Abstract
Double autoimmunostaining by a sequential twice-repeated enzyme-labeled polymer method was examined on archival paraffin sections of formalin-fixed human tissue using an autoimmunostaining apparatus to determine optimal conditions for glycine treatment, to select the best combination of dyes for the horseradish peroxidase–hydrogen peroxide reaction, and to investigate mounting methods for preparing permanent specimens. The optimal glycine treatment determined by changing the incubation time in 0.1 M glycine hydrochloride buffer, pH 2.2, was glycine buffer washing three times for 1 min each, with suppression of nonspecific binding of the primary antibody by protein blocking. Combinations of DAB and AEC, SG and AEC with Ultramount, and DAB and VIP or No-vaRED and SG with the VectaMount were found usable for the double autoimmunostaining, based on color analysis of the dyes. Pairs of primary antibodies, CD68 and anti-fascin antibodies CD3 and CD79a, and anti-Ki-67 antigen and anti-p53 antibodies were applicable in double autoimmunostaining with appropriate antigen retrieval for each pair of primary antibodies. Consequently, good sequential double autoimmunostaining should include masking the nonspecific binding of primary antibodies, optimal glycine treatment, and selection of adequate dyes and mounting methods.
Keywords
D
Many procedures have been established for immunohistochemical (IHC) staining of chemically fixed and paraffin-embedded sections, including suppression of endogenous peroxidase activity (Streefkerk 1972), masking of endogenous biotin (Wood and Warnke 1981), competitive suppression of primary antibodies (Tacha and McKinney 1992), and highly sensitive indirect detection systems such as the avidinbiotin complex (ABC) method (Heggeness and Ash 1977), and retrieval methods for masked antigens are being continuously refined. Enzyme-labeled polymer methods such as the enhanced polymer one-step (EPOS) system (Sano et al. 1996), the EnVision system (Sabattini et al. 1998), and the simple stain system (Nichirei; Tokyo, Japan) have greatly simplified laboratory IHC. Highly sensitive IHC methods (Hasui et al. 2002) using antigen retrieval (AR) (Shi et al. 2001) have opened a new era in surgical pathology and in the study of human histology and histopathology.
Double immunostaining is very complicated, requiring two IHC labelings of two antigens without crossreactivity. The heating method that inactivates reactions in the initial IHC procedure has led to establishment of a combination of the ABC method and the alkaline phosphatase methods (Lan et al. 1995; Bohle et al. 1997; Ezaki 2000). This double immunostaining removes the products of the first two immunoreagent steps and retrieves the target antigen of the second primary antibody. Thus far, only one double autoimmunostaining method has been developed using the Ventana HX System Discovery (Ventana Medical Systems; Tucson, AZ). However, heating in the process of double immunostaining has been regarded as a drawback of the procedure with an autoimmunostaining apparatus that is not equipped with a system for AR by heating at high temperature.
This aim of the present study was to determine optimal conditions for the glycine treatment first reported by Nakane (1968) for double immunostaining of an enzyme-labeled antibody, although other solutions with pH lower than 3 or higher than 9 may be employed to elute the products of the first two immunoreagent steps (Tramu et al. 1978). We used the peroxidase-labeled polymer method (EnVision system; Sabattini et al. 1998), deleting a number of the double autoimmunostaining steps. The best combination of dyes in the horseradish peroxidase (HRP)–hydrogen peroxide (H2O2) reaction and the mounting methods for preparing permanent double-autoimmunostained specimens were determined by analyzing the colors of the dyes in the RGB system and by performing double autoimmunostaining. Finally, we applied double autoimmunostaining for differentiating dendritic cells in lymph follicles, T- and B-cells in lymph nodes, and dysplastic and hyperplastic squamous cells of the tongue, by considering the combination of dyes in the HRP–H2O2 reaction.
Materials and Methods
The materials used were sections of anonymous archival 10% formalin-fixed and paraffin-embedded human tonsil, lymph node, and oral mucosa tissues.
Autoimmunostaining Apparatus
The autoimmunostainer used was a DAKO autostainer (S3400; DakoCytomation Co. Ltd., Tokyo, Japan). The protocols for immunostaining can be easily designed by determining the number of steps needed and by setting the work in each step.
The washing buffer, 0.01 M tris(hydroxymethyl)aminomethane (Tris)-saline buffer, pH 7.6, including 0.1% Tween-20 (polyoxyethylene sorbitan monolaurate) was warmed at 35C by incubating the buffer tank in a hot water bath to control the temperature, while the room temperature was controlled at 25C.
Primary Antibodies and Detection System
The primary antibodies used were CD68 (anti-human CD68, macrophage, PG-M1, M0876; DakoCytomation) and anti-fascin (anti-human fascin, 55K-2, M3567; DakoCytomation) for differentiating monocyte-derived dendritic cells (CD68-and fascin-positive cells) among dendritic cells (fascin-positive cells), CD3 (NCL-CD3-PS1, Novocastra Laboratories, Newcastle upon Tyne, UK) and CD79a (anti-human CD79a, B-cell, M7050; DakoCytomation) for differentiating T- and B-cells, and anti-Ki-67 antigen (Ki-67 antigen, MB-1, M7240; DakoCytomation), and anti-p53 antigen (NCL-p53-1801; Novocastra Laboratories) for differentiating proliferating cells with DNA damage (Ki-67- and p53-positive cells). According to the data sheet for each primary antibody, primary antibody solution was diluted at the adequate level with antibody diluent (Dako ChemMate, S2022; DakoCytomation).
Antigen retrieval (AR) was achieved by incubating sections in 0.01 M citrate buffer, pH 6.0 (DAKO ChemMate Buffer for Antigen Retrieval, S2031), after deparaffinization and suppression of endogenous peroxidase activity. For the AR of the anti-fascin antibody, the 0.01 M citrate buffer retrieval solution was adjusted to pH 8.0 with EDTA (DAKO Target Retrieval solution, high pH, S3307; DakoCytomation). For AR for CD3 and CD79a, digestion by each original, X10−, and X100-diluted solution of trypsin (TRYPSIN Tablet, EC 3.4.21.4; Sigma Chemical, St Louis, MO) for 10 min was added to the usual AR.
IHC reactions were detected by the secondary antibody-and HRP-labeled polymers (ChemMate EnVision, K5027; DakoCytomation) to decrease the number of steps in the procedures for double immunostaining by the autostainer.
Determining the Optimal Conditions for Glycine Treatment
The rinse time in 0.01 M glycine solution, pH 2.2 (Nakane 1968), sufficient to remove the immunoreaction products of the first primary antibody was measured, visualizing the residual immunoreaction products in step 4 of the autoimmunostaining procedure, inserting the glycine treatment (step 5) between steps 3 and 4 (Experiment 1) and between steps 2 and 3 (Experiment 2) in the double immunostaining procedure (Table 1) with deletion of steps 6 to 8.
In Experiment 1, the residue of the reacted primary antibody and the reacted enzyme-labeled polymers after glycine treatment was tested.
In Experiment 2, the residue of the reacted first primary antibody was examined with or without protein block by a nonspecific staining blocking reagent (X090; DakoCytomation). The optimal duration of the glycine treatment was determined by evaluating the residue of the immunoreaction products in Experiments 1 and 2 for treatment times of 1–77 min (Table 2).
The effects of glycine treatment on the retrieved antigen were examined, evaluating the residue of the immunoreaction when steps 2 to 4 in Table 1 were deleted (Experiment 3).
The effects of glycine treatment on the dyes visualizing the immunoreaction were checked, deleting steps 6 to 8 (Table 1).
Steps in double immunostaininga

Steps 2-9 were performed by an autostainer.
Evaluation of glycine treatment
Evaluation of residual immunoreaction products after the glycine treatment. +, obvious residues; ±, faint residues; -, no residues. nd, not done.
With protein block, suppressing nonspecific antibody binding.
Dyes in the HRP-H2O2 Reaction
In selecting the most suitable dye for the HRP-H2O2 reaction for visualizing the immunoreaction products, DAB (3,3′-diaminobenzidine tetrahydrochloride, Liquid system, K3466; DakoCytomation), Vector DAB-Ni (SK-4100), AEC (3-amino-9-ethyl carazole, liquid Substrate Chromogen, K3469; DakoCytomation), Vector NovaRED (SK-4800), Vector VIP (SK-4600), Vector SG (SK-4700), and Vector TMB (SK-4400) from Vector Laboratories (Burlingame, CA) were investigated.
The relationship between the dyes and the following methods for mounting sections was evaluated. The methods used for mounting sections to prepare permanent preparations were the ordinary method of mounting by plastic medium after dehydration by air-drying or by rinsing the sections in 100% ethanol solution three times for more than 5 min and rinsing the sections in xylene solution, Dako Ultramount mounting of moist sections by Ultramount (Aqueous Permanent Mounting Medium, S1964; DakoCytomation) and heating the sections at 70C for more than 30 min, and VectaMount mounting of air-dried sections with Vecta-Mount Mounting Medium (H-5000) and a coverglass.
To determine a good pair of dyes for the double immunostaining, the color of each dye was analyzed in the RGB system in light micrographs from the immunostained specimens taken using a microscopic digital camera (HC-300; Fuji Film, Tokyo, Japan), selecting a region of positive immunoreaction in the digital image, and separating the image of the area into R, G, and B layers (Photoshop 3.0; Adobe Systems, San Jose, CA). The products of the inverted R, B, and G layer images were the mean concentration of darkness in each image (NIH image; http://rsh.info.nihgov/nih-image/).
Double Autoimmunostaining Procedure
The most suitable procedures determined for double autoimmunostaining were applied for double autoimmunostaining of CD68, which labels macrophages and dendritic cells, and anti-fascin antibody, which stains dendritic cells, capillary endothelial cells, and some squamous cells. In this immunostaining, the cells doubly-positive for CD68 and fascin were evaluated as macrophage-derived dendritic cells. In the double autoimmunostaining of CD3 and CD79a, the distribution of T- and B-lymphocytes in lymph nodes was examined. In double autoimmunostaining of Ki-67 antigen, which labels nuclei of proliferating cells, and p53 protein, which labels overexpressed wild-type or mutant p53 gene products, the cells doubly positive for both antigens were evaluated as dysplastic proliferating cells.
Results
Optimal Conditions of Glycine Treatment for Autoimmunostaining
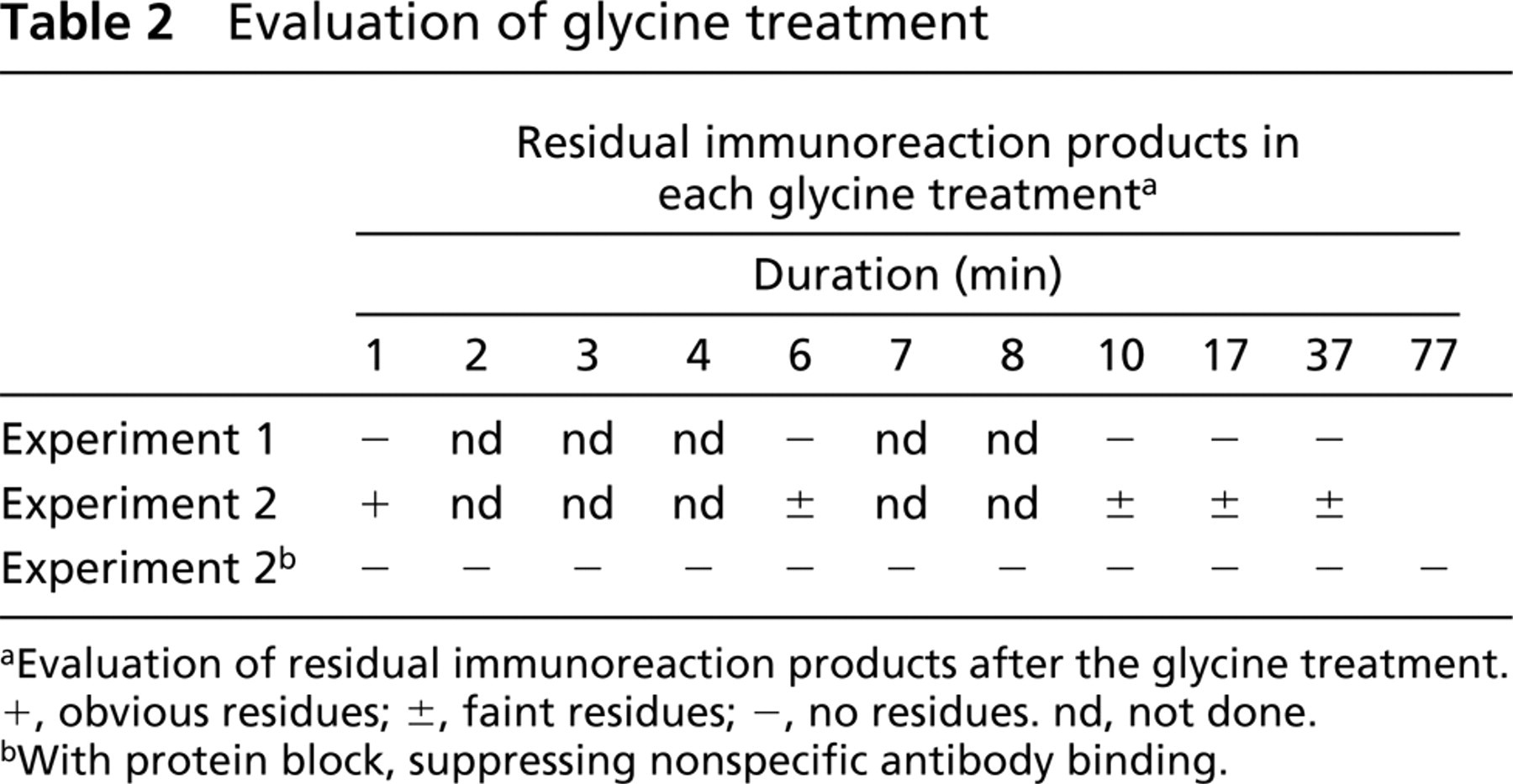
Residual immunoreaction products were not present even after rinsing in glycine solution for 2 min in Experiment 1 (Table 2). However, in Experiment 2, weak staining of the immunoreaction products was noted in macrophages and dendritic cells of germinal centers (Figure 1a), even after rinsing in glycine solution for 77 min (Table 2). The residual staining was removed by introducing a protein blocking agent that competitively suppressed nonspecific binding of the primary antibody (Figures 1b3–1b6). After glycine treatment for 1 min, no residual staining was seen in Experiment 2 (Table 2).
To see the effects of the glycine treatment on retrieved antigens, Experiment 3, using anti-CD68 and anti-Ki-67 antigen antibodies, was performed. The retrieved CD68 remained even after glycine treatment for 77 min (Figure s1c3 and 1c5), whereas the Ki-67 antigen was masked in part after glycine treatment for 7 min (Figures 1c4 and 1c6).
The optimal conditions for glycine treatment were thus determined to be glycine treatment three times for 1 min to induce complete dissociation and washout of the immunoreaction, as indicated in Table 3.
Determination of the Best Dye Combination for the HRP–H2O2 Reaction
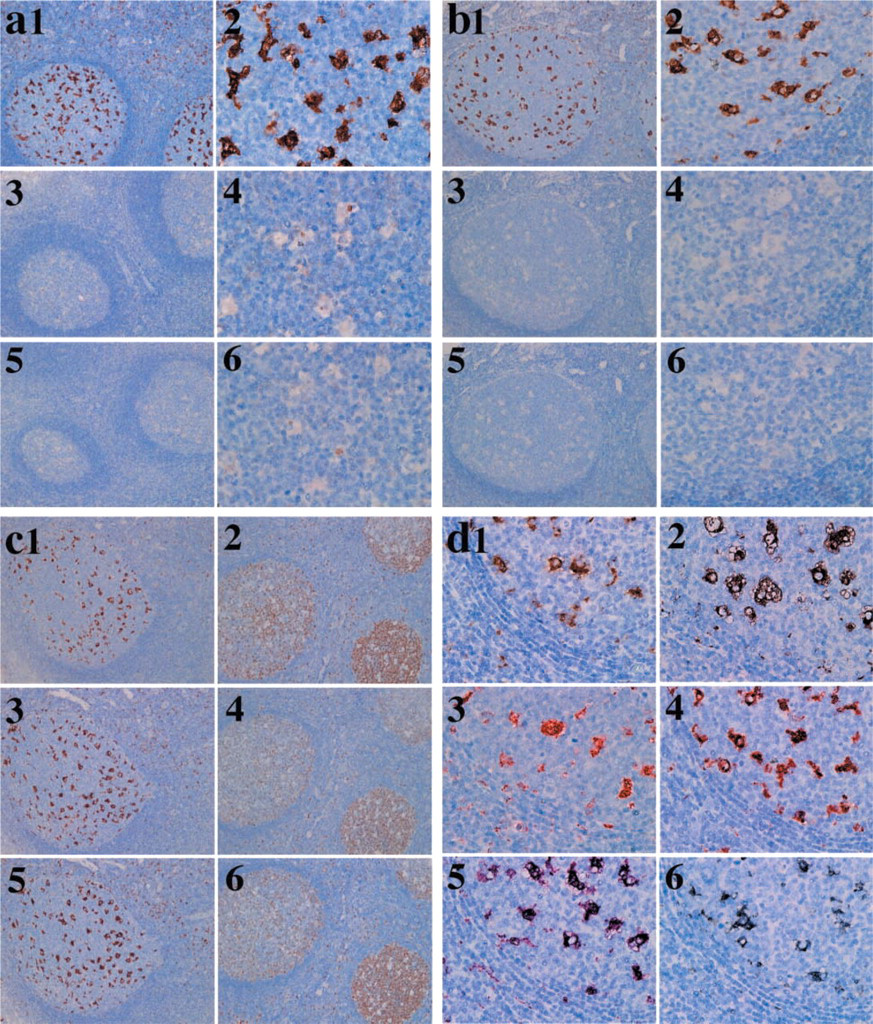
Autoimmunostaining of CD68 with each of the dyes for each of the three mounting methods showed that there was a limitation to the application of the dyes in the HRP–H2O2 reaction. As shown in Table 4, in the ordinary method DAB, DAB–Ni, and SG were used, in the Ultramount method DAB, DAB–Ni, AEC, and SG were used, and in the VectaMount method DAB, DAB–Ni, NovaRED, VIP, and SG were used. TMB could not be used because it disappeared during the mounting process. Sensitivity in visualization of the immunoreaction was the same in the HRP–H2O2 reaction with DAB, DAB–Ni, AEC, NovaRED, and VIP (Figures 1d1–1d5), whereas that of SG was the lowest and did not detect the dendrites of dendritic cells (Figure 1d6).
The real colors of these dyes in the RGB system are shown in Table 4. It was suggested that DAB changed its color, increasing in all the elements in the Ultramount mounting and decreasing in the G and B elements in the VectaMount mounting. Hematoxylin used to counterstain nuclei changed color, revealing high values of the R and G elements in the order of VectaMount, Ultramount and ordinary mounting preparation. The difference in the RGB system between DAB and DAB–Ni was greatest in the B element for VectaMount. For Ultramount, the color of AEC was differentiated from DAB or DAB–Ni, SG, and hematoxylin but faded after glycine treatment. DAB, DAB–Ni, NovaRED, VIP, SG, and hematoxylin all showed characteristic percentages of the R, G, and B elements. Glycine treatment changed the color of NovaRED and VIP to resemble that of DAB.
Results of control experiments. (a) Results of Experiment 2 (CD68) (DAB). (a1,a3,a5) Lymph follicles in a tonsil. (a2,a4,a6) Germinal center in a lymph follicle. (a1,a2) No glycine treatment. (a3,a4) After 7 min of glycine treatment. (a5,a6) After 17 min of glycine treatment. The residual reaction of the CD68 antibody is still visualized in dendritic cells and macrophages in the germinal centers. Original magnifications: a1,a3,a5 × 10; a2,a4,a6 × 40. (b) Results of Experiment 2 (CD68) with protein blocking before reaction with the primary antibody (CD68) (DAB). (b1,b3,b5) Lymph follicles in a tonsil. (
Optimal protocol for double autoimmunostaining

Tris washing buffer: Tris-saline washing buffer with 0.1% Tween-20.
Protein block: protein block by nonspecific staining blocking reagent (X0909; DakoCytomation).
ChemMate EnVision: ChemMate EnVision can be replaced by EnVision-AP (alkaline phosphatase-labeled polymer method) in order to use New Fuchsin as a dye visualizing immunoreaction.
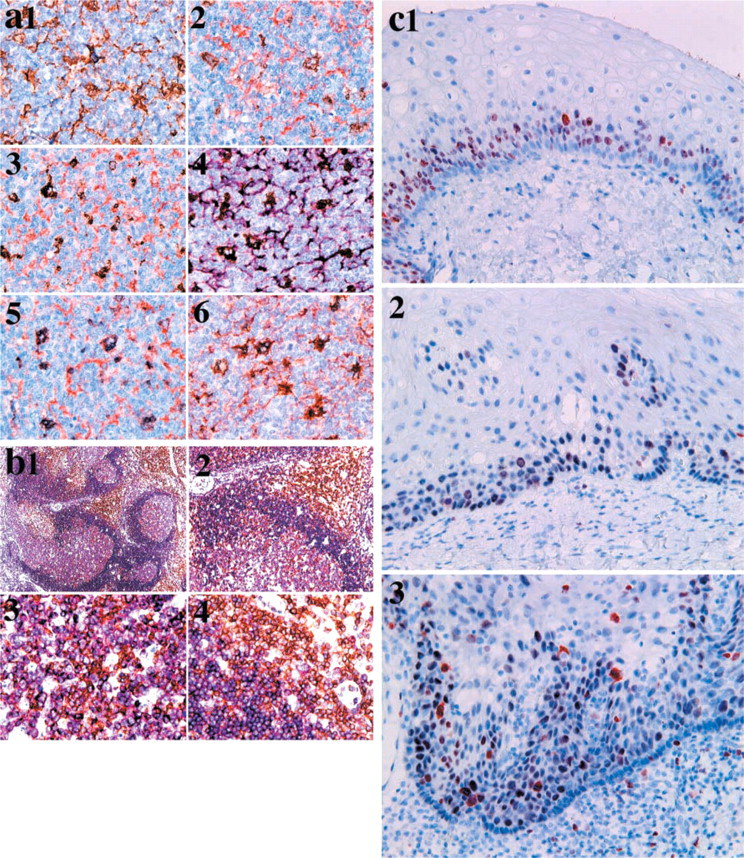
Double autoimmunostaining of anti-CD68 and anti-fascin antibodies with a representative combination of dyes in the appropriate mounting methods was performed (Figure 2a). In the combination of DAB–Ni (CD68) and DAB (fascin) (Figure 2a1), only a few macrophages and dendritic cells showed the bluish-black color of DAB–Ni, but the number of cells thus labeled was extremely low. The combination of DAB– Ni (CD68) and AEC (fascin) (Figure 2a2) suggested that most cells labeled by DAB–Ni showed almost the same color as DAB. In the combinations of DAB (CD68) with AEC (fascin) (Figure 2a3) or VIP (fascin) (Figure 2a4), some cells were labeled by DAB, and the AEC and VIP showed a darker color than DAB. In the combination of SG (CD68) and AEC (fascin) (Figure 2a5) or NovaRED (fascin) (Figure 2a6), smaller numbers of cells showed double labeling of SG and AEC or NovaRED than in Figures 2a3 and 2a4.
Color analysis of the dyes in the HRP–H2O2 reaction
After glycine treatment.
Hematoxylin-counterstained nucleus.
Application of Double Autoimmunostaining
The localization of two antigens can be analyzed by double autoimmunostaining (Table 3).
In double autoimmunostaining using anti-CD68 and anti-fascin antibodies (Figure 2a), some cells showed cytoplasmic staining with both antibodies in the germinal centers of lymph follicles. Double autoimmunostaining with CD3 (anti-pan-T-cells) and CD79a (anti-pan-B-cells) could clearly differentiate the distribution of T- and B-cells in lymph nodes with follicular hyperplasia (Figure 2b). Double autoimmunostaining for p53 protein and Ki-67 antigen in squamous epithelial cells of the tongue (Figure 2c) characterized dysplastic cells and basal cells in comparison with hyperplastic cells.
Discussion
Double immunostaining was first introduced by Nakane (1968) with the enzyme-labeled antibody method, using glycine buffer at low pH to remove the immunoreaction products of the first primary antibody. In this study, we found that the residual immunoreaction products after glycine treatment disappeared when a protein blocker was introduced before incubation with the primary antibody. Although it was believed that glycine treatment needed a long incubation time for complete removal of the immunoreaction products, it was shown that dissociation of antigen–antibody binding happened instantaneously upon glycine treatment. However, the glycine solution we used was strongly acidic. As shown in Figure 1c4 and 1c6, glycine treatment remasked antigens that had been un-masked by AR, although it is unknown whether the strong acidity of the glycine solution remasked the antigen. Because dissociation of the immunoproducts from antigens may be instantaneous, long incubation in the glycine solution remasked the retrieved antigens. Because glycine treatment for 1 min is the shortest reaction for this autostainer, two additional changes of glycine solution were enough to remove the immunoreaction products (Table 2). Then the optimal glycine treatment condition was determined to be more than two washes for 1 min each time to wash out the dissociated immunoreaction products (Table 3) and was expected to have the least effect on AR.
Double autoimmunostaining. (
On the other hand, the heating method is also able to simultaneously remove the immunoreaction products of the first primary antibody and to retrieve the antigens that are the target of the second primary antibody (Lan et al. 1995; Bohle et al. 1997; Ezaki 2000). The Dako autostainer is able to perform double immunostaining with the heating method, performing steps 2 to 4 and 6 to 9 with the heating method to sections set off from the autostainer after step 4 (Table 1), although the handling of the sections is complicated. In the present study, double autoimmunostaining with glycine treatment and the HRP-labeled polymer method (EnVision) was introduced, twice performing serial IHC of the two antigens and deleting certain steps in the protocol. Because of the antigen-remasking effects of the glycine treatment on the retrieved antigen, such antigens must be the ones labeled initially in double autoimmunostaining.
Double immunostaining requires characteristic combinations of dyes that can be differentiated from the nuclear counterstaining, such as hematoxylin. Dyes in the HRP–H2O2 reaction showed various colors. Among such dyes, DAB, DAB–Ni, and SG could be applied to ordinary mounts to prepare permanent specimens. DAB, DAB–Ni, AEC, and SG withstood the Ultramount mounting protocol, whereas DAB, DAB–Ni, NovaRED, VIP and SG withstood the VectaMount mounting protocol. The glycine treatment faded AEC and changed the color of NovaRED and VIP, whereas DAB, DAB–Ni, and SG somewhat changed their R, G, and B elements (Table 4). It was suggested that DAB, DAB–Ni, and SG could be employed in the visualization of the first target antigen in the double immunostaining with glycine treatment. Analyzing the color of the dyes in the RGB system (Table 4) suggested that the combinations of DAB, SG, and hematoxylin, of DAB, AEC, and hematoxylin (Figure 2a3), of DAB, VIP, and hematoxylin (Figures 2a4 and 2b), of DAB, NovaRED, and hematoxylin, and of SG, NovaRED, and hematoxylin (Figure 2a6) were evaluated for use in double autoimmunostaining. The first combination was used for ordinary mounting, the second for Ultramount, and the last three for VectaMount. All combinations, except for those containing SG, can be applied for double immunostaining of two antigens that have different localizations, such as the nucleus, cytoplasm, and cell membrane, nucleus and cell membrane, and in different cells (Figure 2b). These combinations can even be applied for double immunostaining of two antigens showing the same localization in part, such as CD68 and fascin (Figure 2a). Although the sensitivity of SG was low compared with the other dyes except TMB, Figures 2a5 and 2a6 show clear differentiation of colors in combination with AEC or NovaRED. If high expression of antigen has biological or pathological significance, as does p53 protein, the combination of SG with AEC or NovaRED is a powerful analytical approach. As shown in Figure 2c, double immunostaining of p53 protein (SG) and Ki-67 antigen (AEC) may differentiate hyperplastic, dysplastic, and neoplastic squamous cells. The introduction of the alkaline phosphatase-labeled polymer method (EnVision-AP, K4018; DakoCytomation) and the new fuchsin visualization system (New Fuchsin, KO698; DakoCytomation) may facilitate double autoimmunostaining with glycine treatment, if the ordinary mounting method is needed (Table 3).
Any kind of IHC initially needs well-preserved or well-retrieved target antigens. In the field of fluorescence microscopy, new immunofluorescence probes have been introduced. When the target antigens are masked by any kind of tissue fixation, a confocal laser scanning microscope that can detect very weak fluorescence signals must be applied after double immunostaining of antigens in frozen sections with new immunofluorescence probes. In the field of electron microscopy, the freeze-substitution method has been introduced to preserve antigens (Hisano et al. 1984). In the field of light microscopy, especially using archival paraffin-embedded tissue sections from humans, recent advances in antigen retrieval (Shi et al. 2001) have opened a new field of IHC. Double autoimmunostaining of such retrieved antigens is a powerful tool for revealing relationships in the localization of such antigens. The standardized double autoimmunostaining reported here may be introduced in such double immunostaining because this protocol employed coupling dyes in the HRP–H2O2 reaction that is often used in immunohistochemistry.
Footnotes
Acknowledgements
Supported in part by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (KAKENHI, 13576003(B), 13557017(B) to K. Hasui).
We thank Ms Yuki Kamihira (DakoCytomation Co. Ltd.) for valuable information on double immunostaining and Mr Satoru Imamura for technical assistance.
