Abstract
Immunostaining in transgenic mice carrying the lac z gene can be used to map gene and protein distribution in a single tissue. In this study, we examined inner ears from ephrin-B3 homozygous and ephrin-B2 heterozygous mice. Ephrin-B3 lac z expression was limited in these mice. However, immunostaining revealed ephrin-B3 throughout cochlear and vestibular regions. Immunoreactivity was absent in ephrin-B3-homozygous null mutants, demonstrating the specificity of the antibody. Ephrin-B2 lac z reactivity was detected in a limited number of cells in cochlear and vestibular regions. Different immunostaining patterns were found with different antibodies. Comparison with lac z expression indicated which antibody was specific for the transmembrane-bound ephrin-B2 ligand.
I
A number of transgenic mice have been generated in which the lac z gene is substituted for the gene of interest. For example, mice lacking members of the Eph family have been used to examine Eph distribution and function. The Eph family is comprised of ephrin ligands and Eph receptors. The ephrins are membrane-associated ligands. Ephrin-A ligands are attached to the cell membrane by a glycosylphosphatidylinositol (GPI) linkage, whereas ephrin-B ligands are transmembranebound. These ligands bind to the Eph receptor tyrosine kinases. The EphA subclass of receptors binds primarily A ligands, whereas EphB receptors bind primarily B ligands (Gale et al. 1996). A number of Eph family members have been detected in the developing and adult inner ear (Bianchi and Gale 1998; Bianchi and Liu 1999; Cowan et al. 2000; Matsunaga et al. 2000; van Heuman et al. 2000), where they have been associated with ion transport, boundary formation, and axon guidance.
This study examined inner ear tissues of mice homozygous for an ephrin-B3 or heterozygous for an ephrin-B2 null mutation. Specifically, this study evaluated (a) gene expression patterns using lac z histochemical methods, (b) protein expression using IHC analysis, and (c) morphological changes in the inner ear using light microscopy.
Materials and Methods
The lac z gene was used to replace the ligand-coding region of the ephrin-B2 gene or the first coding exon of the ephrin-B3 gene (Gale et al. 2001; Kullander et al. 2001). Adult mice that were heterozygous for ephrin-B2 (n=5) or homozygous for ephrin-B3 (n=6) were sacrificed by inhalation of CO2, following protocols approved by the Oberlin College Institutional Animal Care and Use Committee. Inner ears were then harvested, the stapes was removed, the round window pierced, and 1 ml of 4% paraformaldehyde was infused through the oval window. The inner ears were then immersion-fixed for an additional 30–60 min. After fixation, tissues were rinsed in PBS. Inner ears were decalcified before or after lac z staining (0.12 M EDTA; 24–48 hr). Both methods produced similar results. To detect ephrin-β-Gal activity, inner ears were washed in lac Z wash buffer (2 mM MgCl2, 0.01% deoxycholate, 0.02% NP-40) for 30 min. The inner ears were then incubated overnight (16–24 hr) at 37C in staining buffer (wash buffer with 0.2% X-gal, 5 mM potassium ferrocyanide, 5 mM potassium ferricyanide). Decalcified tissues were then dehydrated through a graded series of alcohols, cleared in xylene, and embedded in low melting temperature paraffin (Paraplast Extra; Fisher Scientific, Pittsburgh, PA). Tissues were sectioned at 4 μm and collected on glass slides (Superfrost; Fisher Scientific). Embryonic day 18 (E18) mice were harvested from sacrificed females and bisected heads were immersion-fixed in 4% paraformaldehyde (3 hr), then embedded in paraffin and sectioned as above.
For IHC, tissue sections were rehydrated through a graded series of alcohols, incubated in 3% H2O2, then blocked in 1% normal serum. Sections were incubated in a primary polyclonal antibody overnight at 4C (anti-ephrin-B3 1:50–1:100 (SC-7281; Santa Cruz Biotechnology, Santa Cruz, CA); anti-ephrin-B2 1:50–1:100 (SC-1010, Santa Cruz Biotechnology); anti-ephrin-B2 1:50–1:100 (AF496; R&D Systems, Minneapolis, MN). The following day, sections were rinsed in PBS, then incubated in biotinylated secondary antibody (1:200; Vector Labs, Burlingame, CA). Sections were then rinsed and treated with the avidin-biotin complex (ABC; Vector Labs). Sites of antibody binding were visualized with diaminobenzidine (DAB; Sigma, St Louis, MO). Ephrin-B2 inner ears were also processed for IHC following lac Z reactivity. Prior processing for lac z histochemistry did not interfere with subsequent IHC results other than reducing the visibility of the DAB signal.
Estimates of neuron numbers in cochlear and vestibular ganglia were made from hematoxylin- and eosin-stained serial sections. The number of neurons with a clearly identifiable nucleus was counted in every fourth section (16-μm intervals) as described previously (Bianchi et al. 1996). Statistical analysis was completed using Student's t-test.
Results
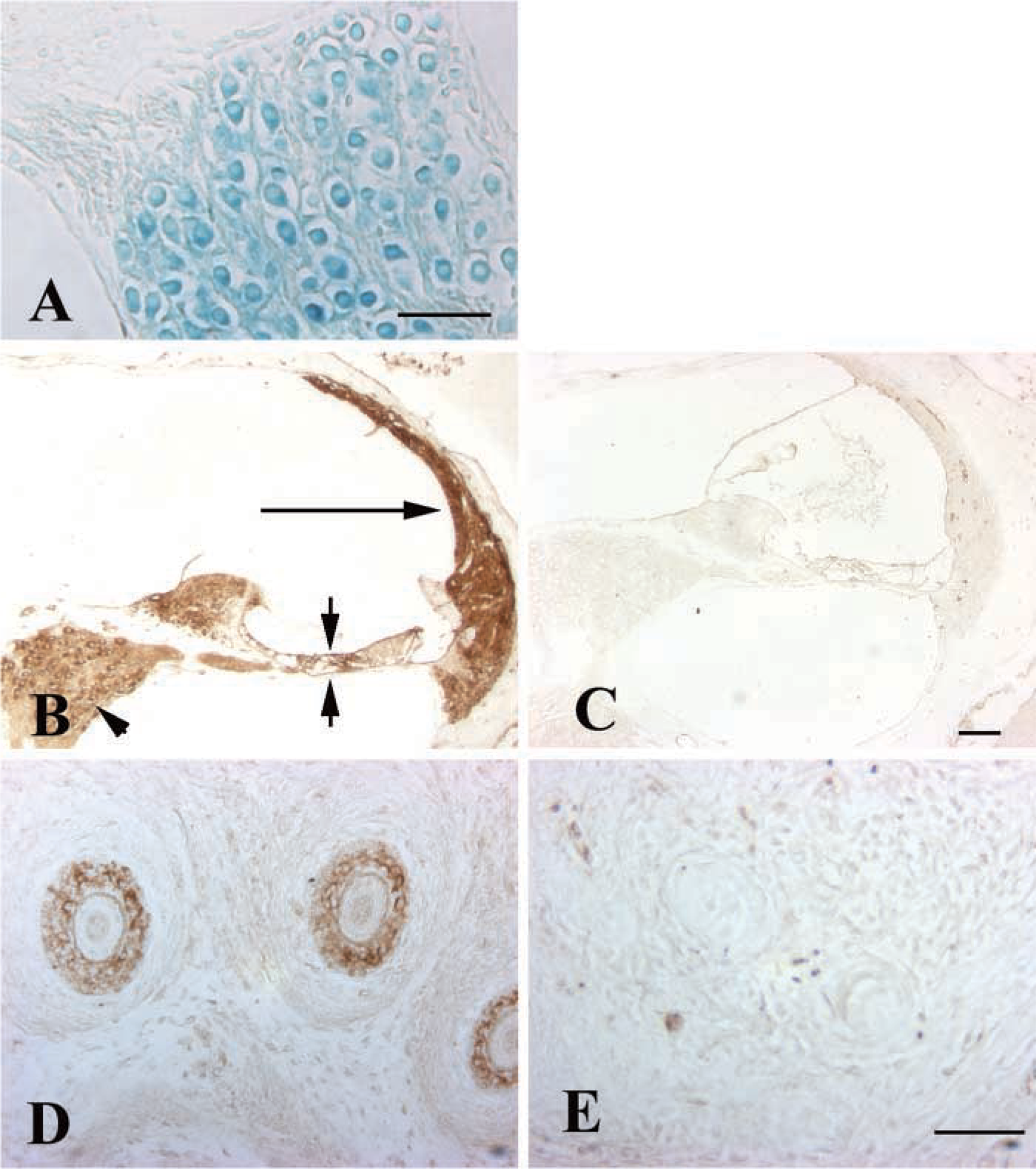
In the adult inner ear, ephrin-B3 lac z reactivity was detected in the cochlear (spiral ganglion) neurons (Figure 1A) and weakly in the stria vascularis (not shown) in four of the six inner ears analyzed. Other regions of the inner ear did not exhibit lac z reactivity. Wild-type mice did not show any reactivity, even after 96 hr in developing solution.
Because the lac z expression was so limited in the inner ear, it was not clear whether the lac z reactivity accurately reflected ephrin-B3 distribution. IHC analysis was therefore completed to examine ephrin-B3 protein production in the adult inner ear. Ephrin-B3 immunoreactivity was widely distributed throughout the inner ear in areas including the cochlear spiral limbus, the organ of Corti, and the lateral wall (Figure 1B). In the vestibular regions, the sensory epithelia and connective tissue areas below the sensory epithelia were reactive (not shown). Both cochlear (Figure 1B) and vestibular neurons (not shown) were also immunoreactive. In contrast to the widespread distribution of ephrin-B3 in the adult inner ear, ephrin-B3 immunoreactivity was absent in the inner ear and brain at E18 (not shown).
The immunoreactivity noted throughout adult inner ear tissues suggested that the antibody was binding nonspecifically. However, immunostaining of ephrin-B3-homozygous mutants revealed that ephrin-B3 immunoreactivity was lost in the null mutants (Figure 1C). Furthermore, in the E18 mouse ephrin-B3 immunoreactivity was detected in whisker follicles of wild-type mice (Figure 1D) but was absent in the whisker follicles of null mutant mice (Figure 1E). Therefore, the IHC studies demonstrated that the anti-ephrin-B3 antibody was specific for ephrin-B3 protein, confirmed that ephrin-B3 protein was not produced in the mutant mice, and allowed more detailed analysis of expression than was possible with lac z histochemistry in these mice.
The gross morphology of the inner ear was normal in the ephrin-B3-null mutants. The hair cells and supporting cells of the cochlear and vestibular epithelia, were present and nerve fibers were seen entering both regions of sensory epithelia, as observed by light microscopy (not shown). Neither cochlear nor vestibular neurons showed a significant decrease in number in the ephrin-B3 null mutant mice (mean number of cochlear neurons in wild-type 1911 ± 169 SE, n=2; homozygous null mutants 2060 ± 57 SE, n=4; mean vestibular neurons in wild-type 1047 ± 54 SE, n=6; homozygous null mutants 934 ± 53 SE, n=6). Although a slight decrease in vestibular neurons was noted in the ephrin-B3-null mutants compared to wild-type mice, no significant difference was noted (p>0.05).
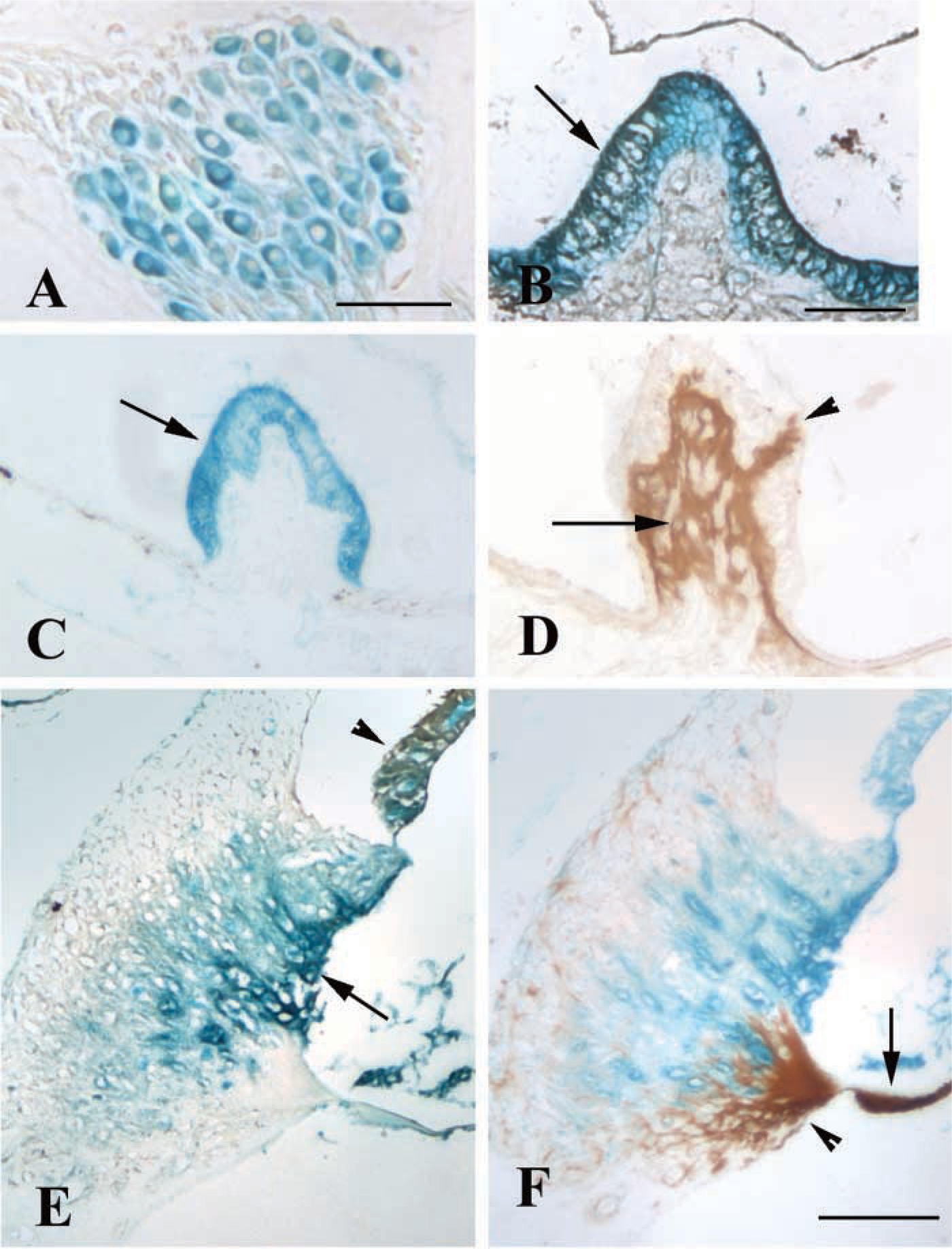
Mice heterozygous for the ephrin-B2 mutation were used to compare lac z and IHC expression in the adult inner ear. Mice homozygous for the ephrin-B2 mutation are embryonic lethal; therefore, they could not be evaluated (Gale et al. 2001). Lac z histochemistry revealed reactivity in both cochlear (Figure 2A) and vestibular neurons (not shown). Various supporting cell types of the cochlea, such as interdental cells, were also reactive, but no reactivity was observed in the sensory hair cells (not shown). Lac z reactivity was also prominent in the outer sulcus cells, their root processes, and the stria vascularis (Figures 2C-2D), areas associated with maintaining ionic homeostasis.
Lac z reactivity revea ed ephrin-B3 expression in the cochlear neurons of the adult inner ear (blue,
IHC studies of ephrin-2-heterozygous mice revealed different results with two different anti-ephrin-B2 antibodies. One antibody (R&D Systems; Figures 2B and C) showed immunoreactivity in areas that overlapped with lac z expression, as would be expected because ephrin-B2 is a transmembrane-anchored protein (Figures 2B and 2C). In contrast, a second anti-ephrin-B2 antibody (Santa Cruz Biotechnology) showed some regions of expression that overlapped with lac z staining, such as cochlear and vestibular neurons, as well as several immunopositive regions adjacent to structures positive for lac z (Figures 2D). The areas of adjacent immunoreactivity were limited to specific sites in the adult inner ear, giving the appearance of immunoreactivity “specific” to certain cell regions such as the spiral limbus, lateral wall, and basilar membrane of the cochlea and the fibrocyte-rich area below the vestibular epithelia. A second lot of this anti-ephrin-B2 antibody (Santa Cruz Biotechnology) failed to stain any region of the inner ear, further suggesting that the areas of adjacent reactivity observed with the first lot of the antibody were largely nonspecific. In summary, lac z reactivity detected ephrin-B2 expression in specific inner ear regions and was useful in determining which commercially available antibody specifically recognized ephrin-B2 in paraffin-embedded tissues. Because ephrin-B2-homozygous mice are not available for study, it was not possible to confirm whether immunoreactivity is lost in homozygous ephrin-B2 mice.
Discussion
The present study demonstrated the need for both lac z histochemistry and IHC to accurately identify the sites of ephrin-B3 and ephrin-B2 expression in the inner ear. Lac z reactivity revealed ephrin-B3 to be only weakly expressed in cochlear and vestibular neurons and in the stria vascularis. Increasing the time in developing solution did not increase the intensity or distribution of the lac z reaction product in the inner ear of the null mutant mice. Further, wild-type mice never showed any reaction product. In contrast, previous studies using the same ephrin-B3-null mutant mice reported that the lac z reactivity was below the level of detection in embryonic brain (Kullander et al. 2001; N. Gale unpublished observations). However, Northern blots of brain tissue showed a decrease in ephrin-B3 in the ephrin-B3-heterozygous mice and a complete loss of ephrin-B3 in ephrin-B3-homozygous mice (Kullander et al. 2001), indicating that the ephrin-B3 gene was disrupted in these mice. Therefore, the lac z gene was only weakly expressed in the ephrin-B3 transgenic mice, for reasons that remain unclear.
Lac z reactivity revea ed eprhin-B2 expression in cochlear neurons (
Because lac z reactivity alone could not be used to map ephrin-B3 expression, IHC analysis was completed. These experiments revealed that ephrin-B3 was widely distributed throughout the adult inner ear of wild-type mice. The observation that this immunoreactivity was absent in the adult inner ear and embryonic whisker follicles of homozygous null mutants indicated that the antibody specifically detected ephrin-B3. These studies also confirmed that the ephrin-B3 protein was not produced in the null mutants.
No gross morphological deficits were noted in ephrin-B3-homozygous mice, suggesting that ephrin-B3 is not required for cell formation and maintenance in the inner ear, or that other ephrins can compensate for the loss of ephrin-B3.
Experiments using heterozygous ephrin-B2 mice revealed lac z expression in vestibular neurons and supporting cells, similar to the distribution reported with another ephrin-B2-null mutant mouse (Cowan et al. 2000). In the cochlea, areas associated with maintaining ionic homeostasis, such as the interdental cells, outer sulcus cells, the root cell processes, and the stria vascularis, were reactive. Previous studies have shown the EphB2 receptor to be associated with ionic homeostasis (Cowan et al. 2000). Therefore, ephrin-B ligands may be necessary for the formation or maintenance of such cell types.
When the lac z expression patterns were compared to the immunoreactivity patterns in the heterozygous ephrin-B2 mice, the specificity of a commercially available antibody was questioned. Whereas one antibody (R&D Systems) overlapped lac z expression, the second antibody (Santa Cruz Biotechnology) showed several areas of immunoreactivity that were adjacent to lac z-positive areas. Although the immunoreactivity with this second antibody looked “specific” in these areas, several of the regions, such as the basilar membrane, have previously been found to nonspecifically bind a variety of antibodies, including antibodies that recognize their appropriate protein in other inner ear regions (B. Schulte, personal communication). Therefore, it appeared that the majority of the immunoreactivity detected with this antibody was nonspecific. The reason for the nonspecific binding to regions adjacent to lac z reactivity is unclear. The immunostaining patterns were not identical to any of the ephrin staining patterns previously observed in paraffin-embedded tissues using antibodies from the same company (Bianchi and Gale 1998; Bianchi and Liu 1999), although the possibility that the antibody detected another ephrin cannot be ruled out. It is also unclear why only one lot of the antibody stained in this manner. The product specification sheet does not indicate that the antibodies have been tested on paraffin-embedded tissues. Therefore, it is possible that the nonspecific binding was related to the method of tissue processing.
In summary, transgenic mice are useful tools for assessing antibody specificity in mice that are homozygous for a null mutation or heterozygous for a mutation in a protein that is not secreted.
Footnotes
Acknowledgements
Supported by NSF IBN 9904566.
We wish to thank S. Anne Hastings for technical assistance, Dr Bradley Schulte for helpful discussion on this project, and Nicole Falk and Annegret Falkner for assisting with analysis of neuron counts.
