Abstract
The basic helix-loop-helix (bHLH-PAS) proteins aryl hydrocarbon receptor nuclear translocator (Arnt) and Arnt2 are transcriptional regulators that function as dimerizing partners for several bHLH-PAS proteins and also some nonrelated partners. They are involved in various biological functions, including regulation of developmental genes. In earlier studies, the developmental expression of Arnt was reported to be almost ubiquitous, whereas Arnt2 expression has been shown to be more limited, comprising neuronal tissues as the main site of expression. Here we provide a detailed description of the expression of Arnt and Arnt2 mRNA in mouse tissues during embryonic and early postnatal development. Arnt and also Arnt2 transcripts, in contrast to earlier reports, are shown to be expressed more widely during development yet show a temporally and spatially specific pattern.
Keywords
A
Arnt and Arnt2 as bHLH-PAS proteins share a conserved sequence structure. The dimerization interface between Arnt and Arnt2 and other bHLH-PAS proteins is provided by PAS and HLH motifs (Reisz-Porszasz et al. 1994; Lindebro et al. 1995). The PAS domain also offers the interaction surface with non-PAS proteins (Sadek et al. 2000). The basic motif is required for sequence-dependent recognition of DNA (Reisz-Porszasz et al. 1994).
Arnt has a central role as a common heterodimerization partner in this family. Interaction with Arnt is a necessity for the ligand-activated aryl hydrocarbon receptor (AhR) to be able to bind to the regulatory DNA sequence XRE (xenobiotic response element) and to mediate biological responses to carcinogenic and toxic environmental chemicals (Denison et al. 1988; Pohjanvirta and Tuomisto 1994). In response to hypoxia, Arnt forms a heterodimer with another bHLH-PAS transcription factor, Hif-1α, and they bind together to HRE (hypoxia response element) of genes responsible for the adaptation to oxygen deprivation (reviewed by Wenger and Gassmann 1997; Semenza 1998). Interestingly, crosstalk between hypoxia and dioxin signal transduction pathways has been shown to occur and Arnt may have a role as the common cell factor (Chan et al. 1999).
Drosophila single-minded (SIM), a bHLH-PAS protein regulating central nervous system midline cell development (Nambu et al. 1991), interacts with Tango, the
Arnt mRNA expression is extensive during murine embryonic development (Abbott and Probst 1995; Jain et al. 1998). Further support of its importance to development is provided by studies of Arnt-deficient transgenic mice (Kozak et al. 1997; Maltepe et al. 1997). The embryos died by E10.5 and had placental and vascular defects, and malformations in the central nervous system.
Arnt2 is a close structural homologue of Arnt (63% identical) and can bind to DNA as a heterodimer with AhR, Hif-1α, and Sim and can also homodimerize in vitro (Ema et al. 1996; Hirose et al. 1996). In vivo, Arnt2 is co-expressed and presumably interacts with Sim1 in controlling the development of neuroendocrine cell lineages in hypothalamic nuclei (Michaud et al. 2000). The developmental expression in previous studies (Hirose et al. 1996; Jain et al. 1998) was stated to be limited to the neuroepithelium, neural crest derivatives such as dorsal root ganglia and the adrenal medulla, to the kidney, and to the inner layer of the retina in mouse embryos.
Homozygous Arnt2 gene knockout mouse embryos die perinatally and exhibit impaired hypothalamic development, similar to those of bHLH-PAS gene Sim1 knockout mice, concordant with the hypothesis that Sim1 and Arnt2 interact in vivo (Michaud et al. 2000; Hosoya et al. 2001; Keith et al. 2001). Arnt2 and Arnt have a strong genetic interaction shown by crossed compound heterozygous mutant Arnt+/-, Arnt2+/- mice embryos (Keith et al. 2001). The embryos with fewer than two wild-type alleles of either Arnt or Arnt2 were under-represented, suggesting overlapping functions for Arnt and Arnt2 during early embryonic development.
Although the number of known related and nonrelated proteins capable of heterodimerization with Arnt and Arnt2 is increasing, we report a detailed distribution of Arnt and Arnt2 mRNA in murine embryonic and postnatal tissues to understand better the in vivo interaction possibilities.
Materials and Methods
In this study we used embryonic (E9-E17) and 36-hr-old (P1.5) NMRI mice (E0 = day of conception). Embryos and tissues were frozen on a block of dry ice and cut with a Microm HM-500 cryostat to serial 14-μm-thick sections, thawed onto polysine (Menzel-Gläser; Braunschweig, Germany) glasses, and stored at −20C until used. At least three embryos for each time point studied were sectioned throughout and adjacent sections from five to ten different levels were used in hybridizations.
In situ hybridization (ISH) was carried out as previously described by Kononen and Pelto-Huikko (1997). The oligonucleotide probes complementary to the mRNAs encoding mouse Arnt [nucleotides 295–339, ctttcctaagagctcctgtggctggtagccaacagtagccacaca and 1078–1118, gcgtaagatggttagcttgtctggttttcgagccagggcactacagg (Li et al. 1994; GenBank accession number NM_0099709)] and mouse Arnt2 [nucleotides 120–166, cgctttcctcctcgggcaggcatggcccctgccattctcacctgtcc, 1839–1885, ggtgaggaggcagctgggctggagagaggactgtaggatgaagggt, and 1977–2023, ccgctctgctgtccgtgatgctggctctgccactgggaccagacctc (Hirose et al. 1996; GenBank accession number NM_007488.1)] were labeled with [α-33P]-dATP (Du Pont-NEN; Boston, MA) to a specific activity of 109 cpm/μg using terminal deoxynucleotidyltransferase (Amersham; Poole, UK). The sections were briefly air-dried at room temperature (RT) before hybridization. Hybridization was done in a humified chamber at 42C for 18 hr with 5 ng/ml of the probe in a mixture containing 4 X SSC (1 X SSC = 0.15 M NaCl, 0.015 M sodium citrate), 50% formamide, 1 X Denhardt's solution (0.02% polyvinyl-pyrroline, 0.02% bovine serum albumin, and 0.02% Ficoll), 1% sarcosyl, 0.02 M phosphate buffer (pH 7.0), and 10% dextran sulfate. After hybridization the sections were washed four times at 55C in 1 X SSC for 15 min each and then left to cool for 1 hr at RT. The sections were rinsed in distilled water, dehydrated with 60% and 90% ethanol, and air-dried. Thereafter the sections were covered with Kodak MR autoradiography film (Kodak; Rochester, NY) and exposed at −20C for 50–60 days. The autoradiography films were developed using LX24 developer and AL4 fixative. Alternatively, the sections were dipped in NTB2 emulsion (Kodak), diluted 1:1 with distilled water, and exposed at 4C. After 60 days of exposure the sections were developed with D19 developer (Kodak), and fixed with G333 fixative (Agfa Gevaert; Leverkusen, Germany). The sections were counter-stained with hematoxylin-eosin and examined with a Nikon FXA microscope equipped with epipolarization and brightfield optics.
The probe sequences exhibited less than 60% similarity to all known sequences in the GenBank database. All probes gave similar expression results when used separately and were usually combined to intensify the hybridization signal. As a control for specificity of ISH, several probes for nonrelated mRNAs with known expression patterns and with similar length and GC content were used as controls. Results with probes to sperm-specific thioredoxin (Miranda-Vizuete et al. 2001a,b), which is exclusively expressed in adult testis, are illustrated in Figure 5. The addition of a 100-fold excess of respective unlabeled probe abolished all ISH signals.
Results
Arnt mRNA Expression
ISH analysis showed the temporally and spatially specific expression of Arnt. Arnt was very widely expressed during mouse embryonic and early postnatal ontogeny (Figure 1; Tables 1 and 2). The expression of Arnt could be detected in a variety of cell types of endodermal, ectodermal, and mesodermal origin.
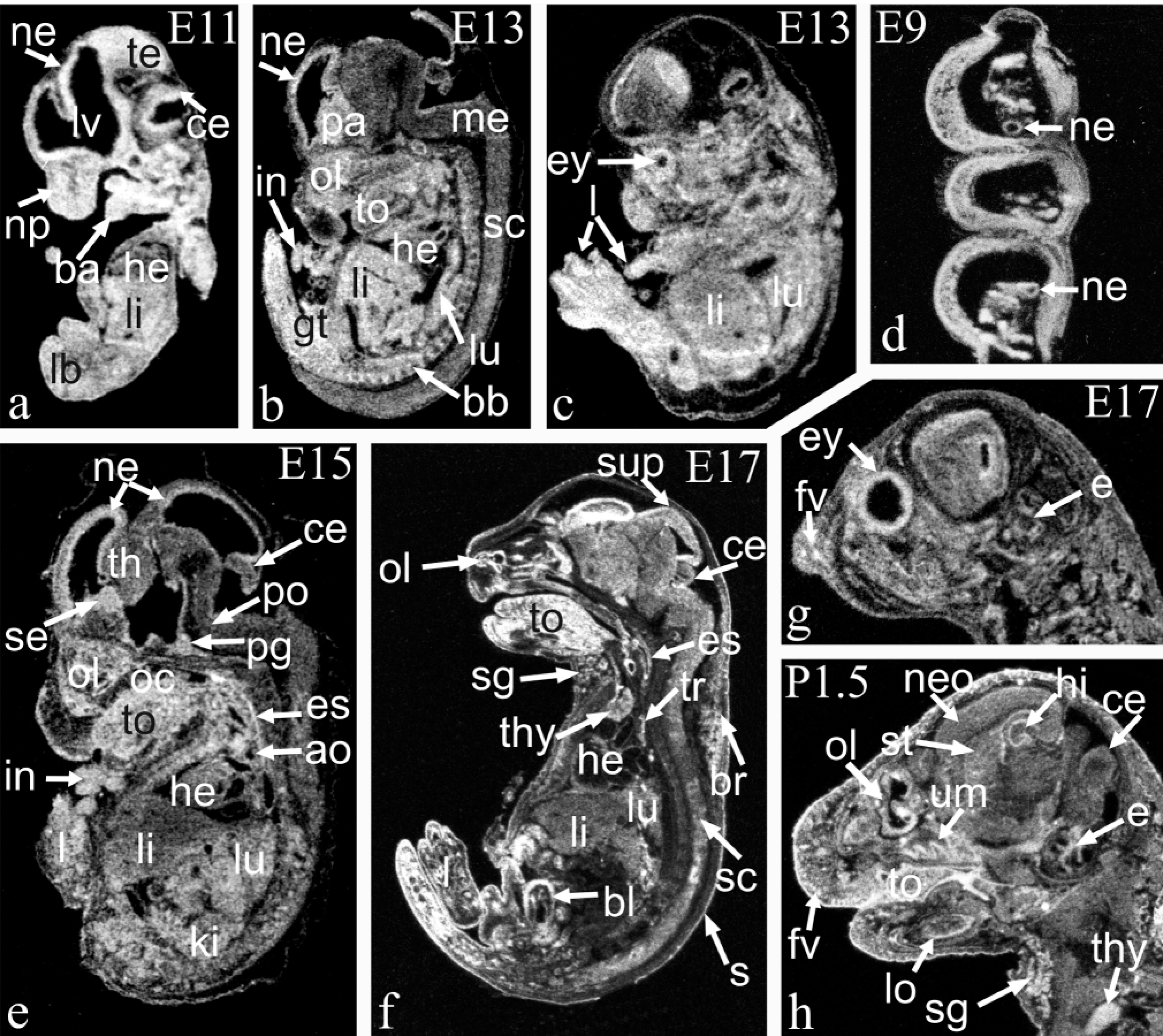
ISH analysis of Arnt mRNA in mouse embryos is shown at E11 (
Expression of Arnt mRNA in murine central nervous system a
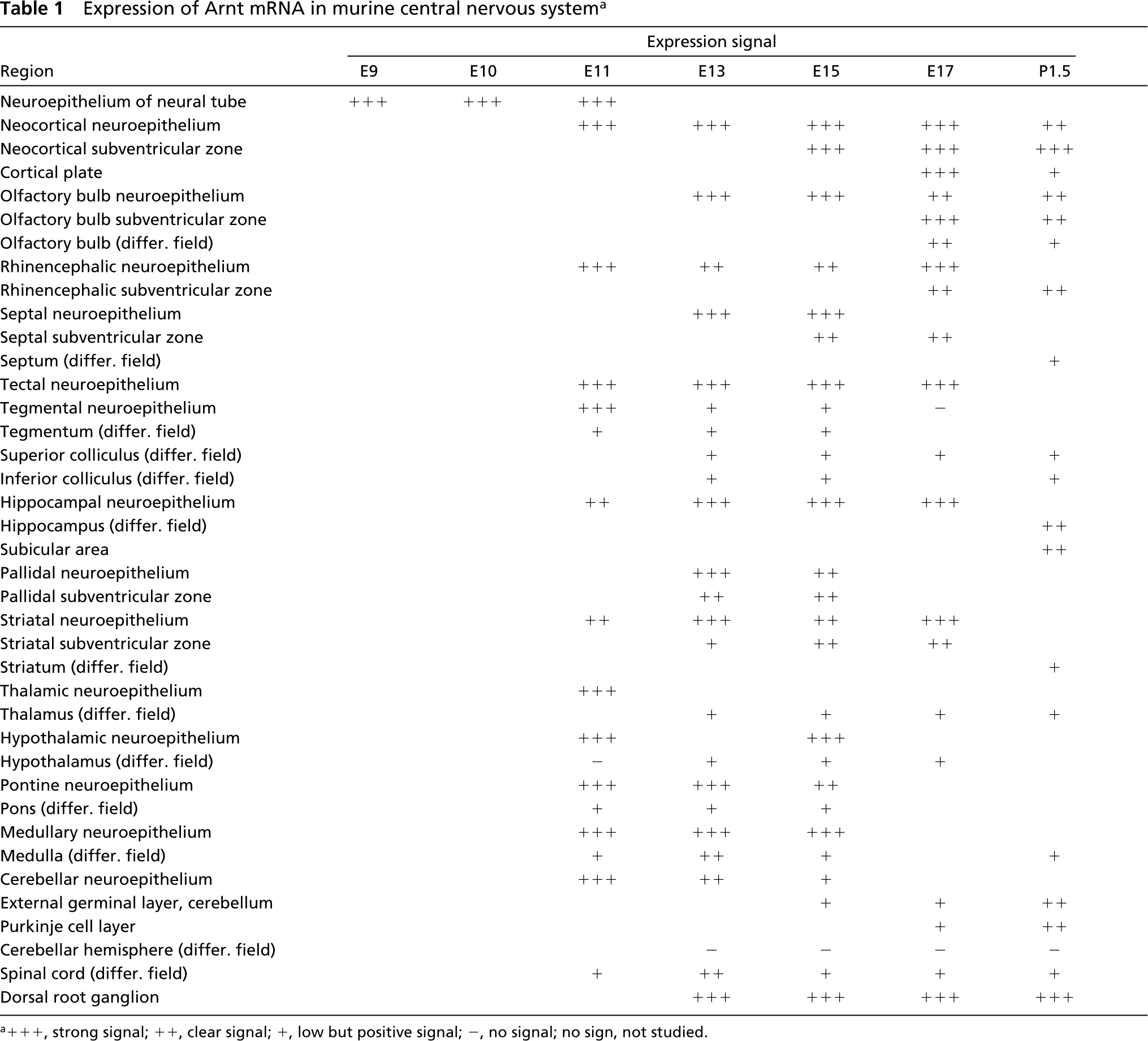
a+++, strong signal; ++, clear signal; +, low but positive signal; -, no signal; no sign, not studied.
Expression of Arnt mRNA in murine peripheral organs a

a+++, strong signal; ++, clear signal; +, low but positive signal; -, no signal; no sign, not studied.
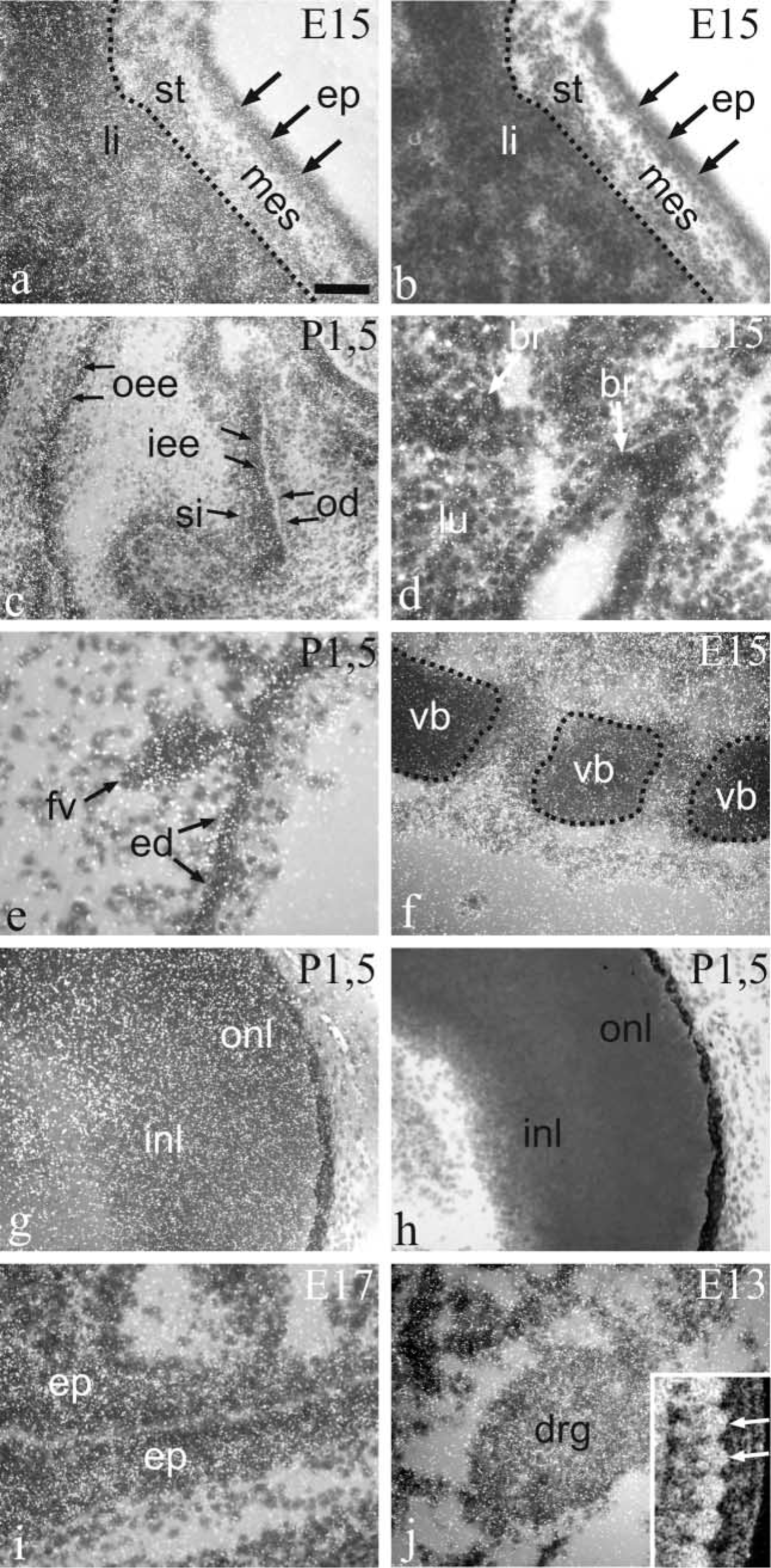
Emulsion autoradiograms of stomach and liver at E15 (
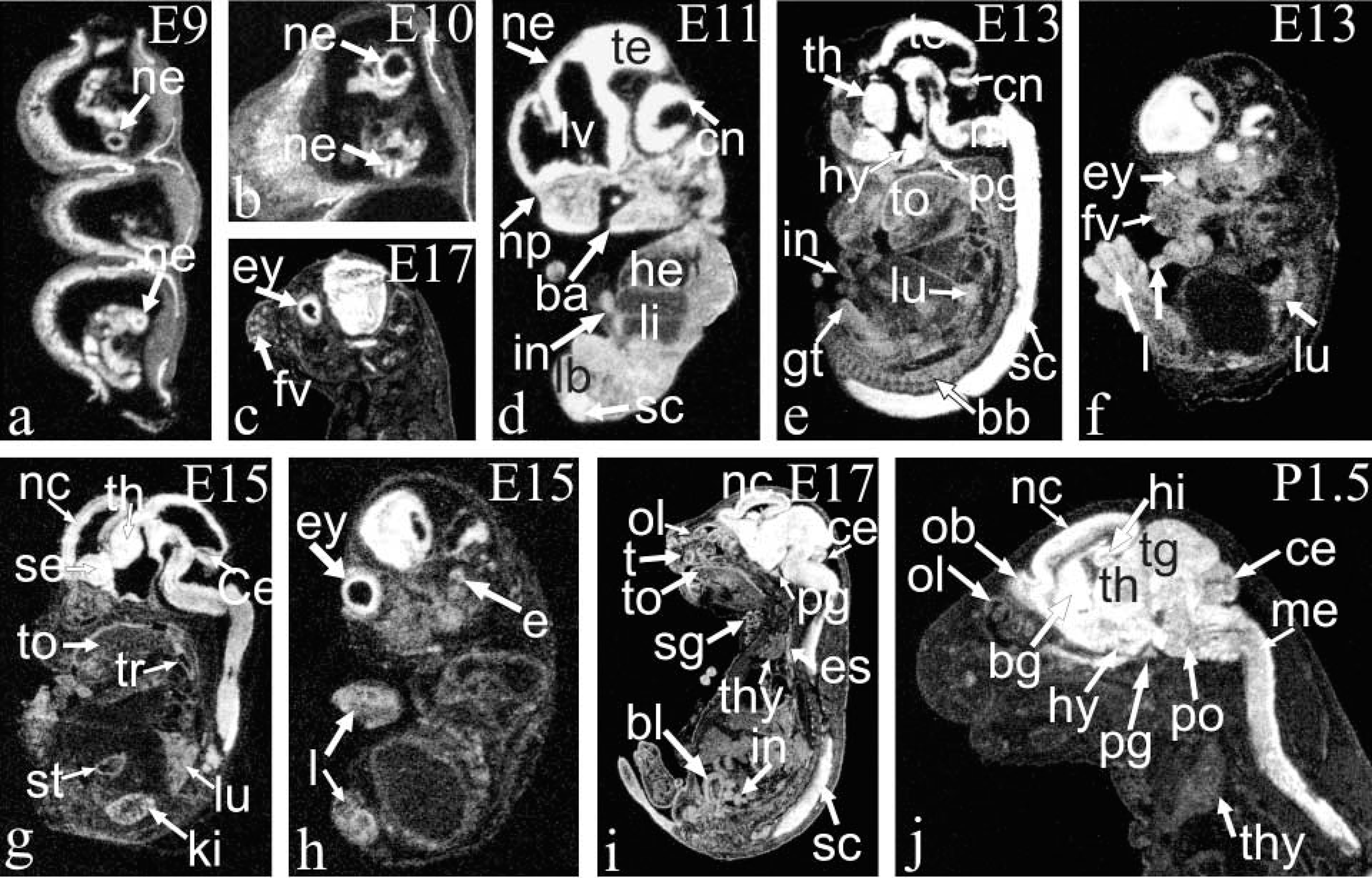
ISH analysis of Arnt2 mRNA in mouse embryos is shown at E9 (
Arnt2 mRNA Expression
The expression of Arnt2 during murine development differs clearly from that of Arnt, although they share several sites of expression (Figure 3; Tables 3 and 4). Arnt2 mRNA is very strongly expressed in the developing nervous system on all the embryonic and postnatal days studied. Outside the nervous system the expression of Arnt2 mRNA is more limited than that of Arnt, and expression levels in the peripheral organs are generally lower than in the nervous system.
Expression of Arnt2 mRNA in murine central nervous system a
a++++, very strong signal; +++, strong signal; ++, clear signal; +, low but positive signal; -, no signal; no sign, not studied.
Expression was already very strong in the neuroepithelium of neural tube at E9 (Figures 3a, 3b, and 3d). High expression was observed in the cerebral cortex on all the days studied. At E17, separate layers of the cortical area exhibited different amounts of Arnt2 transcripts. The signal was strong in the neocortical neuroepithelium and the subventricular zone, weak in the differentiating field, and very strong in the cortical plate in the infra- and supragranular parts (Figures 3c, 3j, 4a, and 4d). By P1.5, expression in the neuroepithelium and subventricular zone had weakened, whereas high expression in the cortical plate still remained (Figure 3j). No signal was seen in the molecular layer of the neocortex. The olfactory bulb, rhinencephalon, septum, and preoptic area all displayed high expression of Arnt2 mRNA. Very strong expression was found in the midbrain in the tectal and tegmental neuroepithelium, the tegmental differentiating field, and the differentiating fields of the superior and inferior colliculus (Figures 3d, 3e, and 3j). In the developing hippocampus at E11, expression was very strong, but at E13 and E15 expression clearly weakened until it became very strong again at E17 and postnatally both in the differentiating areas of the dorsal and ventral hippocampus, the subicular area, and the dentate gyrus (Figure 3j). In the basal ganglia, the pallidum, and striatum, high expression was found (Figure 3j). The thalamus and hypothalamus expressed Arnt2 mRNA very strongly during the embryonic period and strongly postnatally (Figures 3e, 3g, and 3j). In the pontine and medullary areas expression was high, except in the differentiating fields, where it diminished to a moderate level postnatally (Figure 3j). In the cerebellar neuroepithelium, where the Purkinje cells originate, expression was very strong or strong from E11 to E15 but had ceased by E17, as did the mitotic activity in this area (Figures 3d, 3e, 3g, and 3h). In the Purkinje cell layer, expression was moderate at E17 and P1.5 (Figures 3g and 3j). In the external germinal layer, where the birth and migration of the granule neurons occur later than in many other brain areas, a moderate expression of Arnt2 mRNA appeared at E17 and continued postnatally. In the differentiating areas of the cerebellum, weak expression was detectable only at E17. High expression was observed in the spinal cord (Figure 4c). Strong Arnt2 mRNA expression was discovered in the dorsal root ganglia from E13 to P1.5 and in the trigeminal ganglion from E13 to E17 (Figure 4n). The postnatal trigeminal ganglion was not studied.
Expression of Arnt2 mRNA in murine peripheral organs a
a+++, strong signal; ++, clear signal; +, low but positive signal; -, no signal; no sign, not studied.
Discussion
In this study we report the expression of Arnt and Arnt2 mRNAs by ISH in the CNS and peripheral tissues during mouse fetal and early postnatal development. There are earlier reports of the expression of both Arnt and Arnt2 (Abbott and Probst 1995; Hirose et al. 1996; Jain et al. 1998) mRNAs in mouse embryos. However, in our work more detailed distribution is reported, especially concerning expression in the developing CNS. In addition, we found the expression of both Arnt and Arnt2 mRNA in several tissues that have not been reported in previous studies, and we extended the analysis to the early postnatal period.
Arnt mRNA was widely expressed at all stages studied from E9 to postnatal day 1.5, in agreement with previous studies. However, the expression was not ubiquitous and it showed both temporal and spatial specificity. In some organs (certain brain areas, heart muscle, thymus, skin, retina, olfactory epithelium, adrenal gland, and dorsal root ganglion), expression was stable on all the days studied, whereas in some others the level of expression changed from strong to low or undetectable (tegmental neuroepithelium, liver, lung, bones, and muscles). In the same way as with Arnt, the expression of Arnt2 mRNA was temporally and spatially specific and could be detected at all the times investigated. Compared to the earlier studies that have reported Arnt2 gene transcripts being limited to the neuroepithelium, dorsal root ganglia, adrenal medulla, kidney, and the inner layer of the retina (Hirose et al. 1996; Jain et al. 1998), we found a much wider expression pattern. However, the organs reported by these earlier studies showed the most intense expression in our research also.
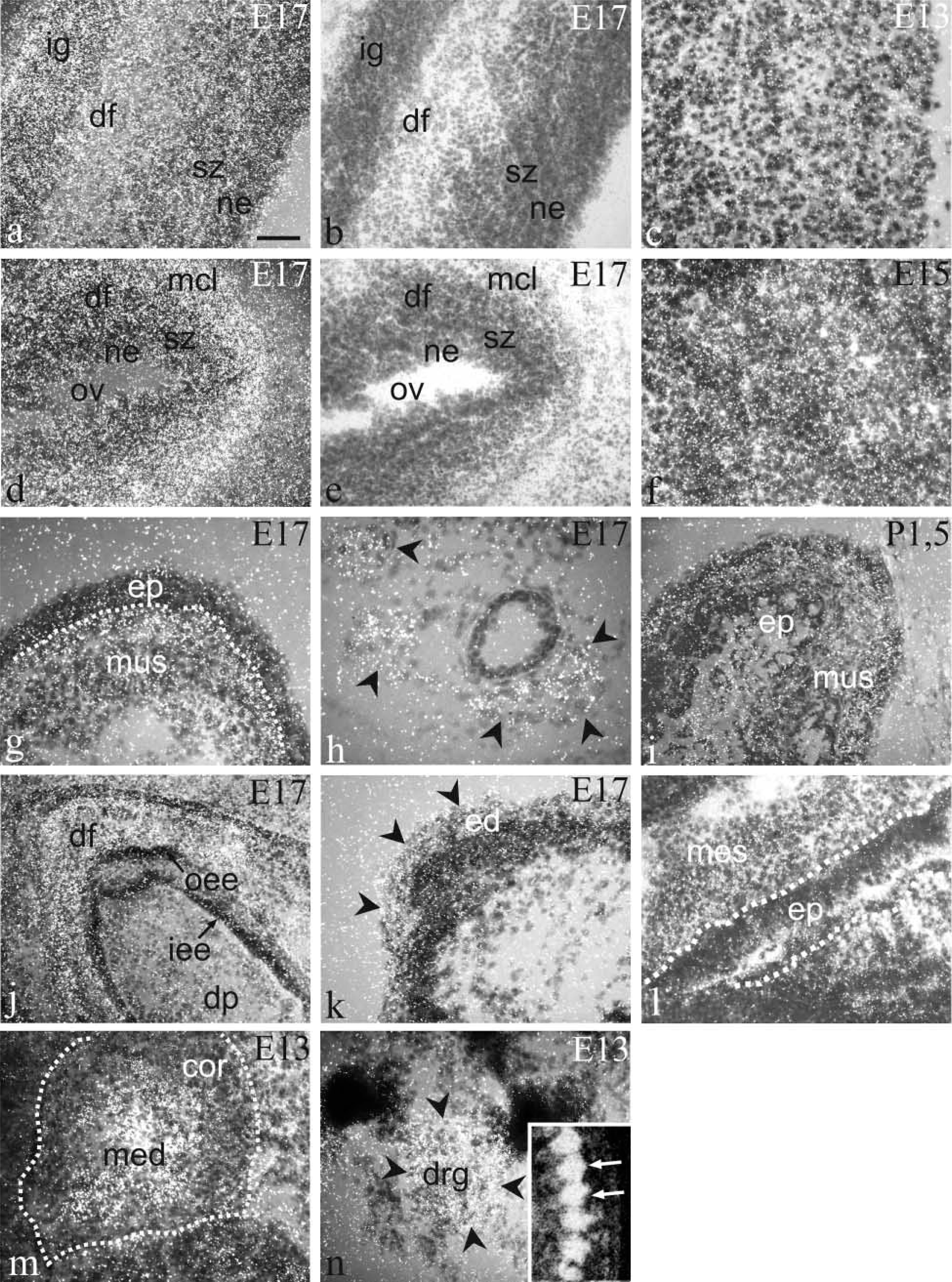
Emulsion autoradiograms showing the distribution of Arnt2 mRNA in E17 cerebral cortex (
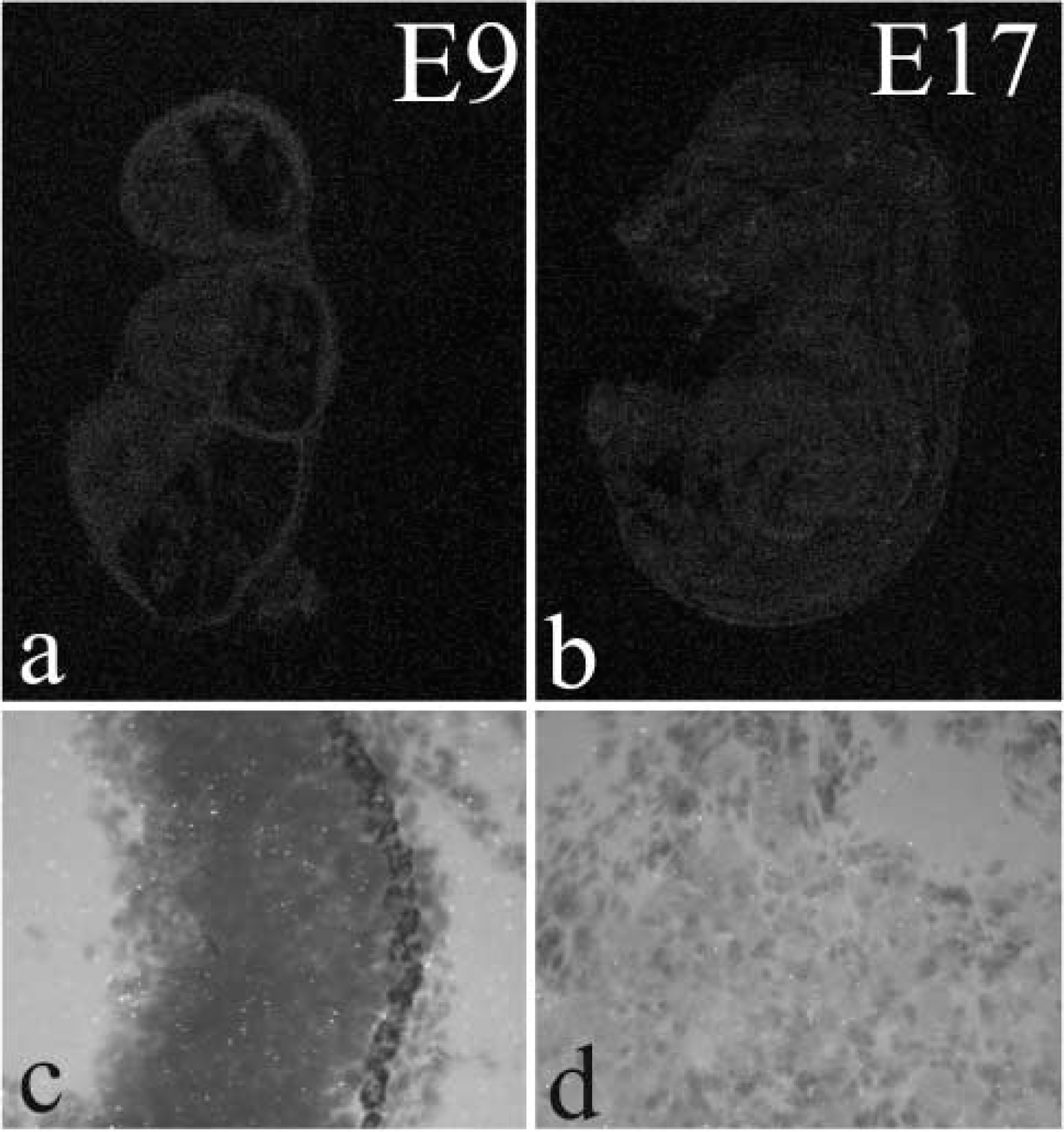
Control hybridizations with probes to sperm-specific thioredoxin. No signal can be seen in E9 (
On a gross scale, the expression of Arnt mRNA in the CNS was weaker (although the intensity reached a strong level in several areas) but almost as extensive as that of Arnt2. The expression of both Arnt and Arnt2 could be seen in the same brain areas, often at the same time, but Arnt mRNA expression showed a tendency to decline as the development of the neurons progressed toward their differentiated state, whereas the levels of Arnt2 stayed mainly at a high level throughout the fetal period and also postnatally. No expression of either Arnt or Arnt2 mRNA was seen in the brain areas containing nerve fibers and glial cells, such as the corpus callosum. This was in agreement with the previous investigations (Drutel et al. 1999) showing that Arnt2 is expressed only in neuronal cells. The prominent expression of Arnt2 transcripts suggests that Arnt2 may be the most prominent bHLH-PAS partner in the developing CNS, whereas Arnt has this role in the peripheral tissues. On the other hand, the severe malformations of the CNS (neural tube closure defect and forebrain hypoplasia) of Arnt−/- knockout mice embryos (Kozak et al. 1997) underline the importance of Arnt for proper brain development. Because Arnt and Arnt2 share several interaction partners, these two proteins may have either a competitive or an additive role in formation of functional heterodimers that regulate the transcription of developmental genes in the brain. They might also mediate distinctive signals in CNS development by interacting with as yet unknown nonrelated heterodimerizing partners.
The shared in vitro interaction partners of Arnt and Arnt2 also expressed in developing mouse CNS include HIF1-α (Jain et al. 1998), AhR (Abbott et al. 1995), and Sim1 and Sim2 (Ema et al. 1996). There is strong evidence of the importance of HIF1-α for brain development, which has been provided by Hif1-α knockout mice studies (Iyer et al. 1998; Kotch et al. 1999) because the unviable Hif1-α−/- embryos manifested neural tube defects. AhR has been detected in the mouse neuroepithelium at E10–13, but levels in the brain decreased as development progressed (Abbott et al. 1995). Presumably, the role of AhR in the CNS is not vital, because the targeted disruption of AhR in mice has revealed that the critical developmental problems are involved with the liver and the immune system, not with the brain area (Fernandez-Salguero et al. 1995; Schmidt et al. 1996).
SIM is known as a master regulator of the
Outside the CNS we were able to show many novel sites of expression for Arnt2 mRNA. For example, Arnt2 has not been previously found in the digestive system. In addition, we show expression in the branchial arch, tongue, salivary gland, stomach, intestines, and teeth. Both Arnt and Arnt2 transcripts were observed in these developing organs and therefore Arnt and Arnt2 may serve as dimerization partners for the same proteins. However, in the teeth they were expressed in different sites, suggesting that they function in dimer formation in separate areas and do not compete in the developing teeth. No Arnt2 transcripts were seen in the liver, where AhR signaling pathways are crucial for proper development, leaving Arnt as a candidate for interaction partner for AhR. No expression of Arnt2 mRNA was seen in the palate either, whereas it has been suggested that AhR and Arnt are important for normal palatogenesis (Abbott et al. 1999).
Our report confirms the expression of Arnt2 transcripts in the lungs, as this remained unclear in the study of Jain and the co-workers (1998) where they reported low or undetectable expression in the lungs. We also report the nasal process and the trachea as novel sites of Arnt2 mRNA expression and these were sites of Arnt expression as well. In addition to the kidney, which has been reported earlier (Jain et al. 1998), we found also the genital tubercle and later the urinary bladder and the urethra as regions exhibiting Arnt2 transcripts. In the heart no Arnt2 mRNA was seen, and also Arnt mRNA expression was weak from E11 to P1.5 in contrast to the results of Abbott and Probst (1995) showing a significant expression in the heart at E11 – although they saw a decline after that.
It has been suggested that Arnt2 is the candidate for the thymus defects of c112K deletion mice (Wines et al. 1998) and, in agreement with that, we show a signal in the developing thymus at E17 and P1.5 suggesting that Arnt2 is important for proper thymus development. Interestingly, expression of Arnt mRNA was even stronger than that of Arnt2 in the thymus, implying that Arnt2 has a distinct role in its development. We found that Arnt and Arnt2 mRNAs are also co-expressed in another lymphatic organ, the spleen, which is a new discovery for both gene transcripts. In the developing spleen, Arnt and Arnt2 may take part in AhR signaling pathways which appear to be important for normal immune system development. AhR−/- mice have shown decreased accumulation of lymphocytes in the spleen and lymph nodes (Fernandez-Salguero et al. 1995).
Further novel locations for both Arnt and Arnt2 transcripts are the skin, the hair follicles and the follicles of vibrissae. Arnt has previously been reported in the bones and muscles (Abbott and Probst 1995), and we showed a somewhat weaker expression of Arnt2 in the same areas at the same time. Both Arnt and Arnt2 were downregulated in the bones and muscles before birth.
We found prominent expression of Arnt expression in the inner and outer nuclear (neuroblastic) layer of the retina and Arnt2 mRNA in the inner nuclear layer. These layers will later form the horizontal cells and the photoreceptor cells (outer nuclear layer) and the ganglion cells (inner nuclear layer). Taken together with the strong neuronal expression in the CNS, the retinal presentation of Arnt and Arnt2 transcripts is not surprising. The earlier results of expression in the eye differ from ours, as Arnt has been either weak or undetectable (Jain et al. 1998) or expressed in the lens (Abbott and Probst 1995). Arnt2 is known to co-express with HIF1α and AhR in the inner layer of the retina (Jain et al. 1998). It is interesting that, in the adult rat retina, a mammalian clock gene, Clock, and BMAL1 (ARNT3) are expressed in the nuclear and ganglion cell layers where the expression of Clock undergoes circadian oscillation while the expression of its partner, BMAL1, is stable (Namihira et al. 1999). However, Arnt and Arnt2 in the developing retina presumably control the neuronal differentiation, thus creating the basis for the light-induced circadian rhythm.
We found expression of both Arnt and Arnt2 mRNA in the olfactory epithelium and in the cochlear epithelial cells and surrounding mesenchyme of the inner ear, which was a new observation. The co-expression of Arnt and Arnt2 has been demonstrated in adrenal gland, which is derived from the neural crest (medulla) and mesoderm (cortex) (Abbott and Probst 1995; Jain et al. 1998). The two transcripts are, however, located in separate sites in our study, because the signal of Arnt was seen in the adrenal cortex and Arnt2 in the medulla, which suggests a distinctive role for them during adrenal development. Expression of Arnt2 mRNA has been previously detected in the neural crest-derived dorsal root ganglia (Jain et al. 1998). In our work, a strong signal was seen not only with the Arnt2 probe but also with the Arnt probe, although the expression of Arnt2 mRNA was higher than that of Arnt.
In conclusion, we have described in detail the expression of Arnt and Arnt2 mRNA during embryonic and postnatal development of the mouse using ISH. We were able to confirm the earlier reported sites of strong expression and in addition could detect organs and tissues where Arnt and especially Arnt2 transcripts were revealed for the first time. Interestingly, we found that Arnt and Arnt2 are expressed in parallel in many tissues, where they may compete or substitute for common heterodimerizing partners, whereas in earlier reports the expression of Arnt2 has been thought to be very limited and Arnt almost ubiquitous. There are also tissues in which they are expressed separately referring to the discrete functions.
Footnotes
Acknowledgements
Supported by the Medical Research Fund of Tampere University Hospital, Research and Science Foundation of Farmos, and the Finnish Medical Foundation.
We are grateful to Ulla Jukarainen and Riika Salmela for skillful technical assistance.
