the effects of oxidative insult can be translated to the retinal pigment epithelial cells through phagocytosis of rod outer segments. Recently, retinal pigment epithelial cells have reported to generate and secrete various cytokines and growth factors such as interleukin-6 (IL-6), interleukin-8 (IL-8), and basic fibroblastic growth factor (bFGF), which has a trophic influence in the development and maintenance of the neural retina (Faktorovich et al. 1992; Elner et al. 1990, 1991; Li and Turner 1991; Park and Hollenberg 1989). Moreover, in the Royal College of Surgeons rat with inherited retinal dystrophy, degeneration of photoreceptors is prevented by translation of normal retinal pigment epithelial cells into the subretinal space (Li and Turner 1991). This effect extends beyond the boundaries of retinal pigment epithelium grafts, suggesting that a diffusable factor might be involved. These findings lead us to speculate that the suppression of damage on the inner retina observed in this study may be caused by production and secretion of ADF in retinal pigment epithelial cell as a protective response against oxidative insult. However, we could not confirm biologically the protective role and mechanism of retinal pigment epithelial cells in this study. Further studies are needed to test this hypothesis.
In summary, we found in this study that OP-1206 treatment can suppress the retinal tissue damage caused by transient ischemia/reperfusion and that ADF immunoreactivity was augmented in retinal pigment epithelial cells in OP-1206-treated rats. These findings prompt us to conclude that OP-1206 and PGE1 may have a cytoprotective function that is involved in ADF induction in retinal pigment epithelial cells. These interrelations and mechanisms must be tested more closely, and such studies are now in progress.
Footnotes
Acknowledgements
We thank Dr Akira Mitsui of the Aji-no-moto Central Laboratory for assistance and for providing the polyclonal antibody against ADF. Limaprost α-cyclodextrin clathrate was donated by Dainippon Pharmaceutical Company, Ltd., Osaka, Japan.
Supported by a Grant-in-Aid for Scientific Research from the Ministry of Education, Science and Culture of Japan (#06671769; AO).
