Abstract
Nitric oxide (NO)-releasing drugs such as glyceryl trinitrate have been used in the treatment of ischemic heart disease for more than a century. Nevertheless, a detailed analysis of the expression of the NO target enzyme soluble guanylyl cyclase (sGC) in the heart is missing. The aim of the current study was to elucidate the expression, cell distribution, and activity of sGC in the rat heart during postnatal development. Using a novel antibody raised against a C-terminal peptide of the rat β1-subunit of sGC, the enzyme was demonstrated in early postnatal and adult hearts by Western blotting analyses, showing maximal expression in 10-day-old animals. Measurements of basal, NO-, and NO/YC-1-stimulated sGC activity revealed an increase of sGC activity in hearts from neonatal to 10-day-old rats, followed by a subsequent decrease in adult animals. As shown by immunohis-tochemical analysis, sGC expression was present in vascular endothelium and smooth muscle cells in neonatal heart but expression shifted to endothelial cells in adult animals. In isolated cardiomyocytes, sGC activity was not detectable under basal conditions but significant sGC activity could be detected in the presence of NO. An increase in expression during the perinatal period and changes in the cell types expressing sGC at different phases of development suggest dynamic regulation rather than constitutive expression of the NO receptor in the heart.
N
sGC represents the primary target of NO-releasing drugs, such as glyceryl trinitrate, that have been used in the treatment of ischemic heart disease for more than a century. Although the role of sGC in the control of vascular tone is widely appreciated, the importance of the enzyme in endothelial cells of blood vessels or myocytes of the heart is less well documented. In cardiomyocytes, sGC is believed to mediate the positive inotropic effects of NO (Vila-Petroff et al. 1999; Müller-Strahl et al. 2000). At high NO concentrations a negative inotropic effect prevails that is not mediated by sGC (Vila-Petroff et al. 1999; Müller-Strahl et al. 2000).
Recently, the development of YC-1 and its derivative BAY41–2272, both NO-independent modulators of sGC, rekindled strong scientific interest in the NO target enzyme (Wu et al. 1995; Friebe and Koesling 1998; Stasch et al. 2001). sGC is a heterodimeric heme-containing enzyme consisting of an α1- and a β1-subunit (Denninger and Marletta 1999). An α2/β1 heterodimeric enzyme that is equally responsive to activation by NO and YC-1 has also been shown to occur in vivo (Russwurm et al. 1998). In contrast the β2-subunit, which is predominantly expressed in kidney and liver, appears to belong to the class of homodimeric nucleotide cyclases (Koglin et al. 2001).
A number of studies focus on cardiac expression of the different isoforms of NO synthases under physiological and pathological conditions, but very little is known about expression of sGC in the heart. The aim of the current study was to elucidate expression, cell distribution, and activity of sGC in the rat heart during postnatal development. Although dynamic regulation of sGC in other tissues than the heart has previously been demonstrated (see Andreopoulos and Papapetropoulos 2000, and references cited therein), we show in the present study that expression of sGC in the heart is regulated during postnatal development. The presence of sGC in endothelial cells of cardiac vasculature and in cardiomyocytes supports the concept that the cardiac functions of the NO-sensitive enzyme go beyond mere control of vascular tone.
Materials and Methods
Tissue Preparation and Determination of Protein Concentrations
Heart tissue was minced in liquid nitrogen and further homogenized in a teflon vessel containing wolfram balls in a microdismembrator (Braun; Melsungen, Germany). A 0.25 ml homogenization buffer (50 mM TEA, pH 7.4, 2 mM DTT, 2 mM EDTA) was used for approximately 50 mg of tissue. Cytosol was obtained by centrifugation at 45,000 × g for 1 hr at 4C. Protein concentrations were determined by the method of Bradford with bovine plasma gamma globulin as standard (Biorad; Hercules, CA).
Animals
All procedures and care of the rats were in accordance with institutional guidelines for the use of animals in research. Male Wistar rats (20 1-day-old, 12 5-day old, eight 10-day old, eight 20-day-old, eight 30-day-old, and eight adult or 90 day-old) were sacrificed by cervical dislocation. Animals were obtained from the animal facilities of the university.
Western Blotting Analyses
SDS-PAGE was performed in 10% slab gels and transferred electrophoretically onto nitrocellulose membranes (Biorad Minigel System). Sixty μg protein was loaded per lane. The membrane was reversibly stained with Ponceau S and unspecific binding sites were saturated by immersing the membrane for 1 hr in TBST buffer (10 mM Tris-HCl, pH 8.0, 150 mM NaCl, 0.05% Tween-20) containing 5% non-fat dry milk. The membranes were then incubated for 1.5 hr in TBST buffer containing sGC-β1-87 antibody (Behrends et al. 2001) in a 1:500 dilution and 0.5% dry milk. Negative control reactions were run in the additional presence of the synthetic peptide used for immunization (2.5 μg/ml). The membranes were washed three times for 10 min with TBST and subsequently incubated for 1 hr with horseradish peroxidase-labeled anti-rabbit IgG antibodies (diluted 1:4000; Sigma, Diesenhofen, Germany). After three washes with TBST the membranes were processed with the ECL Western blotting detection system according to the recommendations of the manufacturer (Amersham; Poole, UK).
Determination of sGC Activity
sGC activity of heart cytosol (∼50 μg protein per assay tube) was determined by incubation for 10 min at 37C in the presence of 1 mM cGMP, 0.5 mM [32P]-GTP (about 0.2 μCi), 3 mM MgCl2, 50 mM TEA-HCl, pH 7.4, 0.25 g/liter creatine kinase, 5 mM creatine phosphate, and 1 mM 3-isobutyl-1-methylxanthine in a total volume of 0.1 ml as described by Schultz and Böhme (1984). Reactions were started by the addition of cytosolic protein and incubation at 37C. All experiments were stopped by ZnCO3 precipitation, and purification of the enzyme-formed cGMP was performed as described (Behrends et al. 2001). Basal enzyme activity measurements were performed in the absence of NO or YC-1. NO-stimulated measurements were performed in the presence of 100 μM of the NO donor DEA/NO, and NO/YC-1-stimulated enzyme activity measurements were performed in the presence of both 100 μM for DEA/NO and 100 μM for YC-1. YC-1 was dissolved in DMSO; the final concentration of DMSO in the samples did not exceed 2.5%, which did not affect sGC activity at this concentration. Results are presented as means ± SEM of at least four independent experiments performed in duplicate.
Statistical Analysis
All results were controlled for their statistical significance by one-way ANOVA followed a Newman-Keuls post test. A value of p<0.05 was considered statistically significant.
Immunohistochemistry
Hearts of Wistar rats were fixed by immersion in 3.7% formaldehyde solution for 24 hr at 20C and subsequently embedded in paraffin. Paraffin sections (6 μm) mounted on Superfrost slides were deparaffinized and rehydrated. The slides were then treated with 1.2% H2O2 in methanol to inhibit endogenous peroxidase activity. Antigen retrieval was performed using microwave cooking for 30 min in 0.1 M citrate buffer, pH 6.0. To block nonspecific binding sites, slides were treated with 2% normal swine serum in PBS. Then sections were incubated in a humid chamber overnight at 4C with an antiserum against the β1-subunit of sGC (see above) diluted 1:600 in PBS, containing 0.2% (w/v) bovine serum albumin and 0.1% (w/v) NaN3. Slides were then treated for 60 min at 20C with biotinylated anti-rabbit IgG (1:250 in PBS; DAKO, Hamburg, Germany) followed by incubations at 20C for 30 min each with rabbit peroxidase anti-peroxidase (PAP, 1:200 in PBS; DAKO) and ABC (1:250 in PBS; Vector, Burlingame, CA). For visualization of the peroxidase activity, the nickel-glucose oxidase approach was used (Behrends et al. 2001). Alternatively, alkaline phosphatase-conjugated complexes as well as fluorescein-conjugated anti-rabbit IgG were used for visualization.
Specificity of the anti-sGC β1 antibody in immunohisto-chemistry had already been tested by analyses of sections from rat lungs of different age (Behrends et al. 2001). For negative controls, sections were used in which the primary antibody was replaced by the incubation buffer (PBS with BSA and NaN3) alone. In addition, sections from mouse hearts were investigated as negative controls because the novel β1 antibody was raised against a C-terminal peptide of the rat β1-subunit (amino acids 605–619; Genbank M22562) which differed from that of mouse (Behrends et al. 2001). Comparative analyses with a primary antibody from another source were also performed to confirm reaction specificity: polyclonal anti-sGC (1:1000; Calbiochem, San Diego, CA, 1:500).
Cardiomyocyte Preparation
Preparation of cardiomyocytes was performed essentially as described earlier (Isenberg and Klockner 1982). In detail, rat hearts were perfused in a retrograde manner through the aorta with a calcium-free 0.1% collagenase (Biochrom; Berlin, Germany) containing MOPS buffer (100 mM NaCl, 10 mM KCl, 1.2 mM KH2PO4, 5 mM MgSO4, 20 mM glucose, 50 mM taurine, 10 mM MOPS saturated with O2, pH 6.9) for 15–18 min at 37C. Atria were then cut off and ventricles were minced with scissors and incubated in 20 ml KB medium according to Isenberg and Klockner (1982) containing 70 mM KCl, 20 mM taurine, 20 mM glucose, 30 mM KH2PO4, 5 mM MgSO4, 5 mM succinic acid, 5 mM creatine, 1 mM EGTA, 5 mM pyruvic acid, 7.3 mM β-hydroxy-butyric acid, 5 mM Na2ATP saturated with O2, pH 7.4, at 37C for 30–60 min. The cell suspension was passed through 200-μm-mesh nylon gauze, centrifuged (50 × g) for 8 min at room temperature (RT), and resuspended in 10 ml MOPS buffer. This cell suspension was centrifuged (50 × g, 8 min at RT) through a gradient of MOPS buffer containing 6% albumin. The resulting cell pellet was washed two times in 10 ml MOPS buffer. Cytosol preparation from cardiomyocytes was performed as described above.
Results
Occurrence and Activity of sGC in Rat Heart and Its Developmental Regulation
To investigate cardiac sGC expression and to screen for a potential difference in expression during development, hearts were harvested from early postnatal (1-, 5-, 10-, 20-, 30-day-old) and adult rats. In the respective cardiac cytosols, sGC activity determinations and Western blotting analyses were performed in parallel. Figure 1 shows the result of four enzyme assay determinations performed independently with separate cytosol preparations under basal, NO-, and NO/YC-1-stimulated conditions. Guanylyl cyclase activity was significantly higher in heart cytosol from 10-day-old vs neonatal or vs adult rats.
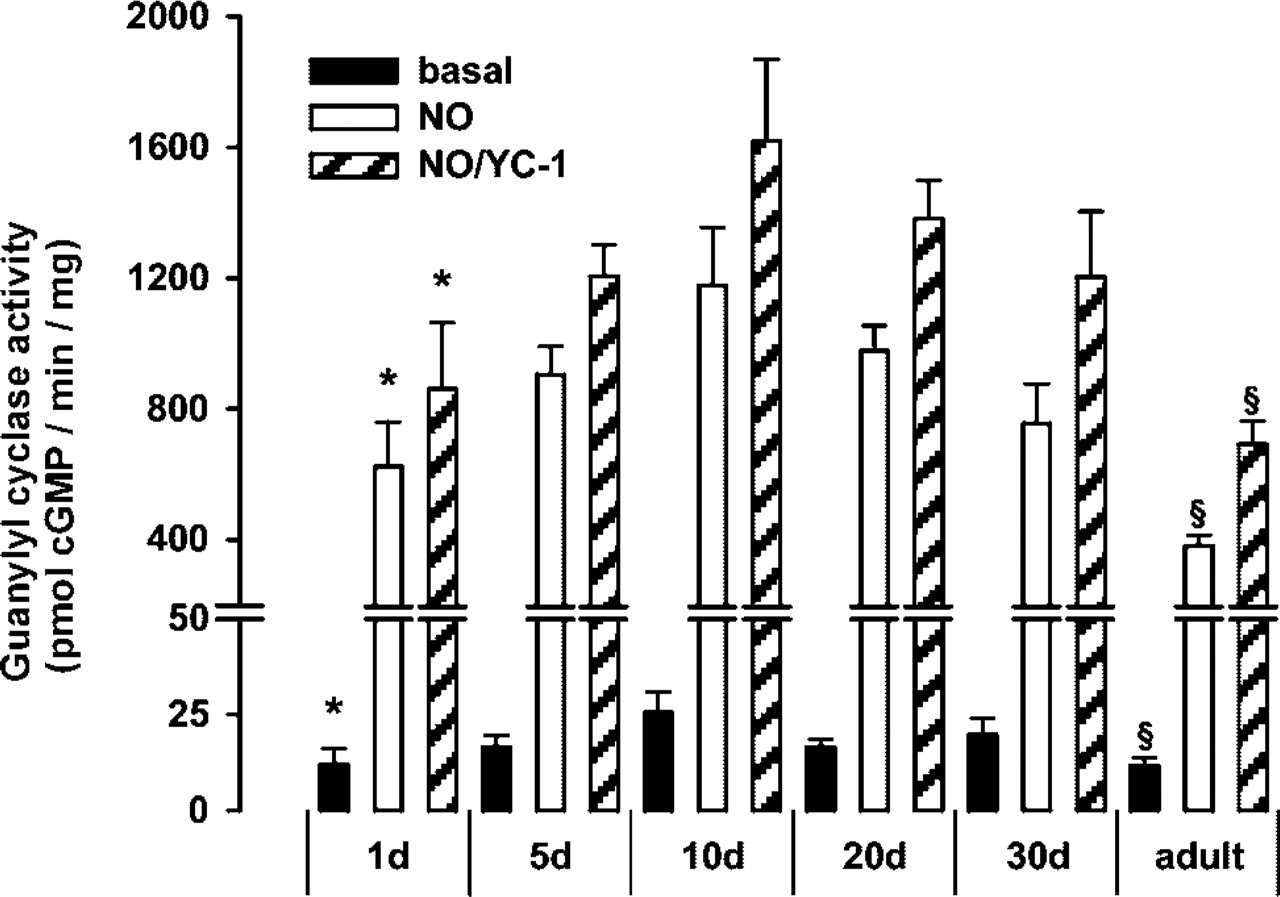
sGC activity in heart during postnatal development. Guanylyl cyclase activity measurements of cardiac cytosol from neonatal to adult rats under basal (black columns), NO-stimulated (white columns), and NO/YC-1 stimulated conditions (gray columns). Data are expressed as mean ± SEM (n=4, ∗ p<0.05 1-day-old vs 10-day-old; § p<0.05 adult vs 10-day-old).
By use of the novel sGC antibody directed against the β1-subunit (Behrends et al. 2001) Western blotting analysis showed a band at the expected molecular weight of 70 kD in cytosolic protein fractions from 1-, 5-, 10-, 20-, 30-day-old and adult rats (Figure 2, below). Analysis of Western blotting autoradiographs by scanning densitometry from four independent cytosol preparations showed peak expression in 10-day-old rats and significantly higher expression in 10-day-old vs adult rats (Figure 2).
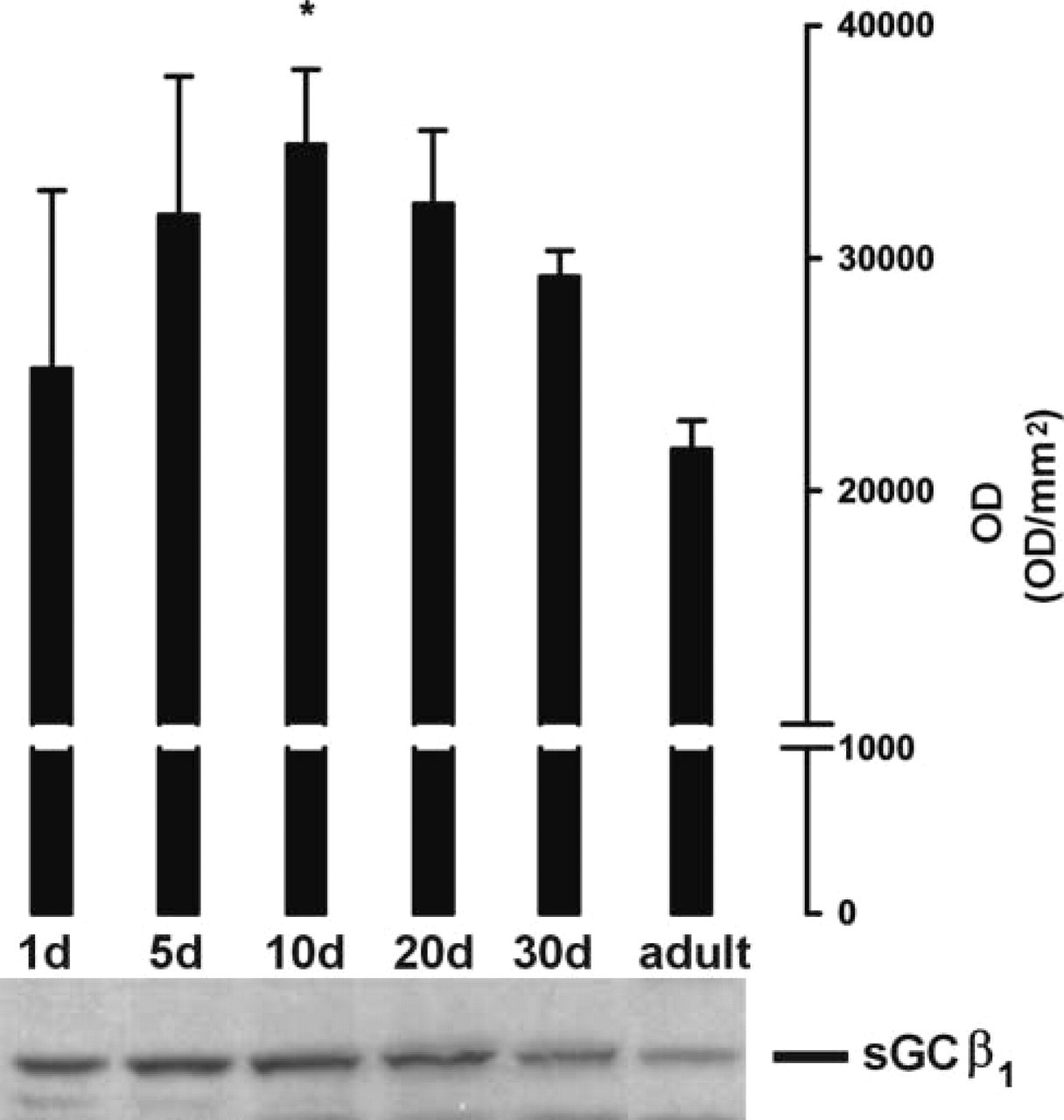
Immunoblotting analysis of cardiac sGC-β1 during postnatal development. Analysis of Western blotting autoradiographs by scanning densitometry from four independent cytosol preparations expressed as optical density (OD) per mm2 ± SEM (above). A representative immunoblot is shown below (∗ p<0.05 cytosol from 10-day-old vs adult rats).
(
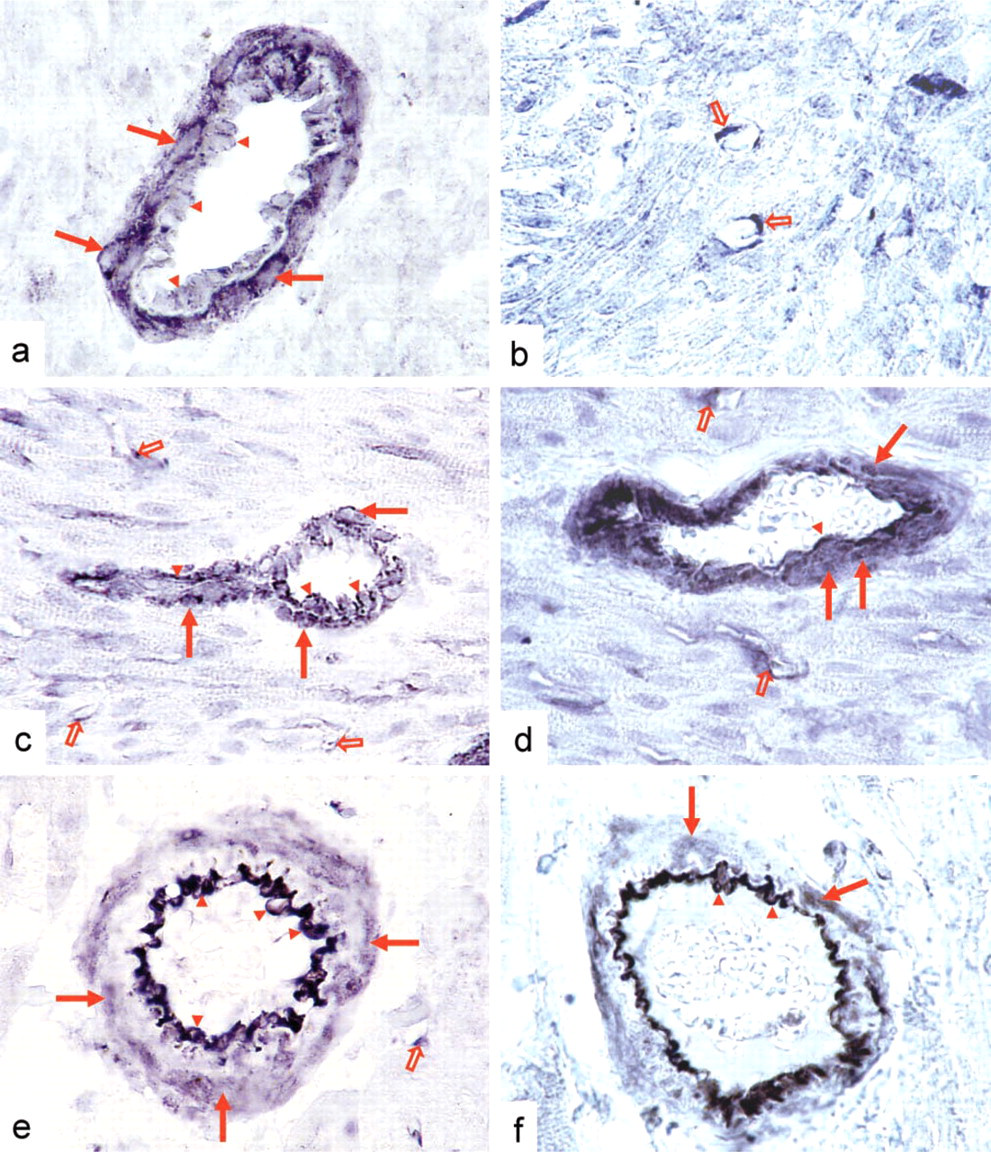
Localization of sGC in Heart. In hearts of adult rats and during each period of postnatal development, the most prominent sGC-specific staining was found in the arteries of the myocardium (Figure 3). At postnatal day 1, cardiac arteries displayed strong sGC immunoreactivity in their smooth muscle cell layer, whereas endothelial cells of these vessels showed only weak staining (Figure 3a). At day 10, staining of comparable intensity was present both in smooth muscle cells and in endothelial cells of cardiac arteries (Figure 3b). Compared to neonatal and 10-day-old rats, a remarkable decrease of sGC expression in arterial smooth muscle cells was apparent in adult heart (Figure 3c). This was accompanied by a parallel increase of sGC-specific staining in endothelial cells of the same cardiac vessels (Figure 3c). During each period of development, capillaries of rat heart also displayed sGC immunoreactivity (Figure 3), whereas sGC expression in cardiomyocytes was barely detectable by immunohistochemical analyses.
Occurrence and Activity of sGC in Cardiomyocytes
In the described immunohistochemical experiments, a subtle staining pattern could be observed in cardiomyocytes throughout the developmental stages. To determine whether cardiomyocytes express sGC, we performed experiments using isolated cardiomyocytes of adult rats. Comparison of sGC activity in cytosol from isolated cardiomyocytes and cytosol from whole heart revealed that sGC activity in cytosol from cardiomyocytes was below the detection limit under basal conditions (Figure 4). In contrast, activity was clearly detectable after activation with NO or YC-1/NO, showing the integrity of the enzyme in isolated cardiomyocytes. The finding of low but detectable expression of sGC in cardiomyocytes compared to whole heart is consistent with the immunohistochemical data showing only faint staining of cardiomyocytes. The combination of YC-1/NO led to a significant increase vs NO alone in whole heart but not in cardiomyocytes.
Discussion
NO-releasing drugs such as glyceryl trinitrate have been used in the treatment of ischemic heart disease for more than a century. Symptoms of angina pectoris are primarily relieved by a decrease in venous tone and a reduction in cardiac preload (Williams et al. 1965). When insufficient amounts of endogenous endothelial NO are produced, glyceryl trinitrate also dilates cardiac vessels (Goldstein et al. 1974; Bache et al. 1975; Cohn et al. 1977; Klassen et al. 1999). The well-described classical role of sGC in the NO-dependent control of vascular tone has led to the prevailing view that the enzyme is constitutively expressed in vascular smooth muscle cells. In a very recent article we have shown that sGC expression in smooth muscle cells of pulmonary arteries decreases in adult animals (Behrends et al. 2001). A decrease of sGC expression in smooth muscle cells of carotid arteries after balloon injury has also been described using Western blotting analysis of microdissected tissue in animals (Chen et al. 2000). Therefore, expression of sGC in arterial smooth muscle cells is not static but depends on developmental and pathophysiological factors. In the present study we find that cardiac vessels show strong smooth muscle sGC expression in neonatal animals, which decreases during postnatal life. This sGC down-regulation in smooth muscle cells may hamper the efficacy of NO-dependent dilatation of the cardiac microcirculation in adult animals. We found that the age-dependent decrease of sGC expression in smooth muscle cells of pulmonary arteries (Behrends et al. 2001) and cardiac arteries (current study) is accompanied by an increase of sGC expression in the endothelial cells of the same vessels. In isolated coronary endothelial cells, the signaling cascade involving NO, sGC, and cGMP-dependent protein kinase has been shown to mediate changes in permeability induced by VEGF (Wu et al. 1996). Studies in other organs confirm these results and indicate a major role of the NO/cGMP pathway in the control of permeability (Morbidelli et al. 1996; He et al. 1998; Mayhan 1999; Russell et al. 2000).
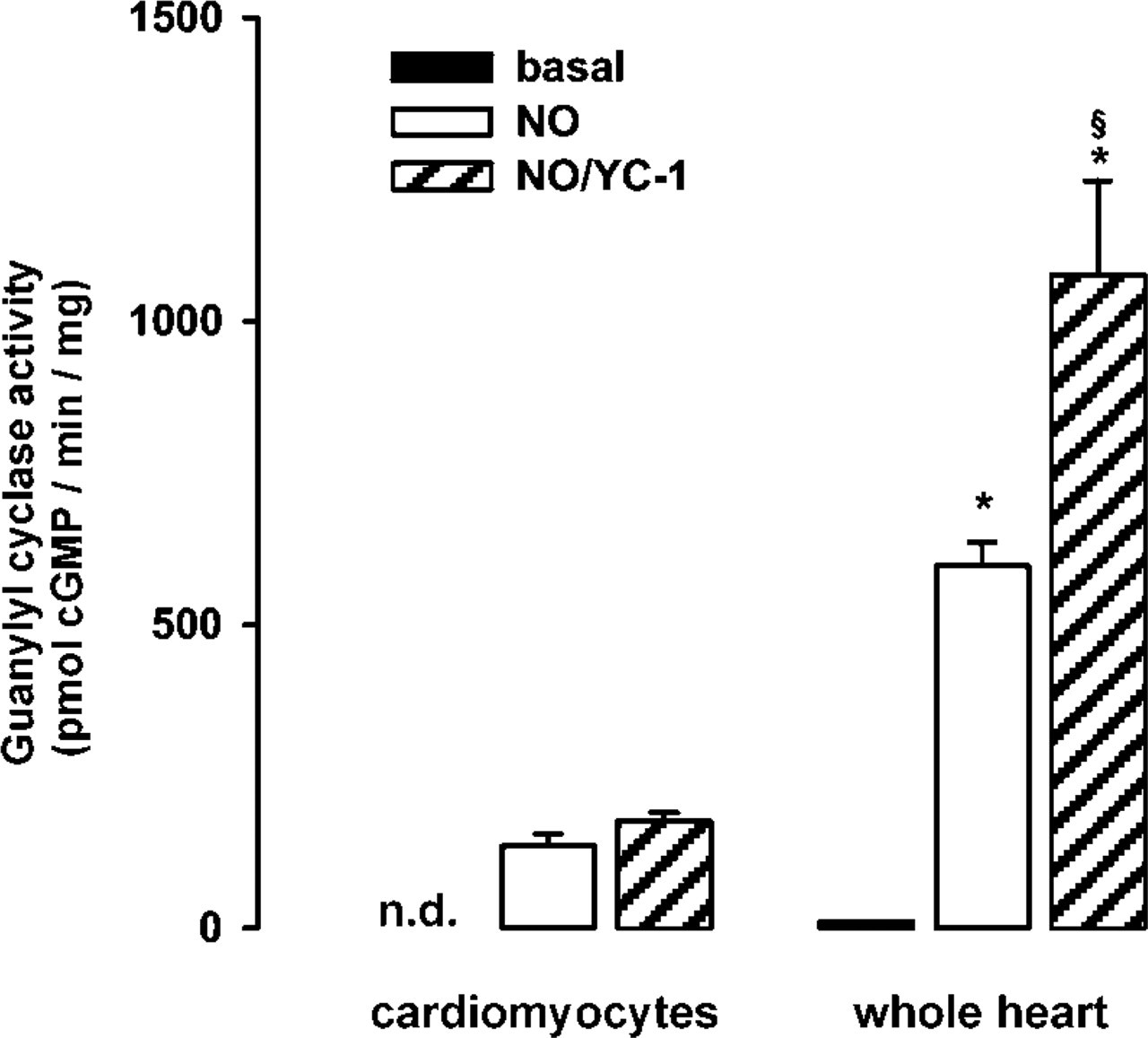
sGC activity in rat heart and adult rat isolated cardiomyocytes. Guanylyl cyclase activity measurements were performed under basal (black columns), NO-stimulated (white columns), and NO/YC-1-stimulated conditions (gray columns). Data are expressed as mean ± SEM (n=4, ∗ p<0.05 isolated cardiomyocytes vs whole heart; § p<0.05 NO/YC-1 vs NO-stimulated conditions in whole heart; n.d., not detectable).
Enzyme activity measurements of isolated cardiomyocytes showed undetectable basal but significant NO-stimulated sGC activity. This finding supports data that postulate a role for cGMP-dependent effects of NO in cardiomyocytes. These include modulation of cardiac contractility (Vila-Petroff et al. 1999; Müller-Strahl et al. 2000), early preconditioning (Rakhit et al. 2000), reduction of ventricular myocyte stunning after simulated ischemia-reperfusion (Gandhi et al. 1999), and regulation of cardiac β-adrenergic response (Balligand 1999). Interestingly, the combination of YC-1/NO led to a significant increase vs NO alone in whole heart but not in cardiomyocytes. This is similar to findings by Wegener et al. (1997) showing a more pronounced activation of sGC activity in smooth muscle cells vs rat myocardium.
In rat lung, a significant increase of sGC expression has been shown during the perinatal period, with a peak at the early postnatal period (days 8–16) (Bloch et al. 1997). In the present study we show that an increase in sGC activitiy during the early postnatal period is not restricted to lung tissue but also occurs in heart. In the heart, the perinatal period is characterized by substantial angiogenesis (Tomanek et al. 1996). During the early postnatal period, marked capillary growth has been demonstrated in rat heart. Capillary volume density increases from 6% on postnatal day 1 to a peak of 15% on postnatal day 11, with a subsequent decrease during further development (Anversa et al. 1978, 1979; Olivetti et al. 1980). Because endothelial cells of capillaries showed sGC immunore-activity throughout the different stages of development, the changes in capillary density could contribute to the peak of sGC activity and expression on postnatal day 10.
In conclusion, changes in expression levels, activity and the cell types expressing sGC at different times of development suggest dynamic regulation rather than static expression of the NO target enzyme in the heart. The shift of expression from smooth muscle cells to endothelial cells during postnatal development and its expression in cardiomyocytes support the concept that the cardiac functions of the NO-sensitive enzyme go beyond mere control of vascular tone.
Footnotes
Acknowledgment
Supported by the Deutsche Forschungsgemeinschaft.
