Abstract
The microarchitecture of the spleen is composed of a meshwork of reticulum cells and their matrix. Heparan sulfates (HS) are important components of this meshwork and are involved in processes such as cell adhesion, cell migration, and cytokine/growth factor binding. The expression of HS epitopes was analyzed using anti-HS antibodies. Four different staining patterns were observed, as exemplified by antibodies RB4EA12, HS4E4, AO4B08, and HS4C3. These antibodies recognize different chemical modifications in HS. In adult spleen, RB4EA12 stained only the reticular meshwork and blood vessels in the red pulp and marginal zone. HS4E4 stained blood vessel-associated basal lamina. AO4B08 and HS4C3 stained the reticular meshwork and blood vessels throughout the spleen, but only AO4B08 strongly stained smooth muscle cells and ring fibers. Interleukin-2 localized in the red pulp and marginal zone and was bound to HS. AO4B08, HS4C3, and RB4EA12 but not HS4E4 co-localized with interleukin-2. In 10-day-old spleen, HS4E4 recognized reticular fibers, which were not stained in the adult stage. Immunoelectron microscopy revealed that HS was restricted to basal laminae and reticular fibers. Taken together, data show that HS epitopes are differentially expressed in the spleen and that this may create specific extracellular environments for immunological processes.
Keywords
H
HS molecules are poorly immunogenic and monoclonal antibodies (MAbs) are difficult to raise. Therefore, we applied the antibody phage display technology and selected single chain variable fragment (scFv) antibodies against HS molecules (van Kuppevelt et al. 1998; Jenniskens et al. 2000; Dennissen et al. 2002). The anti-HS antibodies demonstrated differential staining patterns on tissue sections of kidney and muscle and showed different reactivity patterns with (modified) HS preparations from various organs and with synthetic HS oligosaccharides (van Kuppevelt et al. 1998; Jenniskens et al. 2000; Dennissen et al. 2002). These antibodies therefore define unique epitopes on HS chains.
The spleen is a secondary lymphoid organ composed of red pulp, the site of red blood cell destruction, and white pulp, containing T- and B-cell areas. The spleen has a unique vascular network and its framework is formed by a reticular meshwork consisting of fiber-forming reticular cells. These cells are abundant in the red pulp, where they form ring fibers (embracing the sinusoids) and the cords of Billroth. In the marginal zone, the border between the red and white pulp, they form a wide meshwork. In the white pulp, few reticular cells are found. In rats the marginal zone and white pulp, are sharply demarcated by the marginal sinus, which is covered with endothelium and also contains a layer of myofibroblasts and reticular cells (Steiniger and Barth 2000). Reticular fibers of the spleen are rich in ECM glycoproteins like laminin, collagen, tenascin, fibronectin and vitronectin (Liakka et al. 1991,1995; Liakka and Autio-Harmainen 1992; Ocklind et al. 1993). The expression of these molecules is differential throughout the reticular meshwork. Perlecan, an HS Pg, is located mainly in the red pulp of the spleen and in smooth muscle cells of arterioles (Murdoch et al. 1994; Wrenshall and Platt 1999). Biochemical analysis shows that HS molecules are also found in other lymphoid tissues, such as lymph nodes and thymus. They are synthesized by thymic epithelial cells (Werneck et al. 1999). HS molecules function as storage sites for growth factors and cytokines, and are involved in cell–cell and cell–matrix interactions. They bind immunologically important molecules such as IL-2, −3, −4, −5, −6, −7, −8, −12, GM-CSF, interferon-γ, and TNF-α (Bernfield et al. 1999). They are also involved in processes such as hematopoiesis (by mediating adhesion between stem cells and stromal cells and by presenting IL-7), and in inflammation (by presentation of IL-8 and MIP-1β) (Selvan et al. 1996). IL-2 binds to splenic HS, which occurs mainly in the red pulp and marginal zone but not in the white pulp. The binding of IL-2 to HS in the spleen contributes to the activation of T-cells and primes T-cells for activation-induced cell death (Wrenshall and Platt 1999). The variety of functions of HS suggests the presence of various HS domains and HS chains. To study this, we evaluated the topology of HS epitopes using scFv anti-HS antibodies in infant and adult rat spleen using immunohistochemistry and electron microscopy.
Materials and Methods
Materials
Rats (Wistar, male 8 weeks or 10 days old) and mice (Balb/c, male 4 weeks old) were obtained from the Central Animal Laboratory (UMC Nijmegen). Anti-c-myc tag mouse hybridoma cell line and anti-VSV tag mouse hybridoma cell line were obtained from the American Type Culture Collection (Rockville, MD; IgG, clone 9E10 and P5D4, respectively).
All chemicals used were purchased from Merck (Darmstadt, Germany), unless stated otherwise. Bacterial media (2XTY and LB) was from Life Technology (Paisley, Scotland). Heparinase III (from Flavobacterium heparinum, EC 4.2.2.8) and isopropyl-β-d-thiogalactopyranoside (IPTG), were from Sigma (St Louis, MO). Protease inhibitor cocktail was from Roche (Basel, Switzerland). Mowiol (4-88) was obtained from Calbiochem (La Jolla, CA). Restriction enzymes Not-1 and Nco-1 were from New England Biolabs (Beverly, MA). ABI Prism Big Dye Terminator Cycle Sequencing Ready Reaction Kit was from PE Applied Biosystems (Norwalk, CT).
Anti-HS stub antibody (3G10) was from Seikagaku Kogyo (Tokyo, Japan) (David et al. 1992). Rabbit anti-mouse laminin antibody was from Sigma (L-9393) and rabbit antihuman fibronectin antibody was from Dakopatts (Glostrup, Denmark), both antibodies crossreact with rat. Rat antimouse interleukin-2 antibody (S4B6) was from PharMingen (San Diego, CA). Rabbit anti-c-myc tag polyclonal IgG (A14) was from Santa Cruz Biotechnology (Santa Cruz, CA). Alexa 488-conjugated rabbit anti-mouse and Alexa 594-conjugated goat anti-rabbit IgG and goat anti-rat IgG were from Molecular Probes (Eugene, OR). All experiments were performed at ambient temperature (22C) unless stated otherwise.
Anti-HS scFv Antibodies
Anti-HS antibodies described in this study (HS3A8, HS3B7, HS4A5, HS4C3, HS4E4, AO4B08, AO4F12, RB4EA12, and EV3C3) were selected against HS from various sources. Antibodies with the prefix HS were selected against HS from bovine kidney; antibodies with the prefix AO were selected against HS from mouse skeletal muscle; antibodies with the prefix RB were selected against HS from human skeletal muscle; antibodies with the prefix EV were selected against HS from human lung (van Kuppevelt et al. 1998; Jenniskens et al. 2000; Dennissen et al. 2002). To allow double labeling experiments using two anti-HS scFv antibodies, Not I–Nco I fragments containing the full-length scFv antibody fragment, obtained after digestion of the pHEN phagemid, were subcloned into Not I–Nco I-digested pUC119His-VSV plasmid (J.M.H. Raats, unpublished). This vector is similar to the original pHEN1 vector (Hoogenboom et al. 1991; Nissim et al. 1994; Dennissen et al. 2002) but does not contain the c-myc tag and pIII gene. Instead, it contains a histidine and VSV tag for detection purposes. Subcloning was checked by DNA sequence analysis.
Preparation of scFv Antibodies
To produce scFv antibodies, E. coli clones containing phagemid DNAs expressing anti-HS scFv antibodies (van Kuppevelt et al. 1998; Jenniskens et al. 2000; Dennissen et al. 2002) were grown overnight in 2 × TY medium containing 1% (w/v) glucose and 100 μg/ml ampicillin. Four liters of 2 × TY medium containing 0.1% of glucose and 100 μg/ ml ampicillin was inoculated with 40 ml of overnight culture and grown to OD600 of 0.5–0.8 before protein expression was induced by IPTG. After 3-hr incubation at 30C, the culture was cooled for 15 min on ice and the cells were pelleted (10 min, 3000 × g at 4C). The cells were resuspended in 40 ml of ice-cold 200 mM sodium borate buffer (pH 8.0) containing 160 mM NaCl and EDTA-free protease inhibitor cocktail. The supernatant obtained after centrifugation (30 min, 5000 × g at 4C) was filtered through a 0.45-μm filter and dialyzed overnight at 4C against PBS, pH 7.4. This preparation, dubbed the periplasmic fraction and containing the antibodies, was stored at −20C.
Immunofluorescence Analysis (IFA)
Tissue specimens were snap-frozen in liquid nitrogen and stored at −80C. Tissue sections (5 μm) were incubated with periplasmic fractions of scFv anti-HS antibodies or with anti-laminin, anti-fibronectin, or anti-IL-2 antibodies in 1% (w/v) BSA in PBS with 0.05% Tween-20 (BSA/PBST) for 60 min. Bound antibodies were detected using anti-tag antibodies (anti-c-myc, monoclonal 9E10 or polyclonal A14; anti-VSV, P5D4) in BSA/PBST for 60 min, followed by Alexalabeled (488 or 594) anti-mouse IgG, anti-rabbit IgG, or anti-rat IgG antibodies. Finally, tissue sections were fixed in methanol, air-dried, and embedded in Mowiol [10% (w/v) in 0.1 M Tris-HCl, pH 8.5, 25% (v/v) glycerol, and 2.5% (w/v) NaN3]. As a control, primary antibodies were omitted. Double labeling experiments on rat tissue sections using two anti-HS antibodies were performed using a c-myc-tagged and a VSV-tagged antibody, followed by incubation with the anti-c-myc rabbit polyclonal antibody A14 and the anti-VSV monoclonal antibody P5D4. The staining was visualized by Alexa-labeled (488 and 594) anti-rabbit and anti-mouse IgG antibodies. Double labeling experiments on mouse tissue sections were performed using anti-mouse IL-2 antibodies and c-myc-tagged anti-HS antibodies, followed by the anti-c-myc rabbit polyclonal antibody A14. The staining was visualized by Alexa-labeled (488 and 594) anti-rabbit and anti-rat IgG antibodies. To evaluate the HS specificity of the anti-HS antibodies in rat or mouse spleen, tissue sections were pretreated with heparinase III to digests all HS [0.02 U/ml in 50 mM NaAc, 50 mM Ca(Ac)2, pH 7.0; overnight incubation at 37C]. As a control, cryosections were incubated with reaction buffer without enzyme. After HS digestion, tissue sections were washed in PBS and processed for immunofluorescence analysis as described above. Efficiency of HS digestion was analyzed by incubation with antibody 3G10 (David et al. 1992), which recognizes HS stubs generated after HS digestion. Staining patterns were analyzed by fluorescence microscopy (Axioskop microscope; Zeiss, Göttingen, Germany) and by confocal laser scanning microscopy (Nikon Diaphot inverted microscope attached to a Biorad MRC1024 ES confocal laser scanning microscope; Hemel, Hempstead, UK). Digital images were processed using Confocal Assistant 4.02 and Adobe Photoshop 6.0 software.
Immunoelectron Microscopy
For EM analysis of HS domains in rat spleen, a postembedding immunohistochemical technique was applied on thin sections obtained from material embedded at low temperature in Lowicryl HM20 resin. For this purpose, rats were anaesthestized and perfused with 20 ml 0.9% NaCl, followed by a mixture of 4% paraformaldehyde, 0.1% glutaraldehyde, and picrin acid in 0.1 M phosphate buffer, pH 7.4 (PB). The tissue was immersed overnight in perfusion fluid and sectioned at 100 or 200 μm on a vibratome. Ultrathin Lowicryl sections of the rat spleen were washed for 10 min in PBS (pH 7.4) containing 0.1% sodium borohydride and 50 mM glycine, and for 10 min in PBS containing 0.5% BSA and 0.1% cold water fish skin gelatin (PGB).
For immunolabeling, sections were incubated overnight at 4C in drops of PBG containing anti-HS antibody HS4C3 and HS4E4, diluted 1:30. Sections were washed for 20 min in PBG and incubated with anti-c-myc antibody 9E10. After washing in PBS the sections were incubated in goat anti-mouse immunoglobulin G (IgG H+L) labeled with gold markers (10 nm; Aurion Wageningen, Netherlands). Sections were washed in PBS and postfixed with 2.5% glutaraldehyde in PBS for 5 min to minimize loss of gold label during the contrasting steps. After washing with distilled water, sections were contrasted with uranyl acetate and studied using a JEOL TEM 1010 electron microscope.
Results
Anti-HS Antibodies
A panel of nine anti-HS antibodies (HS3A8, HS3B7, HS4A5, HS4C3, HS4E4, AO4B08, AO4F12, RB4EA1, and EV3C3) was analyzed for their reactivity with rat spleen. These anti-HS antibodies were generated using the phage display technology and were selected for reactivity against HS (van Kuppevelt et al. 1998; Jenniskens et al. 2000; Dennissen et al. 2002). Antibodies HS3A8 and HS4E4 have previously been described as RB4CD12 and RB4CB9, respectively (Jenniskens et al. 2000). All nine anti-HS antibodies reacted differentially with HS in rat spleen. Reaction patterns were divided into four major groups. Antibodies AO4F12, EV3C3, HS3A8 and HS3B7 stained similarly as AO4B08. HS4A5 stained similarly as HS4C3, although subtle differences were observed. For the sake of clarity, we describe the characteristics of the antibodies RB4EA12, HS4E4, AO4B08, and HS4C3 in detail. These four antibodies recognized unique HS domains, as illustrated by the preferred oligosaccharide sequence that they recognize (Table 1; (Dennissen et al. 2002). The antibodies recognize specific patterns of HS modification, such as sulfation, but this is at present still poorly defined.
Characteristics of anti-HS antibodies a
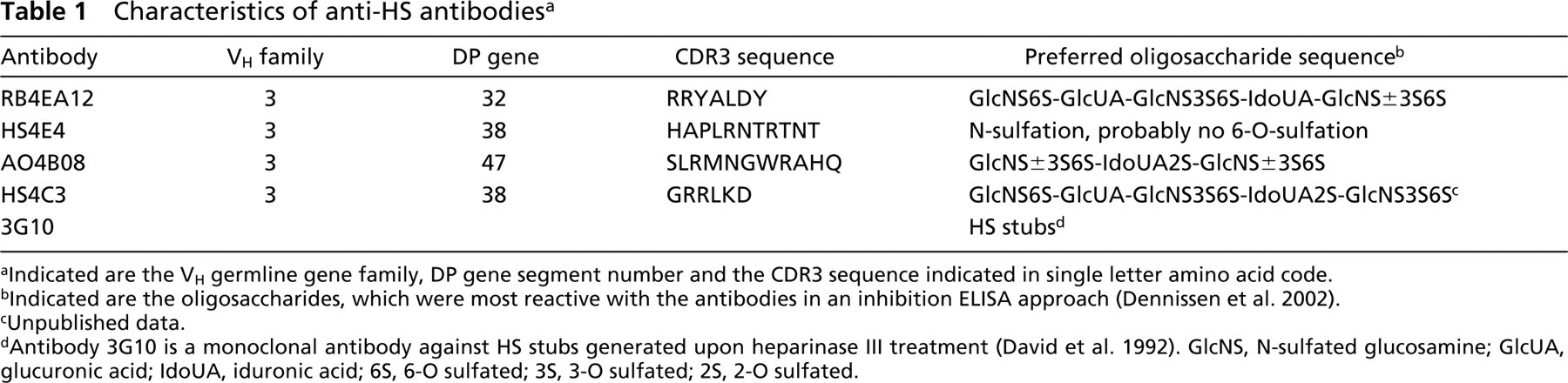
a Indicated are the VH germline gene family, DP gene segment number and the CDR3 sequence indicated in single letter amino acid code.
b Indicated are the oligosaccharides, which were most reactive with the antibodies in an inhibition ELISA approach (Dennissen et al. 2002).
c Unpublished data.
dAntibody 3G10 is a monoclonal antibody against HS stubs generated upon heparinase III treatment (David et al. 1992). GlcNS, N-sulfated glucosamine; GlcUA, glucuronic acid; IdoUA, iduronic acid; 6S, 6-O sulfated; 3S, 3-O sulfated; 2S, 2-O sulfated.
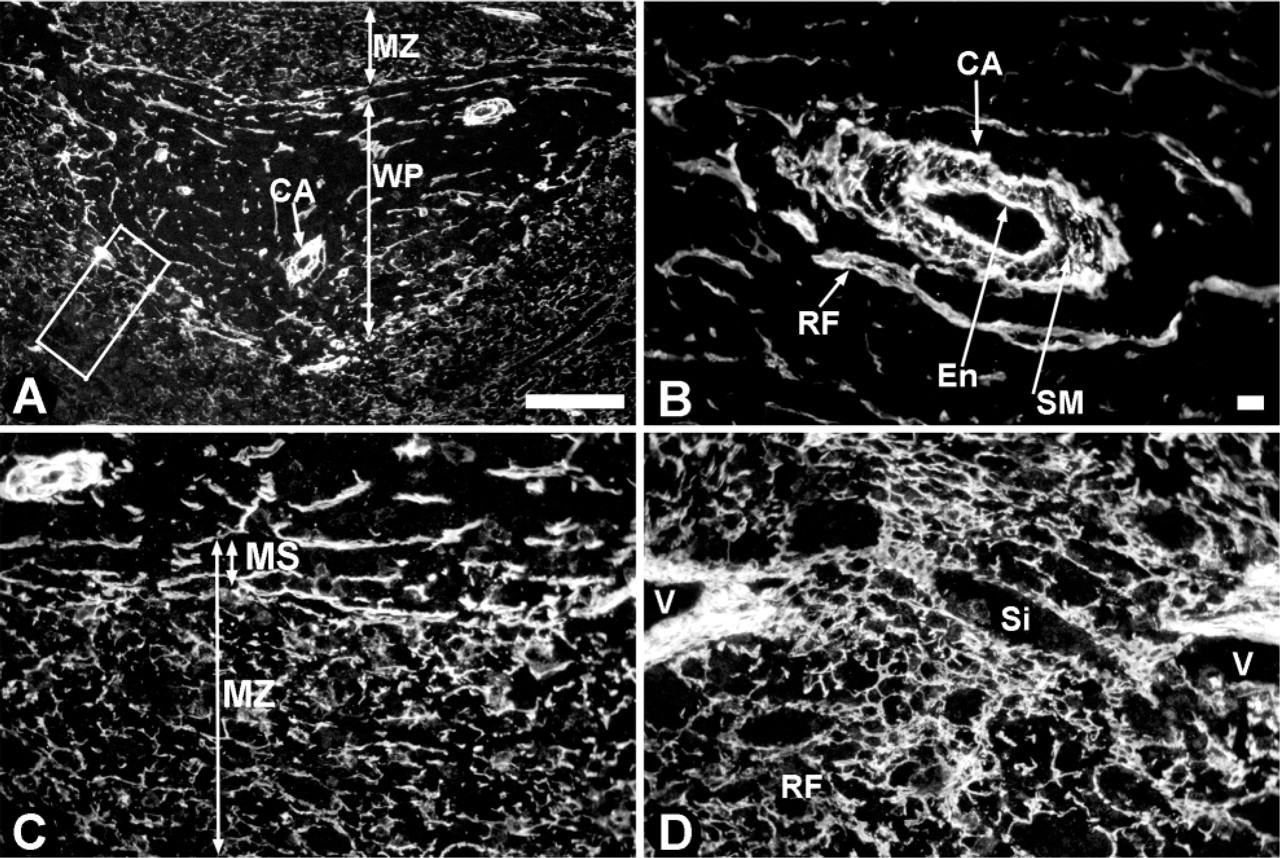
Localization of HS in rat spleen. Cryosections of rat spleen were incubated with heparinase III to digest all HS molecules and the HS stubs generated were detected by the 3G10 antibody. (
Immunofluorescence analysis of rat spleen using anti-HS antibodies
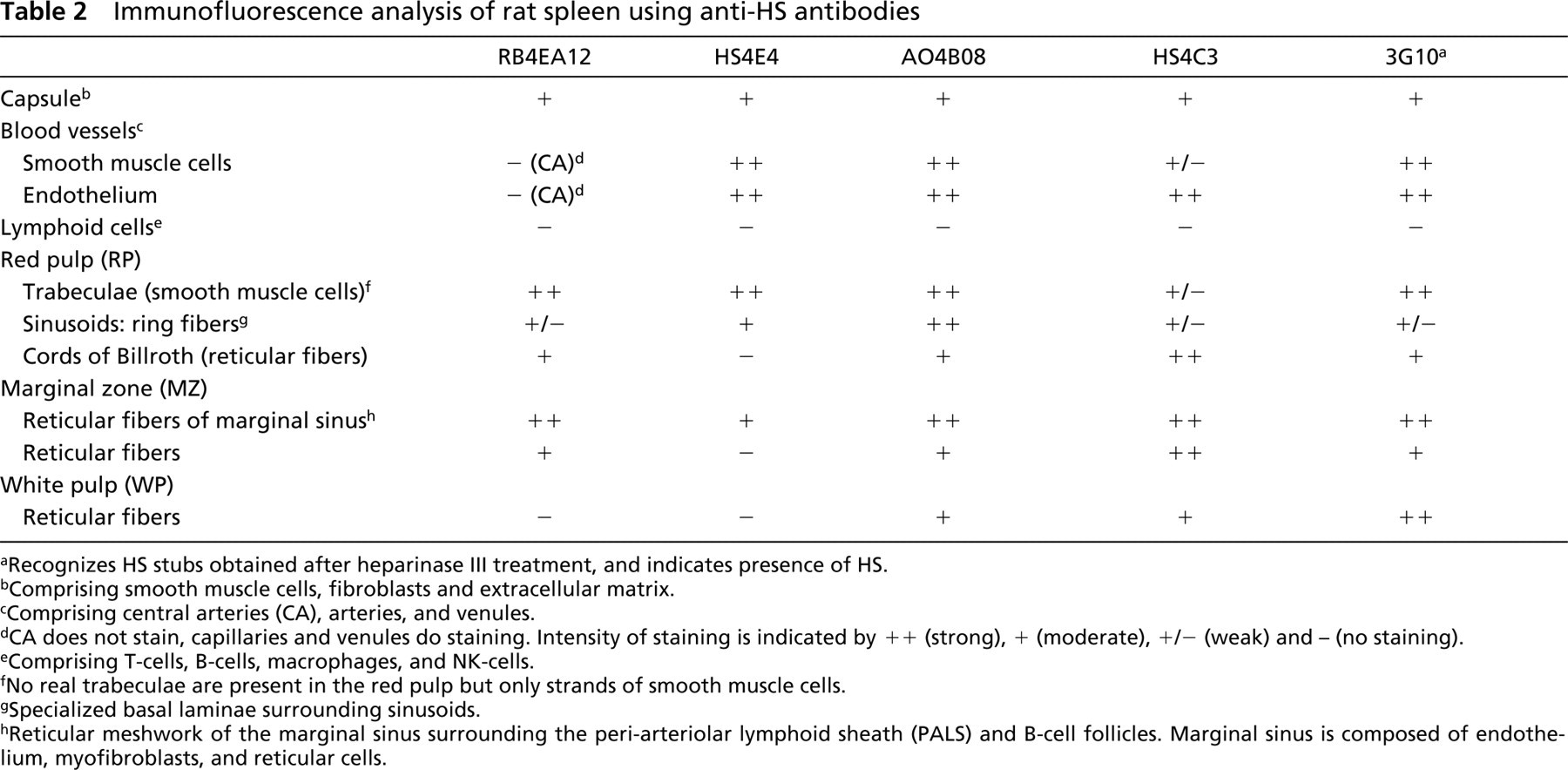
aRecognizes HS stubs obtained after heparinase III treatment, and indicates presence of HS.
bComprising smooth muscle cells, fibroblasts and extracellular matrix.
cComprising central arteries (CA), arteries, and venules.
dCA does not stain, capillaries and venules do staining. Intensity of staining is indicated by + + (strong), + (moderate), +/- (weak) and - (no staining).
eComprising T-cells, B-cells, macrophages, and NK-cells.
fNo real trabeculae are present in the red pulp but only strands of smooth muscle cells.
gSpecialized basal laminae surrounding sinusoids.
hReticular meshwork of the marginal sinus surrounding the peri-arteriolar lymphoid sheath (PALS) and B-cell follicles. Marginal sinus is composed of endothelium, myofibroblasts, and reticular cells.
Heparan Sulfates in Rat Spleen
To identify all HS present in the spleen, sections were treated with heparinase III (Figure 1; Table 2). This enzyme degrades HS and generates stubs, which can be detected by antibody 3G10. This anti-stub antibody was expressed strongly in the white pulp and moderately in the marginal zone and red pulp (Figure 1A). HS was highly expressed in blood vessels, in the endothelium as well as around the smooth muscle cells (see central artery in Figure 1B and venules in Figure 1D). The reticular fibers in the white pulp and marginal sinus stained strongly, whereas the fibers in the marginal zone were moderately stained (Figure 1C). The marginal sinus in rat spleen is composed of endothelium, myofibroblast, and reticular cells. Sinusoids and reticular fibers in the red pulp were also moderately stained (Figure 1D). The trabeculae in the red pulp, which are composed of smooth muscle cell strands (Steiniger and Barth 2000), were labeled strongly (not shown). No staining of lymphoid cells was observed.
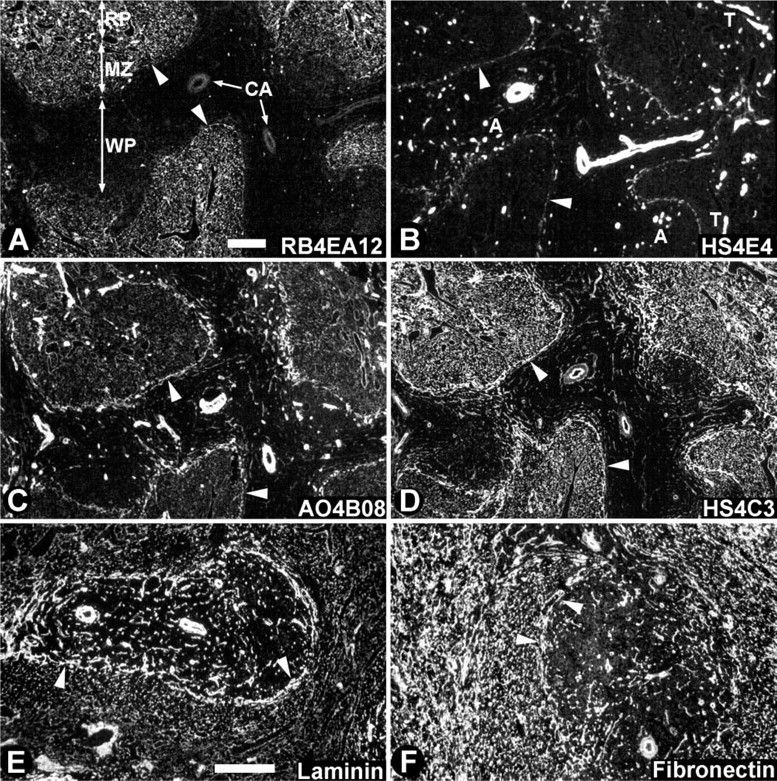
Localization of HS epitopes in rat spleen. Cryosections of rat spleen were incubated with anti-HS antibodies. These antibodies demonstrate a differential staining pattern, indicating a typical HS constitution in various morphological structures. Sections used in
Heparan Sulfate Epitopes in Rat Spleen
All anti-HS antibodies analyzed reacted with HS in rat spleen. Table 2 gives an overview of the morphological structures reactive with the various antibodies. Staining of all antibodies was abolished when sections were pretreated with heparinase III. However, with HS4C3 a very weak staining of nuclei and fibrils in the smooth muscle cell strands was observed (data not shown).
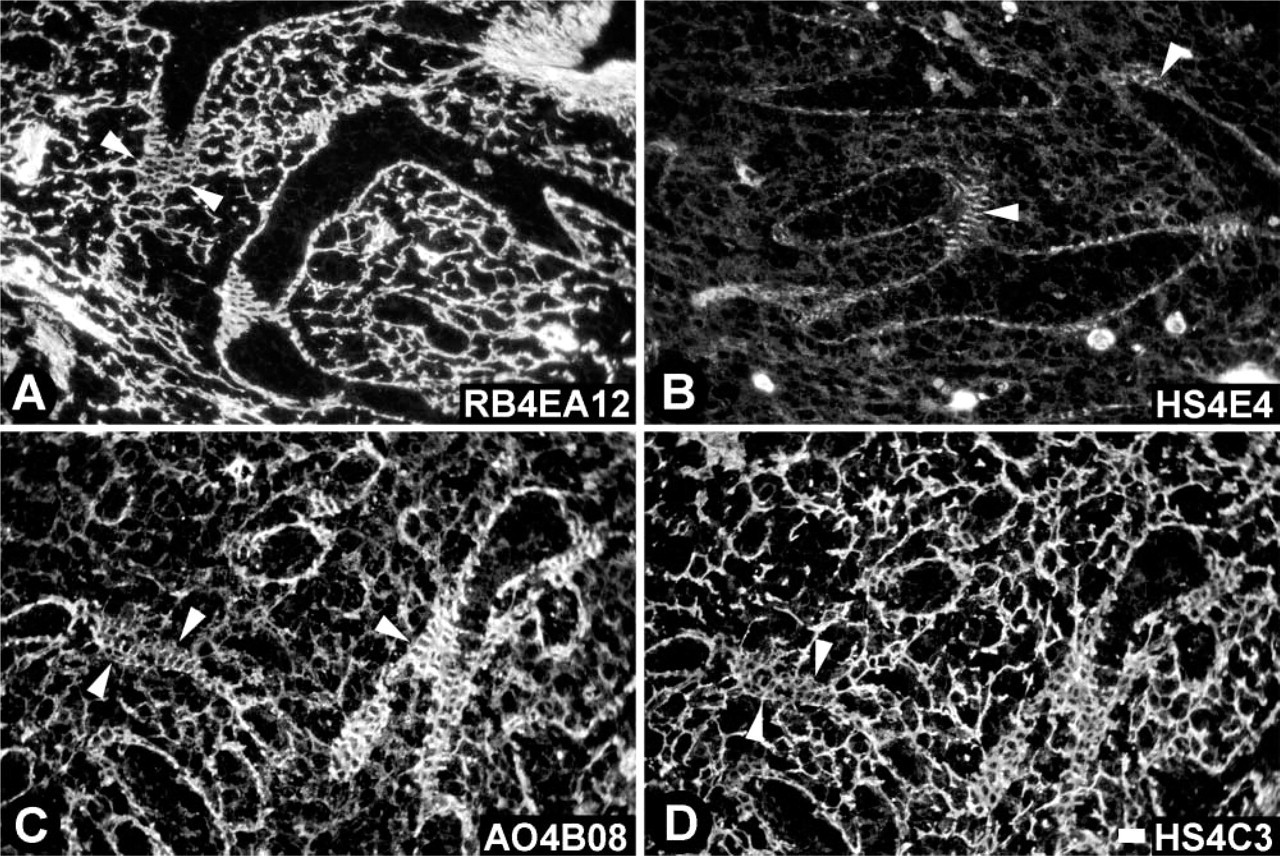
Antibody RB4EA12 (Table 2; Figures 2A and 3A) reacted with the reticular meshwork in the red pulp and marginal zone but not in the white pulp. Cords of Billroth and sinusoids in the red pulp and reticular fibers in the marginal zone and marginal sinus stained positively, whereas the reticular fibers in the white pulp (PALS and B-cell follicle) were negative. The ring fibers in the sinusoids stained only weakly with RB4EA12 (Figure 3A). RB4EA12 did not stain the central artery and other arteries in the white pulp, except for some small arteries near the marginal sinus. Trabeculae and venules stained strongly with RB4EA12. When lower concentrations of antibodies were used, the staining of the smooth muscle cells (in trabeculae and venules) and the reticular fibers in the marginal sinus faded, whereas staining of the reticular fibers in the red pulp and marginal zone remained.
Antibody HS4E4 (Table 2; Figures 2B and 3B) stained strongly with all blood vessels, including the central artery, smaller arteries, and venules. In addition, the fibers in the marginal sinus and the ring fibers (Figure 3B) of the sinusoids stained moderately with HS4E4. Trabeculae throughout the red pulp reacted strongly. All reticular fibers in the red pulp, white pulp, and marginal zone were negative.
Distribution of HS epitopes in ring fibers of sinusoids in rat spleen. Cryosections of rat spleen were incubated with the anti-HS antibodies RB4EA12 (
Co-localization of HS epitopes analyzed by confocal laser scanning microscopy in adult and infant rat spleen. Cryosections of adult (
Antibody AO4B08 (Table 2; Figures 2C and 3C) stained reticular fibers in the red pulp, white pulp, and marginal zone moderately. All blood vessels stained strongly, as did the trabeculae, the fibers of the marginal sinus, and the ring fibers in sinusoids (Figure 3C).
Antibody HS4C3 (Table 2; Figures 2D and 3D) strongly stained reticular fibers in the red pulp, marginal zone, and marginal sinus and moderately stained reticular fibers in the white pulp. Smooth muscle cells in trabeculae and blood vessels stained very weakly, in contrast to the endothelium of the blood vessels, which stained strongly. The ring fibers in sinusoids stained only weakly (Figure 3D).
HS expression patterns were compared with the expression patterns of laminin and fibronectin. Laminin, a large glycoprotein ubiquitously expressed in basal laminae, was strongly expressed in the reticular mesh-work of the marginal sinus and in reticular fibers of the white pulp, being more prominent in the PALS compared to the B-cell follicle. In addition, blood vessels and trabecular smooth muscle cells stained intensely, while the reticular meshwork of the red pulp, including sinusoids, stained moderately (Figure 2E). Fibronectin, a glycoprotein expressed in ECM, was expressed evenly in the reticular meshwork of the red pulp and was expressed slightly less prominently in the white pulp. Blood vessels and smooth muscle cells also expressed fibronectin (Figure 2F).
Co-localization of Heparan Sulfate Epitopes in Rat Spleen
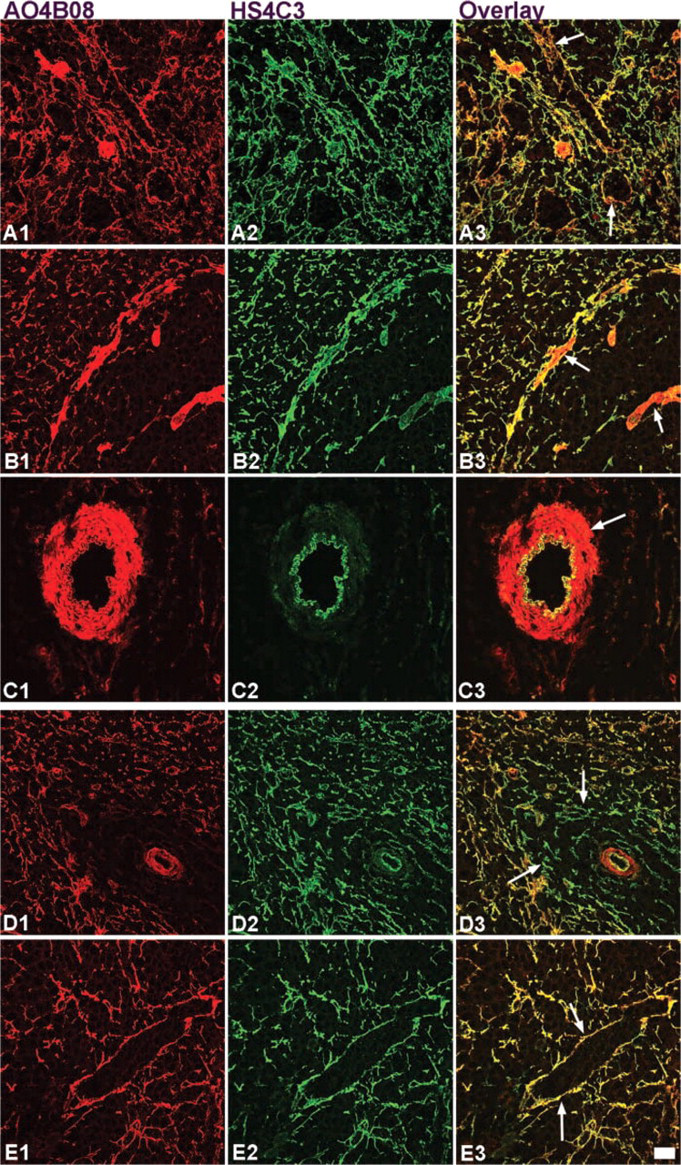
Co-localization studies using confocal laser scanning microscopy further demonstrated that expression of specific HS epitopes occurred at identical as well as at different sites. Staining patterns of the antibodies AO4B08 (Figures 4A1–4C1; red) and HS4C3 (Figures 4A2–4C2; green) combined in the overlay (Figures 4A3–4C3) clearly demonstrated that HS4C3 is barely expressed in the ring fibers of the sinusoids, in contrast to AO4B08 (Figure 4A3). A special type of reticular cells, probably myofibroblast, present in the marginal sinus (Figure 4B3) and smooth muscle cells in the central artery (Figure 4C3) were only weakly stained by HS4C3 but strongly by AO4B08. Reticular fibers throughout the spleen and the endothelium of blood vessels (see CA in Figure 4C3) showed overlap of staining with HS4C3 and AO4B08.
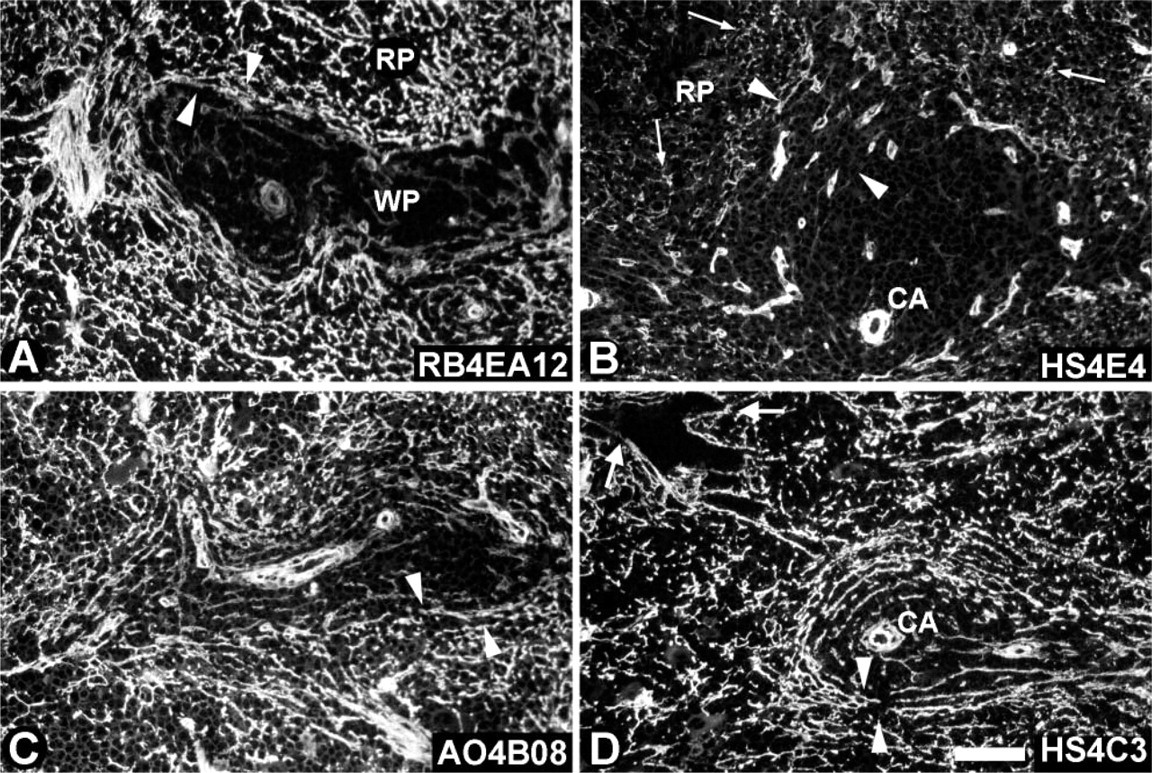
Localization of HS epitopes in infant rat spleen. Cryosections of rat spleen of 10-day-old animals were incubated with anti-HS antibodies. (
Heparan Sulfate Epitopes in Infant Rat Spleen
HS expression in infant rat spleen (10-day-old rats; Figure 5) was compared with HS expression in adult spleen (Figure 2). Some differences observed in staining pattern between infant and adult rat spleen could be ascribed to not fully matured structures in infant spleen, including a not fully immunologically developed white pulp. Staining of sinusoids and marginal sinus in infant spleen was different from that in adult spleen. In infant spleen, sinusoids were often stained as dotted lines or hardly stained at all, the marginal sinus being rough and spacious (arrowheads, Figure 5). In the adult spleen, the sinusoids and the marginal sinus were clearly lined and marked. Most remarkable was the difference observed with HS4E4. This antibody stained fibers in the red pulp and marginal zone in infant spleen (Figure 5B), but no staining of reticular fibers in adult spleen was observed (Figure 2B).
Double labeling experiment and confocal laser scaning microscopy with the antibodies AO4B08 (Figures 4D1–4E1; red) and HS4C3 (Figures 4D2–4E2; green) revealed even more differences. Antibody AO4B08 stained the reticular fibers in the white pulp in adult spleen but not in infant spleen (compare Figures 4B1 and 4D1; Figure 4D3). No differences in staining were observed in the red pulp area (Figures 4E1–4E3), while in adult spleen, ring fibers stained with AO4B08 but not with HS4C3 (compare Figures 4A3 and 4E3).
HS Dependence of Interleukin-2 Binding in Mouse Spleen
To probe the possible functional significance of the differential expression of HS epitopes, we investigated, first, if IL-2 was bound to tissue by HS and, second, if certain HS epitopes co-localized with IL-2. We used mouse spleen instead of rat spleen because of the availability of anti-mouse IL-2 antibodies. Interleukin-2 was located in the red pulp and marginal zone but was absent in the white pulp (Figure 6A; note that the marginal sinus in the mouse spleen is not as clearly demarcated as in the rat spleen). After digestion with heparinase III, staining for interleukin-2 was almost abolished (Figure 6B). Partial co-localization was observed between IL-2 (Figure 6C) and the HS epitope recognized by antibody AO4B08 (Figure 6D). Lymphocyte nests that expressed IL-2 were closely embraced by HS-expressing reticular fibers (as exemplified by AO4B08). The antibodies HS4C3 and RB4EA12 showed similar co-localization patterns (not shown). No co-localization was observed between IL-2 (Figure 6E) and the HS epitope defined by antibody HS4E4 (Figure 6F).
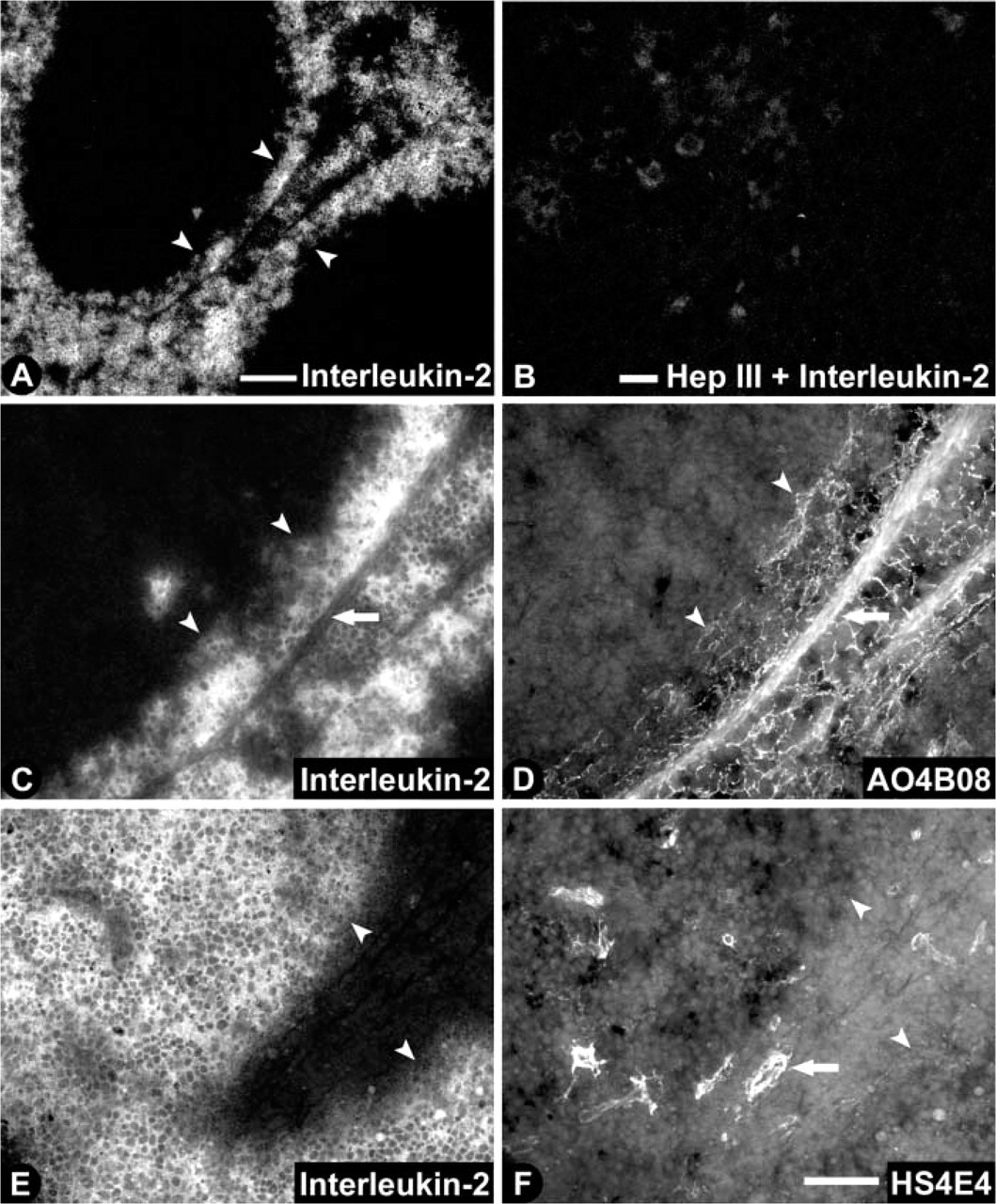
HS dependence of interleukin-2 binding in mouse spleen. Cryosections of mouse spleen were labeled with interleukin-2 without (
Immunoelectron Microscopic Analysis of HS Epitopes in Rat Spleen
Immunoelectron microscopy was performed to reveal the precise location of HS in the spleen. The antibodies HS4C3 and HS4E4 were analyzed for this purpose. Both antibodies demonstrated labeling patterns comparable with the immunofluorescence analysis. HS4C3 showed labeling of reticular fibers in the white pulp (Figure 7A). Intense labeling was found in fibers lining the marginal sinus, surrounding the white pulp (Figures 7B and 7C). Endothelial cells, lining the marginal sinus, and reticular cells were in close contact with the labeled reticular meshwork. In sinusoids, the ring fibers were labeled (Figure 7D). HS4C3 labeling seemed most intense near the contact side of the ring fibers with the reticular cells. Cords of Billroth were also labeled. Figure 7E shows two reticular cell extensions enveloping a labeled reticular fiber. Intracellular labeling was occasionally observed (Figure 7F). Labeled Golgi vesicles and lysosomal vesicles were found in reticular cells. HS4E4 showed very intense labeling of the basal lamina of the central artery (Figure 7G). Labeling was mainly observed in electron-lucent areas of the basal lamina; electron-dense areas were less labeled. Labeling was also found in the basal laminae of smooth muscle cells of the central artery. Smaller arteries were also labeled with HS4E4 but less intensely. Figure 7H shows labeling of a small artery near the marginal sinus. Weak labeling was also found in fibers lining the marginal sinus.
Discussion
Heparan sulfate proteoglycans (HSPGs) are major components of the extracellular matrix (ECM) in the spleen, in addition to glycoproteins such as laminin, fibronectin, type I, III, and IV collagen, tenascin, and vitronectin (Liakka et al. 1991,1995; Liakka and Autio-Harmainen 1992; Ocklind et al. 1993). HS were expressed throughout the spleen, being most prominent in the marginal sinus and in the white pulp area. This expression pattern showed high similarities with laminin, indicating that HS is largely confined to basal laminae structures, which was confirmed by immunoelectron microscopy.
Here we have described the expression of HS epitopes using phage display-derived anti-HS antibodies (Jenniskens et al. 2000; Dennissen et al. 2002). Four major staining patterns were discerned as exemplified by the antibodies RB4EA12, HS4E4, AO4B08, and HS4C3. The RB4EA12 epitope was expressed around smooth muscle cells and by reticular fibers in the red pulp and marginal zone but not in the white pulp. IdoUA-containing disaccharides are probably barely present in the white pulp because this saccharide is thought to be essential for binding of the RB4EA12 antibody (Table 1; Dennissen et al. 2002). Antibody HS4E4 reacted only with basal laminae of endothelial cells and smooth muscle cells, and these structures therefore are probably rich in HS containing GlcNS-IdoUA2S disaccharides. These disaccharides are ubiquitous in HS from human aorta, with which the antibody HS4E4 is highly reactive (Dennissen et al. 2002; Safaiyan et al. 2000). Antibody AO4B08 demonstrated a rather broad staining pattern in the spleen. All structures stained with this antibody, suggesting a common epitope containing 2-sulfated iduronic acid residues because this saccharide is believed to be important for antibody binding (Dennissen et al. 2002). The HS4C3 epitope was also broadly expressed but, in contrast to AO4B08, was only weakly expressed in smooth muscle cells and ring fibers. Antibody HS4C3 recognized HS containing GlcNS3S6S-IdoUA2S disaccharide units [3-O sulfation is of special importance (unpublished data)], suggesting that 3-O sulfation rarely occurs in the latter structures.
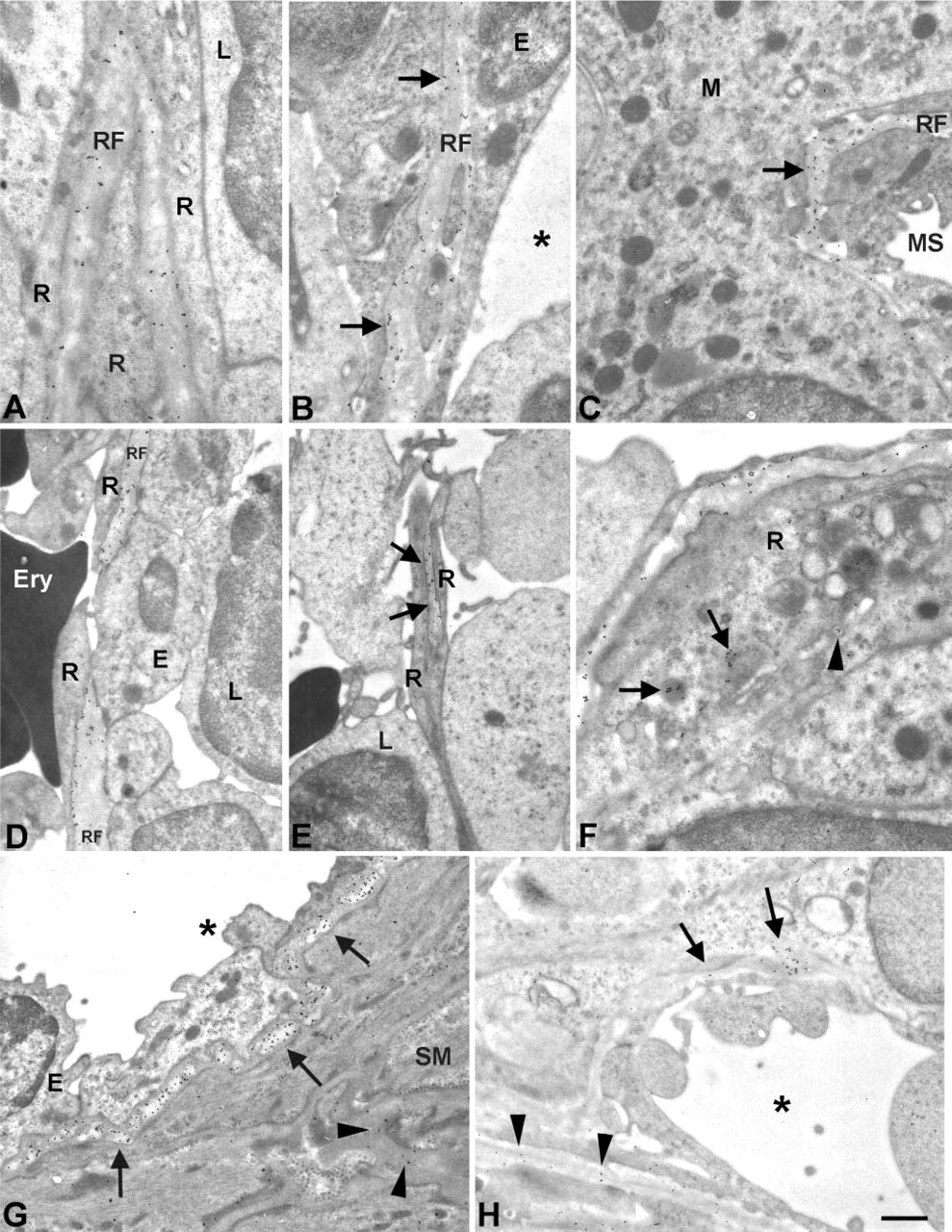
Immunoelectron microscopic analysis of distribution of HS epitopes in rat spleen. Immunoelectron microscopic localization of HS epitopes recognized by HS4C3 (
HS epitopes were differentially distributed throughout the spleen, and so was IL-2. This cytokine was localized in red pulp and marginal zone, and bound to HS (Figure 6; Wrenshall and Platt 1999). Interleukin-2 partially co-localized with HS epitopes AO4B08, HS4C3, and RB4EA12 but not with epitope HS4E4 (Figure 6). This indicates that specific HS structures may be associated with IL-2 binding, and possibly also other immunological effector molecules (Spillmann et al. 1998; Bernfield et al. 1999). Different HS may create specific microenvironments suitable for sequestering various cytokines/growth factors, which would facilitate the interaction of specific molecules at specific sites.
All anti-HS antibodies showed strong labeling of the marginal sinus, indicating the variety of HS epitopes present. This area borders the lymphoid cells in the white pulp and is involved in trafficking of lymphocytes. The marginal sinus lining cells express adhesion molecules such as L1 and Madcam-1 (Liakka et al. 1995; Tanaka et al. 1996; Wang et al. 2000). The strong expression of HS domains in the marginal sinus may imply a role in lymphocyte homing to the white pulp.
When infant and adult spleen were compared differences in staining were noted, especially for epitope HS4E4. This epitope was found in reticular fibers in infant but not in adult spleen suggesting a maturation process of the HS molecules into adulthood, possibly involving 6-O sulfation. An age-dependent increase of 6-O sulfated HS has been noted (Feyzi et al. 1998), and 6-O sulfation is probably inhibitory for binding of the HS4E4 antibody to HS (Dennissen et al. 2002). Differences observed for other epitopes could be attributed to incompleteness of morphological structures in the infant stage. Others have demonstrated that the ring fibers in human infant spleen are immature and that ECM components such as tenascin and vitronectin are distributed differently in infant compared to adult spleen (Liakka et al. 1991,1995; Liakka and Autio-Harmainen 1992).
In conclusion, HS epitopes were differentially expressed in the spleen, and this might create a unique environment specialized for optimal functioning of lymphoid cells. The phage display-derived antibodies used in this study may be instrumental in the analysis of the role of specific HS domains in lymphoid organs.
Footnotes
Acknowledgements
This study was financed by the EU project BIO4 CT 970 538.
We thank Dr J.M.H. Raats (Dept. of Biochemistry, Faculty of Sciences, University of Nijmegen, Nijmegen, The Netherlands) for providing the pUC119His-VSV vector, and IBEX Technologies (Montreal, Quebec, Canada) for providing recombinant heparinase III derived from Flavobacterium heparinum. We thank Dr L.G. Poels for critically reading the manuscript and helpful discussions and Dr P.H.K. Jap for detailed analysis of the immunoelectron microscopy data.
