Abstract
CD44 is a multifunctional adhesion molecule that binds to hyaluronan (HA), type I collagen, and fibronectin. We investigated localization of CD44 and HA in mandibular condylar cartilage compared with the growth plate and the articular cartilage, to clarify the characteristics of chondrocytes. We also performed Western blotting using a lysate of mandibular condyle. In mandibular condyle, CD44-positive cells were seen in the surface region of the fibrous cell layer and in the proliferative cell layer. Western blotting revealed that the molecular weight of CD44 in condyle was 78 to 86 kD. Intense reactivity for HA was detected on the surface of the condyle and the lacunae of the hypertrophic cell layer. Moderate labeling was seen in cartilage matrix of the proliferative and maturative layer. Weak labeling was also seen in the fibrous cell layer. In growth plate and articular cartilage, HA was detected in all cell layers. However, chondrocytes of these cartilages did not exhibit reactivity for CD44. These results suggest that chondrocytes in the mandibular condylar cartilage differ in expression of CD44 from those in tibial growth plate and articular cartilage. Cell-matrix interaction between CD44 and HA may play an important role in the proliferation of chondrocytes in the mandibular condyle.
T
CD44 is an adhesion molecule distinct from cadherin and integrins (Underhill 1992; Goodison et al. 1999; Bajorath 2000; Turley et al. 2002). This protein functions as a hyaluronan (HA) receptor and exhibits homology in its extracellular domain with cartilage link and proteoglycan core proteins (Goldstein et al. 1989; Aruffo et al. 1990; Wolffe et al. 1990; Haynes et al. 1991). CD44 is involved in cell migration (Jacobson et al. 1984; Stamenkovic et al. 1989; Spessotto et al. 2002), metastatic spread (Günthert et al. 1991), and attachment of hematopoietic stem cells to stromal cells (Miyake et al. 1990). Biochemical and histochemical studies indicate that cartilage matrix contains HA, a ligand for CD44 (Asari et al. 1992,1994; Pakkinen et al. 1996; Melrose et al. 2002; Teramoto et al. 2003). Additionally, in vitro studies suggest that CD44 is expressed in chondrocytes (Ishida et al. 1997; Knudson 2003; Wong and Carter 2003). Ostergaard et al. (1997) reported that CD44 expressed in human osteoarthritic cartilage. However, in our previous studies, we did not observe CD44 immunoreactivity in chondrocytes of the growth plate cartilage or articular cartilage of tibia (Nakamura et al. 1995; Nakamura and Ozawa 1996).
In the present study we investigated distribution of CD44-positive chondrocytes and localization of HA in rat mandibular condyle to clarify their characteristics in the process of chondrocyte differentiation by comparison with tibial articular and growth plate cartilage.
Materials and Methods
All animal procedures were performed in accordance with The Guidelines for Animal Experiments, Okayama University Graduate School of Medicine and Dentistry.
Antibody against CD44
A cysteine-conjugated peptide corresponding to c-terminal of CD44 (DQFMTADETRNLQSVDMKIGV) was synthesized and coupled via a terminal cysteine residue to keyhole limpet hemocyanin according to Okamoto et al. (1999). This antigen was subcutaneously injected into rabbits, and antisera were collected. Specific antibody to CD44 was purified using Affi-Gel 10 (Bio-Rad; Hercules, CA) coupled with the peptide.
Protein Extraction, Sodium Dodecyl Sulfate (SDS)-Polyacrylamide Gel Electrophoresis and Western Blotting
Mandibular condyles of 8-week-old rats were resected and then cut into small pieces using razor blades. Extraction of organic material was performed over a 24 hr period at 4C using 1 ml of RIPA buffer (1% NP-40, 0.5% deoxycholic acid and 0.1% SDS in 50 mM Tris-HCl buffer, pH (8.2) containing 10 mg/ml leupeptin, 1 mM phenylmethylsulfonyl fluoride, and 2 mM sodium orthovanadate. The supernatant was separated by centrifugation at 14,000 × g for 10 min and concentrated with acetone. Precipitates were dissolved in 100 μl of sample buffer containing 4% SDS, 20% glycerol and 12% mercaptoethanol in 100 mM Tris-HCl (pH 6.8), followed by heating at 100C. SDS-polyacrylamide gel electrophoresis was performed using 7.5% polyacrylamide gel. Samples were electrophoresed at 150 V for 90 min and then transferred to nitrocellulose membrane. The membrane was immersed in 10% skim milk in 10 mM phosphate-buffered saline (PBS) for 1 hr to block nonspecific binding and was then washed with PBS containing 0.05% Tween 20. The membrane was incubated with rabbit anti-CD44 antibody diluted 1:1000 for 12 hr at 4C, followed by incubation with horseradish peroxidase (HRP)-conjugated anti-rabbit IgG (Sigma; St Louis, MO) for 1 hr at room temperature. Immunoreactivity was visualized using ECL Western blotting detection reagents (Amersham Pharmacia Biotech UK Ltd.; Buckinghamshire, England) according to the manufacturer's instructions.
Preparation of Tissue
Five 8-week-old Wistar rats were used for immunohistochemical and histochemical experiments. The rats were anesthetized with sodium pentobarbital and perfused through the left ventricle with 4% paraformaldehyde and 0.1% glutaraldehyde in 0.05 M phosphate buffer (pH 7.3). Mandibular condyles were resected, immersed in perfusion fixative for 12 hr at 4C, and then decalcified in 5% ethylenediamine-tetraacetic acid (pH 7.3) for 2 weeks at 4C.
Light Microscopic CD44 Immunohistochemistry and HA Histochemistry
Specimens were dehydrated in graded ethanol, embedded in paraffin, and cut into 7-μm-thick sections. For the detection of HA, dewaxed sections were treated with 0.1% trypsin (Sigma) in PBS for 15 min at room temperature or 2 units of chondroitinase ABC (Sigma) in 50 mM Tris-HCl buffer (pH 8.0) containing 60 mM sodium acetate for 1 hr at 37C. They were then immersed in PBS containing 10% bovine serum albumin (BSA) for 15 min and then incubated in mouse anti-rat CD44 monoclonal antibody (Pharmingen; San Diego, CA) diluted 1:500 and biotinylated hyaluronic-acid-binding protein (HABP) (Seikagaku; Tokyo, Japan) diluted to 2 μg/ ml for 12 hr at 4C. They were immersed in AlexaFluor-594-conjugated anti-mouse Ig G (Molecular Probes, Inc.; Eugene, OR) and AlexaFluor-488-conjugated streptavidin (Molecular Probes) diluted 1:200. Fluorescence of specimens was observed under an Axioplan 2 (Carl Zeiss; Oberkochen, Germany) with the appropriate filter combinations.
Electron Microscopic Immunohistochemistry
Approximately 50-μm-thick sections were cut using a Mi-croslicer (D.S.K.; Kyoto, Japan). These sections were incubated in anti-CD44 monoclonal antibody for 24 hr at 4C, followed by incubation with HRP-conjugated anti-mouse Ig G (Sigma) for 24 hr at 4C. Immunoreactivity was visualized by immersion in DAB-H2O2 solution. After postfixation with 1% OsO4 in 0.1 M phosphate buffer (pH 7.4) for 1 hr at 4C, the tissue slices were dehydrated in graded ethanol and embedded in Epon 812 (TAAB; Berkshire, England). Ul-trathin sections were cut using an Ultracut E ultramicrotome (Reichert-Jung; Vienna, Austria) and stained with 1% tannic acid and uranyl acetate. These sections were observed under a Hitachi H-800 transmission electron microscope (Hitachi; Tokyo, Japan) at an accelerating voltage of 100 kV.

Western blotting with anti-CD44 antibody under reduced condition. An intense active band at 78 to 86 kD is detected in the mandibular condyle extract. A weak band is also seen at 42 kD.
Controls
Control sections were incubated with normal mouse Ig G or without a primary antibody. Otherwise, immunohistochemi-cal procedures were performed in the same manner as for the non-control sections. Control sections for HA were digested with 25 mg/ml hyaluronidase (Sigma) in PBS at room temperature for 30 min prior to the incubation with HABP.
Results
Western Blotting
Anti-CD44 antibody reacted with a 78- to 86-kD band and a 42-kD band in the mandibular condyle extract. The reactivity of the 78- to 86-kD band was more intense than that of the 42-kD band (Figure 1).
Localization of CD44 and HA
The mandibular condylar cartilage is divided into four layers: (a) fibrous cell layer, (b) proliferative cell layer, (c) maturative cell layer, and (d) hypertrophic cell layer. We examined the anterior region of sagittal sections of the condyle because these layers were clearly distinguishable in that region.
CD44 immunoreactivity was most pronounced in the proliferative cell layer of mandibular condyle (Figure 2A). Reactivity was detected mainly along plasma membrane of chondrocytes in the proliferative cell layer. The superficial cells in the fibrous cell layer also exhibited moderate reactivity. Several cells in the fibrous cell layer and the maturative cell layer exhibited weak reactivity. Very little immunoreactivity was detected in cells of the hypertrophic cell layer. Strong HA staining was observed on the surface of the condyle and the lacunae of the hypertrophic cell layer. Moderate labeling was also detected in cartilage matrix of the proliferative and maturative layer. Weak labeling was seen in the fibrous cell layer (Figure 2B). Colocalization of CD44 and HA was observed in the surface region of condyle and the proliferative cell layer (Figure 2C).
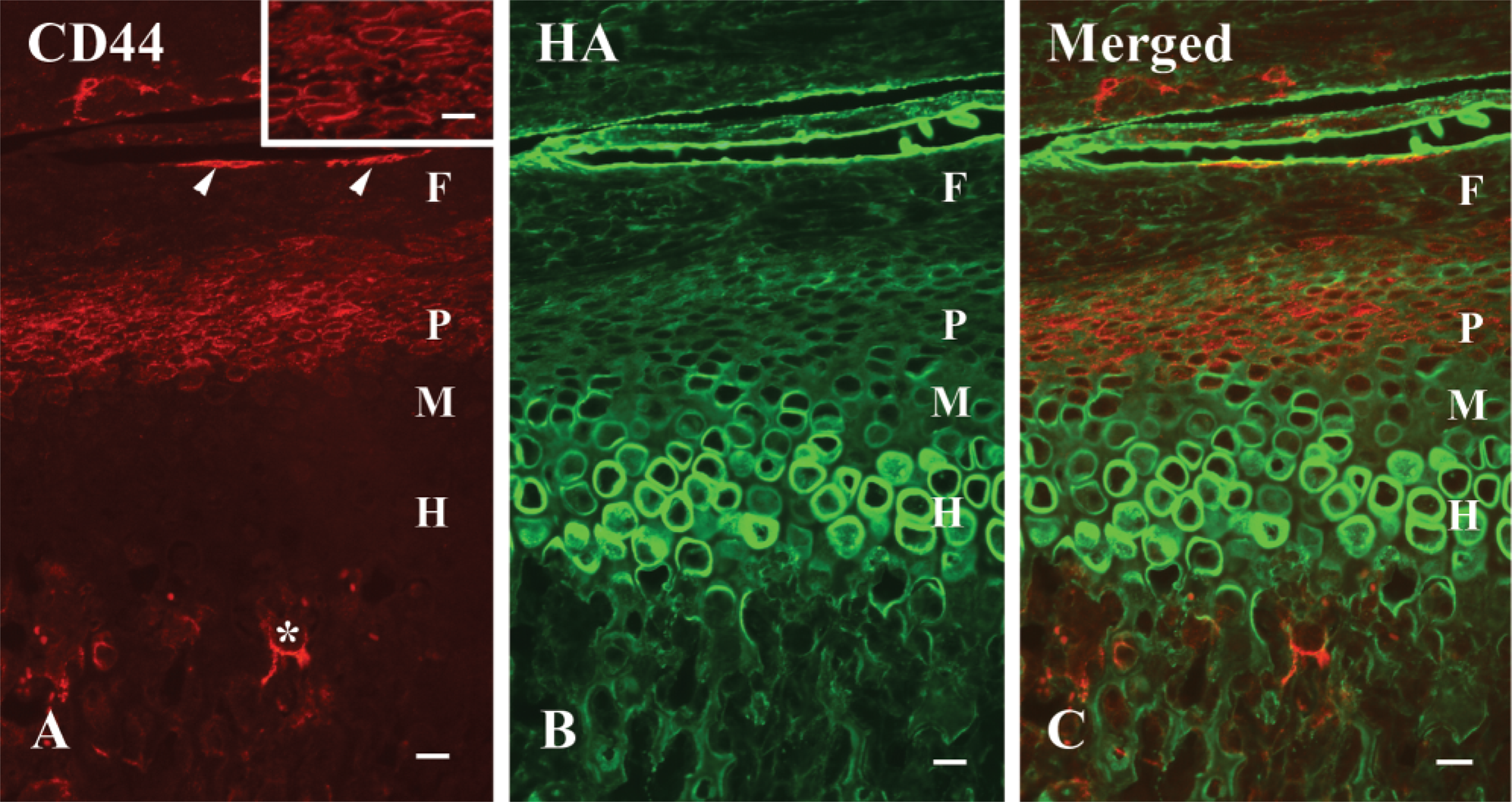
Fluorescent micrographs indicating CD44 and HA in a mandibular condyle. (
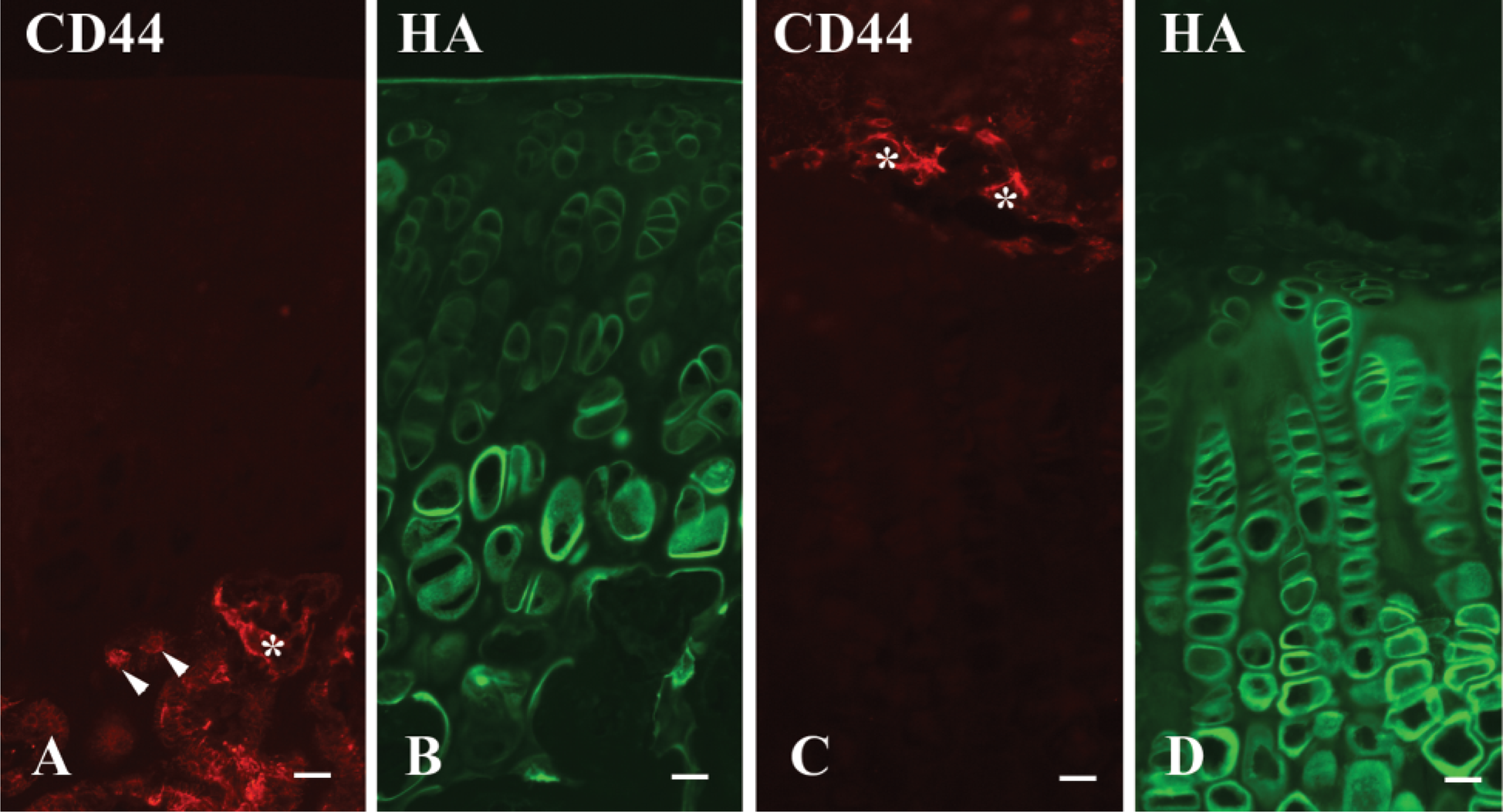
Fluorescent micrographs indicating CD44 and HA in an articular cartilage and a growth plate cartilage. (
In articular cartilage, no reactivity for CD44 was detected in chondrocytes (Figure 3A). Unmasking procedure, such as trypsin, chondroitinase ABC, and hyaluronidase pretreatment, did not change the stainability. HA labeling was most pronounced on the surface of the articular cavity and the hypertrophic cell layer. Other layers showed moderate positive reactivity (Figure 3B). The reactivity was intense in the pericellular matrix around chondrocytes.
In growth plate, no positive CD44 labeling was detected in chondrocytes at any stage of differentiation (Figure 3C). The staining pattern was not changed after unmasking procedure. HA labeling was observed in cartilage matrix of proliferative, maturative, and hypertrophic cell layers (Figure 3D). Particularly, pericellular matrix around chondrocytes showed intense reactivity.
No specific immunoreactivity was detected in the control sections. Staining by HABP was diminished by pretreatment with hyaluronidase.
Immunoelectron Microscopy for CD44
CD44 labeling was detected on the plasma membrane of the superficial cells in the fibrous cell layer (Figure 4A). Immunoreactivity was also detected on the plasma membrane of cells in the proliferative cell layer (Figure 4B). Mitotic cells exhibited CD44 immunoreactivity (Figure 4C). Some maturative chondrocytes adjacent to the proliferative cell layer showed weak CD44 reactivity.
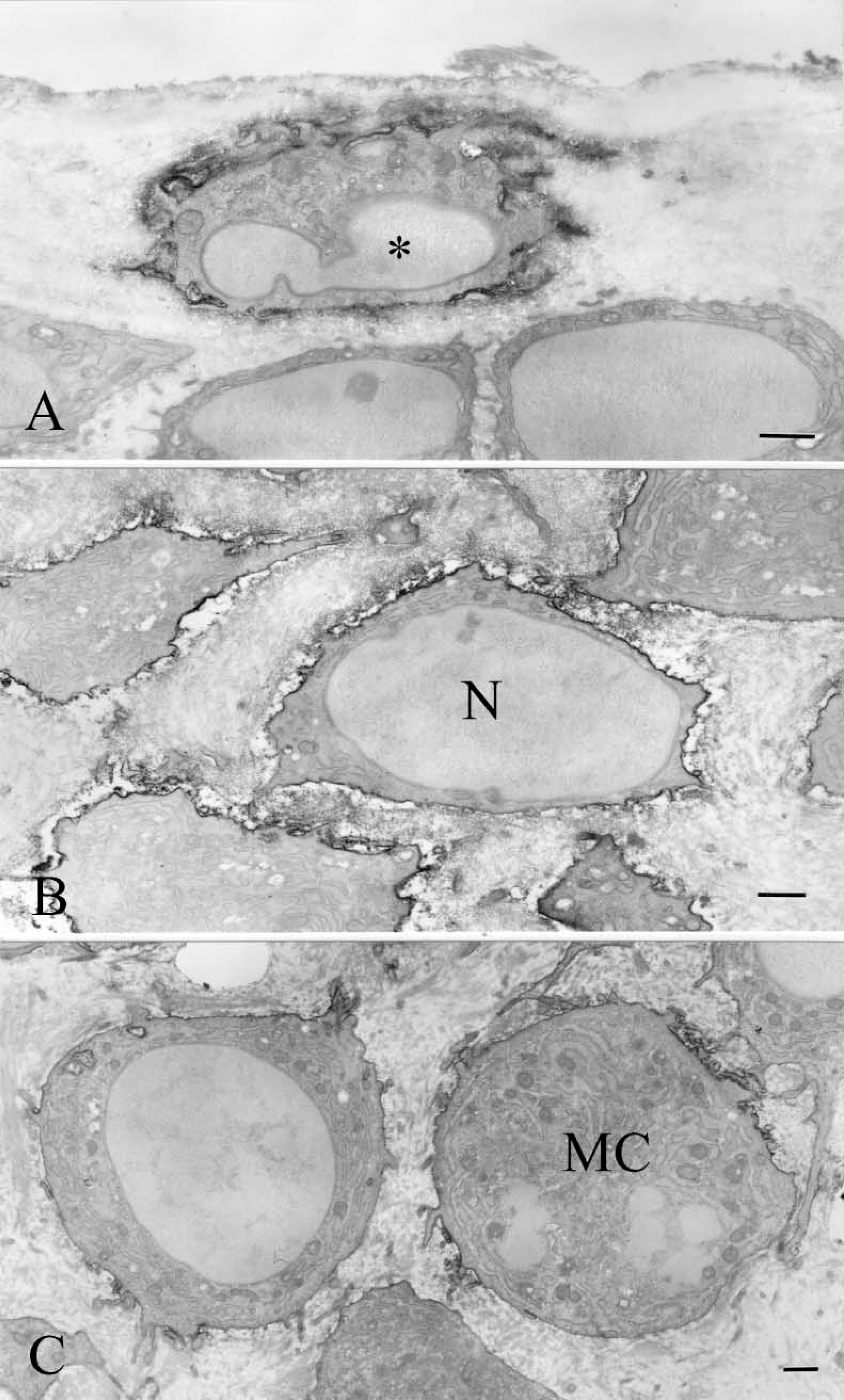
Electron micrographs indicating CD44 localization in a mandibular condyle. (
Discussion
In the present study, the pattern of CD44 expression in mandibular condylar cartilage differed from that of tibial cartilage. In mandibular condylar cartilage, CD44-positive cells were seen in the proliferative cell layer below the fibrous cell layer. However, chondrocytes in the proliferative zone of the growth plate and the articular cartilage of tibia did not exhibit CD44 immunoreactivity. Cranial bone and cartilage, including mandibular condylar cartilage, originate in neural-crest-derived mesenchyme (Helms and Schneider 2003). In contrast, bones and cartilage in limbs are derived from lateral plate mesoderm. Thus, mandibular condylar cartilage has a different developmental origin from tibial cartilage. The different patterns of CD44 expression observed in these two types of cartilage may be due to their different origins. CD44 is used as a marker of neural-crest-derived cells and embryonic stem cells (Corbel et al. 2000). Although differentiated cells such as osteoclasts and osteocytes also express CD44, CD44-positive cells in the proliferative cell layer of mandibular condyle may be undifferentiated mesenchymal cells.
The present Western blotting finding demonstrates that chondrocytes in the condylar cartilage express CD44. The molecular weight of a core protein of CD44 is reportedly ~50 kD in tunicamycin-treated cells (Okamoto et al. 1999). In addition, CD44 could be cleaved by metalloproteinase (Okamoto et al. 1999; Kajita et al. 2001). Therefore, the present 42-kD band corresponds to the core protein or a fragment of CD44. We assume that the 78- to 86-kD band corresponds to CD44 containing glycosaminoglycans. Chondroitin sulfate and/or heparan sulfate chains may be present on CD44 (Brown et al. 1991; Faassen et al. 1992). The type of glycosaminoglycan present in chondrocytes of the proliferative layer has yet to be determined.
Trypsin and chondroitinase ABC pretreatments revealed that HA localized in cartilage matrix at all stages of chondrocytes. Without an unmasking procedure, reactivity for HA was restricted in the surface of condyle and articular cartilage and in the pericellular matrix of the hypertrophic chondrocytes (data not shown). The unmasking procedure is essential for the detection of HA in cartilage as reported by Pakkinen et al. (1996). In the case of CD44 detection, chondroitinase ABC pretreatment slightly increased reactivity in the proliferative layer. However, unmasking procedure did not change the staining pattern for CD44.
Colocalization of CD44 and HA was observed in the proliferative layer of the mandibular condyle, although HA staining was less pronounced in the proliferative cell layer than on the surface of the condyle or the hypertrophic cell layer. CD44 is thought to participate in the internalization and degradation of HA (Knudson et al. 2002). Moreover, the CD44/HA pathway of chondrocytes seems to be involved in cellular signaling that induces cell survival (Brun et al. 2003), cell aggregation (Toole et al. 1972; Knudson et al. 1996), proliferation and expression of c-myc mRNA (Ishida et al. 1997). Furthermore, CD44 is involved in a cytoskeletal organization via the ERM family (Tsukita et al. 1994). Thus, cell-matrix interaction via CD44 and HA in the proliferative cell layer of the condylar cartilage may play an important role in the proliferation of chondrocytes.
We also found colocalization of CD44 and HA in the superficial area of the mandibular condyle. HA in the articular cavity is thought to reduce friction and facilitate joint movement. Thus, HA in the superficial area may play an important role in smooth movement and protection of articular cartilage. HA bound to CD44 in the superficial cells of the fibrous cell layer may serve as a reservoir of HA. Another possibility is that these cells internalize and degrade HA via a CD44-mediated mechanism.
We observed that HA was prominently present in the pericellular matrix around chondrocytes, as reported by previous research (Asari et al. 1992,1994; Pakkinen et al 1996; Melrose et al. 2002). However, chondrocytes in the growth plate and the articular cartilage did not exhibit expression of CD44. These results indicate that CD44 and HA are not involved in cell-matrix interaction of chondrocytes in these cartilage. HA mediates hydrostatic pressure by adsorbing water (Gakunga et al. 2000), indicating that HA is associated with increase in lacunae size of chondrocytes. Calcified cartilage matrix is subsequently resorbed by chondroclasts and osteoclasts in the process of endochondral ossification. These cells express CD44 on their plasma membrane (Nakamura et al. 1995; Nakamura and Ozawa 1996; Spessotto et al. 2002). Hence, HA in the pericellular matrix around chondrocytes may be involved in recognition and adhesion by chondroclasts and osteoclasts during cartilage resorption.
The differences between mandibular condylar cartilage and the articular and growth plate cartilage of long bones observed in the present study are clearly relevant to basic and pathological research of the temporomandibular joint.
Footnotes
Acknowledgements
This study was supported in part by a grant (No. 15591934) for scientific research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
We would like to thank Dr Noriyuki Nagaoka and Tomoko Yamamoto for their technical support.
