Abstract
CD117 is a transmembrane tyrosine kinase growth factor receptor expressed by a variety of normal human cell types, including germ cells, immature myeloid cells, and mast cells. To evaluate the pattern of CD117 expression in dogs and cats, we applied a polyclonal antibody on paraffin sections from 44 samples of normal tissues and 104 tumors. In both species, strong immunoreactivity was observed in mast cells, interstitial cells of Cajal, and in mast cell tumors. Among gastrointestinal mesenchymal neoplasms, tissues from five dogs and one cat revealed strong CD117 expression, enabling us to identify them as gastrointestinal stromal tumors (GISTs).
T
In human medicine, the expression of c-kit has been accurately evaluated in normal tissues and in a large number of neoplastic tissues by molecular techniques, flow cytometry, frozen section immunohistochemistry and, more recently, by paraffin section immunohistochemistry (Matsuda et al. 1993; Tsuura et al. 1994; Arber et al. 1998). Mast cells, melanocytes, breast ductal epithelium, seminiferous tubules, a subset of glial cells, ovarian stroma, follicles, corpora lutea, oocytes, and ICCs comprise the cellular substrate in which the immunohistochemical expression of CD117 is clearly evident, with a prevailing cytoplasmic distribution of the reactivity (Arber et al. 1998; Gibson and Cooper 2002). Among tumors, CD117 expression has been detected in acute myeloid leukemia, malignant melanomas, gastrointestinal stromal tumors (GISTs), small-cell lung carcinomas, germ cell tumors, and systemic mast cell disorders (Izquierdo et al. 1995; Arber et al. 1998; Miettinen and Lasota 2001).
In animal tissues, CD117 expression has been previously demonstrated by IHC in normal and neoplastic canine mast cells (London et al. 1996; Reguera et al. 2000), in the ICCs of dog, mouse, and rat (Komuro 1999), and in canine GISTs (Frost et al. 2003; Bettini et al. 2003). Nevertheless, CD117 distribution has not been extensively studied in canine tissues and has never been examined in the cat.
The present study was performed to further elucidate the IHC expression and distribution of the KIT protein in normal and neoplastic canine and feline tissues, using a polyclonal rabbit antiserum against c-kit, previously used in human (Tsuura et al. 1994; Arber et al. 1998) and canine tissues (London et al. 1996; Bettini et al. 2003; Frost et al. 2003) that specifically recognizes the C' terminus of the c-kit intracellular domains, highly conserved among species. Forty-four formalin-fixed, paraffin-embedded samples of normal tissues (30 dogs and 14 cats) from eight different organ sites (skin, mammary gland, gastrointestinal tract, testis, ovary, uterus, adrenal gland, cerebellum) and 104 samples of different kinds of tumors (74 dogs and 30 cats) were selected. Tumors were histologically classified according to WHO criteria on hematoxylineosin-stained sections. IHC was performed with an avidin-biotin–peroxidase method. Sections were incubated with 0.3% hydrogen peroxide in methanol for 20 min to block endogenous peroxidase activity and microwave-treated for antigen retrieval in citrate buffer solution at pH 6.0. After overnight incubation with the primary antibody (polyclonal rabbit anti-human KIT protein, CD117, DAKO; Dakopatts, Glostrup, Denmark) at a 1:500 dilution in PBS, the sections were incubated with the secondary antibody (anti-rabbit IgG conjugated with biotin) and then with the streptavidin-peroxidase complex. The sections were then immersed for 12 min in DAB chromogenic substrate solution (diaminobenzidine 0.02% and H2O2 0.001% in PBS), counterstained with Papanicolau hematoxylin, dehydrated, and mounted with DPX (Fluka; Riedel-de Haën, Germany). As positive control, the reaction was tested on mast cells in an appropriate tissue section from normal human skin and subcutaneous tissue. As negative control, PBS was used in replicated sections instead of the primary antibody. Tumors were considered positive if more than 5% of neoplastic cells showed immunoreactivity. The immunostaining results were recorded with respect to intensity (strong, moderate, or weak), distribution (focal <30% of cells; intermediate >30% but <70% of cells; and diffuse >70% of cells) and cellular localization of the immunostaining (cytoplasmic, nuclear, membranous, paranuclear, in the luminal side).
The IHC results for CD117 expression are summarized in Tables 1 and 2.
Immunohistochemical detection of CD117 in canine and feline normal tissues compared to human immunoreactivity a
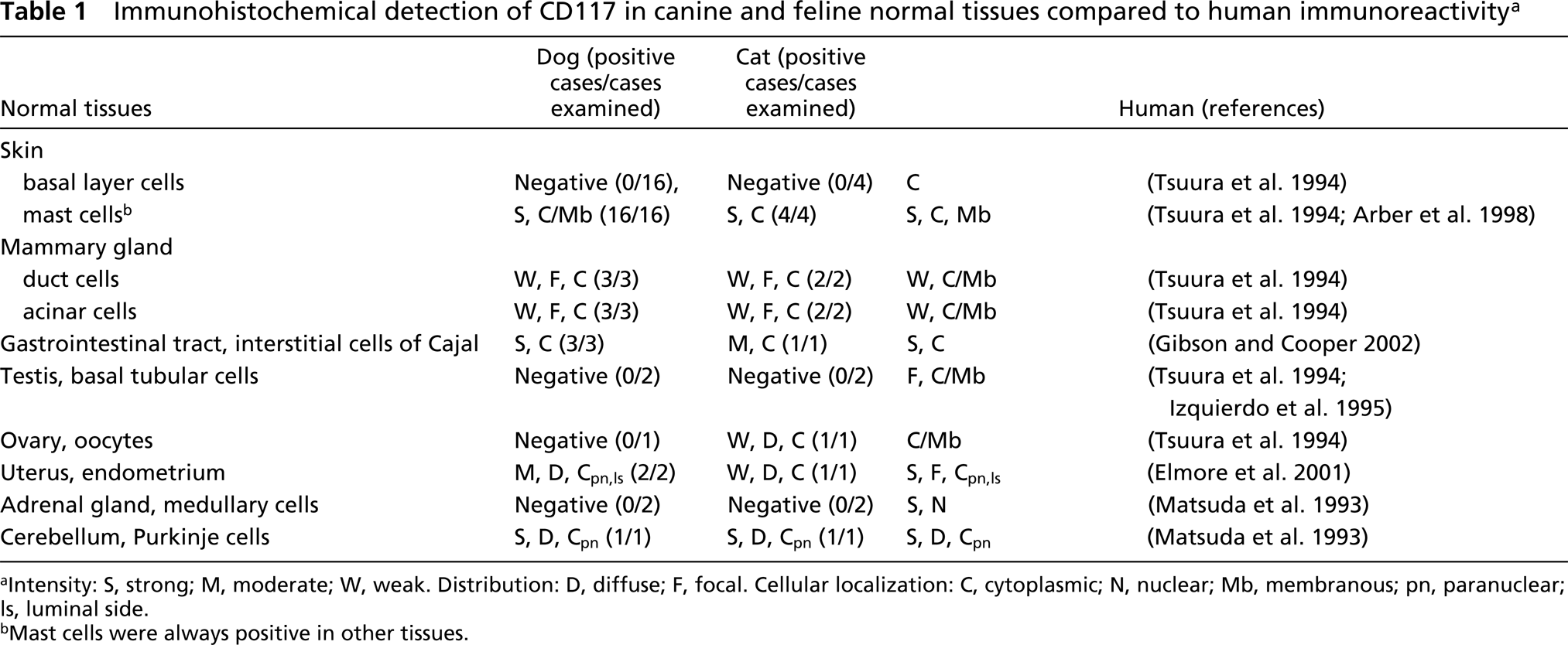
Intensity: S, strong; M, moderate; W, weak. Distribution: D, diffuse; F, focal. Cellular localization: C, cytoplasmic; N, nuclear; Mb, membranous; pn, paranuclear; ls, luminal side.
Mast cells were always positive in other tissues.
Immunohistochemical detection of CD117 in canine and feline neoplastic tissues compared to human immunoreactivity a
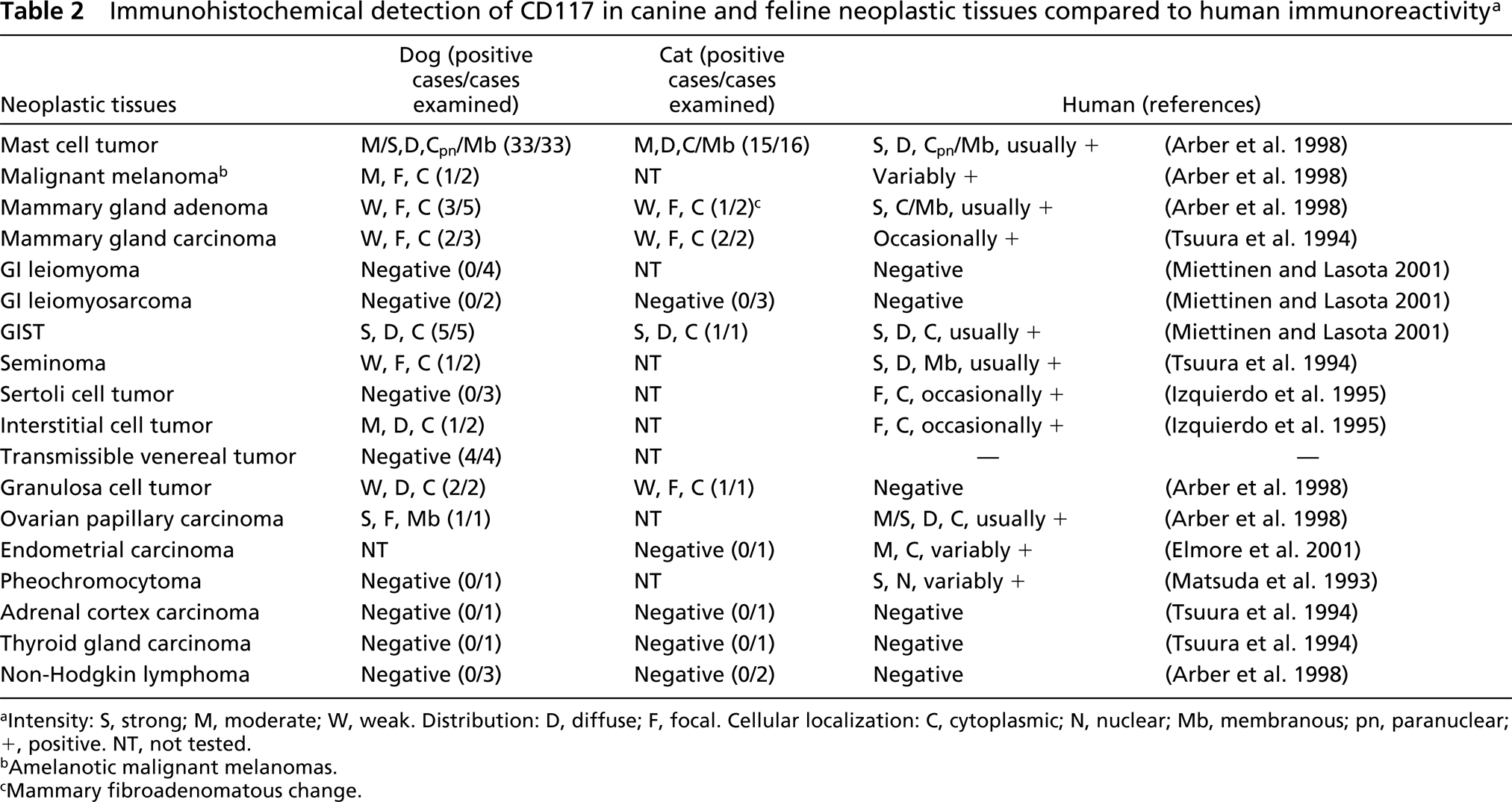
Intensity: S, strong; M, moderate; W, weak. Distribution: D, diffuse; F, focal. Cellular localization: C, cytoplasmic; N, nuclear; Mb, membranous; pn, paranuclear; +, positive. NT, not tested.
Amelanotic malignant melanomas.
Mammary fibroadenomatous change.
Among normal tissues (Table 1), strong CD117 expression was observed in both species, in mast cells (membrane and cytoplasmic, diffuse and paranuclear labeling), Purkinje cells of the cerebellum (cytoplasmic paranuclear labeling) (Figure 1A), and ICCs of the gastrointestinal tract (cytoplasmic labeling) (Figure 1B). Weak to moderate cytoplasmic staining was also detected in ductal and acinar epithelial cells of the mammary gland (dog), oocytes (cat), and endometrium (dog, cat). In one of two dogs the luminal side of the cytoplasm of endometrial cells showed scant immunoreactivity (Figure 1C), with a pattern similar to that seen in human tissues (Elmore et al. 2001).
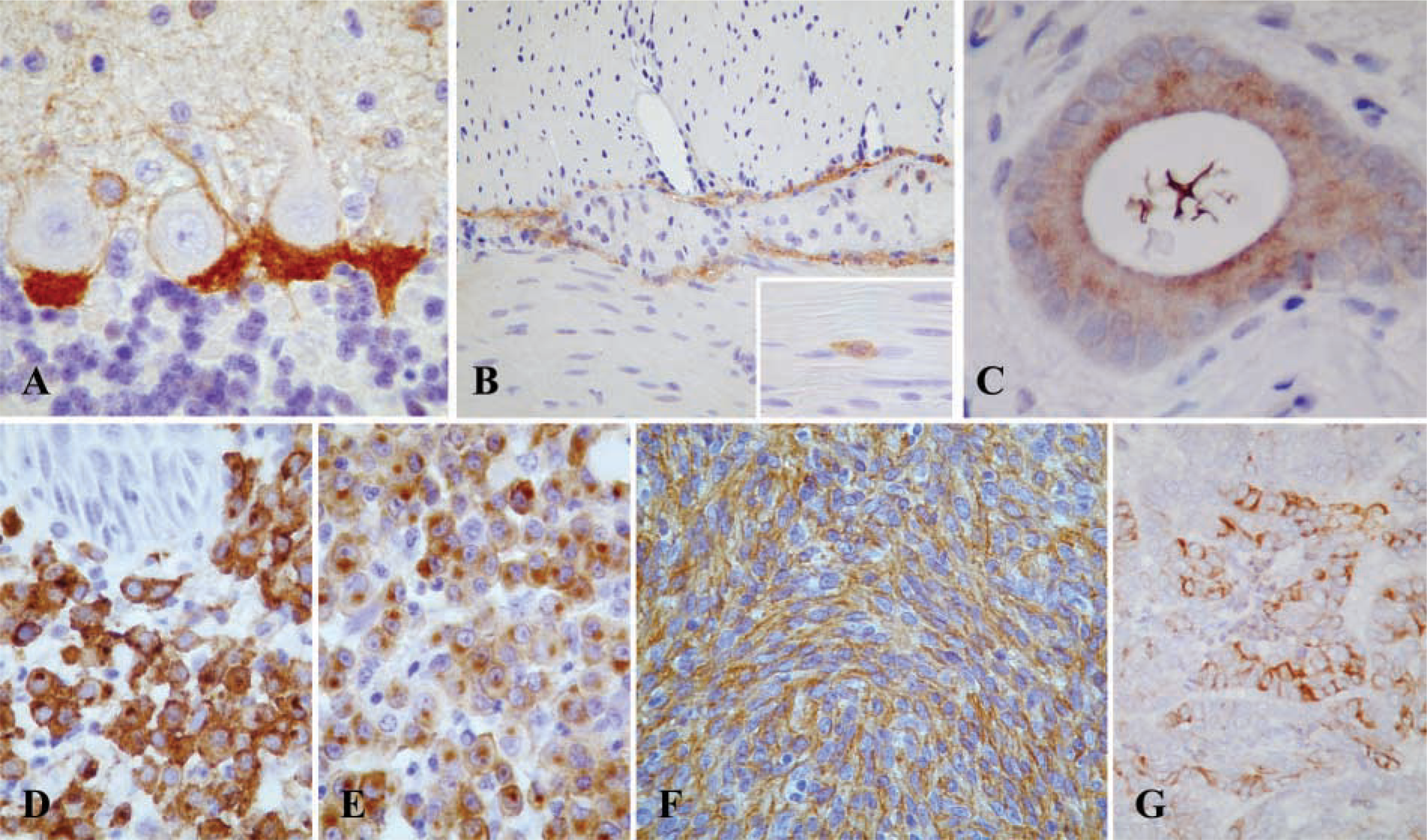
Immunolocalization of CD117 in normal (
Among neoplastic tissues (Table 2), the most relevant CD117 reactivity was observed in mast cell tumors (Figures 1D and 1E), which showed a particularly strong and diffuse immunopositivity (33/33 dogs, membrane and cytoplasmic, mostly paranuclear, labeling; 15/16 cats, membrane and cytoplasmic labeling), and in a subset of gastrointestinal mesenchymal tumors (Figure 1F), i.e., GISTs (5/13 dogs, granular and diffuse cytoplasmic labeling, sometimes with dot-like accentuation; 1/4 cats, diffuse cytoplasmic labeling). Furthermore, a focally moderate to strong membranous staining was observed in a case of canine ovarian papillary carcinoma (Figure 1G). Benign and malignant mammary tumors, melanomas, seminomas, interstitial cell tumors, and granulosa cell tumors stained weakly and inconstantly in both species. Non-Hodgkin lymphomas (dog, cat), pheochromocytoma (dog), adrenal cortical carcinoma (dog), thyroid gland carcinomas (dog, cat), endometrial carcinoma (cat), leiomyomas (dog), leiomyosarcomas (dog, cat), and transmissible venereal tumors (dog) were negative.
Our results have shown that CD117 IHC in normal tissues of dogs and cats has a similar pattern of reactivity. The KIT receptor is expressed by a limited number of cells, with a distribution comparable to that of human tissues in mast cells, ICCs, Purkinje cells, endometrium, and epithelial cells (ducts and acini) of the mammary gland. Nevertheless, epidermal basal cells, germinal cells of the testis, oocytes, and medullary cells of adrenal gland stain moderately to strong in human tissue, whereas in our samples these compartments did not react to CD117 IHC.
Mast cell tumors expressed CD117 with constant and significant intensity. Neoplasms of mast cells are the most common cutaneous malignant tumors of dogs, representing between 7–21% of all tumors, an incidence much higher than that found in humans (London et al. 1996). In our cases, CD117 immuno-positivity was found in both cell membrane and cytoplasm, sometimes with paranuclear aggregates of focal strong accentuation of the positivity. Although further studies are needed to elucidate the correlation between the pattern of IHC expression of KIT and the biological behavior of mast cell tumors, our preliminary data suggest that, in the dog, the cytoplasmic-paranuclear staining is correlated with a significantly shortened survival time (personal observation).
In gastrointestinal (GI) tract pathology, a subset of primary mesenchymal tumors, called GISTs, has been recently recognized and defined as CD117-positive spindle or epithelioid neoplasms (Miettinen and Lasota 2001). GISTs have been also documented in dogs, but not yet in cats, by their KIT immunopositivity (Bettini et al. 2003; Frost et al. 2003), and histologically they appeared as densely packed spindle cells, quite similar in features to smooth muscle cell tumors and commonly misdiagnosed as leiomyomas or leiomyosarcomas. The ICCs, a complex cellular network postulated to act as pacemaker cells of the GI tract and responsible for slow and rhythmic contractions of the smooth muscle, are believed to represent the cells of origin for GISTs (Miettinen and Lasota 2001). Therefore, as in humans, CD117 may be a useful diagnostic marker for canine and feline GISTs.
In our study, other sporadic examples of tumors have shown weak to moderate focal staining for CD117 (benign and malignant mammary tumors, amelanotic melanomas, testicular and ovarian tumors). In human pathology, seminoma is another tumor, in addition to GISTs and mast cell disorders that reveals constant strong, diffuse, and membranous KIT positivity, whereas in our results only one case (of two) disclosed weak, focal and cytoplasmic immunore-activity. However, the meaning of these discrepancies between human and animal findings is not still clear and requires further investigation. In a previous report (Matsuda et al. 1993), pheochromocytoma also showed a significant positivity for CD117 (seemingly nuclear), but this could be related to the use of different antibodies (monoclonal vs polyclonal) because in later studies (Tsuura et al. 1994; Arber et al. 1998), by the use of a polyclonal antibody, this neoplasm did not react against CD117.
The results of our study show that CD117 can be considered a reliable and useful marker in canine and feline tissues. Therefore, it may play a decisive role as a diagnostic marker for canine and feline GISTs and as a significant prognostic factor for canine and feline mast cell tumors.
Footnotes
Acknowledgements
We are grateful to Dr Claudio Ceccarelli for technical assistance.
