Abstract
A family of growth factors highly specific for endothelial cells was identified more than 10 years ago, in which the receptor of vascular endothelial growth factor C (VEGFR-3) is implicated in the regulation of lymphatic development and regeneration. Comparative studies on the lymphatic network and lymphangiogenesis have been done mainly using combined 5′-nucleotidase (5′-Nase) enzyme and VEGFR-3 immunohistochemical approaches in adult and fetal gastric walls. Developing lymphatic networks represented fewer blind ends and branches than mature networks in whole-mount preparations. Many circular lymphatic-like structures exhibited VEGFR-3 expression and weak 5′-Nase activity in the early embryonic stage, showing visible morphological properties in the lymphatic endothelium. These newly formed lymphatics showed an obvious accumulation in the submucosa and serosa and a variation in the intensity of VEGFR-3 binding to endothelial cells among samples. A reaction product for anti-VEGFR-3 was found on the luminal surface of endothelial cells and on the membrane of some organelles and intraluminal lymphocytes. These findings indicate that an active proliferating feature of the clustered developing lymphatics may create a favorable environment for their sprouting and growth, which may serve as a functional requirement for lymph drainage in the region.
T
Materials and Methods
Three adult (4–6 kg), two young (660–720 g), and three fetal (170–500 g) Japanese monkeys (Macaca fuscata) of both sexes were anesthetized with ketamine (10 mg/kg, IM) with the approval of the Animal Research Ethics Committee of Oita Medical University.
Enzyme Histochemistry
Samples for histochemical identification and analysis were frozen fresh or fixed with 4% paraformaldehyde in 0.1 M PBS, pH 7.4, or 0.1 M cacodylate buffer (pH 7.2). After fixation, whole-mount preparations were processed in intact or peeled gastric walls. Tissue blocks were cut into small pieces and refixed for 3–4 hr for electron microscopic observation.
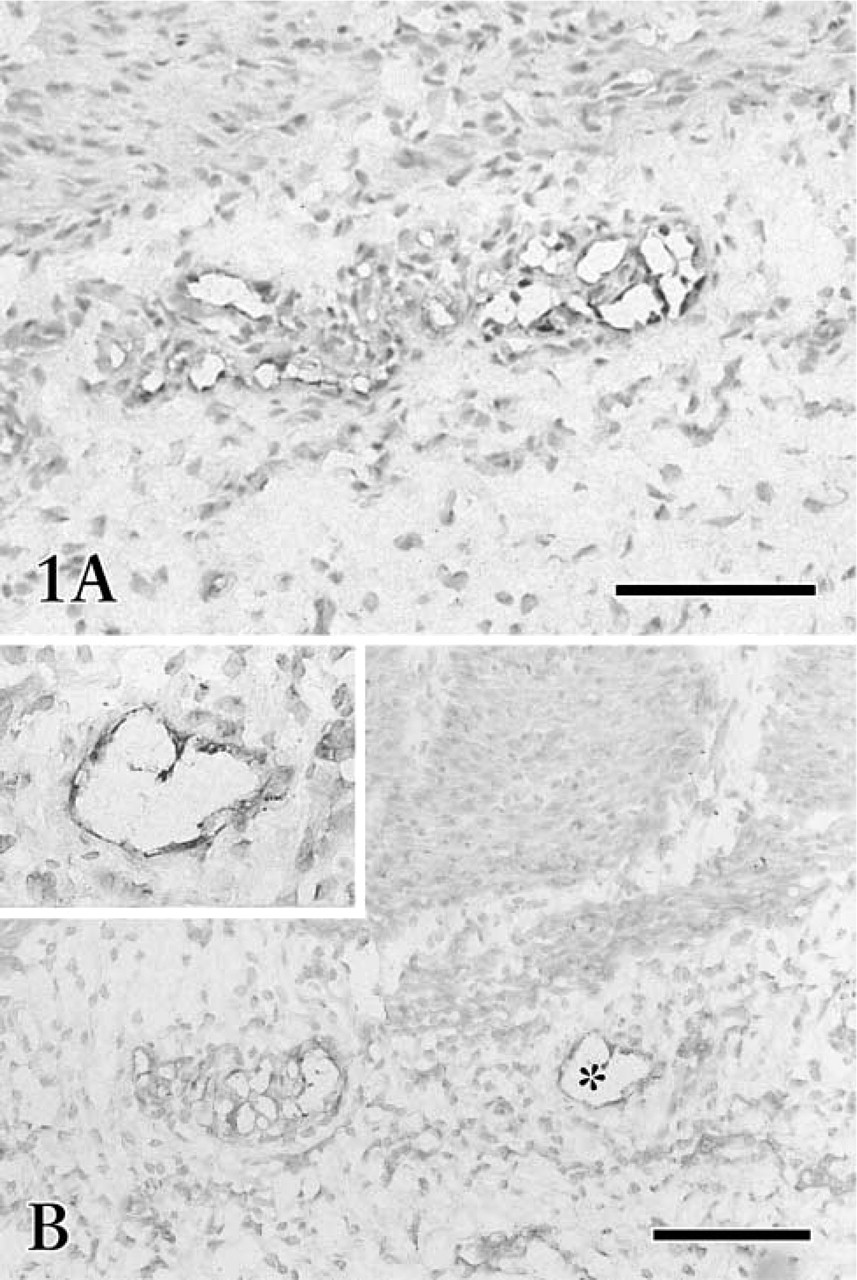
Photomicrographs of fetal gastric cryosections treated with VEGFR-3 staining. VEGFR-3-expressing lymphatic-like structures show an obvious accumulation in the submucosa (
5′-Nucleotidase (5′-Nase) Staining. 5′-Nase activity for demonstrating lymphatics in whole-mount preparations and sections was conducted in the standard medium in addition to 2 mM L-tetramisole for 30–40 min at 37C. The lead-based medium contained 1.45 mM adenosine 5′-monophosphate (AMP; Sigma, St Louis, MO) and 1.8 mM Pb(NO3)2. Samples were immersed in 1% ammonium sulfide solution for 1–2 min at room temperature (RT) and then examined under a light microscope. The cerium-based staining was treated in the ultrathin sections for transmission electron microscopic (TEM) analyses. The medium consisted of 1 mM AMP and 2 mM CeCl3. The incubation lasted for 25–30 min at 37C (Ji and Kato 2000).
Alkaline Phosphatase (ALPase) Staining. ALPase activity for identifying blood vessels was applied in whole-mount preparations and sections for 40–50 min at 4C. The medium contained naphthol AS-MX phosphate and Fast Blue BB salt. For controls, the substrates were omitted from the reaction medium (Kato et al. 1996).
Immunohistochemistry
Successive 5–7-μm cryosections were air-dried and fixed in cold pure acetone or 4% paraformaldehyde for 10 min. Samples were rinsed with 0.1 M PBS and incubated with 0.3% H2O2 for 30 min to remove endogenous peroxidase activity. The sections were incubated with 10% blocking serum (normal goat serum, NGS or normal rabbit serum, NRS) and then with rabbit anti-human and rabbit anti-mouse polyclonal antibodies against the receptor of vascular endothelial growth factor C (VEGFR-3) (Santa Cruz Biotechnology; Santa Cruz, CA; Alpha Diagnostic International; San Antonio, TX) at a concentration of 1:50–1:200, rabbit anti-mouse laminin, rabbit anti-bovine type IV collagen (LSL Co; Tokyo, Japan) in dilutions of 1:1000–1:2500, mouse anti-human monoclonal antibody against α-smooth muscle actin (Sigma) diluted 1:500, and rabbit anti-bovine endothelial nitric oxide synthase (eNOS) (Wako Industries; Osaka, Japan) diluted 1:500 for 60 min at RT or overnight at 4C. A subsequent incubation for 40 min in biotinylated goat anti-rabbit IgG or rabbit anti-mouse IgG, diluted 1:100, was followed by a 40-min incubation using reagents of the streptavidin-biotinylated peroxidase complexes. The samples were rinsed in PBS and peroxidase was visualized by incubation with 0.03% 3,3′-diaminobenzidine tetrahydrochloride (DAB) solution in 0.05 M Tris-HCl, pH 7.4. Sections were lightly counterstained with 1% methyl green or hematoxylin and examined by light microscopy. Negative controls were done by using nonimmune serum or by omitting the primary antibodies.
The antigenic site of VEGFR-3 was further examined in ultrathin sections (85–90 nm) by a postembedding immunogold staining method. Fixed samples were dehydrated in a graded ethanol series at 4C, with a 1:1 LR White:100% ethanol mixture for 1 hr before being transferred to 100% LR White for 1 hr with two changes. Tissues were then embedded in LR White by using a gelatin capsule. The resin was polymerized at 60C for 24 hr. LR White sections mounted on Formvar-coated nickel grids were preincubated for 5 min face down on drops of buffer containing 1% bovine serum albumin (BSA) and transferred to a drop of 10% NGS in rinsing buffer for 15 min. Sections were then incubated on a drop of anti-VEGFR-3 diluted 1:100. Unbound primary antibody was then rinsed off the grids with buffer, and incubation was continued with goat anti-rabbit IgG bound to 5 nm gold particles (Auroprobe EM-GAR G5; Janssen, Beerse Belgium) in a 1:70 dilution. Silver enhancement was performed in some samples after they were rinsed to remove excess unbound gold particles (Ji and Kato 2001). The sections were counterstained for 3–6 min with diluted aqueous uranyl acetate and lead citrate and examined with a JEM 1,200 EX II electron microscope.
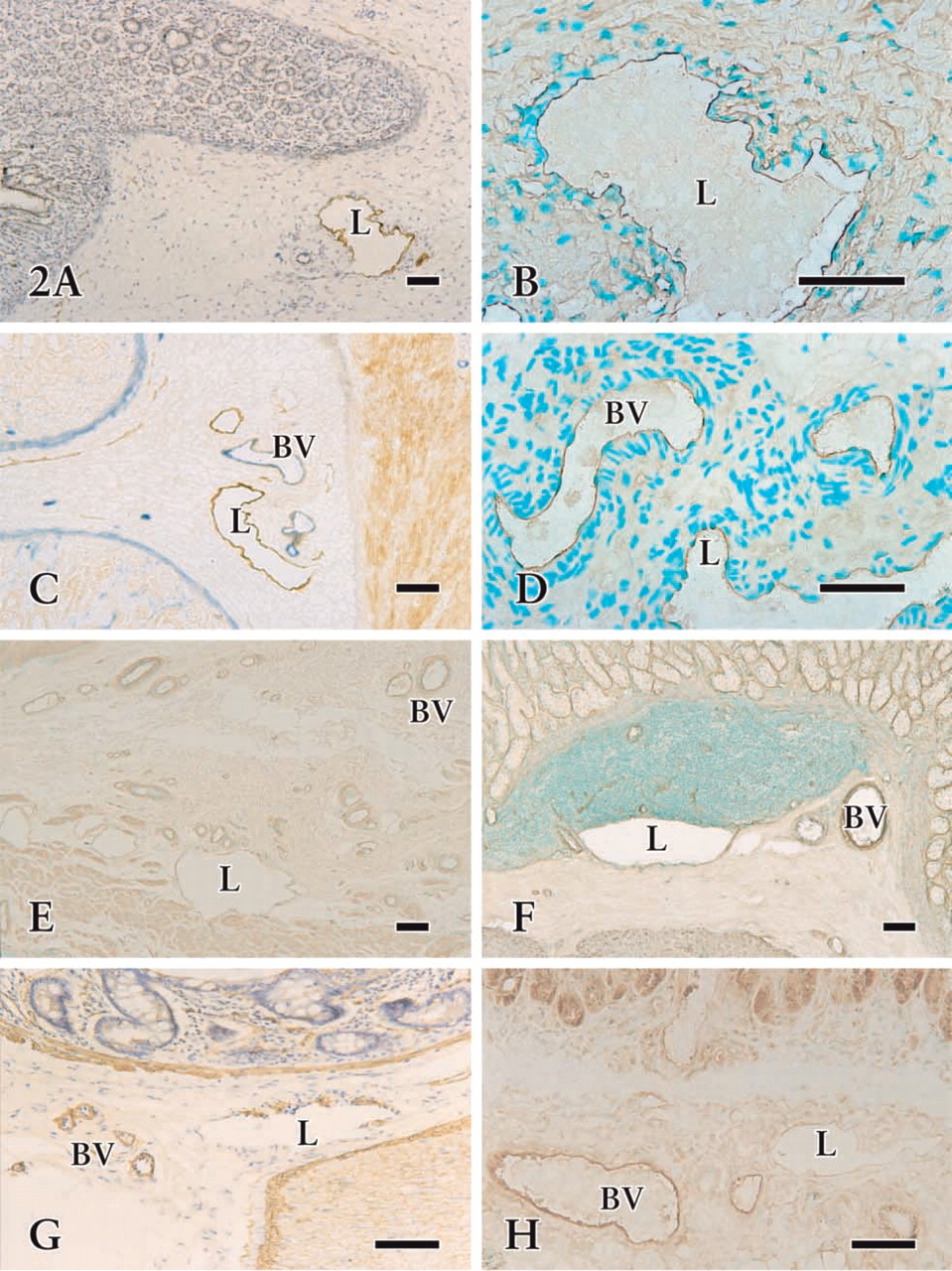
Photomicrographs of cryosections in fetal (
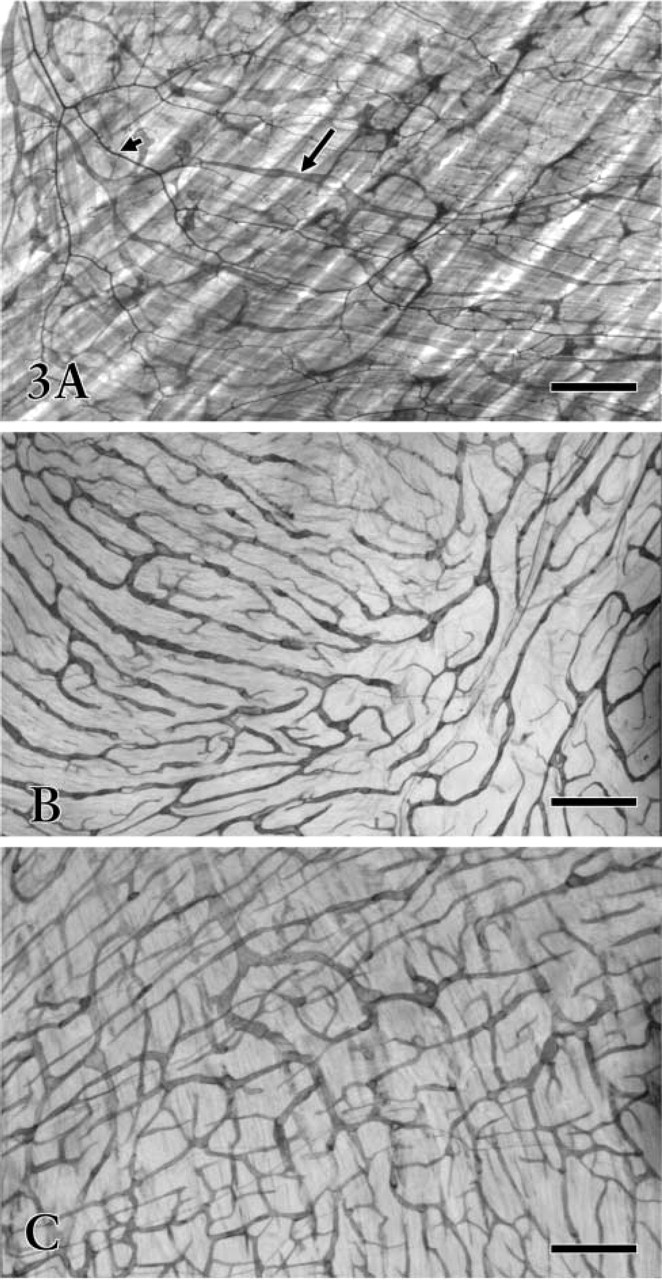
Whole-mount preparations of fetal (
Scanning Electron Microscopy (Backscattered Electron Imaging; BEI-SEM)
The tissue sections incubated in the 5′-Nase-lead-based medium were dehydrated in an ascending concentration of ethanol and treated using the t-butyl alcohol freeze-drying method. Dried samples were mounted on an aluminum stub, coated with carbon, and then examined in an SEM S-800 with a GW type 30 backscattered electron detector (Hitachi Science Systems; Tokyo, Japan).
Lymphatic Corrosion Casts
Under anesthesia, the Mercox medium (CL-2B or CL-2R; Japan Vilene Hospital, Tokyo, Japan) diluted to 40–50% (v/v) with methyl methacrylate monomer was intramurally injected into the submucosa of the gastric wall and directly into the superior mesenteric artery. The injected organs were removed and placed in a hot water bath (60C) for more than 2 hr. They were transferred into 15% NaOH at 60C until tissue components were completely corroded away. The corrosion casts of lymphatic and blood vessels were gently washed and coated with gold, then observed in a Hitachi S-800.
Results
Lymphangiogenesis in the Fetal Stomach
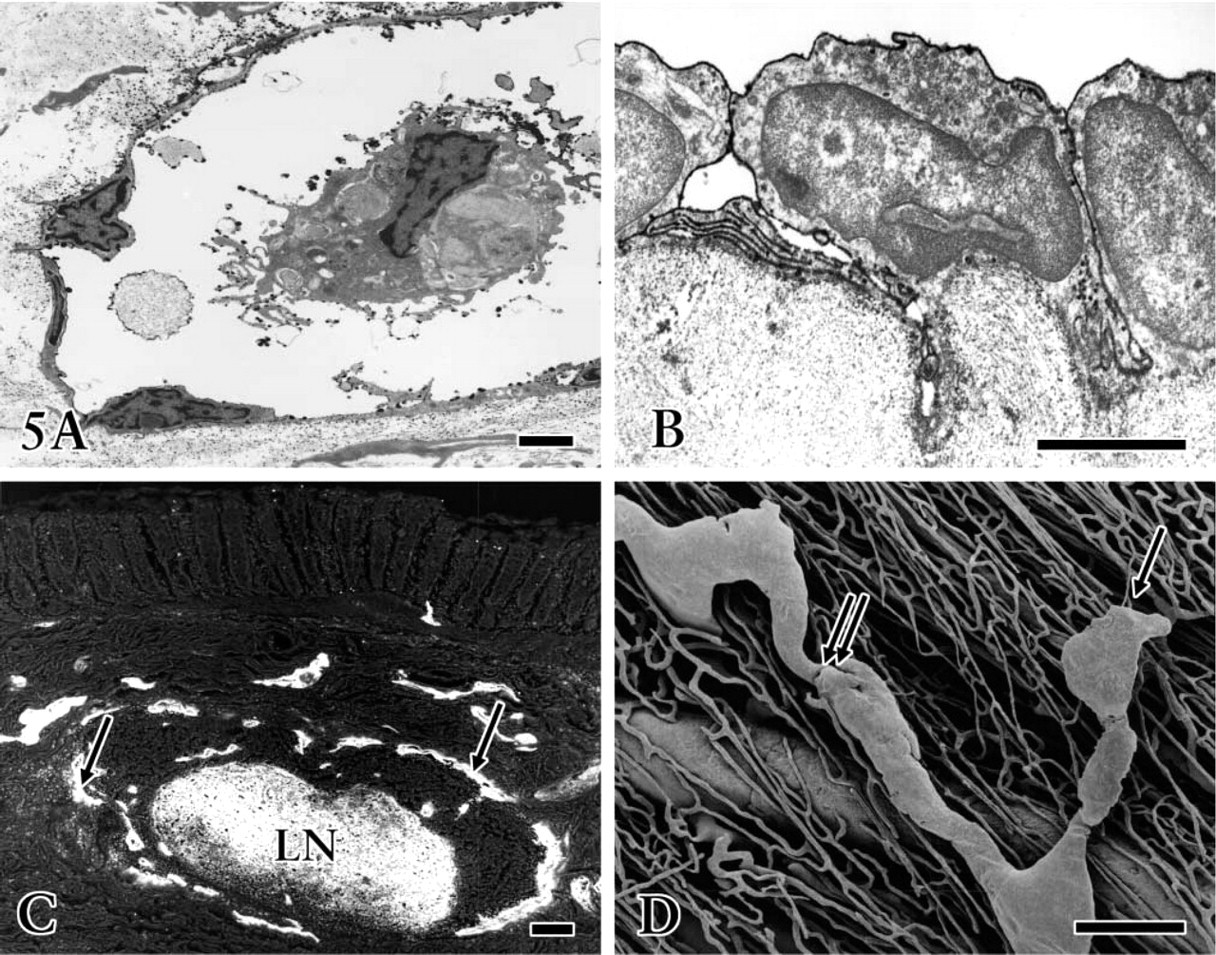
Immunohistochemical staining of the cryosections revealed that the irregularly outlined lymphatic-like structures reacting with anti-VEGFR-3 were mainly restricted to the submucosa and serosa (Figures 1 and 2A-2D). In the early developing gastric wall, anti-VEGFR-3 was expressed in a cluster of circular lymphatic-like structures, which were gathered together in several groups (Figure 1). VEGFR-3 binding to endothelial cells showed a variation in staining intensity in the lymphatic wall and among samples. Immature lymphatic vessels were usually stained more intensely than typical lymphatic vessels. Quite a number of small blood vessels expressed similar immunoreactivity to lymphatics (Figures 2C and 2D), although the endothelium of large blood vessels was virtually absent in the reactivity. The developing lymphatic network was seen to have fewer blind ends and branches than the mature network in the whole-mount preparations. These networks underwent obvious morphological changes in mesh density, communicating branches, valve-like structures, and blind ends from the fetal to adult stages (Figures 3 and 4). Initial lymphatics were several times larger in diameter than blood capillaries (Figure 3A).
Developing lymphatic vessels demonstrated 5′-Nase activities with different intensities in the intrinsic gastric wall. Interrupted weak or absent staining of endothelial cells was seen on newly formed lymphatic-like structures in the early stage. In contrast to lymphatic endothelium, no 5′-Nase reaction product was observed in the blood vascular endothelial cells. Sometimes the developing lymphatic vessels had a spacious cavity (Figure 5A) filled with lymphatic fluid and lymphocytes. The lymphatic wall became slender and irregular as the endothelial cells fully matured. The endothelium protruded into the lumen and adjacent connective tissue, and unique junctions (end-to-end, overlapping, and interdigitating) appeared between the endothelial cells. A reaction product for anti-VEGFR-3 was found on the luminal membrane of endothelial cells and microprocesses, and occasionally on the surface of organelles and intraluminal lymphocytes (Figure 6).
Lymphatic Networks in the Adult Stomach
Intramural lymphatics with strong 5′-Nase activity represented rich networks, abundant initial blind ends, and valve-like structures in the whole-mount preparations and tissue sections. The lymphatic vessels were found in all layers of the gastric wall (Figure 4). In the lamina propria mucosae, 5′-Nase-positive lymphatics (20–40 μm) with fewer valve-like structures revealed a fine network and branches like antlers, surrounding the basal portion of gastric glands (Figure 4A). The lymphatics of the submucosa possessed typical blind ends and valvelike structures (Figure 4B), varying greatly from 40 to 120 μm in caliber. Lymphatics in the lamina muscularis mucosae formed a wide communicating channel between the mucosa and submucosa. Fine lymphatics in the mucosa drained into lymphatic vessels in the submucosa by communicating branches (Figure 4C). Lymphatics in the muscular layer were seen to have a very irregular distribution, paralleling or crossing the muscle bundles (Figure 4D). Large collecting lymphatics in the submucosa ran through the muscle bundles into the serosa, where a well-developed network formed an effective extra-organic lymph drainage route (Figure 3C).
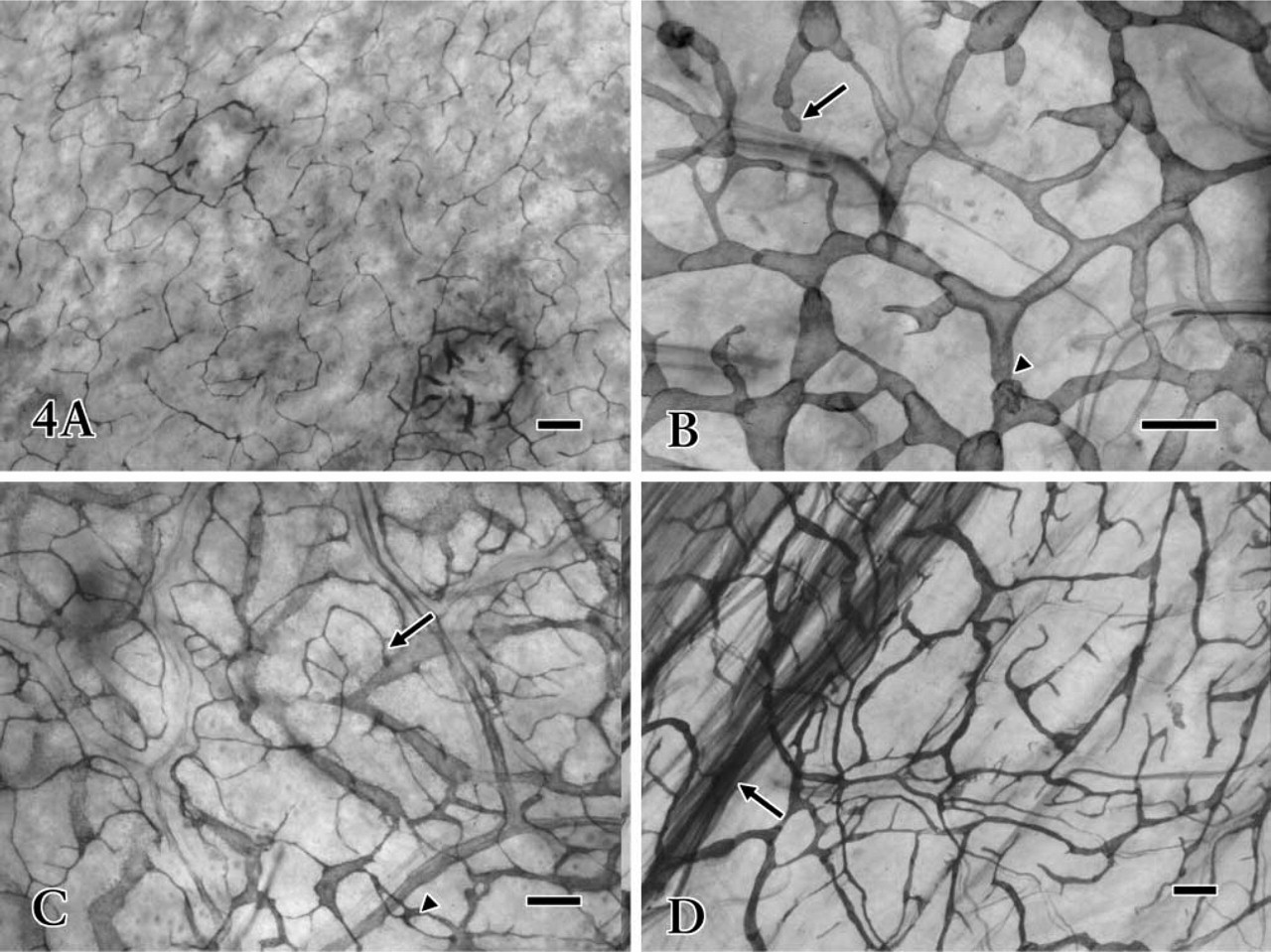
Whole-mount preparations treated with 5′-Nase staining. The 5′-Nase-positive lymphatic networks are widely distributed in various layers of the stomach. (
Transmission (
The staining pattern of lymphatic vessels for laminin, type IV collagen, a-SMA, and eNOS was different from that of blood vessels in the tissue sections. Immunoreactivity for these antibodies was generally stronger in the blood vessels than in the lymphatics (Figures 2E-2H). Endothelial cells of the initial lymphatics, in contrast to those of the blood capillaries, exhibited a relatively weak reaction, if any detectable activity for these antibodies did exist. The vessels with weak laminin, type IV collagen, and eNOS immuno-reaction revealed obvious 5′-Nase activity, whereas the vessels with significant immunoreactivity did not show any positivity with 5′-Nase. a-SMA immuno-staining showed uneven expression in the collecting lymphatic walls (Figure 2G).
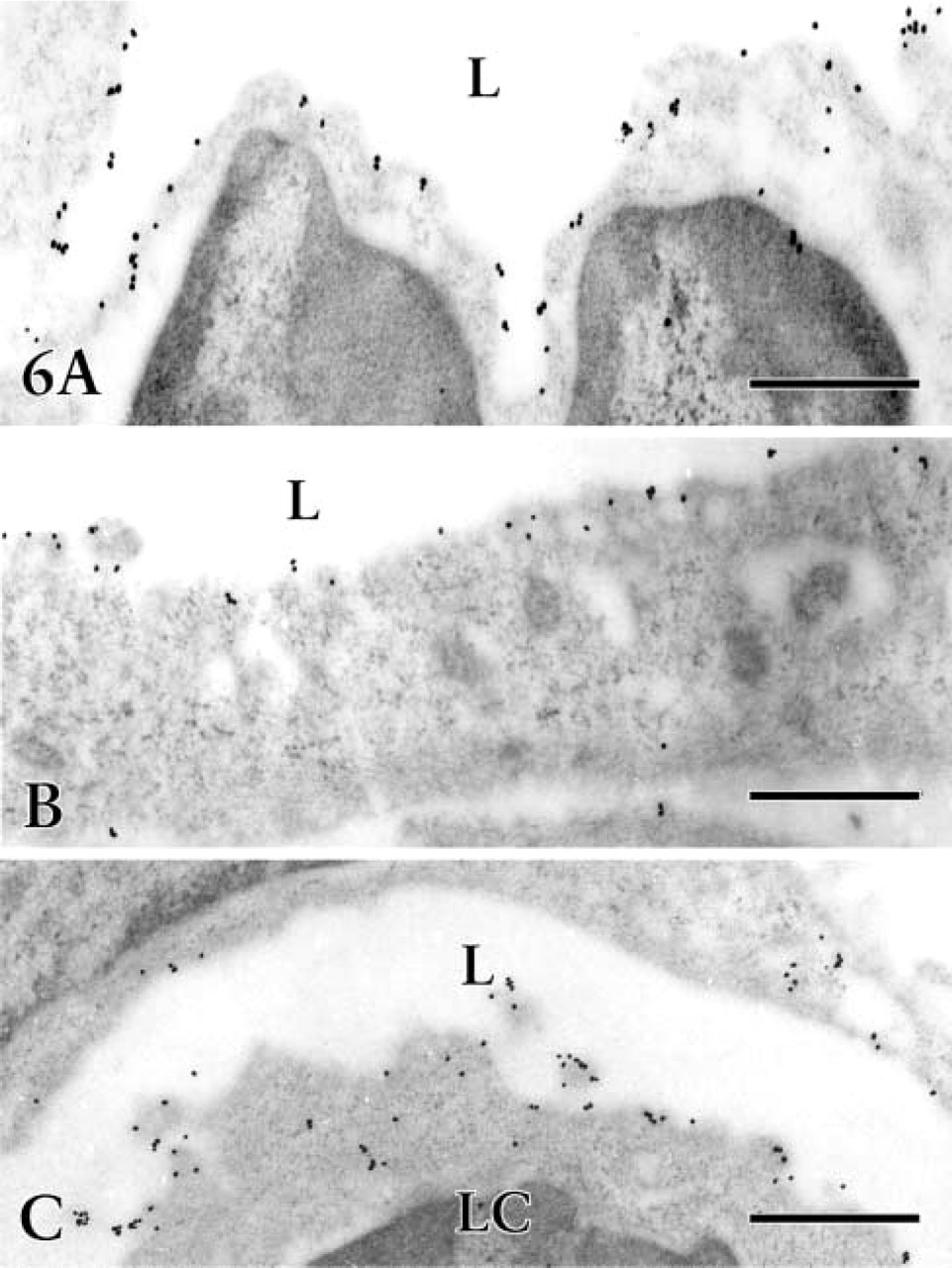
VEGFR-3 immunoelectron micrographs of fetal stomach. Immunogold labeling with silver enhancement by a postembedding technique reveals the cytochemical localization of VEGFR-3 on the luminal membrane of lymphatic endothelial cells and on the surface of some organelles and intraluminal lymphocytes (LC). L, lumen of lymphatic vessels. Bars = 0.5 μm.
In the TEM, lymphatic endothelium identified by specific deposition of 5′-Nase-cerium reaction particulates constantly showed overlapping and interdigitating junctions (Figure 5B). In the SEM-BEI, the solitary lymphatic nodule in the submucosa was surrounded by many lymphatic vessels, visible as a strong highlight in both the lymphatics and the nodule (Figure 5C). Corrosion casts further revealed the morphological characteristics of both lymphatic and blood vessels and their three-dimensional relationship (Figure 5D).
Discussion
Methodological problems have greatly hampered the study of lymphatics, especially the lack of suitable markers for differentiating lymphatic from blood vascular endothelial cells. The present study demonstrated that combined 5′-Nase enzyme and VEGFR-3 immunohistochemical approaches are the most reliable means to illustrate functional and morphological features of the lymphatic networks and lymphangiogenesis in wholemount preparations and in tissue sections of the stomach. For 5′-Nase, a membrane-bound protein, enzyme histochemical staining has been widely used to investigate organ specificity and structural organization of the lymphatic capillary networks in postnatal and adult mammals (Ji 1998; Kato 2000). In early embryonic tissue, endothelial cells of newly formed lymphatic-like structures usually show extremely low 5′-Nase activity. Lymphatic vessels in the rat stomach reveal increased 5′-Nase activity as the animals grow (Ji and Kato 1997). 5′-Nase staining appears to be impractical for distinguishing developing lymphatic and blood vasculature at the light microscopic level during early life, although 5′-Nase-cerium electron microscopic observation can localize fine and unevenly distributed reaction precipitates on the cell membrane of initial lymphatics. The mechanism of lymphangiogenesis was almost unknown until VEGF-C, the high-affinity ligand for VEGFR-3, was ascertained to be a specific lymphangiogenic growth factor in embryonic and differentiated tissues. VEGFR-3, as a most favorable molecular marker of endothelial cells, makes it possible to identify developing and regenerating lymphatic vasculature by localizing the antigen, and has set off a chain reaction of lymphatic studies in recent years. Developing endothelial cells of lymphatic and blood vasculatures react with VEGFR-3 during the early embryonic stage. For this reason, the staining results for cell proliferation should be carefully analyzed. With regard to adhesion, differentiation, and migration of endothelial cells, some elements, including 5′-Nase and eNOS, the extracellular matrix of basement membrane, and components of surrounding connective tissues, are very important factors in the formation of developing vasculature (Grant et al. 1981; Kleinman et al. 1985; Kubota et al. 1988; Ji and Kato 2001). More recently, several other proposed markers for lymphatic endothelial cells, e.g., podoplanin, LYVE-1, and Prox-1, have emerged, but there still remain questions about their reliability and specificity, and they need further study (Breiteneder-Geleff et al. 1999; Wigle and Oliver 1999; Prevo et al. 2001; Sleeman et al. 2001). However, co-expression of these new markers with so-called routine differentiating reagents, such as laminin, collagen type IV, and α-smooth muscle actin, is definitely helpful in analyzing the functional-structural properties of endothelial cells of both lymphatic and blood vessels.
The present findings reveal that rich lymphatic networks are located in various layers of the intrinsic gastric wall, undergoing obvious morphological changes from the fetal to the adult stage. Fewer blind ends and branches were found in developing than in mature lymphatic networks in whole-mount preparations and lymphatic corrosion casts. The most remarkable finding was that many circular and incomplete lymphatic-like structures expressing VEGFR-3 show an obvious accumulation, indicating that lymphangiogenesis occurs sequentially in definite regions in the early embryonic stage. The active proliferating feature of clustered developing lymphatics may create a favorable environment for their sprouting and growth, which serves as a functional requirement for lymph drainage. These initial developing lymphatics are characterized by an extremely permeable thin endothelial lining devoid of a basal lamina. The results also showed that VEGFR-3 is co-expressed in closely distributed lymphatic and blood vasculature, suggesting that lymphatic endothelial cells have similar molecular physiological features to blood vascular endothelium, and probably originate from the sprouting of small venous structures. This opinion is not contrary to the observation that lymphangiogenesis originates from lymphatic vessels (Witmer et al. 2001). Interestingly, VEGF-C expression was found in glandular cells, and in endothelial cells of blood vessels rather than lymphatics in fetal tissue (data not shown). Meanwhile, a reaction product for anti-VEGFR-3 was found on the luminal membrane of lymphatic endothelial cells and on the surface of intraluminal lymphocytes. These findings indicate that VEGF-C acts on VEGFR-3-expressing endothelial cells in a paracrine and/or endocrine fashion, i.e., the endothelial cell is stimulated by the growth factor delivered through the neighboring tissue matrix or the circulatory system. VEGF-C works as a differentiation factor for lymphatic endothelial cells, selecting the population of endothelial cells that differentiates and forms the lymphatic vessels, which then maintain VEGFR-3 expression (Taipale et al. 1999). Local circulatory lymphocytes with VEGFR-3 expression might participate in the progressive growth of endothelial cells, although this should be confirmed by further experimental evidence.
Based on the endothelial expression profile and its binding to VEGFR-3, VEGF-C is implicated in the stimulation of lymphangiogenesis, and to some extent angiogenesis, in fetal tissue. In the postnatal stage, two independent lymphatic networks were encountered in the gastric wall, the submucosa and serosa, from which branches are given off extending into the deep lamina propria and muscularis externa. The presence of lymphatic networks in the deep lamina propria implies that lymphatic development in the gastric wall is being perfected (Ji and Kato 1997). In this respect, it seems extremely important to decide whether these delicate lymphatics are existing lymphatics that have enlarged in response to VEGF-C or are newly formed initial lymphatics. With lymphatic modification, an obvious change of interendothelial junctions appears, going from simple end-to-end to overlapping and interdigitating. The pinocytotic vesicles and open junctions, two major passages for fluids and particles across the endothelium, greatly increase in the lymphatic vessels of the mucosa and submucosa (Ji and Kato 1997). Immunohistochemically, the present findings are in close agreement with the viewpoint that VEGFR-3 is widely expressed in early embryonic vasculature but becomes restricted to the lymphatic endothelium at later developmental stages and in postnatal life (Wilting et al. 1999). However, the study of VEGFR-3 expression on regenerating lymphatic capillaries, which occur in tumors and wound healing tissue, should not be neglected. The differential analysis in the structure and VEGFR-3 molecular identification between developing and regenerating lymphatics will become most invigorating and productive fields in the near future.
