Abstract
We have shown that dopamine (DA), an inhibitor of prolactin secretion from anterior pituitary lactotrophs, stabilizes the cortical actin cytoskeleton. DA-induced cortical actin stabilization is accompanied by cytoplasmic actin cable disassembly and cell rounding up. Our aim was to identify the mechanisms involved in DA-induced stabilization of the lactotroph's actin cytoskeleton. Here we show that DA increased the association of myosin II with the cell cortex, suggesting that DA facilitates actin-myosin interaction to stabilize cortical actin filaments. This notion was supported by the finding that inhibitors of actin-myosin interaction blocked DA-evoked morphological responses. In addition, our results showed that DA-induced myosin association with the cell periphery may be mediated by inhibition of Rac1/Cdc42-dependent pathways, whereas, DA-induced cytoplasmic actin filament disassembly may be mediated by the inhibition of MLCK- and RhoA-dependent pathways. In conclusion, the present results provide evidence that myosin II is involved in the DA-induced remodeling of actin filaments in lactotrophs, and that DA-induced cortical actin filament assembly and stabilization involve the translocation of myosin II to the cell cortex. This effect requires, among other things, inhibition of the Rac1/Cdc42-dependent signaling pathway.
D
Actin dynamics are controlled by actin-binding proteins. Among the several actin-binding proteins that can be involved in the stabilization of cortical actin filaments, we have chosen myosin II. This choice is based on the following facts. (a) Myosin is involved in actin-membrane interactions and has been implicated in the control of actin filament cross-linking (Wilson et al. 2001) and of actin filament stiffness (Cai et al. 1998). (b) Myosin is implicated in the regulation of cell shape, cell contractility (García et al. 1999; Zeng et al. 2000), cell volume (Nilius et al. 2000), and cell spreading (Cramer and Mitchison 1995). Non-muscle myosin II consists of two 200-kD heavy chains (MHC) non-covalently bound to two pairs of light chains, of which one pair is essential and the other pair is regulatory. In mammalian cells, non-muscle myosins (A and B) are regulated mainly by phosphorylation of their regulatory light chains (MLC). This phosphorylation allows the myosin ATPase to be activated by actin, and therefore it constitutes a critical regulatory mechanism for actin dynamics.
Here we studied the role of myosin II in DA-induced cortical actin remodeling in cultured lactotrophs. We aimed to test the hypothesis that the remodeling of cortical actin filaments by DA requires an interaction between actin and myosin. The present data show that DA increases the association of myosin II with the cell cortex and that inhibition of actin-myosin interaction blocked DA-induced morphological effects in lactotrophs. In addition, our results indicate that this effect may require, among other events, inhibition of the Rac1/Cdc42-dependent signaling pathway.
Materials and Methods
Preparation of Anterior Pituitary Cell Cultures and Lactotroph-enriched Cell Cultures
Randomly cycling Sprague-Dawley female rats (Charles River; St Constance, PQ, Canada) were used as a source of anterior pituitary cells. The experimental protocol was approved by the Université de Montréal Animal Care Committee. Anterior pituitary lobes were dissected, diced in small pieces, and dispersed into a single cell population by incubation with Mg2+/Ca2+-free Locke's solution (154 mM NaCl, 2.6 mM KCl, 2.15 mM K2HPO4, 0.85 mM KH2PO4, 10 mM HEPES, 10 mM glucose, pH 7.2) containing 0.15% trypsin, 0.3% collagenase D, and 0.3% BSA for 2-3 hr at 37C. Digestion was stopped by addition of a volume of DMEM containing 0.3% of soybean trypsin inhibitor. Preparation of lactotroph-enriched cultures was performed by using a discontinuous Percoll gradient as described by Burris and Freeman (1993), with slight modifications (Nguyen et al. 1999). The lactotroph cell-enriched layer was localized in the interface 50%-35% (79 ± 7% lactotrophs) and was re-suspended in culture medium (DMEM supplemented with 2.5% fetal calf serum, 12.5% horse serum, antibiotics, and antifungi). When cells were incubated with DA, the medium was supplemented with ascorbic acid (100 μM final concentration). Cells were plated on poly-
Preparation of the Membrane Fraction
Lactotroph-enriched cell cultures were rinsed with cold PBS (137 mM NaCl, 3 mM KCl, 8 mM Na2HPO4, 1.5 mM KH2PO4, pH 7.4). Next, lysis buffer (50 mM Tris-HCl, pH 7.4, 1 mM MgCl2, 1 mM EDTA, 1 mM PMSF, 2 μg/ml leupeptin, 2 μg/ml aprotinin) was added to the dishes and cells were scraped off and homogenized. Homogenates were centrifuged at 600 rpm (Beckman GS-6R; Beckman Canada, Mississauga, ON, Canada) for 4 min. Pellets were discarded and supernatants were centrifuged at 15,000 × g for 25 min (Beckman microfuge E). Pellets corresponding to the membrane-enriched fraction were rinsed with lysis buffer and re-suspended in electrophoresis buffer. Proteins in the supernatants were precipitated with 10% TCA, washed with acetone, and resuspended in electrophoresis buffer.
Electrophoresis and Immunoblotting
Proteins were separated by SDS-PAGE and transferred onto nitrocellulose membranes. Membranes were saturated with BLOTTO medium and tested for the presence of MLC or Rac-1 with specific antibodies diluted in PBS containing 5% skim milk: monoclonal anti-MLC (1:500 dilution), monoclonal anti-Rac-1 (1:200 dilution). The secondary antibody was coupled to HRP. Antigen-antibody complexes were detected by enhanced chemiluminescence.
Treatment of Lactotroph Cells with Clostridium botulinum C3 Exoenzyme/Lipofectamine Mixture
C3 exoenzyme-containing Lipofectamine liposomes were prepared by incubating 1 volume solution A (10 μl lipofectamine/100 μl OPTI-MEM) with 1 volume solution B (10 μg C3 exoenzyme/100 μl OPTI-MEM) for 45 min at room temperature (RT). The resulting mixture was diluted 1:5 with serum-free OPTI-MEM. Cells were exposed to this medium for 2 hr at 37C. By using calcein as a fluorescent tracer we established that cells had been exposed to the lipofectamine liposomes in a serum-free medium, otherwise there was very low incorporation of the dye, and that of the living lactotroph cells, 98% incorporated the dye. The actin cytoskeleton and the lactotroph cell shape were not affected by either incubation with lipofectamine liposomes or serum starvation for 2 hr.
Fluorescence Microscopic Studies
After the specific treatment, cells were rinsed and immediately fixed with 3.7% formaldehyde, permeabilized with acetone, and processed for fluorescence microscopy as previously described (Carbajal and Vitale 1997). Cells were incubated for 1 hr at RT with 3% non-fat milk in PBS to block unspecific labeling. To analyze F-actin and myosin II distribution in cultured lactotrophs, we triple-labeled the cells by incubating them with an MLC monoclonal antibody (1:100 dilution) together with a PRL antibody (1:1500 dilution). Next, the cells were incubated with FITC-conjugated anti-mouse IgM (1:150 dilution), AMCA (7-amino-4-methyl-coumarin-3-acetic acid)-conjugated anti-rabbit IgG (1:400 dilution), and rhodamine-phalloidin (1:1000). After rinsing with PBS, coverslips were mounted in PBS:glycerol (1:1) containing 5% 1,4 diazabiscyclo (2,2,2) octane (DABCO). Cells were observed with a Carl Zeiss Axioskop 2 fluorescence microscope. We chose to use anti-MLC to label myosin II rather than antibodies against the heavy chains to avoid potential problems arising from a different distribution of the two non-muscle cell myosin heavy chain isoforms. To evaluate the impact of the treatments on lactotroph morphology, the shape, i.e., rounded vs flattened, of the PRL-positive cells was recorded as described previously (Nguyen et al. 1999). In addition, the distribution of F-actin and MLC in cells subjected to different treatments was evaluated. All these procedures, which were done without knowing the type of treatment applied to the cells (single-blind design), were performed in 100 PRL-immunopositive cells per coverslip. Therefore, every final datum for a given experimental condition is the result of the observation of not less than 600 lactotrophs from at least three different experiments. Photographs were taken with 400 ASA TMAX Kodak film.
Statistics
Data were evaluated by one-way ANOVA and differences between means were analyzed by the Keuls multiple-range test or the Student's í-test according to the number of groups.
Sources of Reagents
Enzymes for anterior pituitary cell dispersion and chemiluminescence kits were purchased from Roche (Laval, QC, Canada). DMEM and OPTI-MEM were purchased from GIBCO-BRL (Burlington, ON, Canada). Percoll was from Pharmacia (St Anne de Bellevue, QC, Canada). Antibiotics, protease inhibitors, soybean trypsin inhibitor, poly-
Results
Most lactotrophs incubated in a DA-free medium have a polygonal shape (Nguyen et al. 1999). Incubation of these flat cells with a medium containing different concentrations of DA, from 50 to 1000 nM, for 48 hr causes cell rounding up (Nguyen et al. 1999). To further characterize the impact of DA in the morphology of lactotrophs, we performed a time course study. Lactotrophs were isolated, incubated in a DA-free medium for 24-48 hr, and next treated with 100 nM DA for increasing periods of time (Figure 1). Most cells incubated in the absence of DA were polygonal (Figure 1, time 0). When DA (100 nM final concentration) was added to the medium, there was an increase in the number of rounded lactotrophs. Within 2 hr of exposure to DA there was a significant increase in the percentage of rounded cells. After 4 hr in the presence of DA, 80% of the cells were rounded and this percentage did not increase even with longer exposures to DA (Figure 1).
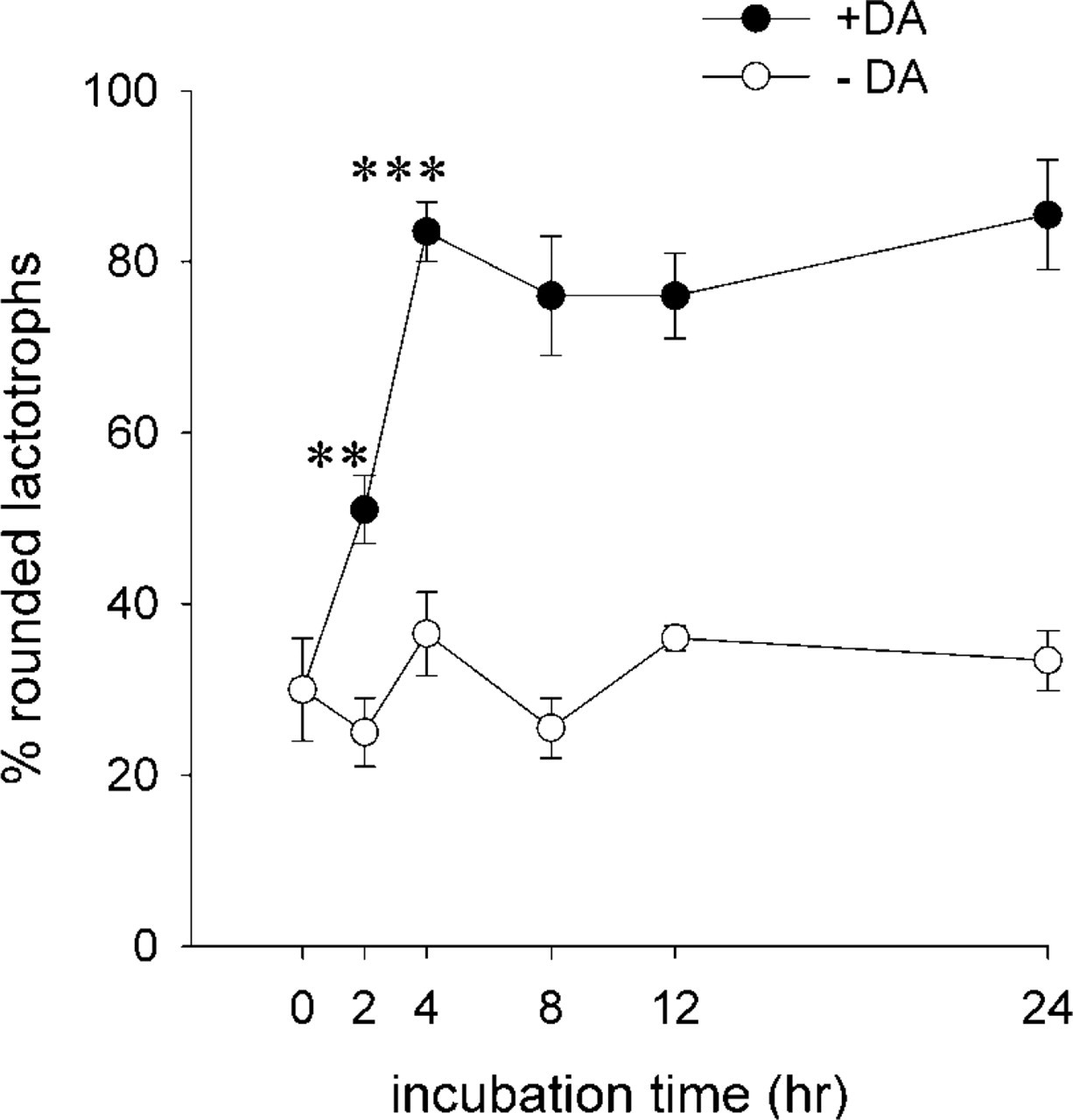
Time course of DA-induced morphological changes in cultured lactotroph cells. Lactotroph-enriched cultures incubated in DA-free culture medium were treated either with medium alone (∘) or containing 100 nM DA (•) for increasing periods of time. After each period, preparations were processed for fluorescence microscopy with PRL antibodies and rhodamine-phalloidin to analyze the lactotroph shape. One hundred lactotroph cells (PRL-positive cells) were analyzed and their shape defined as rounded or flattened. The percentage of rounded lactotrophs was then calculated. Each value (mean ± SEM) corresponds to the analysis of nine coverslips (900 cells). The experiment was repeated three times. ∗∗p<0.005 2 hr +DA vs 2 hr -DA; ∗∗∗p<0.0005 4 hr +DA vs 4 hr -DA.
Most changes in cell shape are a consequence of changes in the organization of the actin cytoskeleton. We have previously shown that DA, at concentrations ranging from 50 to 1000 nM, has noticeable effects on the lactotroph's actin cytoskeleton. These effects consist mainly of the reassembly and stabilization of cortical actin filaments (Carbajal and Vitale 1997) and of the disassembly of cytoplasmic actin filaments (Nguyen et al. 1999). The molecular mechanisms underlying these DA-evoked morphological effects remain to be elucidated. We investigated here whether the actin-crosslinking protein myosin II participates in DA-induced actin remodeling and cell retraction in lactotrophs. Fluorescence microscopic studies were performed in cells triple-labeled to visualize actin and myosin distribution in lactotrophs. Cells were incubated in DA-free medium for 48 hr and then they were challenged with 100 nM DA for 0 hr (Figures 2A, 2A′, and A”), 2 hr (Figures 2B, 2B′, and 2B”) or 4 hr (Figures 2C, 2C′, and 2C”). As shown in Figure 2A, lactotrophs incubated in the absence of DA were flat and PRL staining was granular and associated mainly with the Golgi region. These cells possessed a cortical and cytoplasmic actin cytoskeleton (Figure 2A′, arrows); MLC staining displayed a diffuse cytoplasmic distribution (Figure 2A”, arrowheads). Addition of DA to the medium modified the lactotroph cell shape. Figures 2B and 2C show that the presence of DA induced the lactotrophs to round up. DA-induced lactotroph cell rounding up was accompanied by the disassembly of cytoplasmic actin cables and by the maintenance of cortical actin filaments (Figures 2B′ and 2C, arrows). In addition, there was a concomitant redistribution of myosin II (MLC) from a diffuse cytoplasmic staining in flat cells (Figure 2A”, arrowheads) to a cortical ring in DA-treated cells (Figures 2B” and 2C”, arrowheads). A time course study of DA-evoked myosin II (MLC) redistribution revealed that 2 hr after the addition of DA there was a significant increase in the number of lactotrophs with MLC being associated with the cell periphery, which reached a plateau after 4 hr in the presence of DA (Figure 2D). To further characterize the association of myosin II with the cell cortex, membrane-enriched fractions were prepared from cell cultures treated with 100 nM DA for 0, 1, 2, and 4 hr and subjected to immunoblotting analysis with MLC antibodies. These analyses showed that there was an increased association of MLC with membranes after a 2-hr incubation period with DA (Figure 2E).
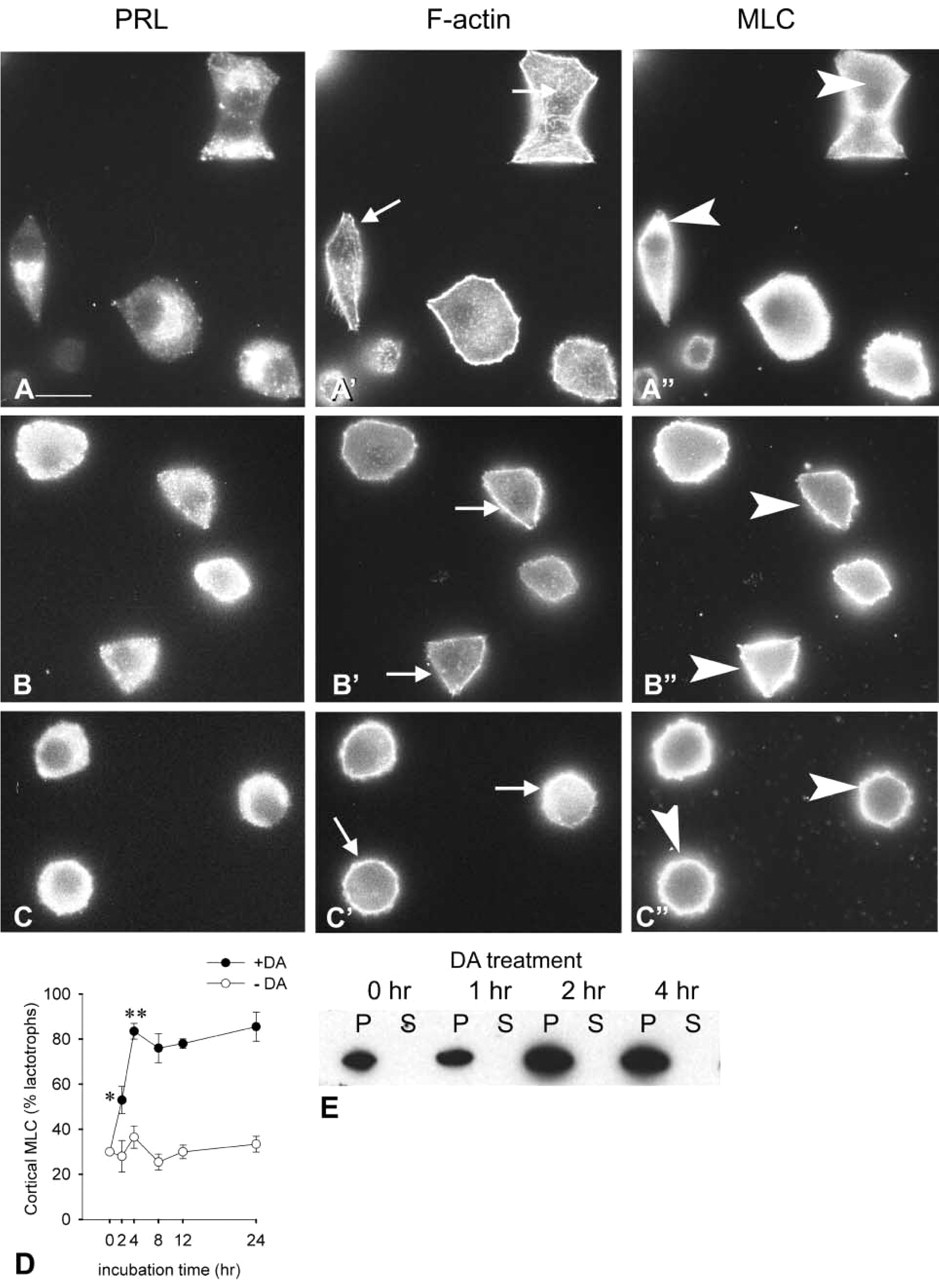
Effect of DA on lactotroph cell shape, F-actin organization, and myosin II subcellular distribution. Lactotroph cell-enriched cultures were incubated for 48 hr in DA-free culture medium (
To investigate whether myosin II is involved in DA-induced F-actin reorganization, cells were treated with 2,3-butanedione 2-monoxime (BDM) an inhibitor of actin-myosin interaction. Cells cultured in DA-free medium were treated with 15 mM BDM for increasing time periods (30 min to 8 hr) either in the absence or in the presence of DA. As described above, lactotrophs incubated in DA-free medium possessed a cortical and cytoplasmic actin cytoskeleton, diffuse cytoplasmic MLC staining, and a polygonal shape (Figures 3A and 3A′). Lactotrophs incubated in the presence of BDM displayed diffuse cytoplasmic F-actin staining (Figure 3B). MLC labeling was cytoplasmic and associated with the perinuclear region (Figure 3B′). Although the cell limits are not easy to define, it is apparent that, despite the disorganization of the cytoskeleton, the cells remained polygonal. When cells were simultaneously incubated with BDM and DA, the distributions of F-actin and MLC were similar to those observed in cells incubated with BDM alone. There was strong MLC labeling in the perinuclear region cells incubated with DA + BDM (Figure 3C). DA + BDM-treated cells remained flattened. Figure 3E shows the time course of the inhibition of DA-induced cell rounding up by BDM.
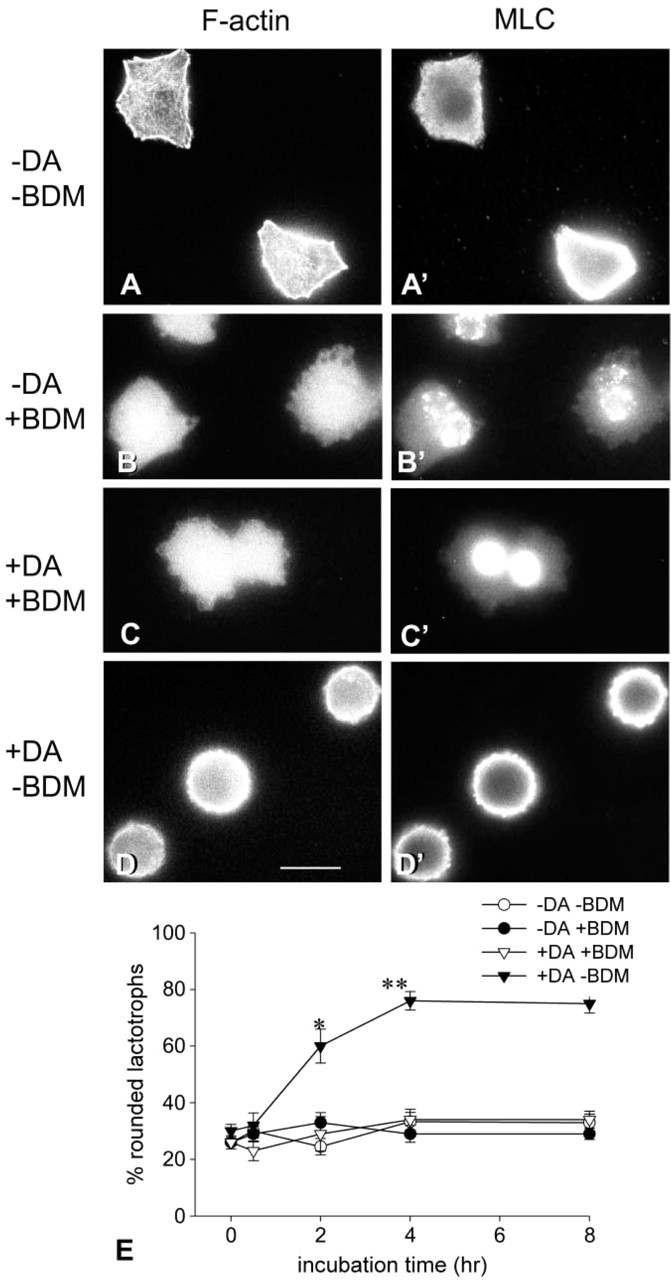
Effect of BDM, an inhibitor of myosin II ATPase, on lactotroph cell morphology. Lactotrophs incubated for 48 hr in the absence of DA were treated with medium alone (-DA-BDM) or with medium containing either 15 mM BDM (-DA + BDM), or 100 nM DA +15 mM BDM (+DA+BDM), or 100 nM DA (+DA-BDM) for 2 hr. Next, cells were tripled-labeled with anti-PRL, anti-MLC, and phalloidin. In (-DA+BDM)- and (DA + BDM)-treated cells, F-actin staining was diffuse and no actin filaments were apparent in the cortex or in the cytoplasm (B,C). MLC staining was observed in the perinuclear region of (-DA+BDM) and DA + BDM-treated cells (B′,C′). Bar = 15 μm. (E) Time course studies on BDM-induced morphological effects. Lactotroph-enriched cultures were incubated in a DA-free medium for 48 hr. After this period (time 0), cells were incubated in a medium with or without 100 nM DA in the presence or absence of 15 mM BDM for increasing periods of time (0-8 hr). After the incubations, cells were immediately fixed and processed for double-labeling fluorescence microscopy with PRL antibodies/FITC-IgG and rhodamine-phalloidin, and the cell shape was recorded. One hundred lactotroph cells per coverslip were visualized and their shape classified as rounded or flat. The percentage of rounded cells was calculated for each coverslip. Values shown are the mean ± SEM. Each experimental condition is the result of the analysis of at least six cover-slips (600 cells) from three different cultures. ∗p<0.005 2 hr (+ DA-BDM) vs 2 hr (-DA + BDM) and vs 2 hr (+ DA + BDM); ∗∗p<0.0001 4 hr (+ DA-BDM) vs 4 hr (-DA + BDM) and 4 hr (+DA + BDM).
Taken together, the present results suggest that the interaction of actin and myosin II is central for DA-induced morphological changes in lactotrophs. The interaction between myosin II and F-actin depends largely on the phosphorylation of the regulatory MLC. MLC is a substrate for many kinases, particularly MLCK, ROCK, and PAK. MLCK has been considered as the major kinase for MLC. To evaluate the participation of MLCK, 48 hr-cultured lactotroph cells were incubated with increasing concentrations of ML-9, a specific inhibitor of MLCK. Concentrations of ML-9 below 10 μM affected neither actin-myosin distribution nor lactotroph shape. Cells treated with ML-9 concentrations of 10 μM and above displayed a thin cortical ring of F-actin that was weaker than the one observed in DA-treated cells (compare Figure 4A with Figure 2C′). In ML-9-treated cells, cytoplasmic F-actin staining was diffuse (Figure 4A) and MLC staining was not associated with the cell cortex but with the perinuclear area (Figure 4A′, arrowheads). ML-9 caused lactotroph cell rounding up (Figure 4B).
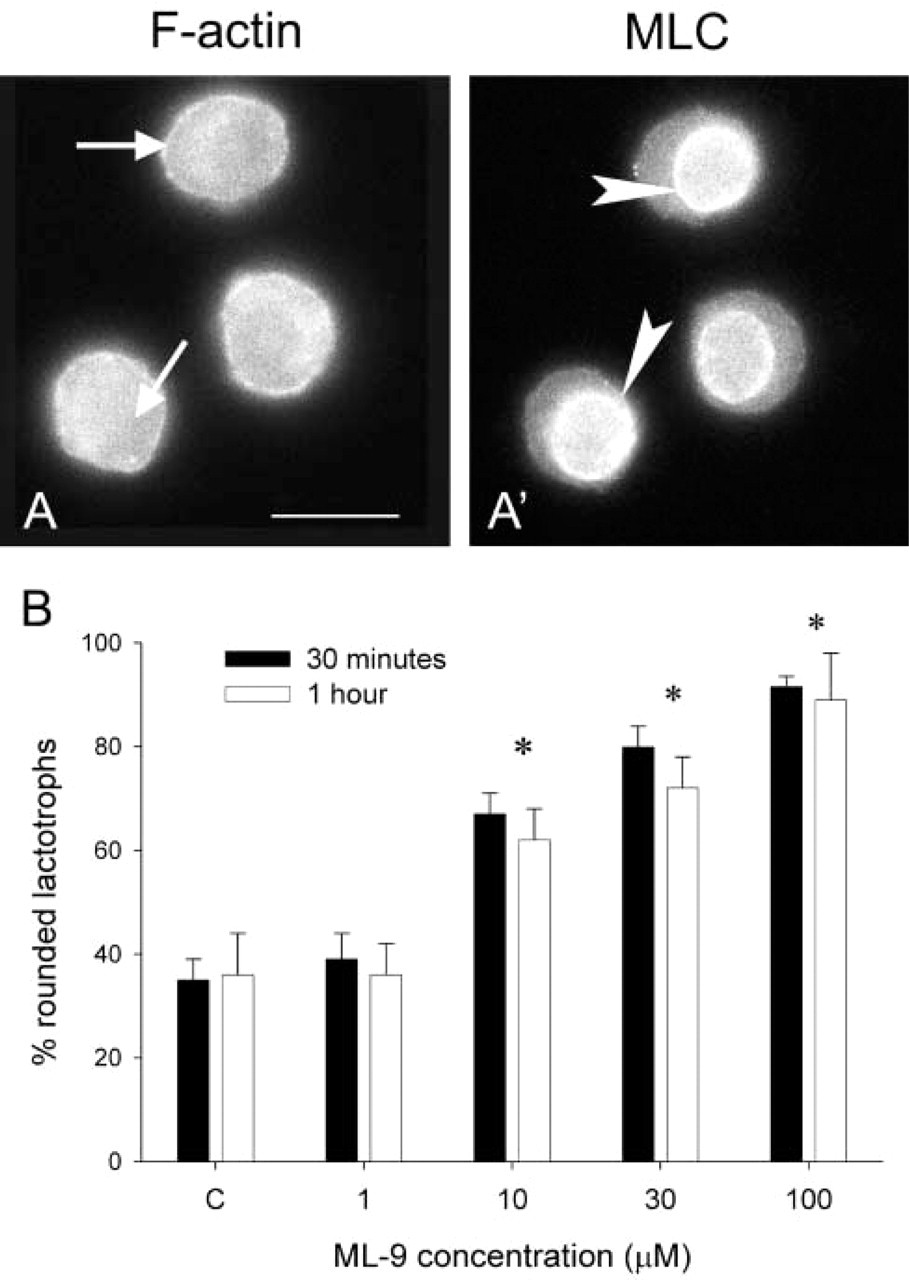
Effect of ML-9, an inhibitior of MLCK activity, on cultured lactotroph cell shape. Lactotrophs were cultured for 48 hr in the absence of DA and next challenged with increasing concentrations of ML-9 for 30 min or 1 hr. After the treatments, cells were triple-labeled for PRL, F-actin, and MLC. The micrographs in
The impact of small GTP-binding proteins in the regulation of the actin cytoskeleton is well documented. We have shown that long-term incubation of lactotrophs with C3 exoenzyme, a procedure that inhibits RhoA-dependent pathways, leads to lactotroph cell rounding up (Nguyen et al. 1999). Here we confirm these results by incubating flat lactotroph cells with 10 μg/ml C3 exoenzyme in the presence of lipofectamine for 2 hr. This method has the advantage of introducing low amounts of C3 exoenzyme and therefore prevents side effects due to an abnormally high intracellular concentration of the toxin (Hirao et al. 1996; Mills et al. 1998; García et al. 1999). Immunofluorescence studies showed that in C3 exoenzyme-lipofectamine-treated cells, F-actin (Figure 5A, arrows) and MLC (Figure 5A′, arrowheads) were diffusely distributed in the cytoplasm. No MLC staining was apparent either at the cortical region or in the perinuclear area of C3-treated cells (Figure 5A”, arrowheads). C3-lipofectamine treatment induced cell rounding up (Figure 5B).
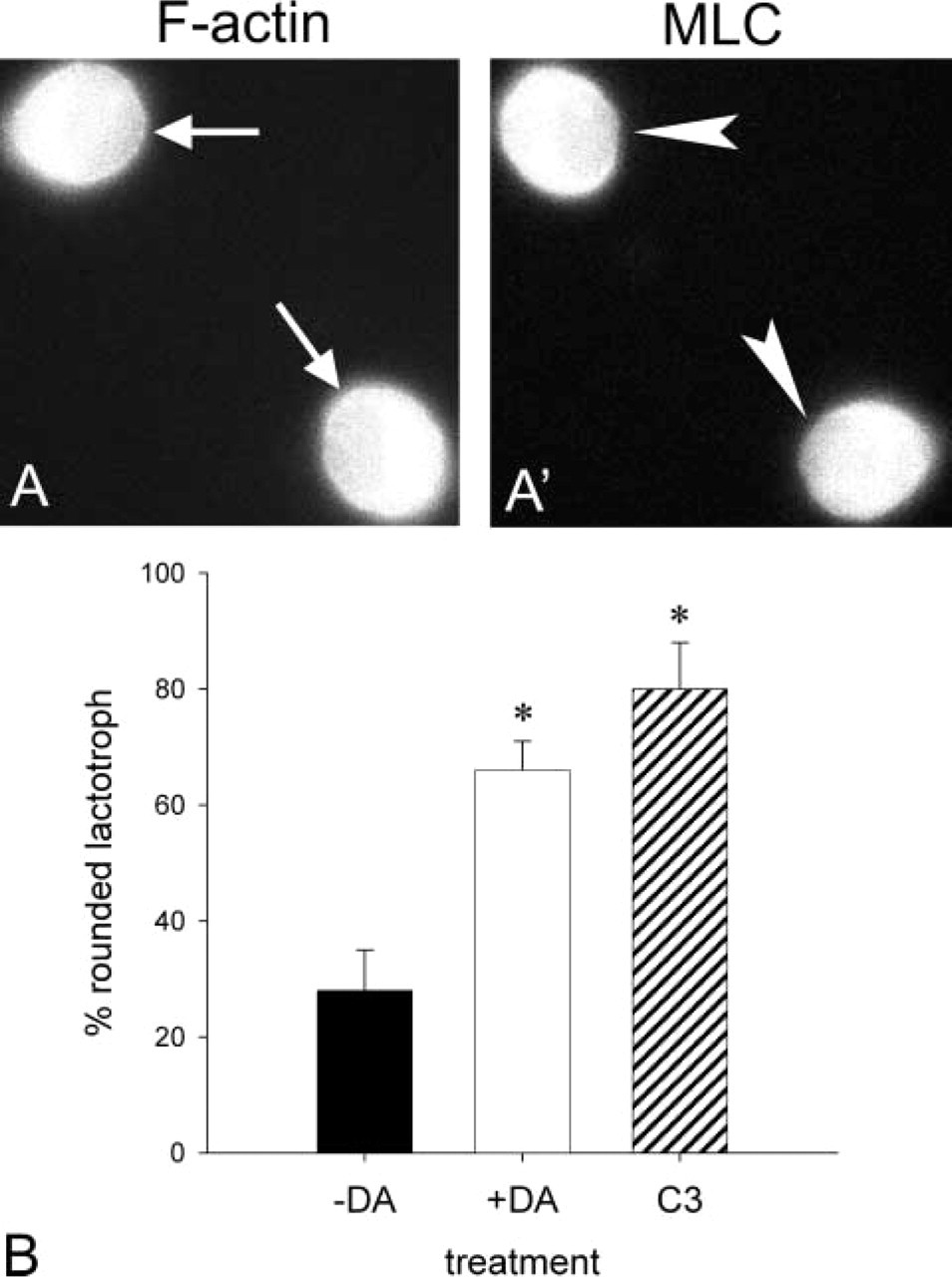
Effect of C3 exoenzyme on DA-induced morphological changes in cultured lactotroph cells. Lactotroph cells were cultured for 48 hr and next challenged with medium alone (-DA) or containing 100 nM DA for 4 hr (DA) or C3/lipofectamine for 2 hr (C3). After the incubations, cells were immediately fixed and processed for double-labeling fluorescence microscopy with PRL antibodies/FITC-IgG, rhodamine-phalloidin, and MLC antibodies to analyze the lactotroph cell shape and F-actin and myosin II distribution. The microphotographs in
Modulation of actin-myosin interactions also implicates other members of the Rho family, such as Cdc42 and Rac-1. To affect Cdc42- and/or Rac-1-dependent pathways, we used C. difficile toxin B which glycosylates and thereby inactivates endogenous Rho, Cdc42, and Rac-1. Treatment of flattened lactotroph cells with 5 ng/ml C. difficile toxin B caused cortical actin reassembly (Figure 6A, arrows), MLC association with the cortical region of lactotroph cells (Figure 6A′, arrowheads), and cell rounding up. In view of the similarity of the morphological effects induced by C. difficile toxin B and by DA, we performed a time course study of toxin B-induced lactotroph cell rounding up, cortical F-actin reassembly, and MLC association with the cell cortex. Figure 6B shows the results of the study. C. difficile toxin B-induced kinetics of cell rounding up, cortical actin reassembly, and MLC association with the cell cortex were similar to the kinetics of DA-induced phenomena. To estimate whether DA affected Rac-1, cells were treated for increasing periods of time with DA and the subcellular localization of Rac-1 was evaluated by immunoblotting with anti-R-1-specific antibodies. The results showed that Rac-1 intracellular distribution was affected by DA. In cells incubated with DA for 2 hr, cytosolic Rac-1 increased (Figure 7).
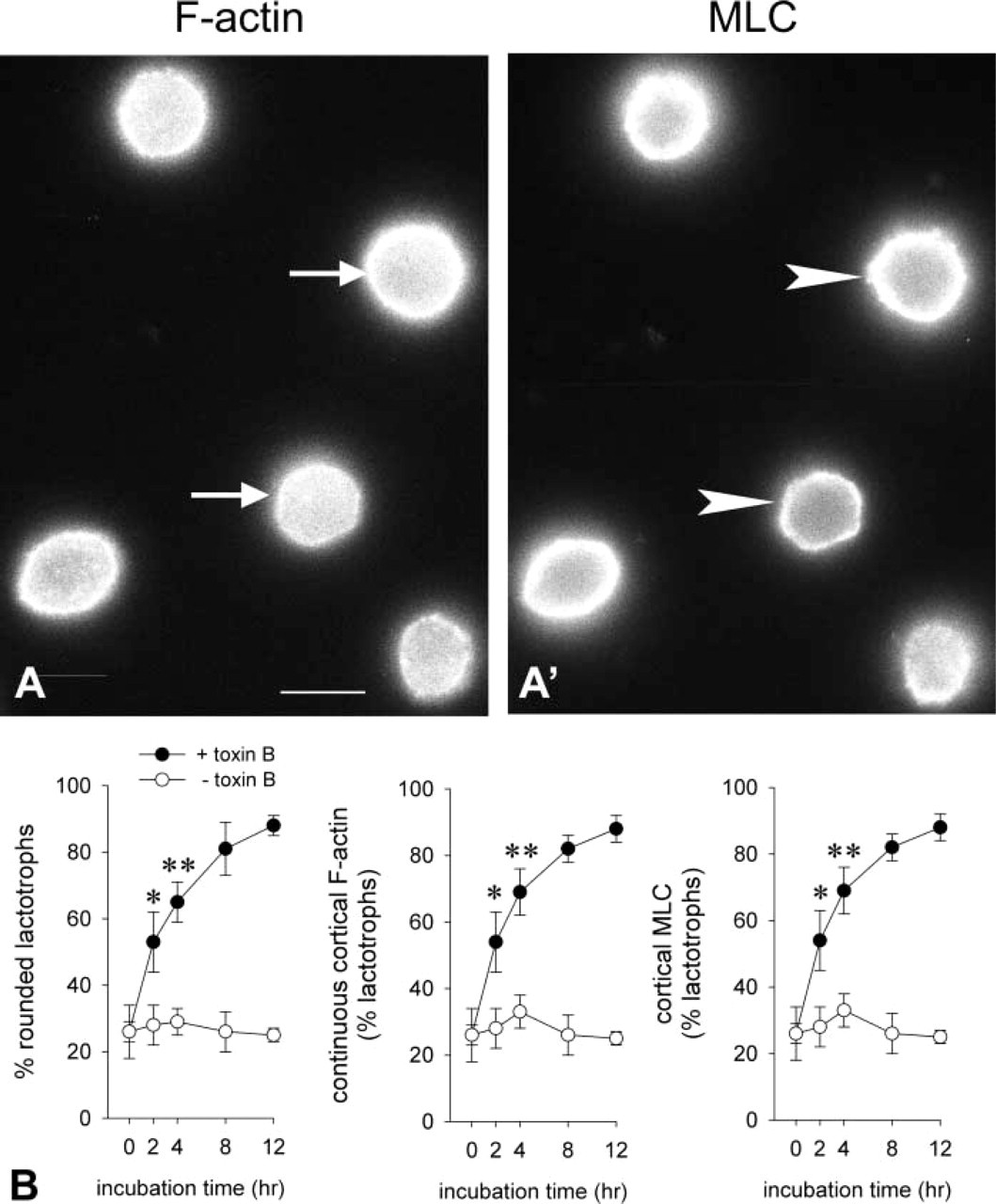
Effect of C. difficile toxin B on lactotroph cell shape, cortical actin reassembly, and association of MLC with the cell cortex. Lactotroph-enriched cultures were allowed to recover for 48 hr. After this period (time 0) the cells were incubated with medium alone or with medium containing 5 μM C. difficile toxin B for increasing periods of time. After treatment, cell preparations were labeled for PRL, F-actin, and MLC. Micrographs
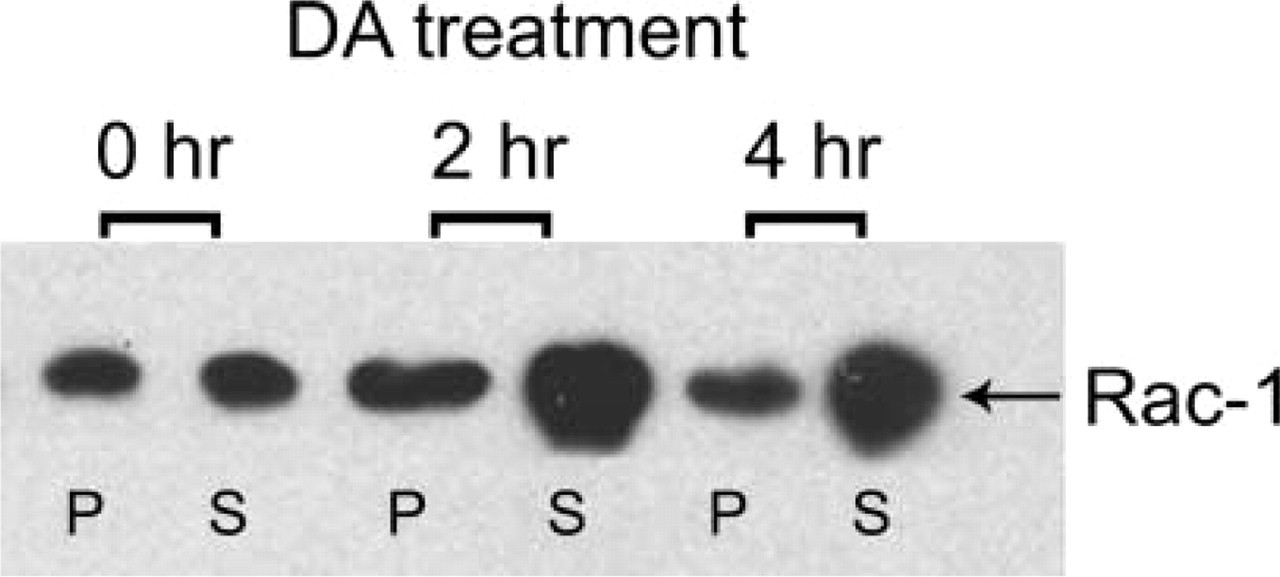
Effect of DA on the intracellular distribution of Rac-1. Lactotroph cultures were incubated for increasing periods of time (0-4 hr) with 100 nM DA. After the treatments, cells were scraped off and membrane (P)- and cytosol (S)-enriched fractions were prepared and subjected to electrophoresis and immunoblotting with Rac-1 antibodies. A band of 21-23 kD corresponding to Rac-1 was observed in both fractions. However, the intensity of the band in the cytosolic fraction in cells treated with DA was stronger than in non-treated cells. The figure shows a representative immunoblot.
Discussion
We have shown that DA, the main inhibitor of PRL secretion, induces significant remodeling of the lactotroph's actin cytoskeleton (Carbajal and Vitale 1997). DA induces the stabilization of the cortical actin cytoskeleton, the disassembly of cytoplasmic actin filaments, and cell rounding up in lactotrophs (Carbajal and Vitale 1997; Nguyen et al. 1999). The molecular mechanisms by which DA modifies the cytoskeleton of lactotrophs remain unclear. The dynamics of the actin cytoskeleton involve actin-binding proteins. Myosin II, an actin-binding protein, plays a central role in actin-based morphology and membrane-based phenomena in non-muscle cells. In particular, myosin II has been associated with the control of cell shape and contractility (Chrzanowska-Wodnicka and Burridge 1996; Majumdar et al. 1998; van Leeuwen et al. 1999) and with actin filament stiffness (Cai et al. 1998). These characteristics of myosin II prompted us to assess its potential role in DA-evoked morphological responses in lactotrophs. Here we show by fluorescence microscopy that DA increased myosin II association to the cell cortex. This result was confirmed by a biochemical approach. It should be pointed out that the effects of DA on the lactotroph's cytoskeleton are slow. The cells must be exposed to DA for at least 2 hr for any significant morphological change to be observed. Interestingly, the time course of DA-induced myosin II association with the cell cortex is similar to the time course of DA-induced stabilization of cortical F-actin (Carbajal and Vitale 1997). Furthermore, the concentration of DA that affects myosin II distribution is within the range of concentrations of DA that induce cortical actin stabilization (Carbajal and Vitale 1997). The increased association of myosin II with the cell cortex after DA treatment suggests that DA may stimulate the formation of actin-myosin complexes at the cell periphery to strengthen cortical actin filaments.
BDM in concentrations ranging from 10 mM to 20 mM (Chrzanowska-Wodnicka and Burridge 1996; Cheresh et al. 1999; Sanders et al. 1999; Torgerson and McNiven 2000; Sullivan et al. 2000) has been widely used as an inhibitor of actin-myosin interactions. Here we found that BDM blocked DA-induced reorganization of actin filaments, myosin association with the cell cortex, and cell rounding up. This indicates that the interaction between actin and myosin is required for DA to evoke its morphological effects in lactotrophs.
The interaction between actin and myosin is modulated by phosphorylation of the myosin light chains in non-muscle cells. MLC is a substrate for different kinases. The most studied kinase is myosin light chain kinase (MLCK), a Ca2+/calmodulin-dependent kinase (Chew et al. 1998). MLC is also phosphorylated by citron kinase (Madaule et al. 1998) during cytokinesis (Yamashiro et al. 2003) and by Rho-kinase (ROCK) (Amano et al. 2000), two RhoA-dependent kinases. In addition, ROCK phosphorylates and thereby inactivates the myosin-binding subunit (MBS) of the myosin light chain phosphatase (Kimura et al. 1996; Suzuki et al. 1999). P21-activated kinase (PAK) stimulated by Rac-1 phosphorylates MLC, causing cytoplasmic F-actin depolymerization and cell retraction in several cell types (Chew et al. 1998; Manser et al. 1998; Kiosses et al. 1999; Sells et al. 1999; Zeng et al. 2000). Recent studies showed that, depending on the kinase involved and on the cell type, MLC phosphorylation may induce either assembly (Kolega 1999; Suzuki et al. 1999; Totsukawa et al. 2000) or disassembly of actin fibers (Manser et al. 1998; Sells et al. 1999; Sullivan et al. 1999). Because of the known effect of DA on intracellular Ca2+ in lactotrophs, we first evaluated the role of MLCK, a Ca2+/calmodulin-dependent kinase, in DA-induced morphological effects. DA decreases intracellular Ca2+ (Ben-Jonathan and Hnasko 2001), and we have previously shown that a Ca2+ channel blocker causes lactotrophs to round up, mimicking the DA-induced effect (Nguyen et al. 1999). Here we found that ML-9, an inhibitor of MLCK, also caused lactotroph contraction. However, ML-9-treated lactotrophs showed a rather weak cortical ring of actin filaments and no accumulation of myosin II at the periphery. Therefore, inhibition of MLCK may not mediate DA-induced actomyosin interactions at the cell cortex. However, inhibition of MLCK did induce disassembly of cytoplasmic actin cables which, in turn, may explain the assembly of cytoplasmic actin filaments in lactotroph cells incubated in the absence of DA, a condition that is accompanied by increased Ca2+ entry. Inhibition of MLCK activity by ML-9 has also been reported to disrupt actin stress fibers in astrocytes, (Koyama and Baba 1996). Conversely, in fibroblasts, the activation of MLCK is responsible for cortical actin assembly without affecting cytoplasmic actin fibers (Totsukawa et al. 2000). The results indicate that the discrimination between cortical and cytoplasmic actin cytoskeleton that is regulated by MLCK is cell-specific.
The influence of GTPases of the Rho family in the dynamics of the actin cytoskeleton is well established. RhoA/C mediates stress fiber formation (Ridley and Hall 1992), Rac-1 induces lamellipodia and cortical actin polymerization (Ridley et al. 1992), and Cdc42 induces filopodia (Nobes and Hall 1995) in several cell lines, principally fibroblasts. How Rho proteins induce actin cytoskeleton remodeling is unclear, but evidence shows that these GTPases modulate the phosphorylation state of specific actin-binding proteins. We found that inhibition of the Rho signaling pathway with C3 exoenzyme in lactotrophs caused cytoplasmic actin cable disassembly and cell rounding up. Myosin distribution in C3-treated cells was cytoplasmic and diffuse. Therefore, inactivation of Rho may be responsible for the cell rounding up by disassembling cytoplasmic actin cables without mediating DA-induced cortical actin-myosin interaction in lactotrophs. Similar results have been reported in fibroblasts and melanocytes, in which Rho inactivation disassembles the actin cytoskeleton and causes cell rounding up (Chrzanowska-Wodnicka and Burridge 1996; Dong et al. 1998; Totsukawa et al. 2000).
The two other members of the Rho family of GTP-ases, Rac-1 and Cdc42, have been reported to affect peripheral actin filament organization in fibroblasts (Ridley et al. 1992; Nobes and Hall 1995). Rac-1 modulates the activity of PAK which, in turn, modifies the phosphorylation state of MLC (Chew et al. 1998; Zeng et al. 2000). PAK also phosphorylates and inhibits MLCK (Sanders et al. 1999; Goeckeler et al. 2000). Our results show that inhibition of Rho GTPases with C. difficile toxin B caused cytoplasmic F-actin disassembly, assembly of a cortical ring of actin filaments, and association of MLC with the cell cortex. These results differ from those induced by inhibition of Rho alone with C3, which caused only cytoplasmic actin cable disassembly. Therefore, it is the inhibition of Rac-1/Cdc42 pathways in lactotrophs that probably caused cortical actin cytoskeleton assembly and actin-myosin II interaction at the cell periphery. Interestingly, in PC12 and N1E-115 neuroblastoma cells, activation of Rac-1 causes the loss of cortical myosin and cell spreading (van Leeuwen et al. 1999).
Of the several signaling pathways evaluated here, only the inhibition of Rac-1/Cdc42 reproduced DA-induced actin-myosin interaction at the cell periphery. However, to our knowledge there is no information on DA effects on the Rho family of GTP-binding proteins. Because of the slowness of DA at inducing actin-myosin interaction at the lactotroph cell cortex, it is difficult to correlate DA-induced morphological effects with changes in the GTP:GDP ratio of Rho proteins. However, we found that DA treatment of lactotrophs increased the amount of Rac-1 in the cytosol. Even though Rac-1 association with membranes cannot always be interpreted as its activation and the cytoplasmic localization as its inactivation, the present results suggest that Rac-1 is a target for DA and therefore that DA may affect Rac-1-dependent pathways to modulate actin-myosin interaction and thus to reorganize the actin cytoskeleton in lactotrophs.
Footnotes
Acknowledgements
Supported by the Natural Sciences and Engineering Research Council of Canada (MLV). MLV is supported by a scholarship from Fonds de la Recherche en Santé du Québec.
We thank Dr A. Parlow and the National Hormone and Pituitary Program of the NIDDKD for the gift of anterior pituitary hormone antibodies.
