Abstract
Calcium-activated chloride channels (CLCAs) are a family of multifunctional proteins that are widely distributed in tissues. To investigate the distribution of human CLCA-2 (hCLCA2) in human epithelia at the light and electron microscopic levels, we raised a primary antibody against a synthetic polypeptide sequence from natural hCLCA2. Corneal, skin, vaginal, esophageal, and laryngeal epithelia were immunopositive for hCLCA2 at the cytosolic aspect of the basal cells adjacent to the basement membrane. Epithelia of stomach and small intestine showed no hCLCA2 immunoreactivity. This study reports the cellular distribution of hCLCA2 in human epithelia and suggests its possible involvement in epithelial stratification and cell-substrate adhesion.
C
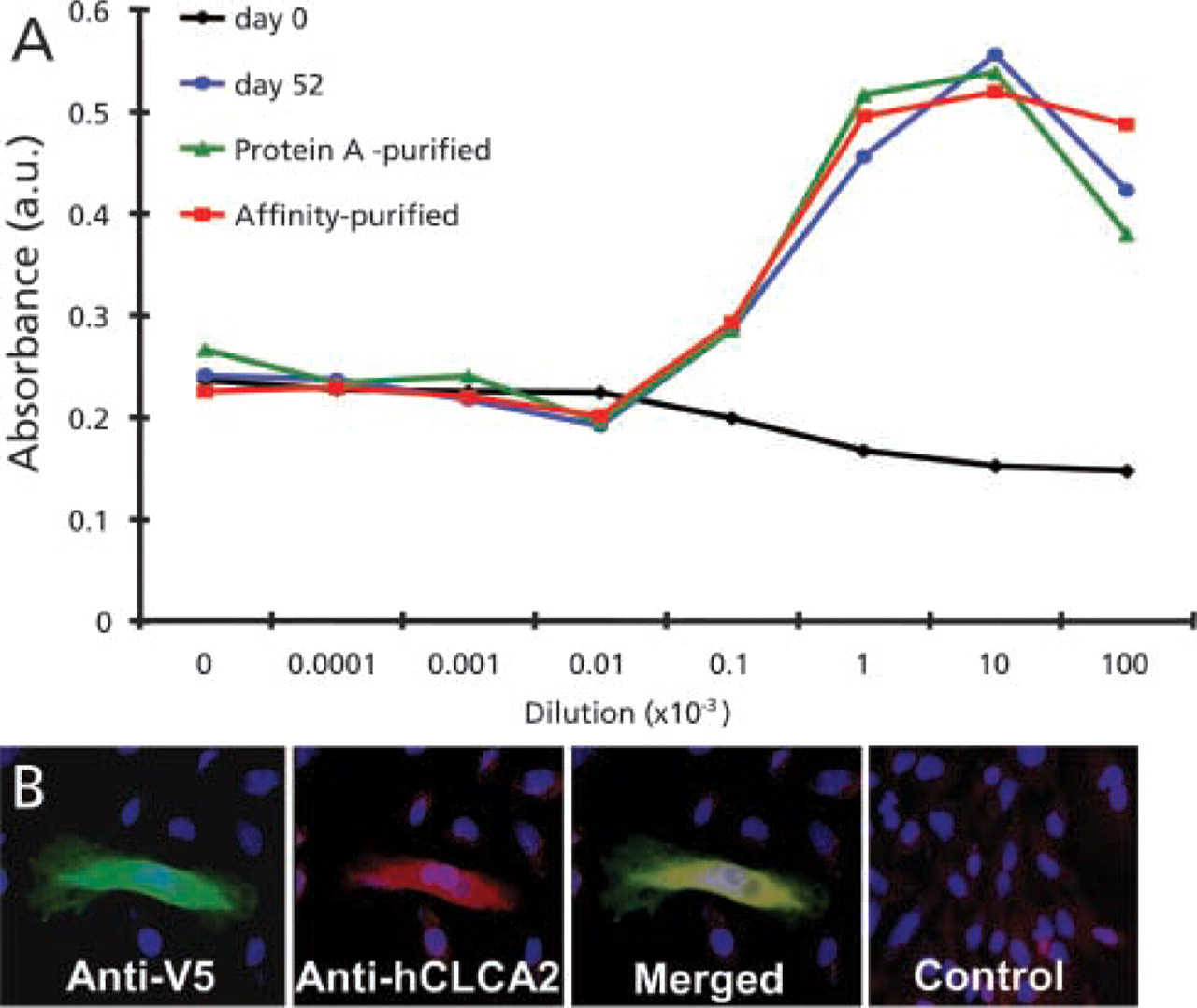
The hCLCA2 antibody was prepared from a synthetic peptide corresponding to a non-conserved region on the internal loop between transmembrane domains 3 and 4 (TVEPETGDPVTLRLC) (Takara Bio; Tokyo, Japan). The synthetic peptide was conjugated to keyhole limpet hemocyanin via a C-terminally added cysteine and injected under the skin of a rabbit. After 52 days the IgG fraction of the sera was purified with a protein A column and then affinity-purified with a column filled with a Sepharose-immobilized hCLCA2 synthetic peptide. The reactivity of the anti-hCLCA2 antibody was quantified against immobilized hCLCA2 (Figure 1A) and its specificity was shown by successfully labeling HeLa cells transfected with recombinant hCLCA2 (Figure 1B). To further prove the specificity of the anti-hCLCA2 antibody, Western blotting of hCLCA2-transfected HeLa and E. coli cells was attempted. However, despite successful transfection of hCLCA2, shown by positive Coomassie Blue staining in E. coli (data not shown), it could not be subsequently detected by the anti-hCLCA2 antibody. Nevertheless, the V5 tag, which was fused to the recombinant hCLCA2 protein during cloning, was detected by antibody staining. This suggests that although the cells were successfully transfected with recombinant hCLCA2, the anti-hCLCA2 epitope was denatured during the Western blotting procedure.
(
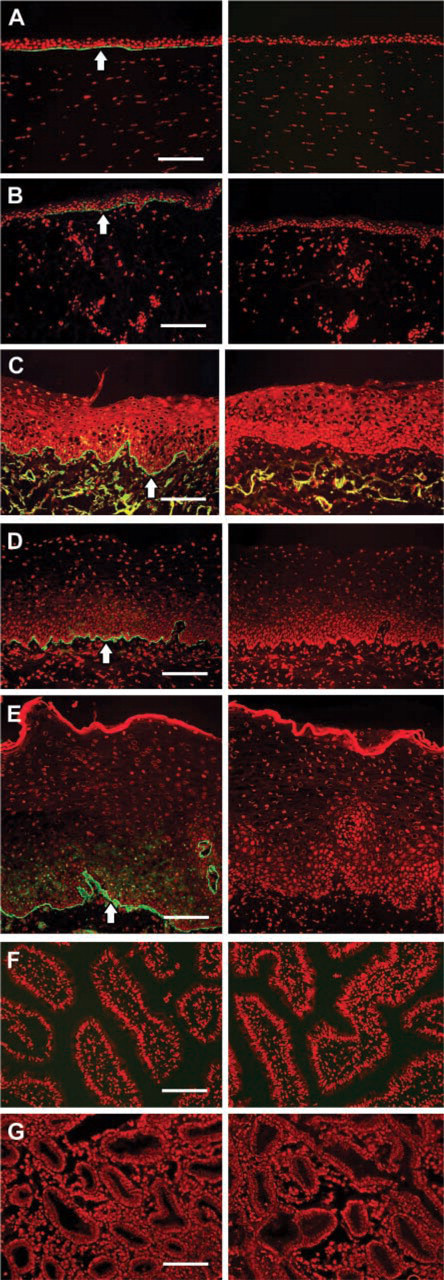
Tissue samples for immunohistochemistry (IHC) were obtained from healthy tissue adjacent to tumors excised during surgery on seven individual patients at the Kyoto Prefectural University of Medicine, Japan. Prior informed consent was obtained from each patient after a detailed explanation of the procedures, in accordance with the tenets of the Declaration of Helsinki. Immediately after excision, specimens were frozen in Tissue Tek (Miles; Elkhart, IN). Cryosections 7 μm thick were collected on silanized glass slides and fixed in Zamboni fixative at 4C for 10 min, followed by multiple washes in 0.01 M PBS. Nonspecific antibody-binding sites were blocked by incubation for 30 min with 0.01 M PBS containing 1% BSA, after which sections were incubated with anti-hCLCA2 antibody (× 10,000 dilution) for 1 hr at 25C, followed by three 5-min washes in 0.01 M PBS. The sections were then incubated at room temperature (RT) for 1 hr with AlexaFluor 488-conjugated anti-rabbit IgG antibody (Molecular Probes; Eugene, OR). After washing with PBS the sections were mounted in medium containing propidium iodide (Vector; Burlingame, CA) and examined by fluorescent microscopy (Olympus AX70). Negative controls were performed by replacing the primary antibody with nonspecific normal rabbit IgG (DAKO; Kyoto, Japan) at the same concentration. The affinity-purified anti-hCLCA2 antibody detected hCLCA2 in each of the stratified epithelia examined. The expression was primarily limited to the basal membrane of basal cells where the cells were in contact with the basement membranes of the cornea, skin, larynx, vagina and esophagus (Figures 2A-2E). Non-stratified epithelia of the small intestine and stomach do not express detectable levels of hCLCA2 (Gruber et al. 1999) and were included as positive controls. No hCLCA2 immunoreactivity was seen in these tissues (Figures 2F and 2G), confirming the validity of our antibody.
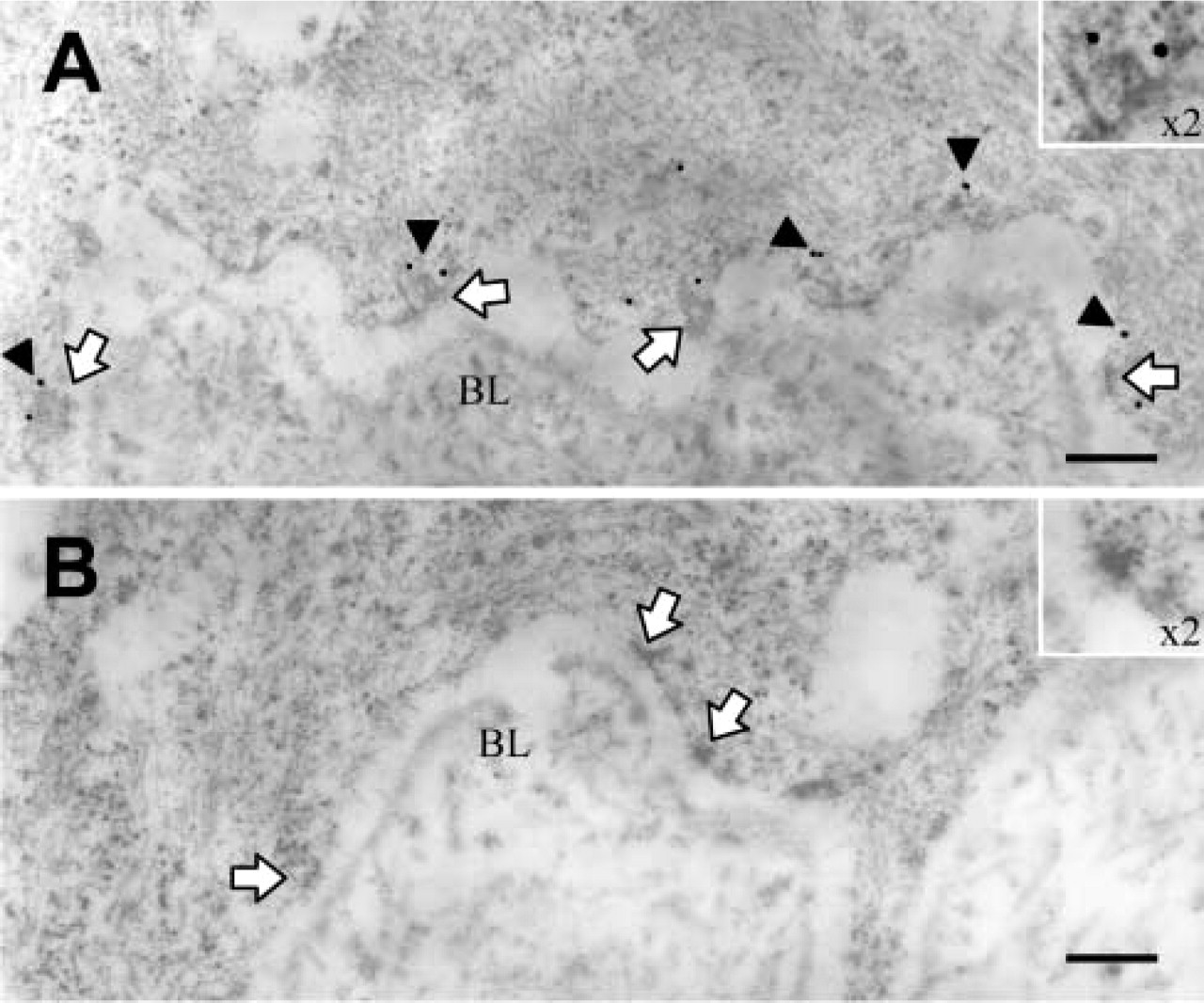
Corneal tissue was further examined by immuno-electron microscopy to elucidate more fully the specific location of the hCLCA2 antibody-binding site. For this, the central portion of a human cornea was dissected into small (1 × 2-mm) samples and fixed in 4% paraformaldehyde in 0.1 M PBS for 2 hr at 4C. After dehydration in ethanol (70% and 95% for 15 min and 2 × 100% for 30 min) and infiltration of Unicryl (British Biocell International; Cardiff, UK) overnight, the samples were polymerized in 100% Unicryl at 55C for 48 hr. Ultrathin sections (70-nm) were collected on nickel grids and placed section side down on blocker buffer (0.1 M PBS, 1% goat serum, 0.1% Tween-20, and 1% BSA, adjusted to pH 8.2) for 30 min to reduce nonspecific binding. The grids were then transferred to anti-hCLCA2 antibody (×5000 dilution) overnight at 4C, followed by five 5-min washes in blocker buffer and 12-nm gold-labeled anti-rabbit immunoglobulins (×25 dilution; Jackson ImmunoResearch, West Grove, PA) for 1 hr at 25C. This was followed by three further washes in blocker buffer, and then sections were allowed to dry overnight. Next day, sections were counterstained with 2% uranyl acetate for 10 min followed by 2% lead citrate for 5 min. Incubations were completed in a moist chamber on 50-μl drops. Grids were observed in a Hitachi H-7000 transmission electron microscope at 75 kV. The resulting immunoelectron microscopy of the corneal epithelium disclosed hCLCA2 localization solely to the cytosolic side of the basal aspect of the basal cells, adjacent to the basement membrane. Furthermore, the gold particles appeared primarily in close apposition to the hemidesmosomes, which are positioned regularly along the basal membrane (Figure 3A).
hCLCA2 was situated alongside the basal epithelial cells (arrows) of cornea (
(
Previously, mRNA expression of hCLCA2 has been found in lung, mammary gland, and trachea (Gruber et al. 1999), as well as in cornea (Itoh et al. 2000). Here we disclose the presence of hCLCA2 protein in cornea, skin, vagina, esophagus, and larynx. The tissue distribution of hCLCA2 is perhaps an indicator of possible function. Consistent with previous work (Gruber et al. 1999), we show that hCLCA2 is not present in the non-stratified epithelia of the stomach or small intestine (Figures 2F and 2G). However, it is found in all stratified epithelia investigated here and with a similar distribution pattern, thus implying some structural role in epithelial stratification. The integrity of stratified epithelia is maintained by a variety of adhesion mechanisms, including contact via desmosomes at the sites of cell-cell interaction and by hemidesmosomes at the sites of cell-substrate interaction. These sites, in turn, have been linked to the presence of integrin receptors including integrin-β4, an integral component of the hemidesmosome (Carter et al. 1990; De Luca et al. 1990; Stepp et al. 1990,1993). In cornea, integrin-β4 is localized specifically to the basal aspect of the basal epithelium (Stepp et al. 1993), where it has been shown to form an adhesion partner with hCLCA2 during cell-cell contact (Abdel-Ghany et al. 2001). Our disclosure of hCLCA2 at the basal aspect of the basal cells in the stratified epithelia examined raises the possibility of an involvement with integrin-β4 and a role in basal cell-basement membrane adhesion. This view may go some way to explaining the lack of hCLCA2 in non-stratified epithelia, such as the stomach and small intestine, as shown here, or the colon, spleen, pancreas, and placenta, as shown previously (Gruber et al. 1999), if we consider the likelihood that basal cell-basement membrane adhesion in such tissues will encounter lower shearing forces than those encountered by stratified epithelia.
In conclusion, this study provides the first comprehensive results showing hCLCA2 localization at the cellular level in human epithelial tissues. The data indicate a specific localization of hCLCA2 towards the basal side of the basal cells of stratified epithelium, and, in cornea, apparent co-localization with hemidesmosomes. These observations are in marked contrast to those from the non-stratified epithelia investigated here, which indicate an absence of hCLCA2. The physiological role of hCLCA2 requires further elucidation. Nevertheless, the current results point to the involvement of hCLCA2 in epithelial stratification and possibly in cell-substrate adhesion.
Footnotes
Acknowledgements
Supported by the Japanese Society for the Promotion of Science (CJC) and by a Royal Society 2+2 Fellowship (CJC).
